Abstract
This study was made to assess the possibilities and limits of minimally invasive transoral approach to the nasopharynx using the Da Vinci surgical robot. It was conducted on eleven corpses, without need for palatine split; using surgical robots Da Vinci models S HD and Si HD. We have defined “anatomical key landmarks” on all sides of the nasopharynx, to confirm our hypothesis. All of the nasopharynx could be visualized and transorally reached by the robot with a validation of all the key landmarks. The advantages and shortcomings of this technique were discussed, as well as the ability to use those results on human subjects. This study allowed us to show the possibility to access the nasopharynx by minimally invasive transoral robotic surgery. This new technique opens a new field for surgery of the skull base or the nasopharynx.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Classically, radiotherapy or concurrent chemoradiotherapy are the most common treatments in nasopharynx cancer, depending on its stage. In case of local recurrence or failure, which affects approximately 18–58% of patients [1], re-irradiation protocols exist but with important morbidity: osteoradionecrosis, neurological injuries, trismus, chronic otitis media or radio-induced malignancies. In some teams, surgical access of the nasopharynx is processed through external [2, 3] or endonasal [4] approaches, with a major risk of complications. The external path is more aesthetically offensive since it creates a skin scar, but allows a better control of the deep invasion of the parapharyngeal space. This is the reason why the endonasal way has been used for the last 20 years, using optical instruments to perform some resections with a minimal invasive approach. This technique requires two operators and allows an excellent visualization in two dimensions, it also creates new difficulties: the difficulty to achieve complex gestures in a confined location like the nasal cavity or the nasopharynx and the impossibility to stitch. The procedure also generates morbidity, especially because of the requirements to perform resections in the nasal cavity (septum, turbinates) with functional respiratory after-effects.
Robotic surgery with the Da Vinci system in Otorhinolaryngology [5] was developed in 2005 after its use in other surgical specialties (Urology, gynaecology, heart surgery…). The robot used a so called minimally invasive surgery with several advantages: suppression of physiological tremor, use of both hands allowing many different movements through a small incision or even without any opening, visualization in three dimensions; all of this without lengthening classical operating times. Thus, the surgical management of cancers of the upper aero-digestive tract is amended, including the reduction of morbidity related to the procedure since the resumption of normal feeding can be carried out sooner while the hospital stay is convergingly shorter [6]. The safety of Transoral Robotic Surgery (TORS) in some areas such oropharynx, hypopharynx and superior part of the larynx first was established through several animal studies [7] and then works on corpses [8], clinical trials using this method were carried out in France [9] and recently a multicenter research trial [10]. However, the nasopharynx, or rhinopharynx, or cavum, remains a difficult zone to access transorally and many different techniques [11, 12] were considered for its approach but is possible only by splitting the soft and the hard palate [13] for some authors. On the other hand, the interest is multidisciplinary: neurosurgical transorally exploration of the skull base has been the subject of several publications [14,15,16,17]. The objective of this study is to establish the feasibility of the access to the nasopharynx and to define the limits of minimally invasive transoral robotic surgery through the use of anatomical subjects.
Materials and methods
Eleven corpses from the Centre du Don du Corps de l’Université Paris Descartes have been included in this retrospective study between April 2013 and February 2016. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study. Corpses were fresh and not fixed nor frozen to ensure tissue quality. Gender, age, height, mouth opening, and dentition were recorded for each corpse. The opening of mouth of the subject was measured clinically by the intermaxillary length, the hard palate was measured by fluoroscopy before the procedure using a C-arm fluoroscope BV Pulsera (Philips®, Andover, MA, USA).
The subjects were settled in supine position, a block under the shoulders, head in extension. To maintain the mouth opening, three spreaders were considered: Doyen-Jansen, Boyle-Davis and Feyh-Kastenbauer simulating an oro-tracheal intubation, while two rubber catheters inserted nasally and retrieved orally maintained a good superior retraction of the soft palate, as well as a pull suture median on mobile tongue and if the palatine uvula was too long, a stitch was placed in its center to pull it within the oral cavity: this manipulation made it possible to insert the camera along with two arms of the robot beyond the hard palate, mouth and teeth of the corpse without damaging them (Fig. 1).
The dissections were carried out using the Da Vinci Surgical robots models S HD and Si HD (Intuitive Surgical®, Sunnyvale, CA, USA) within the Ecole Européenne de Chirurgie. An 8.5 mm 30 degree 3D endoscopic camera, a 5 mm EndoWrist® monopolar cautery as well as a 5 mm EndoWrist® Maryland dissector have been used in three of the four arms purposed by the robotic system to simulate a surgical procedure. A fourth arm with an 8 mm Harmonic® ultrasonic clamp (Ethicon Endo-Surgery ®, Cincinnati, OH, USA) has been installed. The photographs and videos were recorded with an acquisition system MediCap USB300 (MediCapture®, Philadelphia, PA, USA).
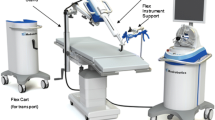
The Control panel was located away from the patient; the robot was placed on the right side of the table, leaving room at the head of the patient to the assistant for the aspiration of the surgical field and physical conflict prevention between the arms of the robot and the oral cavity (Figs. 2 and 3).
Schematic view of the surgical setup. Chauvet et al. [14]
For each patient, we have established a dissection protocol including fourteen anatomical key landmarks distributed on the different areas of the nasopharynx. The upper and posterior parts have been defined by the lower and posterior faces of the sphenoid body, the sphenoidal sinus, the sphenoidal clivus, the remnants of the pharyngeal tonsils, and the median recess of the pharynx. The anterior and lower parts have been defined by the choanaes, the rear edge of the vomer, the posterior part of the turbinates, and the dorsal side of the soft palate. Finally, the lateral side has been defined by the ostium of the Eustachian tube, the torus tubarius, the pharyngeal recess or fossa of Rosenmüller, the salpingopharyngeal and salpingopalatine folds and the junction with the soft palate. These anatomical key landmarks had to be viewed and reached without conflict by the instruments to be validated.
Results
The Average installation time of the robot was 20 min (between 11 and 37 min). Differences in gender, size, sex, mouth opening, length of the hard palate as well as the dentition between the anatomical subjects did not interfered with the procedure, as other studies have suggested [18] (Table 1).
The entire nasopharynx was visualized using the 30 degree angulation of the endoscope placed just behind the rear edge of the hard palate and reached by instruments (Figs. 4 and 5). The possibility to dissect tissues has been validated on the eleven bodies for the fourteen anatomical key points. However, we note pharyngeal tonsils removal and a history of velopharyngoplasty in two subjects.
The dissection of the sphenoidal sinus to the sella turcica, the pituitary gland, and even the optic chiasm as well as an implementation of a mucosal flap using the monopolar cautery and its closure by stitches have been carried out successfully; this subject is treated in an annex study [14].
There were no oral lesions or major conflict between the instruments limiting the functioning of the robot. The use of a 0 degree endoscope or 8 mm instruments were found irrelevant given the angulation and the narrowness of the surgical field.
Discussion
For the last 20 years, the nasopharyngectomy described by Wei [19] has proved valuable after radio-chemotherapy failures in nasopharynx malignancies. More recently, endoscopic approach has superior carcinological outcomes for low-sized tumors with minimal lateral extension [20]. Those surgical interventions seem to allow a better local control than re-irradiation protocols.
In a 2008 anatomical study, Ozer and Waltonen [11] have already suggested to use the Da Vinci System in TORS on human corpse. The Ohio State University team had divided the soft palate along the middle line and retracted the two parts on both sides.
At the Queen Mary Hospital in Hong Kong, Wei and Ho [12] described in 2010 the first clinical trial of TORS, with a slightly modified procedure compared to the previous authors. The two teams performed different technique, but needed the soft palate split. Splitting the soft palate can lead to major after-effects, especially after radio-chemotherapy failure: velopharyngeal insufficiency, Eustchian tube dysfunction, osteonecrosis…
Through this preliminary study on eleven corpses, a minimally invasive transoral access is possible thanks to robotic surgery. In addition, the gear miniaturization allows a complete approach of structures and their dissection without need for palatine split. Furthermore, the use of two instruments without tremor as well as 3D visualization makes this technique more efficient. Removal of nasopharynx lesions, whether they are malign or not, can be achieved with minimal oral or nasal aftereffects.
In our specialty, the clinical applications of this study can be multiple: resection of benign lesions (Tornwaldt cyst, naso-pharyngeal fibroma…) or malignant tumor (Undifferentiated Carcinoma of Nasopharyngeal Type UCNT, lymphoma…), surgery of the sphenoidal sinus (mucocele…) and of the skull base [21].
However, some aspects still have to be criticized. First of all, this robot was not designed for a transorally nasopharynx access. Because of this, it requires a rigorous installation allowing the surgeon to reach for and use his instruments without impediment at all time, also 5 mm instruments which size is maximal for the surgical field. New robots are being designed like single-arm robots Medrobotics Flex® and Intuitive Surgical Da Vinci SP®, cadaveric studies were recently published [22, 23].
Second, the lack of force feedback changes the surgical sensations and perceptions, two features that are essential in this region with the proximity of noble structures, including the internal carotid artery. This study did not have to consider the flesh depth; studies of injection of the internal carotid artery to find it through the lateral wall of the nasopharynx and its integration within the console of the robot have been made [24]. However, this lack of sensibility is offset by the absence of tremor and especially the visualization in three dimensions of all the surgical field. In addition, Simon and al. had calculated in 2016 the position of the internal carotid artery in relation to reliable anatomical landmarks [25]. Through a cadaveric study of maxillary swing, the artery was located in a reproducible way next to the lateral pharyngeal recess, the isthmus of Eustachian tube and the torus tubarius.
Third, although this is a cadaveric study, the cost of this procedure must be taken into account in a context of clinical practice. In this capacity, a surgical procedure even with the robot will be more efficient than re-irradiation protocols and frequently after-effects care. Nevertheless, the multiplication of possible procedures in various surgical specialties allows the investment required for robotic surgery.
Finally, it is important to check our cadaveric results on patients. Indeed, the anatomical data for teeth, mouth opening and thickness of living flesh can vary, as much as the actual operating conditions such as bleeding or possibly fogging. It may be viable to associate an endoscopic approach with a TORS [26].
The innovation from transoral robotic surgery allows access to the nasopharynx by a novel infero-superior approach through natural ways without the need for additional incision, thus avoiding morbidity associated with the procedure. These benefits have allowed the first drilling of the sella turcica [27] and the removal of the pituitary gland transorally [28].
Further studies are needed to compare the results on anatomical subjects with those on human patients and assess the possibilities of minimally invasive nasopharynx access by transoral robotic surgery in humans.
Conclusion
The use of the Da Vinci robots models S HD and Si HD allowed a complete approach of the nasopharynx by minimally invasive transoral robotic surgery on eleven corpses. The advanced technology offered by the robot gives place to a unique access of the nasopharynx as compared to conventional ways. This preliminary cadaveric study is to be confirmed by clinical trials.
References
Lee AW, Law SC, Foo W, Poon YF, Cheung FK, Chan DK, Tung SY, Thaw M, Ho JH (1993) Retrospective analysis of patients with nasopharyngeal carcinoma treated during 1976–1985: survival after local recurrence. Int J Radiat Oncol Biol Phys 26(5):773–782
Wei WI, Ho CM, Yuen PW, Fung CF, Sham JS, Lam KH (1995) Maxillary swing approach for resection of tumors in and around the nasopharynx. Arch Otolaryngol Head Neck Surg 121(6):638–642
King WW, Ku PK, Mok CO, Teo PM (2000) Nasopharyngectomy in the treatment of recurrent nasopharyngeal carcinoma: a 12-year experience. Head Neck 22(3):215–222
Yoshizaki T, Wakisaka N, Murono S, Shimizu Y, Furukawa M (2005) Endoscopic nasopharyngectomy for patients with recurrent nasopharyngeal carcinoma at the primary site. Laryngoscope 115(8):1517–1519
Mc Leod IK, Mair EA, Melder PC (2005) Potential application of the Da Vinci minimally invasive surgical robotic system in otolaryngology. Ear Nose Throat J 84:483–487
Hans S, Delas B, Gorphe P, Ménard M, Brasnu D (2012) Transoral robotic surgery in head and neck cancer. Eur Ann Otorhinolaryngol Head Neck Dis 129(1):32–37
Weinstein GS, O’malley BW Jr, Hockstein NG (2005) Transoral robotic surgery: supraglottic laryngectomy in a canine model. Laryngoscope 115(7):1315–1319
Hockstein NG, O’Malley BW Jr, Weinstein GS (2006) Assessment of intraoperative safety in transoral robotic surgery. Laryngoscope 116(2):165–168
Hans S, Badoual C, Gorphe P, Brasnu D (2012) Transoral robotic surgery for head and neck carcinomas. Eur Arch Otorhinolaryngol 269:1979–1984
De Almeida JR, Li R, Magnuson JS, Smith RV, Moore E, Lawson G, Remacle M, Ganly I, Kraus DH, Teng MS, Miles BA, White H, Duvvuri U, Ferris RL, Mehta V, Kiyosaki K, Damrose EJ, Wang SJ, Kupferman ME, Koh YW, Genden EM, Holsinger FC (2015) Oncologic outcomes after transoral robotic surgery: a multi-institutional study. JAMA Otolaryngol Head Neck Surg 141(12):1043–1051
Ozer E, Waltonen J (2008) Transoral robotic nasopharyngectomy: a novel approach for nasopharyngeal lesions. Laryngoscope 118(9):1613–1616
Wei WI, Ho WK (2010) Transoral robotic resection of recurrent nasopharyngeal carcinoma. Laryngoscope 120(10):2011–2014
Tsang RK, Mohr C (2013) Lateral palatal flap approach to the nasopharynx and parapharyngeal space for transoral robotic surgery: a cadaveric study. J Robot Surg 7(2):119–123
Chauvet D, Missistrano A, Hivelin M, Carpentier A, Cornu P, Hans S (2014) Transoral robotic-assisted skull base surgery to approach the sella turcica: cadaveric study. Neurosurg Rev 37(4):609–617
O’Malley BW Jr, Weinstein GS (2007) Robotic anterior and midline skull base surgery: preclinical investigations. Int J Radiat Oncol Biol Phys 69(2 Sup):S125–S128
O’Malley BW Jr, Weinstein GS (2007) Robotic skull base surgery: preclinical investigations to human clinical applications. Arch Otolaryngol Head Neck Surg 133:1215–1219
Lee JY, O’Malley BW Jr, Newman JG, Weinstein GS, Lega B, Diaz J, Grady MS (2010) Transoral robotic surgery of the skull base: a cadaver and feasibility study. ORL J Otorhinolaryngol Relat Spec 72:181–187
Amelot A, Trunet S, Degos V, André O, Dionnet A, Cornu P, Hans S, Chauvet D (2015) Anatomical features of skull base and oral cavity: a pilot study to determine the accessibility of the sella by transoral robotic-assisted surgery. Neurosurg Rev 38(4):723–730
Chan JY, Wei WI (2012) Critical appraisal of maxillary swing approach for nasopharyngeal carcinoma. Expert Opin Ther Targets 16(Suppl 1):S111–S117
Vlantis AC, Lee DL, Wong EW, Chow SM, Ng SK, Chan JY (2016) Endoscopic nasopharyngectomy in recurrent nasopharyngeal carcinoma: a case series, literature review, and pooled analysis. Int Forum Allergy Rhinol. https://doi.org/10.1002/alr.21881
Trévillot V, Garrel R, Dombre E, Poignet P, Sobral R, Crampette L (2013) Robotic endoscopic sinus and skull base surgery: review of the literature and future prospects. Eur Ann Otorhinolaryngol Head Neck Dis 130(4):201–207
Richmon JD (2015) Transoral palate-sparing nasopharyngectomy with the Flex® System: preclinical study. Laryngoscope 125(2):318–322
Tsang RK, Holsinger FC (2016) Transoral endoscopic nasopharyngectomy with a flexible next-generation robotic surgical system. Laryngoscope 126(10):2257–2262
Tsang RK, Sorger JM, Azizian M, Holsinger CF (2015) Real-time navigation in transoral robotic nasopharyngectomy utilizing on table fluoroscopy and image overlay software: a cadaveric feasibility study. J Robot Surg 9(4):311–314
Simon F, Vacher C, Herman P, Verillaud B (2016) Surgical landmarks of the nasopharyngeal internal carotid using the maxillary swing approach: a cadaveric study. Laryngoscope 126(7):1562–1566
Yin Tsang RK, Ho WK, Wei WI (2012) Combined transnasal endoscopic and transoral robotic resection of recurrent nasopharyngeal carcinoma. Head Neck 34(8):1190–1193
Chauvet D, Hans S, Missistrano A, Rebours C, Bakkouri WE, Lot G (2016) Transoral robotic surgery for sellar tumors: first clinical study. J Neurosurg 23:1–8
Chauvet D, Hans S, Lot G (2016) Abord transoral des adénomes hypophysaires assisté par le robot: premiers patients. Ann Endocinol Paris 77(4):298
Acknowledgements
The authors wish to thank Intuitive Surgical and Antoine Missistrano, the Ecole Européenne de Chirurgie as well as the Centre du Don des Corps de l’Université Paris Descartes, the Ligue contre le Cancer and the Fondation des « Gueules Cassées » for their help, and Mélodie Garcia for her proof-reading.
Author information
Authors and Affiliations
Ethics declarations
Conflict of interest
Dr. Amine Harichane, Dr. Dorian Chauvet, and Pr. Stéphane Hans declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MP4 443620 kb)
Rights and permissions
About this article
Cite this article
Harichane, A., Chauvet, D. & Hans, S. Nasopharynx access by minimally invasive transoral robotic surgery: anatomical study. J Robotic Surg 12, 687–692 (2018). https://doi.org/10.1007/s11701-018-0804-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11701-018-0804-7