Abstract
Background
Several studies have found that metabolic surgery can significantly improve glucose homeostasis; however, the intrinsic mechanisms remain unclear. Accumulating evidence suggests that duodenal bypass plays a crucial role in the treatment of type 2 diabetes mellitus (T2DM). Here, we aimed to evaluate the effect of duodenal reflux on glucose metabolism in T2DM.
Methods
A high-fat diet and low-dose streptozotocin (STZ) administration were used to induce T2DM in male rats, which were assigned to three experimental groups: sham operation (SO; n = 10), new duodenal-jejunal bypass (NDJB; n = 10), and new duodenal-jejunal bypass with a tube (NDJBT; n = 10). Weight, food intake, oral glucose tolerance test (OGTT) results, glucagon-like peptide 1 (GLP-1) levels, and histopathology were assessed before or after surgery. Plain abdominal radiography was performed 1 week after the operation.
Results
Plain abdominal radiography indicated the occurrence of contrast agent reflux into the duodenum. The body weight and food intake in all three groups did not significantly differ before and after surgery. The NDJB and particularly the NDJBT groups exhibited better glucose tolerance, lower fasting blood glucose (FBG) levels, lower area under the curves for OGTT (AUCOGTT) values, and higher GLP-1 levels, as compared with the sham group postoperatively. The villus height and crypt depth were both shorter in the biliopancreatic limb after NDJBT, as compared with those after SO and NDJB.
Conclusions
Thus, exclusion of the duodenum alone and tube placement can effectively prevent duodenal reflux and improve glucose homeostasis, which further suggests that the duodenum plays an important role in T2DM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetes is a multifactorial metabolic disease characterized by high fasting blood glucose levels and impaired insulin secretion or insulin resistance [1]. In 2017, the International Diabetes Federation (IDF) reported that about 451 million individuals had diabetes worldwide. According to the current growth rate, this number is expected to reach 653 million by 2045 [2]. The complications of T2DM can lead to disabilities and can also be life threatening [3]. The primary treatment methods for T2DM include rational dietary habits, increased physical activity, and medication [4]. In the 1950s, bariatric surgery was primarily used to address severe obesity and reduce weight [5]. In the 1980s, a clinical follow-up study found that the treatment of obesity via bariatric surgery also relieved the high glucose levels to varying extents [6]. Furthermore, abundant evidence suggests that obesity-related surgery has a major effect on resolving T2DM and may be better than traditional therapy [7]. At present, metabolic surgery is recommended as the treatment method for T2DM by the American Diabetes Association (ADA) and the IDF [8].
Biliopancreatic diversion (BPD) and Roux-en-Y gastric bypass (RYGBP) currently appear to be the most effective in the treatment of T2DM among the different types of metabolic surgery; the remission rates are about 95 and 75%, respectively [9, 10]. The bypassed intestine in BPD, which is much longer than that in RYGBP, almost prevents the chyme from refluxing into the duodenum. In particular, the exclusion of a shorter portion of the small intestine leads to a larger amount of intestinal content reflux into the duodenum. Moreover, the hypoglycemic effect of RYGBP is superior to that of Billroth II (BII) reconstruction in our clinical study. In fact, a postoperative barium meal examination confirmed that duodenal reflux was more likely to occur after BII gastrojejunostomy. In a previous study, Silvio et al. reported the occurrence of reflux into the biliopancreatic limb after RYGBP [11]. In addition, BII reconstruction after subtotal gastrectomy for cancer or intractable ulcers was more effective in terms of improving T2DM, as compared with Billroth I reconstruction [12]. The reflux of intestinal contents into the duodenum may be the key factor responsible for the difference in effects among these operation methods. Thus, we speculate that isolating the duodenum from food plays an important role in blood glucose regulation. To further assess these mechanisms, we assigned streptozotocin (STZ)-induced T2DM rats to undergo sham operation (SO), new duodenal-jejunal bypass (NDJB), or new duodenal-jejunal bypass with a tube (NDJBT) and compared the metabolic outcomes after these surgeries. Accordingly, we aimed to evaluate the effects of excluding the duodenum alone and inhibiting intestinal content refluxing to the duodenum on glucose metabolism.
Materials and Methods
Animals
All 5-week-old male Sprague-Dawley rats (146 g on average) were purchased from the Laboratory Animal Center of Fuzhou University (Fujian, China). Animals were housed in a controlled room at 22 °C with a 12-h day/night cycle (lights on at 07:00 and lights off at 19:00). The rats were given free access to tap water and fed on a high-fat diet (HFD; 10.0% lard, 2.5% cholesterol, 20.0% saccharose, and 67.5% basic diet) for 4 weeks and were then intraperitoneally injected with a low dose of STZ (30 mg/kg; Sigma, USA) to induce hyperglycemia. Seventy-two hours and 1 week after the injection, random blood glucose measurements were performed using a glucometer (Bayer Health Care LLC, Germany) from the tail veins. A total of 30 rats with random blood glucose levels ≥ 16.7 mmol/l were considered diabetic and were assigned to the SO (n = 10), NDJB (n = 10), and NDJBT (n = 10) groups. Each group finally had eight rats after excluding the two rats with minimum weight. All the experimental procedures were approved by the Animal Care and Utilization Committee of Fuzhou General Hospital.
Surgical Interventions
Operations were performed at the rat surgery laboratory of the Animal Center of Comparative Medicine at Fuzhou General Hospital. Rats were provided access to water only for 12 h before the surgery. During the surgery, all animals were anesthetized with 5% ketamine hydrochloride (0.1 g/kg). Their abdominal hair was shaved, and the site was prepared with aqueous betadine. A middle abdominal incision of approximately 4 cm in length was made using a surgery scissor following localization of the stomach and small intestine. All the procedures lasted for 95 min on average, and a total of 24 rats were included after surgery.
NDJB
The duodenum was transected at a site 5 mm distal to the pylorus, and the duodenal stump was closed using 5–0 polypropylene (Shanghai Jinhuan Medical Needle, China). The jejunum was transected at a site 1 cm distal to the Treits, thus creating two ends. The distal end was used to create a duodeno-jejunostomy with the proximal end of the duodenum in an end-to-end manner. The biliopancreatic end was anastomosed to the alimentary limb at a site 15 cm distal to the duodeno-jejunal anastomosis via a Roux-en-Y anastomosis (Fig. 1a). An X-ray visual mark line was fixed on each side of the biliopancreatic limb and anastomoses.
Surgical model. a Duodenum exclusion is performed in the NDJB model. b In the NDJB model, a polyethylene tube (4 cm in length, 0.06 mm in thickness, and 0.4 cm in diameter) is inserted into the conjunction of the duodenum and jejunum and is fixed to the wall of the duodenum using a 5–0 silk suture at the base. c In the SO group, transection and re-anastomosis of the gastrointestinal tract were performed at all sites where enterotomies were performed as part of NDJB
NDJBT
The procedure was similar to the NDJB, except for the additional placement of a polyethylene tube (Fig. 1b). An X-ray visual mark line was fixed at the proximal end of the tube which was then placed in the biliopancreatic limb, and also on each side of the biliopancreatic limb and at the site of anastomosis.
SO
The abdominal cavity was kept open for the same duration required to perform the NDJBT procedures. Transection and re-anastomosis of the gastrointestinal tract were performed at all sites where enterotomies were performed as part of NDJB (Fig. 1c).
Measurement of Body Weight, Food Intake, and Glucagon-Like Peptide-1 Levels
In all the groups, body weight and food intake were measured at baseline, and at 1, 2, 4, and 8 weeks postoperatively. Blood samples were collected from the retrobulbar venous plexus of rats after overnight fasting at baseline, and at 2, 4, and 8 weeks postoperatively, and were stored in chilled EDTA tubes (MF Lab, 0.9–1.1 mm) containing a dipeptidyl peptidase IV inhibitor (Merck KGaA, Darmstadt, Germany). After centrifugation (3000 rpm) at 4 °C for 5 min, the supernatant was immediately collected. The samples were stored at − 80 °C for future analysis. Active glucagon-like peptide-1 (GLP-1) concentrations were measured using an enzyme-linked immunosorbent assay (ELISA) kit (Cusabio Biotech Co.).
Oral Glucose Tolerance Test and Fasting Blood Glucose Levels
Oral glucose tolerance test (OGTT) was performed preoperatively and at 2 and 8 weeks postoperatively. A fixed dose of 2 g/kg of 50% glucose was administered via gavage to animals following overnight fasting. The blood glucose (BG) levels were measured from the tail vein before and at 15, 30, 60, 90, and 120 min after the gavage. The fasting blood glucose (FBG) level was also measured prior to gavage. The area under the curves for OGTT (AUCOGTT) was calculated using the formula: 15 × BG0+ 30 × BG30 + 45 × BG60 + 30 × BG120.
Radiography
Plain abdominal radiography was performed 1 week after surgery to determine the presence of reflux into the biliopancreatic limb. After 12 h of overnight fasting, the rats were gavaged with 60% meglumine diatrizoate (1.5 ml/g) using a stomach tube (6.5 cm in length and 1.2 mm in diameter), and then anesthetized with 5% ketamine hydrochloride. The rats then underwent plain abdominal radiography.
Histological Analysis of the Intestine
Tissue samples were collected from the biliopancreatic limbs at 8 weeks after surgery. The specimens were fixed overnight in formalin and embedded in paraffin wax. Sections of 4-μm thickness were deparaffinized and stained using hematoxylin phloxine saffron. Histologic structures were observed under a light microscope. Each slide was scanned using the Aperio ScanScope CS System (Leica Microsys-temes SAS). Morphometric analyses were performed using the Calopix Software (TRIBVN) by measuring villus height and crypt depth. The mean values were used for statistical analyses.
Statistical Analysis
Categorical variables were analyzed using Fisher’s exact test. To compare the means between groups, we used one-way analysis of variance (ANOVA) to analyze continuous variables, followed by the Turkey test. All data were normally distributed. Overall analysis was performed using SPSS 20, and P < 0.05 was considered significant.
Results
Body Weight and Food Intake
As observed in Fig. 2, due to perioperative food restriction and surgical stress, the body weight and food intake in all rats markedly decreased during the first postoperative week, and then gradually increased. Compared with that in the SO group, the postoperative body weight of the NDJB and NDJBT groups (Fig. 2a), as well as the food intake (Fig. 2b), was not significantly different.
OGTT and AUCOGTT
No difference in OGTT and AUCOGTT values was observed among the three groups preoperatively (Fig. 3a, d). However, the NDJB and particularly the NDJBT group exhibited better glucose tolerance, as compared with the SO group, at 4 and 8 weeks postoperatively (Fig. 3b, c). Compared with the SO group, both the NDJB and NDJBT groups showed significantly lower AUCOGTT values at all postoperative time points (Fig. 3d). There was a difference in the AUCOGTT values at 8 weeks but no at 4 weeks postoperatively between the NDJB and NDJBT group.
OGTT and AUCOGTT. a OGTT values before operation. The OGTT values of rats did not significantly differ before surgery among the three groups. b OGTT values at 4 weeks after operation. c OGTT values at 8 weeks after operation. Compared with the values in the SO group, the OGTT values were significantly reduced at 4 and 8 weeks postoperatively in the NDJB and NDJBT groups. d The AUCOGTT values did not differ between the NDJB and NDJBT groups, although the values in both groups were significantly different as compared with those in the SO group at 4 and 8 weeks postoperatively. N.S. indicates no difference. * indicates P < 0.05
FBG Levels
There was no difference in the FBG levels among the groups before surgery (Fig. 4). Compared with the SO group, the NDJB and NDJBT groups exhibited a significant reduction in the FBG levels after duodenal exclusion at 4 and 8 weeks postoperatively. However, there was no significant difference in the FBG levels between the NDJB and NDJBT groups.
Serum GLP-1 Levels
There was no difference in the fasting serum GLP-1 levels before and 2 weeks after surgery. Compared with the levels in the SO group, higher GLP-1 levels were observed after overnight fasting in the NDJB and NDJBT groups at 4 and 8 weeks postoperatively (Fig. 5). However, there was no significant difference between the NDJB and NDJBT groups.
Radiography of the Abdomen
The occurrence of contrast agent duodenal reflux was easily detected in the NDJB group but not in the NDJBT group (Fig. 6). In particular, duodenal reflux was detected in two rats in the NDJBT group, as compared with eight rats in the NDJB group; the difference was significant (P < 0.05).
Intestinal Histology
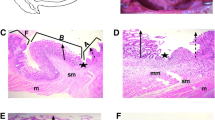
Histologic examination was performed at 8 weeks postoperatively. Adaptive changes after NDJBT were observed in the excluded duodenum segments. Under a microscope (staining with hematoxylin and eosin, × 100), the villus and crypt were both found to be significantly atrophied in the biliopancreatic limb after NDJBT, as compared with that in the SO group (Fig. 7). Although the length of the villus and crypt in the NDJB group was shorter than that in the SO group, the difference was not significant.
Intestinal histology (hematoxylin and eosin, × 100). a Micrograph of a representative section of the biliopancreatic limb in the NDJBT group. b Micrograph of a representative section of the biliopancreatic limb in the NDJB group. c Micrographs of representative sections of the biliopancreatic limb in the SO group. d The villus height was significantly shorter in the NDJBT group than in the SO and NDJB groups. e Crypt depth was significantly greater in the NDJBT group than in the SO group. N.S., no difference. *P < 0.05
Discussion
Duodenal-jejunal bypass (DJB), which is reportedly effective for resolving diabetes and does not require a change in the normal physiological structure and capacity of the stomach, is used to assess the effect of the duodenum and proximal jejunum in the treatment of diabetes. Several studies indicate that the duodenum and proximal jejunum does play a key role in the anti-diabetic effect [13, 14]. In the present study, NDJBT was newly derived from DJB and involved the exclusion of the duodenum along with the placement of a silicone tube at the anastomosis site of the duodenum and jejunum. The tube effectively prevented the reflux of the intestinal contents into the duodenum, as proven via contrast agent gavage. Moreover, we found that the intestinal mucosa of the biliopancreatic limb after NDJBT had a shorter villus length and depth as compared with that after SO. As there was no stimulation by food for a long duration, the intestinal mucosa of the biliopancreatic limb had atrophied after NDJBT. Compared with the SO group, there was a significant difference in the OGTT and GLP-1 values after NDJBT and NDJB, independent of weight and food intake. The OGTT value in the NDJBT group was slightly better improved as compared with that in the NDJB group, which can be explained by the lack of reflux. Thus, the present study shows that duodenal reflux influences the anti-diabetic effects, as supported by the lower AUCOGTT values and FBG levels in NDJBT group. The NDJBT and NDJB groups did not show any significant difference in serum GLP-1 levels, as the same length of the biliopancreatic limb was excluded and the same length of the common limb was retained.
The use of metabolic surgery by experts and scholars has gradually become more widespread over the last 60 years [9]. The mechanisms underlying metabolic surgery have been extensively studied, and the foregut and the hindgut hypotheses are primarily reported [14,15,16]. In 2003, Rubino et al. suggested that the duodenum and proximal jejunum played an important role in weight-loss surgery and hypothesized that diabetes may be a small intestinal disease [17]. Exclusion of the proximal intestine could relieve the symptoms of diabetes and this effect disappeared after the physiological structure was restored. Several studies assessed DJB, and found that bypassing the duodenum and jejunum can improve glucose homeostasis, independent of weight loss [14, 18, 19]. Moreover, research has shown that food can stimulate the proximal intestine, duodenum, and jejunum to produce an inhibitor to glucose metabolism [20]. In a previous study, Jun et al. suggested that the exclusion of the duodenum alone is sufficient to reduce the FBG levels in non-obese diabetic Goto-Kakizaki (GK) rats [21]. Furthermore, the study of Bin et al. showed that the BG levels of these rats were significantly reduced after duodenal bypass liner (DBL) implantation for duodenal isolation. The success of DBL implantation in the treatment of T2DM supports the hypothesis that bypassing the duodenum, which could avoid contact with food, prevents detrimental metabolic effects. Thus, the underlying mechanism of glucose regulation may involve a receptor on the duodenal mucosa that can be activated by reflux contents.
GLP-1 is mainly secreted by L cells of the small intestine. The increase in GLP-1 levels is thought to be induced by the rapid delivery of undigested nutrients to the distal bowel after ileal transposition (IT) [22, 23]. The blend time of pancreatic juices and food directly affects the digestion and absorption of nutrients [24], whereas the undigested food stimulates the secretion of GLP-1 via premature contact with the distal small intestine, followed by blood glucose regulation [25]. The changes in the GLP-1 levels in the present study can be explained by excluding the foregut and shortening the hindgut [26]. The role of GLP-1 in improving insulin sensitivity has been previously confirmed [27, 28], and GLP-1 receptor agonists have been used for the treatment of type 2 diabetes [29, 30].
The present study has certain limitations. First, this experimental study of rats differs from that of humans due to the biological diversity. Hence, the results cannot be directly extrapolated to humans. Second, we did not detect any insulin resistance hormones. Moreover, the tube in NDJBT may not only prevent duodenal reflux, but may also decrease the level of pancreatic juice, which could affect the pH of the intestinal tract and result in a change in the intestinal microflora. Several studies have found that intestinal flora, pH, and bile acid enterohepatic circulation are related [31, 32], and hence, these factors should be studied further. However, in the present study, we could not measure the extent of the reflux and gastric pH due to certain experimental limits.
Conclusions
Thus, we found that the exclusion of the duodenum alone can effectively improve glucose metabolism in T2DM rats, and the placement of a polyethylene tube can prevent reflux, which could further enhance the remission effect of hyperglycemia. Our study showed that duodenal reflux could represent a potential mechanism of T2DM.
References
Chan JCN, Malik V, Jia W, et al. Diabetes in Asia: epidemiology, risk factors, and pathophysiology. JAMA. 2009;301(20):2129–40.
Jeannette Aldworth, Chris Patterson, Esther Jacobs, Anoop Misra, Elizabeth B Snouffer, Lorenzo Piemonte, et al. IDF diabetes Atlas—8th edition. International diabetes federation. 2017.
Home P, Mant J, Diaz J, et al. Management of type 2 diabetes: summary of updated NICE guidance. BMJ. 2008;336(7656):1306–8.
PAVY BFWP. On the principles of the treatment of diabetes mellitus. BMJ. 1890:392–394.
Kremen AJ, Linner J, Nelson CH. An experimental evaluation of the nutritional importance of proximal and distal small intestine. Ann Surg. 1954;140(3):439–47.
Abbatini F, Rizzello M, Casella G, et al. Long-term effects of laparoscopic sleeve gastrectomy, gastric bypass, and adjustable gastric banding on type 2 diabetes. Surg Endosc. 2010;24(5):1005–10.
Pories WJ, Hicky MS, Macdonald KG, et al. Prevention and control of type 2 diabetes mellitus with gastric bypass surgery. International J. Obesity. 1998;22(3 Suppl):S85–S8.
Buchwald H, Oien DM. Metabolic/bariatric surgery worldwide 2011. Obes Surg. 2013;23(4):427–36.
Mingrone G, Panunzi S, Gaetano AD, et al. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366(17):1577–85.
Mingrone G, Panunzi S, De Gaetano A, et al. Bariatric–metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet. 2015;386(9997):964–73.
Däster S, Borbély Y, Peterli R. Acute pancreatitis after Roux-en-Y gastric bypass surgery due to reflux into biliopancreatic limb. Surg Obes Relat Dis. 2012;8:37–9.
Kwon Y, Jung Kim H, Lo Menzo E, et al. A systematic review and meta-analysis of the effect of Billroth reconstruction on type 2 diabetes: a new perspective on old surgical methods. Surg Obes Relat Dis. 2015;11(6):1386–95.
Levy P, Fried M, Santini F, et al. The comparative effects of bariatric surgery on weight and type 2 diabetes. Obes Surg. 2007;17:1248–56.
Rubino F, Marescaux J. Effect of duodenal-Jejunal exclusion in a non-obese animal model of type 2 diabetes. Ann Surg. 2004;239(1):1–11.
Thaler JP, Cummings DE. Hormonal and metabolic mechanisms of diabetes remission after gastrointestinal surgery. Endocrinology. 2009;150(6):2518–25.
Vetter ML, Cardillo S, Rickels MR, et al. Narrative review: effect of bariatric surgery on type 2 diabetes mellitus. Ann Intern Med. 2009;150:94–103.
Rubino F, Forgione A, Cummings DE, et al. The mechanism of diabetes control after gastrointestinal bypass surgery reveals a role of the proximal small intestine in the pathophysiology of type 2 diabetes. Ann Surg. 2006;244(5):741–9.
Imoto H, Shibata C, Ikezawa F, et al. Effects of duodeno-jejunal bypass on glucose metabolism in obese rats with type 2 diabetes. Surg Today. 2014;44(2):340–8.
Buchwald H, Estok R, Fahrbach K, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009;122(3):248–56e5.
Goh YM, Toumi Z, Date RS. Surgical cure for type 2 diabetes by foregut or hindgut operations: a myth or reality? A systematic review. Surg Endosc. 2017;31(1):25–37.
Ke J, Wang Y. Duodenum exclusion alone is sufficient to reduce fasting blood glucose in non-obese diabetic Goto-Kakizaki rats. Obes Surg. 2014;24(3):433–4.
Chai J, Zhang G, Liu S, et al. Exclusion of the distal ileum cannot reverse the anti-diabetic effects of duodenal-Jejunal bypass surgery. Obes Surg. 2016;26(2):261–8.
Mason EE. Llial transposition and enteroglucagon/GLP-1 obesity (and diabetic?) surgery. Obes Surg. 1999;9:223–8.
Keller J, Layer P. Human pancreatic exocrine response to nutrients in health and disease. Gut. 2005;54 Suppl 6:vi1–28.
Feng X, Zhong S, Yang J, et al. Effects on glucagon-like peptide-1 secretion by distal ileal administration of nutrients. Obes Surg. 2013;23(11):1774–82.
Deane AM, Chapman MJ, Fraser RJL, et al. The effect of exogenous glucagon-like peptide-1 on the glycaemic response to small intestinal nutrient in the critically ill: a randomised double-blind placebo controlled cross over study. Crit Care. 2009;13(3):R67.
Tourrel C, Bailbe D, Portha aB, et al. Persistent improvement of type 2 diabetes in the Goto-Kakizaki rat model by expansion of the β-cell mass during the prediabetic period with glucagon-like peptide-1 or exendin-4. Diabetes. 2002;51:1443–52.
Kindel TL, Yoder SM, Seeley RJ, et al. Duodenal-jejunal exclusion improves glucose tolerance in the diabetic, Goto-Kakizaki rat by a GLP-1 receptor-mediated mechanism. J Gastrointest Surg. 2009;13(10):1762–72.
Esquivel MA, Lansang MC. Optimizing diabetes treatment in the presence of obesity. Cleve Clin J Med. 2017;84(7 Suppl 1):S22–S9.
Paschou SA, Dede AD, Anagnostis PG, et al. Type 2 diabetes and osteoporosis: a guide to optimal management. J Clin Endocrinol Metab. 2017;102(10):3621–34.
Aron-Wisnewsky J, Dore J, Clement K. The importance of the gut microbiota after bariatric surgery. Nat Rev Gastroenterol Hepatol. 2012;9(10):590–8.
Liu H, Hu C, Zhang X, et al. Role of gut microbiota, bile acids and their cross-talk in the effects of bariatric surgery on obesity and type 2 diabetes. J Diabetes Investig. 2018;9(1):13–20.
Funding
This study was supported by the Fujian Provincial Key Science and Technology Project (2009Y0039), the Fujian Provincial Science Cooperation Project (2012I0007), and the Fujian Provincial Natural Science Fund Project (2017J01316).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All study protocols were approved by the Animal Care and Utilization Committee of Comparative Medicine at Fuzhou General Hospital.
Conflict of Interest
The authors declare that they have no conflict of interest.
Statement of Informed Consent
Does not apply.
Statement of Human and Animal Rights
All applicable institutional and national guidelines for the care and use of animals were followed.
Additional information
Weihang Wu and Li Lin are first co-authors.
Rights and permissions
About this article
Cite this article
Wu, W., Lin, L., Lin, Z. et al. Duodenum Exclusion Alone Is Sufficient to Improve Glucose Metabolism in STZ-Induced Diabetes Rats. OBES SURG 28, 3087–3094 (2018). https://doi.org/10.1007/s11695-018-3291-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-018-3291-z