Abstract
This investigation explores memory performance using the California Verbal Learning Test in relation to morphometric and connectivity measures of the memory network in severe traumatic brain injury. Twenty-two adolescents with severe traumatic brain injury were recruited for multimodal MRI scanning 1–2 years post-injury at 13 participating sites. Analyses included hippocampal volume derived from anatomical T1-weighted imaging, fornix white matter microstructure from diffusion tensor imaging, and hippocampal resting-state functional magnetic resonance imaging connectivity as well as diffusion-based structural connectivity. A typically developing control cohort of forty-nine age-matched children also underwent scanning and neurocognitive assessment. Results showed hippocampus volume was decreased in traumatic brain injury with respect to controls. Further, hippocampal volume loss was associated with worse performance on memory and learning in traumatic brain injury subjects. Similarly, hippocampal fornix fractional anisotropy was reduced in traumatic brain injury with respect to controls, while decreased fractional anisotropy in the hippocampal fornix also was associated with worse performance on memory and learning in traumatic brain injury subjects. Additionally, reduced structural connectivity of left hippocampus to thalamus and calcarine sulcus was associated with memory and learning in traumatic brain injury subjects. Functional connectivity in the left hippocampal network was also associated with memory and learning in traumatic brain injury subjects. These regional findings from a multi-modal neuroimaging approach should not only be useful for gaining valuable insight into traumatic brain injury induced memory and learning disfunction, but may also be informative for monitoring injury progression, recovery, and for developing rehabilitation as well as therapy strategies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Impaired memory function following traumatic brain injury (TBI) has long been known as the most common, persisting cognitive deficit (Schacter & Crovitz, 1977). The hippocampal formation is the centerpiece for learning and memory with its extensive afferent and efferent connections with the rest of the brain, but its medial temporal lobe location makes it particularly vulnerable to traumatic injury and TBI-induced memory impairment (C. N. Smith et al., 2013). From a quantitative neuroimaging perspective, the volume of the hippocampus as a marker of hippocampal damage has been consistently related to various memory deficits, especially in severe-TBI. The relationship is however not exceptionally robust (Bigler et al., 1997; Himanen et al., 2005; Tomaiuolo et al., 2004).
There are several possible explanations why assessing the size of the hippocampus as a predictor of memory outcome following TBI falls short of being a robust indicator of the level of memory impairment. The memory network is complex including not only the hippocampus, but fornix, mammillary bodies, anterior thalamic projections and aspects of the cingulum bundle (Budson & Price, 2005). Furthermore, the hippocampus is a structure that interfaces with all primary sensory and motor cortical regions as well as association cortices and therefore, poses a complex neural structure throughout the brain (Dalton et al., 2022; Ekstrom & Ranganath, 2018; Lavenex & Amaral, 2000; Maller et al., 2019; Raut et al., 2020).
In the past decade, with advanced methods for image analysis, it has become important to not only examine where a specific TBI-induced lesion or focal abnormality may be identified, but also how neural networks are damaged and where in the network may be most affected (Gordon et al., 2018; Rangaprakash et al., 2018; Yan et al., 2016). In terms of traumatically-induced hippocampal pathology, a multimodality neuroimaging approach is most appropriate as it takes into account not only the target structure, but also the neural network associated with the hippocampus (Irimia et al., 2017). As such, in the current study we employed volumetric as well as structural and functional neuroimaging analyses that examined the memory network in relation to neuropsychological learning and memory performance on the California Verbal Learning Test (CVLT-C/II) in children recovering from severe traumatic brain injury and typically developing controls.
Methods
Participants
Children 11–18 years old and enrolled in the Approaches and Decisions in Acute Pediatric TBI (ADAPT) trial were recruited for inclusion in this study. ADAPT enrolled 1000 severe TBI children (post-resuscitation Glasgow Coma Scale (GCS) < 8 (Bell et al., 2022; Ferrazzano et al., 2019; Kochanek et al., 2022)). Typically developing (TD) participants without history of TBI or neuropsychiatric diagnoses were recruited at the University of Wisconsin – Madison (UW). The TBI group consisted of 22 patients (12 females) between the ages of 11.6 and 18.9 years (mean ± SEM, 15.7 ± 2.1 years) at time of MRI scanning, recruited from thirteen sites. These were subjects that met the inclusion criteria from a subset of ADAPT participating sites and agreed to follow up assessments. The mechanism of injury was predominantly motor vehicle accident (77%) with the remainder due to falls and other accidental injuries. The TD group had 49 subjects (25 females) between the ages of 9.0 and 18.0 years (mean 13.45 ± 2.8 years).
California verbal learning test
The California Verbal Learning Test is a standardized list learning task, for both children—Children’s Edition (CVLT-C; (Delis et al., 1994)) and those 16 and older – Second Edition (CVLT-II; (Delis et al., 2000)). Since the ADAPT cohort examined in this investigation spanned children 11 to 18 years of age, those under 16 received the CVLT-C and those 16 and older, received the CVLT-II. While the word lists are different, the format is similar and only the total recall T-score from trials 1–5, based on the normative data based on the child’s age was used as a memory index. The CVLT-C/CVLT-II is a widely used and well-validated test of learning and memory that involves learning a standardized list of words read to examinees over several trials, followed by several conditions of recall and recognition. The total recall during trials 1–5 (“CVLT T-score”), chosen as our variable of interest, is considered a measure of verbal learning and immediate memory. Eighteen TBI subjects completed CVLT testing and were included in this analysis.
Brain imaging
Prior to subject enrollment, sites were provided scanner-specific protocols to be implemented on their system. Protocols were harmonized to conform to those similar in the multi-site Transforming Research and Clinical Knowledge in TBI (TRACK-TBI) study. Scanning procedures were disseminated to all participating sites. Sites were required to collect phantom data using the provided protocol and send to UW to verify compliance. Once approved, sites enrolled adolescent TBI participants. Imaging was performed 12–25.5 months post injury with a mean interval between injury and MRI scanning of 20 ± 4.44 months. Data from T1-weighted images, diffusion tensor imaging (DTI), and resting-state functional MRI (rs-fMRI) were used in this analysis. Based on visual quality inspection, three participants had unusable diffusion and rs-fMRI data due to severe susceptibility distortion artifacts from what appeared to be wearing of braces. We assessed and reported (Guerrero-Gonzalez et al., 2022) cross site variability in diffusion measurements using the NIST PVP diffusion (Boss et al., 2015) phantom at 6 sites which demonstrated consistency across sites. Given the stability in these metrics and the small sample size, site was not included as covariate in subsequent analyses. Relevant vendor-specific scanning parameters are shown in Table 1.
Memory network morphometry and connectivity
The multi-modal framework detailing several pre- and post-processing steps, and specific commands used is presented in the Supplement. Major relevant steps of the multimodal approach are provided next.
Hippocampal volume was derived from FreeSurfer (Fischl et al., 2002) segmentations of the T1-weighted images (recon-all command). Mean diffusion tensor fractional anisotropy (FA) was sampled (MRtrix’s tcksample command) from TractSeg (Wasserthal et al., 2018) fornix segmentations (TractSeg command) of pre-processed DTI scans. At this stage, one TBI participant was excluded due to a large lesion in region of the fornix precluding accurate segmentation. Another TBI subject was excluded due to visually-assessed excessive motion artifact on the DTI scan.
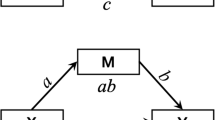
Hippocampus functional connectivity (FC) was produced using the AFNI software package from averaged pre-processed fMRI data over hippocampus masks, followed by computing the temporal Pearson’s correlation with all other voxel time series. The network of brain regions significantly connected to each hippocampus (“hippocampal network”, Fig. 1) was determined by computing a one-sample t-test of Fisher-Z transformed voxel-wise connectivity maps in the control subjects. Voxels where the group-level FC exceeded Bonferroni corrected p-value of 0.05 were considered significantly connected. The hippocampal FC in each TBI subject was then averaged over each hemisphere’s hippocampal network.
Functional and structural hippocampal networks in TD group. A,B: Network of brain regions with significant functional connectivity to the hippocampus. C: Left hippocampal structural connectome (6 of the same regions were found for the right hippocampus). Abbreviations: parahippocampal gyrus (PG, note, the FreeSurfer definition of PG includes also the entorhinal cortex), thalamus (Thal), the medial occipito-temporal and lingual sulcus (collateral sulcus, CS), amygdala (Amyg), temporal pole (TP), calcarine sulcus (CalcS), anterior transverse collateral sulcus (ATCS), lingual part of the medial occipito-temporal gyrus (lingual gyrus, LG)
A hippocampus-specific structural connectome was derived by first producing individual connectomes using spherical-deconvolution informed filtering of tractograms-2 (SIFT2) for whole-brain probabilistic fiber tracking (Smith et al., 2015; Tournier et al., 2019) combined with 164 gray matter regions from the Destrieux atlas (Destrieux et al., 2010; Fischl et al., 2002). The measure of structural connectivity was the fiber bundle capacity (FBC), which represents the ability of a white matter pathway to carry information (Smith et al., 2020). Full-brain structural connectomes were averaged across participants in the TD group. Subsequently, hippocampus-specific connections were extracted from the mean connectome. This connectome was sorted in descending order according to FBC. Finally, the top 5% connections were selected to form the hippocampus connectome. This definition was used to study connectivity in the TBI cohort.
Group comparisons of neuroimaging measures (hippocampus volume, fornix FA) and associations with CVLT (hippocampus volume, fornix FA, FBC, functional connectivity) were tested using linear regression. Six out of the 8 regions representing the top 5% in the left hippocampus connectome (Fig. 1) also appear on the right. The lingual and medial gyri and the anterior transverse collateral sulci were found only in the left hippocampal connectome, while the putamen and the pallidum were found only in the right. See Figure S3 (Supplement) for all regions included in the right-side hippocampal connectome. Note, the FreeSurfer definition of the parahipocampal gyrus includes also the entorhinal cortex and the thalamus encompasses the whole of the thalamus proper region.
Results
Hippocampal volume
Volume group comparisons are shown in Fig. 2. Group comparisons included twenty-two TBI participants. Adjusted for age, sex, brain and intracranial volumes, hippocampal volume was smaller in TBI (left-hippocampus: se = 1.080 × 102, t = 4.516, p < 0.001; right-hippocampus: se = 8.997, t = 5.126, p < 0.001). Figure 2 also shows partial residual plots for CVLT. This part of the analysis includes all eighteen TBI participants that completed CVLT testing. Adjusted values reflect CVLT-fitted value plus the residual. Positive significant associations were found in TBI (left-hippocampus: se = 4.983, t = 4.065, p < 0.001, R2 = 0.3732; right-hippocampus: se = 4.525, t = 2.188, p = 0.03, R2 = 0.3488). Associations for TD participants were non-significant. For reference, a group comparison revealed a significantly lower CVLT score in the TBI group (TBI mean: 38.4, TD mean: 53.5; Welch Two Sample t-test: t = -4.0352, df = 30.836, p-value = 0.0003337).
Hippocampus volume by group and memory performance score. A-B, group comparison of left and right hippocampus volume between TBI (yellow) and control cohort (gray), adjusted for age and sex; *p < 0.05. C-D, left and right hippocampus volume as a function of CVLT T-score for TBI (yellow) and control cohorts (gray), adjusted for age and sex
Fornix FA
Fornix FA group comparisons are shown in Fig. 3 (age- and sex-adjusted). Group comparisons included seventeen TBI participants. Adjusted-FA was significantly smaller in TBI (left-hippocampus: se = 0.01871, t = -7.121, p < 0.001; right-hippocampus: se = 0.016198, t = -7.398, p < 0.001). Out of the seventeen TBI participants in the group analysis, fifteen had completed CVLT testing. For these participants, Fig. 3 also shows partial residual plots and model fits for CVLT. Adjusted values reflect CVLT-fitted value plus the residual. Significant positive associations were found in TBI (left-hippocampus: se = 0.001520, t = 2.845, p = 0.0159, R2 = 0.3275; right-hippocampus: se = 0.001230, t = 2.662, p = 0.0221, R2 = 0.3642). Associations for TD participants were non-significant.
Fornix FA by group and memory performance. A-B, group comparison of left and right fornix FA between TBI (yellow) and control cohort (gray), adjusted for age and sex; *p < 0.05. C-D, left and right fornix FA as a function of CVLT T-score for TBI (blue) and control cohorts (gray), adjusted for age and sex
Hippocampal connectivity
After correcting for age and sex, significant associations were found for CVLT in TBI with left-hippocampus FBC to ipsilateral calcarine sulcus (R2 = 0.4333) and thalamus (R2:0.3625) (Fig. 4). These associations were statistically significant to p < 0.05 after FDR multiple-comparisons correction. Also, average hippocampus FC in TBI was significantly associated with CVLT (Fig. 4) in the left hemisphere (p = 0.039, R2 = 0.027), and associated at a trend-level in the right hemisphere (p = 0.057, R2 = 0.023). A significant correlation was found for TD participants in the left hippocampus (p < 0.001, R2 = 0.25). These analyses included sixteen participants that had diffusion and rsfMRI imaging data and also had completed CVLT assessments.
Hippocampal connectivity and CVLT. Top panel: Structural connectivity and memory performance. Fiber bundle capacity as a function of CVLT T-score for TBI (yellow) and control cohort (gray), adjusted for age and sex, for hippocampus with (A) thalamus and with (B) calcarine sulcus. Bottom panel: Association between hippocampus functional network connectivity and memory performance for left (C) and right (D) hippocampi. Spearman rank correlation between Hippocampal FC and CVLT T-score is shown for TBI (yellow) and control (gray) subjects
Discussion
This work employed a multi-modality MRI approach to study the memory network in relation to neuropsychological memory and learning performance on the CVLT following severe TBI.
Hippocampal volumetrics after TBI
We found significant reductions in hippocampal volume in TBI subjects compared to controls, and in TBI subjects the hippocampal volume was associated with memory and learning performance on the CVLT. The hippocampus has been consistently identified as a brain structure affected by TBI, and the extent of hippocampal atrophy has been associated with severity of injury (Beauchamp et al., 2011; King et al., 2019; Tasker et al., 2005; Tate & Bigler, 2000). Importantly, our analysis adjusted for brain volume thus reflects hippocampal-specific atrophy. Biomechanically, the position of the hippocampus within the medial temporal lobe in association with the boney structures of the middle cranial fossa and the inferior horn of the lateral ventricular system, make it especially vulnerable to shear-strain-deformation injury from TBI (Zhou et al., 2022). Selective hippocampal atrophy was similarly found by (Wilde et al., 2007) in their study of 16 children with moderate-to-severe TBI. Given its central role in memory formation and retrieval, hippocampal volume loss has been investigated as a marker of TBI-induced memory dysfunction, though most prior studies have been performed in adults, so information on children with severe-TBI is limited. In a study of 86 subjects age 16–65 years with any-severity TBI, (Tate & Bigler, 2000) found significant reductions in hippocampal volume in TBI compared to controls 2 months post-injury. The degree of atrophy in these regions correlated with injury severity and the General Memory Index of the Wechsler Memory-Scale. Similarly, a study of 129 children age 8–15 years with any-severity TBI (15 severe-TBI subjects), (DeMaster et al., 2017) found atrophy in hippocampal subregions was associated with TBI injury severity and memory performance 6 weeks post-injury. Adding to these studies performed early after injury, our findings suggest that the association between hippocampal atrophy and memory dysfunction is sustained up to 1–2 years post-injury in children with severe TBI.
Alterations in fornix microstructure
FA in the fornix of the TBI group significantly decreased compared to the TD group. In the TBI group, but not in the TD cohort, reduced fornix FA was associated with memory and learning impairment on the CVLT. Being the major carrier of efferent projections from the hippocampus and connecting it to structures such as mammillary bodies, thalamus, and temporal lobe, the fornix plays an important role in the hippocampal network and in memory function (Bigler et al., 2010). The fine fornix tracts straddling both cerebral hemispheres are especially vulnerable to physical shearing forces (Tate & Bigler, 2000), and TBI has long been associated with fornix atrophy (Gale et al., 1993). Decreased fornix FA at the chronic stage of TBI may reflect disruptions in tissue microstructure such as loss of axonal fiber coherence or density as well as demyelination. To investigate this, in a secondary analysis we assessed fornix-specific apparent fiber density (AFD) voxel maps, using the afdconnectivity tool within MRtrix (Tournier et al., 2019) with the ‘-afd_map’ option. This uses the input fornix streamlines to identify the fixels belonging to the tract-of-interest, computes the fixel-specific AFD as the fiber orientation distribution (fODF) lobe integral and outputs it as a 3D scalar map. As described in this publication (Smith et al., 2020), integrating a discrete fODF lobe is proportional to the MR-visible intra-cellular tissue volume at high b-values in the direction of that lobe. We compared fornix AFD between the TBI and TD groups and found a significant reduction in AFD for the TBI group. See Figure S5 (Supplement). Mechanisms such as loss of axonal fiber coherence or density may be related to direct injury in the fornix or atrophy nodes connected by it like the hippocampus or mammillary bodies. Our findings of decreased fornix FA associated with worse performance on CVLT in TBI are consistent with a study of DTI that found abnormal FA in several regions of the brain in adult TBI patients, but only fornix FA correlated with performance in associative memory and learning tasks (Kinnunen et al., 2011). In another study of diffuse TBI, adult patients were observed to have globally decreased FA in the brain, but regional analyses revealed that lower fornix FA was associated with poorer declarative memory (Palacios et al., 2011). More generally, (Adnan et al., 2013) suggested a critical role for fornix integrity in the development of memory impairments after moderated-to-severe TBI in adults. Their findings consistently showed lower fornix FA accompanied memory deficits compared to healthy controls. These numerous findings including our own linking decreased FA in the fornix to declining memory function in TBI are indicative that fornix FA may provide a robust imaging marker for TBI-induced memory deficits.
Hippocampal network connectivity
Statistically significant linear associations were found for memory and learning with respect to connectivity strength of hippocampus to the thalamus and the calcarine sulcus. These were found for the left hemisphere only, and the majority of the subjects in our study were right-handed. CVLT, as its name implies is an auditorily presented, verbal learning and memory task. The material-specificity theory posits that left and right hippocampus differentially process verbal and visual memory, supported by many studies, including elegant studies of temporal lobe epilepsy in children which demonstrate reductions specifically in verbal memory after temporal lobe resections of the left hippocampus (Law et al., 2017; Sepeta et al., 2018), and broader functional imaging studies of medial temporal lobe and hippocampus (Dalton et al., 2016; Golby, 2001). CVLT is therefore dependent on the integrity of left hemisphere language and memory networks (Lezak, 2012), and would be expected to relate to left hemisphere hippocampal network analyses as we found in the current investigation of children with severe-TBI. Interestingly, hippocampal connectivity with the calcarine sulcus, a visual processing region, was significantly associated with verbal memory performance in our study. These findings are consistent with those from recent investigations of hippocampal structural connectivity that report strong patterns of connectivity between the hippocampus and these visual cortex regions (Dalton et al., 2022; Maller et al., 2019). Auditory attention often engages visual cortical areas (Cate et al., 2009), especially when the child “visualizes” the word as an association in an attempt to recall the word – i.e. in response to retaining the word “boat,” the child may visualize an image of the boat. Additionally, visual cortical areas in association with parietal association cortical regions are part of the dorsal stream network intimately involved in guided attention (Deco & Rolls, 2005).
We also found that hippocampal-thalamic connectivity was associated with CVLT. Thalamic projections have been shown to be particularly vulnerable to biomechanical, shear-strain deformation in TBI (Bian & Mao, 2020; Cosgrove et al., 2022; Dennis et al., 2021; Mofakham et al., 2022), where even in mild-TBI, diminished integrity of thalamic radiations occurs and relates to behavioral and cognitive outcome (Ware et al., 2020, 2022). The original Papez circuit delineates projections from hippocampus to mammillary bodies via the fornix, then to anterior thalamic nuclei (ATN) of the thalamus, and on to cingulate and posterior parietal cortex, and finally back to the hippocampus through parahippocampal cortex. The ATN are believed to be a part of an ‘extended hippocampal system’, which is involved in memory encoding (Aggleton et al., 2010; Sweeney-Reed et al., 2021). Lesions or degeneration of the ATN lead to impairments along this tract, impairing memory encoding (Sweeney-Reed et al., 2021). Thus, it is not surprising that reduced connectivity between the hippocampus and thalamus among TBI patients in our study, was associated with reduced memory performance.
Our structural connectivity findings are supported by our functional connectivity analysis, which similarly demonstrated a positive association between hippocampal network connectivity and memory performance. Similar to our structural connectivity analysis, the FC associations were found in the left hippocampal network. Consistent with prior studies, the hippocampal FC network in our analysis overlaps with the Default Mode Network, a set of brain regions that tend to be more active during “rest” and involved in, among other functions, episodic memory (Buckner et al., 2008; Raichle et al., 2001; Vincent et al., 2006). The TBI subjects with the highest CVLT-scores showed hippocampal functional connectivity that was similar to control subjects at comparable performance. This relationship with CVLT is consistent with prior studies of resting-state FC in children (Riggins et al., 2016) and older adults (Wang et al., 2010) that show a positive association of within-network hippocampal FC and memory performance.
Importance of multimodality neuroimaging approach
2hile the vulnerability of the hippocampus to severe-TBI has been well established (Bigler et al., 2002; Dennis et al., 2021; Wilde et al., 2007), the current investigation is the first to take a multimodality neuroimaging analysis approach to examine hippocampal connectivity and network factors that most robustly relate to verbal memory in an adolescent severe-TBI sample. As mentioned in the introduction, merely assessing hippocampal volume or whether a lesion has been present within the hippocampal formation post-TBI has not necessarily predicted memory outcome. As demonstrated in the current investigation, the hippocampus, while a critical, central component to the memory network, happens to be just one component of the network. The participants in the current investigation, while having sustained a severe TBI had, nonetheless, sufficiently recovered to be able to cooperate with the cognitive assessment process and were between 1–2 years post injury. As such, the neuropsychological assessment process was undertaken at a point where sufficient plasticity, adaptation, accommodation and/or recovery of memory networks had occurred. What the current investigation demonstrates is that memory performance relates to the integrity of the entire memory network, not just a single component. Following injury, the hippocampus and related medial temporal lobe structures have some capacity for plasticity (Schumm et al., 2022) and there is also the potential for neurogenesis within the injured hippocampus (Rizk et al., 2021). How well hippocampal neurons may still participate in generating neural connectivity within the network may be key to how other systems and pathways in the memory network react to injury and potentially compensate to whatever hippocampal pathology may be present (M. Dennis et al., 2014; Sta Maria et al., 2019). Also, as shown in Fig. 2, even though this was a restricted sample of severe-pediatric-TBI, a number of the severe-TBI cases had hippocampal volumes that were entirely within the range of the TD control sample. Accordingly, interrogating the entire hippocampal network provides a more comprehensive view of injury-induced dysfunction and compensatory changes. From a practical perspective, however, hippocampal volume and/or fornix FA measures are the most straightforward to calculate on a clinical basis and appear to represent valid biomarkers of memory impairment in severe-TBI in children.
Some limitations must be considered when interpreting the results of our study. First, the relatively small sample size in our TBI cohort affects estimates of the quantitative neuroimaging measures in terms of variance and reliability, which in turns limits the generalizability of the findings. Also, site effects must be considered when interpreting results from any multi-site neuroimaging study. To minimize site-to-site variation, imaging protocols were standardized across sites. We also conducted an analysis of the NIST PVP diffusion phantom (Boss et al., 2015) scanned across 6 of the sites. The findings of that analyses (Guerrero-Gonzalez et al., 2022) did not reveal significant variations in diffusion measures across sites. Nonetheless, measurement variation across scanners and sites cannot be entirely ruled out as a contributing factor in this study. On the other hand, severe-TBI tends to produce significant changes in brain structure, manifested as generalized and regional volume loss (Beauchamp et al., 2011; Wilde et al., 2007), and MRI has been successfully used in other multi-site studies of adults (Palacios et al., 2022) and children (Bigler et al., 2013) with TBI. Normative control data and/or human traveling phantoms at each site could be employed in future studies to further address harmonization across sites but were not feasible here. Regardless, the group differences we observed in volumetric and DTI measures were robust and occurred in the expected direction, and the associations between hippocampal imaging findings and memory dysfunction we identified are biologically plausible.
Conclusion
This analysis included hippocampal volume and fornix microstructure, as well as structural and functional hippocampal connectivity in relation to memory and learning. We found significant reductions in hippocampal volume in TBI subjects compared to controls, and in TBI subjects the hippocampal volume was associated with memory and learning performance on the CVLT. FA in the fornix of the TBI group significantly decreased with respect to the TD group. Also, in TBI reduced fornix FA correlated with CVLT performance. Further, we found statistically significant linear associations for CVLT and FBC of hippocampus to thalamus and calcarine sulcus. Functional connectivity analysis similarly demonstrated a positive association between left hippocampal network connectivity and memory performance. This multi-modal interrogation of the hippocampal network provides a more comprehensive look into injury-induced dysfunction and compensatory changes.
Data availability
The participants of this study did not consent to public sharing of data. Thus, data cannot be published on public repositories. The authors are open to explore data sharing alternatives with proper procedures and documentation that comply with relevant review protocols.
Code availability
All relevant functions and libraries that were used from existing packages (e.g. MRtrix3, TractSeg, etc.) are detailed in the pertinent sections of the manuscript or Supplement.
References
Adnan, A., Crawley, A., Mikulis, D., Moscovitch, M., Colella, B., & Green, R. (2013). Moderate-severe traumatic brain injury causes delayed loss of white matter integrity: Evidence of fornix deterioration in the chronic stage of injury. Brain Injury, 27(12), 1415–1422. https://doi.org/10.3109/02699052.2013.823659
Aggleton, J. P., O’Mara, S. M., Vann, S. D., Wright, N. F., Tsanov, M., & Erichsen, J. T. (2010). Hippocampal-anterior thalamic pathways for memory: Uncovering a network of direct and indirect actions: Hippocampal-thalamic pathways for memory. European Journal of Neuroscience, 31(12), 2292–2307. https://doi.org/10.1111/j.1460-9568.2010.07251.x
Beauchamp, M. H., Ditchfield, M., Maller, J. J., Catroppa, C., Godfrey, C., Rosenfeld, J. V., Kean, M. J., & Anderson, V. A. (2011). Hippocampus, amygdala and global brain changes 10 years after childhood traumatic brain injury. International Journal of Developmental Neuroscience, 29(2), 137–143. https://doi.org/10.1016/j.ijdevneu.2010.12.003
Bell, M. J., Rosario, B. L., Kochanek, P. M., Adelson, P. D., Morris, K. P., Au, A. K., Schober, M., Butt, W., Edwards, R. J., Zimmerman, J., Pineda, J., Le, T. M., Dean, N., Whalen, M. J., Figaji, A., Luther, J., Beers, S. R., Gupta, D. K., Carpenter, J., & Sarnaik, A. (2022). Comparative effectiveness of diversion of cerebrospinal fluid for children with severe traumatic brain injury. JAMA Network Open, 5(7), e2220969. https://doi.org/10.1001/jamanetworkopen.2022.20969
Bian, K., & Mao, H. (2020). Mechanisms and variances of rotation-induced brain injury: A parametric investigation between head kinematics and brain strain. Biomechanics and Modeling in Mechanobiology, 19(6), 2323–2341. https://doi.org/10.1007/s10237-020-01341-4
Bigler, E. D., Blatter, D. D., Anderson, C. V., Johnson, S. C., Gale, S. D., Hopkins, R. O., & Burnett, B. (1997). Hippocampal volume in normal aging and traumatic brain injury. AJNR. American Journal of Neuroradiology, 18(1), 11–23.
Bigler, E. D., Anderson, C. V., Blatter, D. D., & Andersob, C. V. (2002). Temporal lobe morphology in normal aging and traumatic brain injury. AJNR. American Journal of Neuroradiology, 23(2), 255–266.
Bigler, E. D., McCauley, S. R., Wu, T. C., Yallampalli, R., Shah, S., MacLeod, M., Chu, Z., Hunter, J. V., Clifton, G. L., Levin, H. S., & Wilde, E. A. (2010). The temporal stem in traumatic brain injury: Preliminary findings. Brain Imaging and Behavior, 4(3–4), 270–282. https://doi.org/10.1007/s11682-010-9105-0
Bigler, E. D., Abildskov, T. J., Petrie, J., Farrer, T. J., Dennis, M., Simic, N., Taylor, H. G., Rubin, K. H., Vannatta, K., Gerhardt, C. A., Stancin, T., & Owen Yeates, K. (2013). Heterogeneity of brain lesions in pediatric traumatic brain injury. Neuropsychology, 27(4), 438–451. https://doi.org/10.1037/a0032837
Boss, M. A., Chevenert T., Jackson E. (2015). Multicenter study of reproducibility of wide range of ADC at 0 C. In: Proceedings of the RSNA Annual Meeting.
Buckner, R. L., Andrews-Hanna, J. R., & Schacter, D. L. (2008). The brain’s default network: Anatomy, function, and relevance to disease. Annals of the New York Academy of Sciences, 1124, 1–38. https://doi.org/10.1196/annals.1440.011
Budson, A. E., & Price, B. H. (2005). Memory dysfunction. The New England Journal of Medicine, 352(7), 692–699. https://doi.org/10.1056/NEJMra041071
Cate, A. D., Herron, T. J., Yund, E. W., Stecker, G. C., Rinne, T., Kang, X., Petkov, C. I., Disbrow, E. A., & Woods, D. L. (2009). Auditory attention activates peripheral visual cortex. PLoS ONE, 4(2), e4645. https://doi.org/10.1371/journal.pone.0004645
Cosgrove, M. E., Saadon, J. R., Mikell, C. B., Stefancin, P. L., Alkadaa, L., Wang, Z., Saluja, S., Servider, J., Razzaq, B., Huang, C., & Mofakham, S. (2022). Thalamo-prefrontal connectivity correlates with early command-following after severe traumatic brain injury. Frontiers in Neurology, 13, 826266. https://doi.org/10.3389/fneur.2022.826266
Dalton, M. A., Hornberger, M., & Piguet, O. (2016). Material specific lateralization of medial temporal lobe function: An f MRI investigation. Human Brain Mapping, 37(3), 933–941. https://doi.org/10.1002/hbm.23077
Dalton, M. A., D’Souza, A., Lv, J., & Calamante, F. (2022). New insights into anatomical connectivity along the anterior–posterior axis of the human hippocampus using in vivo quantitative fibre tracking. eLife, 11, e76143. https://doi.org/10.7554/eLife.76143
Deco, G., & Rolls, E. T. (2005). Attention, short-term memory, and action selection: A unifying theory. Progress in Neurobiology, 76(4), 236–256. https://doi.org/10.1016/j.pneurobio.2005.08.004
Delis, D. C., Kramer, J. H., Kaplan, E., & Ober, B. A. (1994). The California verbal learning test-Children’s version. San Antonio, TX: The Psychological Corporation.
Delis, D. C., Kramer, J. H., Kaplan, E., & Ober, B. A. (2000). The California verbal learning test—second edition. San Antonio, TX: The Psychological Corporation.
DeMaster, D., Johnson, C., Juranek, J., & Ewing-Cobbs, L. (2017). Memory and the hippocampal formation following pediatric traumatic brain injury. Brain and Behavior, 7(12), e00832. https://doi.org/10.1002/brb3.832
Dennis, M., Spiegler, B. J., Simic, N., Sinopoli, K. J., Wilkinson, A., Yeates, K. O., Taylor, H. G., Bigler, E. D., & Fletcher, J. M. (2014). Functional plasticity in childhood brain disorders: When, what, how, and whom to assess. Neuropsychology Review, 24(4), 389–408. https://doi.org/10.1007/s11065-014-9261-x
Dennis, E. L., Caeyenberghs, K., Hoskinson, K. R., Merkley, T. L., Suskauer, S. J., Asarnow, R. F., Babikian, T., Bartnik-Olson, B., Bickart, K., Bigler, E. D., Ewing-Cobbs, L., Figaji, A., Giza, C. C., Goodrich-Hunsaker, N. J., Hodges, C. B., Hovenden Aa, E. S., Irimia, A., Königs, M., Levin, H. S., Wilde, E. A. (2021). White Matter Disruption in Pediatric Traumatic Brain Injury: Results from ENIGMA Pediatric Moderate to Severe Traumatic Brain Injury. Neurology, https://doi.org/10.1212/WNL.0000000000012222.
Destrieux, C., Fischl, B., Dale, A., & Halgren, E. (2010). Automatic parcellation of human cortical gyri and sulci using standard anatomical nomenclature. NeuroImage, 53(1), 1–15. https://doi.org/10.1016/j.neuroimage.2010.06.010
Ekstrom, A. D., & Ranganath, C. (2018). Space, time, and episodic memory: The hippocampus is all over the cognitive map. Hippocampus, 28(9), 680–687. https://doi.org/10.1002/hipo.22750
Ferrazzano, P. A., Rosario, B. L., Wisniewski, S. R., Shafi, N. I., Siefkes, H. M., Miles, D. K., Alexander, A. L., & Bell, M. J. (2019). Use of magnetic resonance imaging in severe pediatric traumatic brain injury: Assessment of current practice. Journal of Neurosurgery: Pediatrics, 23(4), 471–479. https://doi.org/10.3171/2018.10.PEDS18374
Fischl, B., Salat, D. H., Busa, E., Albert, M., Dieterich, M., Haselgrove, C., van der Kouwe, A., Killiany, R., Kennedy, D., Klaveness, S., Montillo, A., Makris, N., Rosen, B., & Dale, A. M. (2002). Whole Brain Segmentation. Neuron, 33(3), 341–355. https://doi.org/10.1016/S0896-6273(02)00569-X
Gale, S. D., Burr, R. B., Bigler, E. D., & Blatter, D. (1993). Fornix degeneration and memory in traumatic brain injury. Brain Research Bulletin, 32(4), 345–349. https://doi.org/10.1016/0361-9230(93)90198-K
Golby, A. J. (2001). Material-specific lateralization in the medial temporal lobe and prefrontal cortex during memory encoding. Brain, 124(9), 1841–1854. https://doi.org/10.1093/brain/124.9.1841
Gordon, E. M., Lynch, C. J., Gratton, C., Laumann, T. O., Gilmore, A. W., Greene, D. J., Ortega, M., Nguyen, A. L., Schlaggar, B. L., Petersen, S. E., Dosenbach, N. U. F., & Nelson, S. M. (2018). Three distinct sets of connector hubs integrate human brain function. Cell Reports, 24(7), 1687-1695.e4. https://doi.org/10.1016/j.celrep.2018.07.050
Guerrero-Gonzalez, J. M., Yeske, B., Kirk, G. R., Bell, M. J., Ferrazzano, P. A., & Alexander, A. L. (2022). Mahalanobis distance tractometry (MaD-Tract) – a framework for personalized white matter anomaly detection applied to TBI. NeuroImage, 260, 119475. https://doi.org/10.1016/j.neuroimage.2022.119475
Himanen, L., Portin, R., Isoniemi, H., Helenius, H., Kurki, T., & Tenovuo, O. (2005). Cognitive functions in relation to MRI findings 30 years after traumatic brain injury. Brain Injury, 19(2), 93–100. https://doi.org/10.1080/02699050410001720031
Irimia, A., Goh, S.-Y.M., Wade, A. C., Patel, K., Vespa, P. M., & Van Horn, J. D. (2017). Traumatic Brain Injury Severity, Neuropathophysiology, and Clinical Outcome: Insights from Multimodal Neuroimaging. Frontiers in Neurology, 8, 530. https://doi.org/10.3389/fneur.2017.00530
King, D. J., Ellis, K. R., Seri, S., & Wood, A. G. (2019). A systematic review of cross-sectional differences and longitudinal changes to the morphometry of the brain following paediatric traumatic brain injury. NeuroImage: Clinical, 23, 101844. https://doi.org/10.1016/j.nicl.2019.101844
Kinnunen, K. M., Greenwood, R., Powell, J. H., Leech, R., Hawkins, P. C., Bonnelle, V., Patel, M. C., Counsell, S. J., & Sharp, D. J. (2011). White matter damage and cognitive impairment after traumatic brain injury. Brain, 134(2), 449–463. https://doi.org/10.1093/brain/awq347
Kochanek, P. M., Adelson, P. D., Rosario, B. L., Hutchison, J., Miller Ferguson, N., Ferrazzano, P., O’Brien, N., Beca, J., Sarnaik, A., LaRovere, K., Bennett, T. D., Deep, A., Gupta, D., Willyerd, F. A., Gao, S., Wisniewski, S. R., Bell, M. J., ADAPT Investigators, Agarwal, S., Pineda, J. (2022). Comparison of intracranial pressure measurements before and after hypertonic saline or mannitol treatment in children with severe traumatic brain injury. JAMA Network Open, 5(3), e220891. https://doi.org/10.1001/jamanetworkopen.2022.0891
Lavenex, P., & Amaral, D. G. (2000). Hippocampal-neocortical interaction: A hierarchy of associativity. Hippocampus, 10(4), 420–430. https://doi.org/10.1002/1098-1063(2000)10:4%3c420::AID-HIPO8%3e3.0.CO;2-5
Law, N., Benifla, M., Rutka, J., & Smith, M. L. (2017). Verbal memory after temporal lobe epilepsy surgery in children: Do only mesial structures matter? Epilepsia, 58(2), 291–299. https://doi.org/10.1111/epi.13635
Lezak, M. D. (Ed.). (2012). Neuropsychological assessment (5th ed). Oxford University Press.
Maller, J. J., Welton, T., Middione, M., Callaghan, F. M., Rosenfeld, J. V., & Grieve, S. M. (2019). Revealing the hippocampal connectome through super-resolution 1150-direction diffusion MRI. Scientific Reports, 9(1), 2418. https://doi.org/10.1038/s41598-018-37905-9
Mofakham, S., Liu, Y., Hensley, A., Saadon, J. R., Gammel, T., Cosgrove, M. E., Adachi, J., Mohammad, S., Huang, C., Djurić, P. M., & Mikell, C. B. (2022). Injury to thalamocortical projections following traumatic brain injury results in attractor dynamics for cortical networks. Progress in Neurobiology, 210, 102215. https://doi.org/10.1016/j.pneurobio.2022.102215
Palacios, E. M., Fernandez-Espejo, D., Junque, C., Sanchez-Carrion, R., Roig, T., Tormos, J. M., Bargallo, N., & Vendrell, P. (2011). Diffusion tensor imaging differences relate to memory deficits in diffuse traumatic brain injury. BMC Neurology, 11(1), 24. https://doi.org/10.1186/1471-2377-11-24
Palacios, E. M., Yuh, E. L., Mac Donald, C. L., Bourla, I., Wren-Jarvis, J., Sun, X., Vassar, M. J., Diaz-Arrastia, R., Giacino, J. T., Okonkwo, D. O., Robertson, C. S., Stein, M. B., Temkin, N., McCrea, M. A., Levin, H. S., Markowitz, A. J., Jain, S., Manley, G. T., Mukherjee, P., Zafonte, R. (2022). Diffusion tensor imaging reveals elevated diffusivity of white matter microstructure that is independently associated with long-term outcome after mild traumatic brain injury: A TRACK-TBI study. Journal of Neurotrauma, 39(19–20), 1318–1328. https://doi.org/10.1089/neu.2021.0408
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001). A default mode of brain function. Proceedings of the National Academy of Sciences of the United States of America, 98(2), 676–682. https://doi.org/10.1073/pnas.98.2.676
Rangaprakash, D., Dretsch, M. N., Venkataraman, A., Katz, J. S., Denney, T. S., & Deshpande, G. (2018). Identifying disease foci from static and dynamic effective connectivity networks: Illustration in soldiers with trauma. Human Brain Mapping, 39(1), 264–287. https://doi.org/10.1002/hbm.23841
Raut, R. V., Snyder, A. Z., & Raichle, M. E. (2020). Hierarchical dynamics as a macroscopic organizing principle of the human brain. Proceedings of the National Academy of Sciences of the United States of America, 117(34), 20890–20897. https://doi.org/10.1073/pnas.2003383117
Riggins, T., Geng, F., Blankenship, S. L., & Redcay, E. (2016). Hippocampal functional connectivity and episodic memory in early childhood. Developmental Cognitive Neuroscience, 19, 58–69. https://doi.org/10.1016/j.dcn.2016.02.002
Rizk, M., Vu, J., & Zhang, Z. (2021). Impact of pediatric traumatic brain injury on hippocampal neurogenesis. Neural Regeneration Research, 16(5), 926–933. https://doi.org/10.4103/1673-5374.297057
Schacter, D. L., & Crovitz, H. F. (1977). Memory function after closed head injury: A review of the quantitative research. Cortex; a Journal Devoted to the Study of the Nervous System and Behavior, 13(2), 150–176. https://doi.org/10.1016/s0010-9452(77)80006-3
Schumm, S. N., Gabrieli, D., & Meaney, D. F. (2022). Plasticity impairment exposes CA3 vulnerability in a hippocampal network model of mild traumatic brain injury. Hippocampus, 32(3), 231–250. https://doi.org/10.1002/hipo.23402
Sepeta, L. N., Berl, M. M., & Gaillard, W. D. (2018). Imaging episodic memory during development and childhood epilepsy. Journal of Neurodevelopmental Disorders, 10(1), 40. https://doi.org/10.1186/s11689-018-9255-8
Smith, C. N., Frascino, J. C., Hopkins, R. O., & Squire, L. R. (2013). The nature of anterograde and retrograde memory impairment after damage to the medial temporal lobe. Neuropsychologia, 51(13), 2709–2714. https://doi.org/10.1016/j.neuropsychologia.2013.09.015
Smith, R. E., Tournier, J.-D., Calamante, F., & Connelly, A. (2015). SIFT2: Enabling dense quantitative assessment of brain white matter connectivity using streamlines tractography. NeuroImage, 119, 338–351. https://doi.org/10.1016/j.neuroimage.2015.06.092
Smith, R., Raffelt, D., Tournier, J.-D., & Connelly, A. (2020). Quantitative streamlines tractography: Methods and inter-subject normalisation [Preprint]. Open Science Framework. 10.31219/osf.io/c67kn
Sta Maria, N. S., Sargolzaei, S., Prins, M. L., Dennis, E. L., Asarnow, R. F., Hovda, D. A., Harris, N. G., & Giza, C. C. (2019). Bridging the gap: Mechanisms of plasticity and repair after pediatric TBI. Experimental Neurology, 318, 78–91. https://doi.org/10.1016/j.expneurol.2019.04.016
Sweeney-Reed, C. M., Buentjen, L., Voges, J., Schmitt, F. C., Zaehle, T., Kam, J. W. Y., Kaufmann, J., Heinze, H.-J., Hinrichs, H., Knight, R. T., & Rugg, M. D. (2021). The role of the anterior nuclei of the thalamus in human memory processing. Neuroscience & Biobehavioral Reviews, 126, 146–158. https://doi.org/10.1016/j.neubiorev.2021.02.046
Tasker, R. C., Salmond, C. H., Westland, A. G., Pena, A., Gillard, J. H., Sahakian, B. J., & Pickard, J. D. (2005). Head circumference and brain and hippocampal volume after severe traumatic brain injury in childhood. Pediatric Research, 58(2), 302–308. https://doi.org/10.1203/01.PDR.0000169965.08854.25
Tate, D. F., & Bigler, E. D. (2000). Fornix and hippocampal atrophy in traumatic brain injury. Learning & Memory (Cold Spring Harbor, N.Y.), 7(6), 442–446. https://doi.org/10.1101/lm.33000
Tomaiuolo, F., Carlesimo, G. A., Di Paola, M., Petrides, M., Fera, F., Bonanni, R., Formisano, R., Pasqualetti, P., & Caltagirone, C. (2004). Gross morphology and morphometric sequelae in the hippocampus, fornix, and corpus callosum of patients with severe non-missile traumatic brain injury without macroscopically detectable lesions: A T1 weighted MRI study. Journal of Neurology, Neurosurgery, and Psychiatry, 75(9), 1314–1322. https://doi.org/10.1136/jnnp.2003.017046
Tournier, J.-D., Smith, R., Raffelt, D., Tabbara, R., Dhollander, T., Pietsch, M., Christiaens, D., Jeurissen, B., Yeh, C.-H., & Connelly, A. (2019). MRtrix3: A fast, flexible and open software framework for medical image processing and visualisation. NeuroImage, 202, 116137. https://doi.org/10.1016/j.neuroimage.2019.116137
Vincent, J. L., Snyder, A. Z., Fox, M. D., Shannon, B. J., Andrews, J. R., Raichle, M. E., & Buckner, R. L. (2006). Coherent spontaneous activity identifies a hippocampal-parietal memory network. Journal of Neurophysiology, 96(6), 3517–3531. https://doi.org/10.1152/jn.00048.2006
Wang, L., Laviolette, P., O’Keefe, K., Putcha, D., Bakkour, A., Van Dijk, K. R. A., Pihlajamäki, M., Dickerson, B. C., & Sperling, R. A. (2010). Intrinsic connectivity between the hippocampus and posteromedial cortex predicts memory performance in cognitively intact older individuals. NeuroImage, 51(2), 910–917. https://doi.org/10.1016/j.neuroimage.2010.02.046
Ware, A. L., Shukla, A., Goodrich-Hunsaker, N. J., Lebel, C., Wilde, E. A., Abildskov, T. J., Bigler, E. D., Cohen, D. M., Mihalov, L. K., Bacevice, A., Bangert, B. A., Taylor, H. G., & Yeates, K. O. (2020). Post-acute white matter microstructure predicts post-acute and chronic post-concussive symptom severity following mild traumatic brain injury in children. NeuroImage. Clinical, 25, 102106. https://doi.org/10.1016/j.nicl.2019.102106
Ware, A. L., Yeates, K. O., Tang, K., Shukla, A., Onicas, A. I., Guo, S., Goodrich-Hunsaker, N., Abdeen, N., Beauchamp, M. H., Beaulieu, C., Bjornson, B., Craig, W., Dehaes, M., Doan, Q., Deschenes, S., Freedman, S. B., Goodyear, B. G., Gravel, J., Ledoux, A.-A., Pediatric Emergency Research Canada A-CAP Study Team. (2022). Longitudinal white matter microstructural changes in pediatric mild traumatic brain injury: An A-CAP study. Human Brain Mappinghttps://doi.org/10.1002/hbm.25885
Wasserthal, J., Neher, P., & Maier-Hein, K. H. (2018). TractSeg—Fast and accurate white matter tract segmentation. NeuroImage, 183, 239–253. https://doi.org/10.1016/j.neuroimage.2018.07.070
Wilde, E. A., Bigler, E. D., Hunter, J. V., Fearing, M. A., Scheibel, R. S., Newsome, M. R., Johnson, J. L., Bachevalier, J., Li, X., & Levin, H. S. (2007). Hippocampus, amygdala, and basal ganglia morphometrics in children after moderate-to-severe traumatic brain injury. Developmental Medicine and Child Neurology, 49(4), 294–299. https://doi.org/10.1111/j.1469-8749.2007.00294.x
Yan, H., Feng, Y., & Wang, Q. (2016). Altered Effective Connectivity of Hippocampus-Dependent Episodic Memory Network in mTBI Survivors. Neural Plasticity, 2016, 6353845. https://doi.org/10.1155/2016/6353845
Zhou, Z., Li, X., Domel, A. G., Dennis, E. L., Georgiadis, M., Liu, Y., Raymond, S. J., Grant, G., Kleiven, S., Camarillo, D., & Zeineh, M. (2022). The Presence of the Temporal Horn Exacerbates the Vulnerability of Hippocampus During Head Impacts. Frontiers in Bioengineering and Biotechnology, 10, 754344. https://doi.org/10.3389/fbioe.2022.754344
Additional author information
The ADAPT MRI Biomarkers Investigators are: Ranjit Chimam, Robert Clarkn, Nikki Fergusono, Mary Hilfikerp, Kerri LaRovereq, Iain Macintoshr, Darryl Miless, Kevin Morrist, Nicole O’Brienu, Jose Pinedav, Courtney Robertsonw, Karen Walsonx, Nico Westy, Anthony Willyerdz, Jerry Zimmermanh, Brandon ZielinskiA,
mCincinnati Children’s Hospital;
nUniversity of Pittsburgh;
oVirginia Commonwealth University;
pUniversity of California—San Diego;
qBoston Children’s;
rUniversity Hospital Southampton, Southampton, UK;
sUniversity of Texas Southwestern;
tBirmingham Children’s Hospital, Birmingham, UK;
uNationwide Children’s Hospital;
vWashington University;
wJohns Hopkins University;
xChildren’s Healthcare of Atlanta;
yUniversity of Tennessee;
zPhoenix Children’s Hospital;
AUniversity of Utah Primary Children’s;
hSeattle Children’s Hospital
Funding
Support for this work was provided by the UW-Madison Office of the Vice Chancellor for Research, the Wisconsin Alumni Research Foundation, and NIH grant U01 NS081041 (Bell) and RO1 NS092870 (Ferrazzano). This study was also supported in part by a core grant to the Waisman Center from the National Institute of Child Health and Human Development P50HD105353. JG’s effort was supported in part by the Medical Physics Radiological Sciences Training Grant NIH T32 CA009206. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Consortia
Contributions
Author contributions included:
JGG: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Software, Validation, Visualization, Writing – original draft, Writing – review & editing, Interpretation of results.
PF: Funding acquisition, Investigation, Methodology, Project administration, Resources, Writing – review & editing, Interpretation of results.
AA: Conceptualization, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Writing – original draft, Writing – review & editing, Interpretation of results.
GK: Formal analysis, Writing – review & editing.
RB: Formal analysis, Writing – review & editing, interpretation of results.
EB: Conceptualization, Methodology, Validation, Writing – original draft, Writing – review & editing, Interpretation of results.
KB: Writing – review & editing, Interpretation of results.
AB: Formal analysis, Writing – review & editing.
BR: Writing – review & editing.
WB: Writing – review & editing.
SB: Writing – review & editing.
MB: Writing – review & editing.
All authors revised the manuscript critically for important intellectual content and approval of final version to be published and agreement to be accountable for the integrity and accuracy of all aspects of the work.
Corresponding author
Ethics declarations
Ethics approval
The study was conducted in accordance with ethics guidelines in the Declaration of Helsinki. The study was approved by the Health Sciences Institutional Review board at the University of Wisconsin – Madison and all participating sites.
Consent to participate
Informed consent was obtained from the subject or legal guardian when appropriate.
Consent for publication
Consent for publication was obtained from the subject or legal guardian when appropriate.
Competing interests
The authors have no conflicts or competing interests to disclose that are relevant to the current work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guerrero-Gonzalez, J.M., Kirk, G.R., Birn, R. et al. Multi-modal MRI of hippocampal morphometry and connectivity after pediatric severe TBI. Brain Imaging and Behavior 18, 159–170 (2024). https://doi.org/10.1007/s11682-023-00818-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-023-00818-x