Abstract
This study’s objective was to correlate the abnormalities in brain MRIs performed at corrected-term age for minor or moderate neurocognitive disorders in children school-age born extremely premature (EPT) and without serious sequelae such as autism, cerebral palsy, mental impairment. Data were issued from a cross-sectional multicenter study (GP-Qol study, number NCT01675726). Clinical examination and psychometric assessments were performed when the children were between 7 and 10 years old during a day-long evaluation. Term-equivalent age brain MRIs on EPT were analyzed with a standardized scoring system. There were 114 children included in the study. The mean age at the time of evaluation, was 8.47 years old (± 0.70). 59% of children with at least one cognitive impairment and 53% who had a dysexecutive disorder. Only ten EPT (8.7%) presented moderate to severe white and grey matter abnormalities. These moderate to severe grey matter abnormalities were associated with at least two abnormal executive functions [OR 3.08 (95% CI 1.04–8.79), p = 0.04] and language delay [OR 3.25 (95% CI 1.03–9.80), p = 0.04]. These results remained significant in the multivariate analysis. Moderate to severe ventricular dilatation abnormalities (15%, n = 17) were associated with ideomotor dyspraxia [OR 7.49 (95% CI 1.48–35.95), p = 0.02] and remained significant in multivariate analysis [OR 11.2 (95% CI 1.45–131.4), p = 0.02]. Biparietal corrected diameters were moderate abnormal in 20% of cases (n = 23) and were associated to visuo spatial integration delay [OR 4.13 (95% CI 1.23–13.63), p = 0.02]. Cerebral MRI at term-equivalent age with scoring system analysis can provide information on long-term neuropsychological outcomes at school-age in EPTs children having no severe disability.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There has been a decline in severe sensorimotor or cognitive disorders among extremely preterm infants (EPT) (Adams-Chapman et al., 2018). This decline is notable in conjunction with cerebral palsy rates (Pierrat et al., 2014). Nonetheless, EPT children continue to pose an increased risk for specific neuropsychological and behavioural disorders (Woodward et al., 2011; Hutchinson et al., 2013). Learning impairments, dysexecutive syndromes, attention disorders, minor motor disorders and/or behavioural disorders, are more common with EPT children than with term-born children (Johnson et al., 2016; Wolke et al., 2015). These neuropsychological and behavioural impairments, classified in the literature as minor to moderate, are likely to have a strong impact on the child and his family (Gire et al., 2018). Hence, there is value in screening for these disorders in order to begin early management (Gire et al., 2018).
Very preterm births (in particular extremely preterm births) pose risks for normal brain development even without any major perinatal brain injury complications such as periventricular leukomalacia, intraventricular haemorrhage and periventricular haemorrhagic infarction. The non-invasive nature of an MRI is valuable for tracking brain development. In addition to any major brain injury detected on the cranial ultrasound, brain MRIs provide better information regarding brain maturation and size (De Vries & Volpe, 2013).
Adverse outcomes can occur even with the absence of any significant brain abnormalities and can be related to subtle alterations in brain development (Tich et al., 2011; Wood et al., 2005). The MRI findings (diffuse white matter injuries, nuclear grey matter, cerebellum lesions and decreased brain size of grey and white matter) are associated with neurodevelopmental deficits, which have led to the concept of encephalopathy of prematurity (Anderson et al., 2015). Diffuse Excessive High Signal Intensity is very common increased signal intensity in the cerebral white matter on T2-weighted images at term-equivalent age, but has no prognostic value (Volpe, 2009; Spittle et al., 2011; Hinojosa-Rodriguez et al., 2017). Recently, studies have evaluated the relationship between brain abnormalities on cerebral MRIs at term-equivalent age in preterm infants born before 30 weeks of gestation and executive dysfunction (Anderson et al., 2015) and cognitive disorders at school age (Hintz et al., 2018). However, these studies focus on a small-sized population, which is not always consistent with gestational age and with short neurodevelopmental assessments (Hinojosa-Rodriguez et al., 2017; Anderson et al., 2015). So far, no studies have investigated possible relationships between neonatal brain anomalies in EPT and neurodevelopmental outcomes in these school-age children (7–10 years old), with no severe disabilities. Thus, the value of brain MRIs at term equivalent age to determine the clinical significance of subtle brain structural differences for school-age outcomes remains unclear, especially in EPT with no major impairments (autism, mental retardation or CP). Our study’s objective was to examine the association between brain MRI at a term-equivalent age in EPT and cognitive impairments at school age.
Methods
General framework and participants
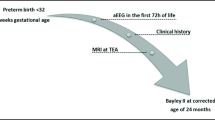
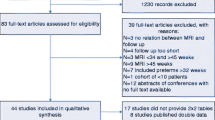
A cross-sectional, multicentre, observational study was carried out between 2012 and 2015 (GPQOL study) within five French Level III facilities authorised to care for EPT less than 28 week’s gestational age (GA) (Gire et al., 2018). Preterm children were recruited among 302 EPT children who were included in the GPQOL study (Fig. 1), the mean GA was 26.2 (± 0.8), birth weight was 879 (± 181) g and 51.5% were male. Inclusion criteria were enrolment in the GPQOL study with no severe disabilities (autism, mental delay, cerebral palsy), and participation in a day-long evaluation with complete data and an MRI scanning at term-equivalent age. MRIs at term-equivalent age were performed without sedation between 39- and 42-weeks corrected age. The children were placed in a 1.5 T MRI scanner and sagittal, coronal and axial, 3 mm thickness sections and T1 and T2 weighted sequences were obtained.
Then, these children, aged seven to 10 years, received a clinical examination, and an assessment of their motor, cognitive functions and quality of life (QoL). Motor skills were assessed by the Touwen Infant Neurological Examination (Hadders-Algra et al., 2010). A psychometric assessment was performed, using the WISC-IV (Canivez, 2014), the Rey’s figure (Senese et al., 2015), a short perceptual organization and memory test, and the NEPSY (NEuroPSYchological assessment) with subtests evaluating attention and executive functions (Korkman et al., 2001).
Psychometric assessment data
Disability was defined according to (Full scale Intelligent Quotient) FSIQ scores and the results of the Touwen Infant neurological examination:
-
no disability: FSIQ ≥ 89 and Touwen normal,
-
mild disabilities: FSIQ < 89 and ≥ 79 or Touwen abnormal,
-
moderate disability: FSIQ < 79 and ≥ 65 (Farooqi, Adamsson, Serenius & Hägglöf, 2016).
A “specific cognitive impairment" was considered if at least one of the following five specific neuropsychological mental illness disorders were observed: language delay, ideo-motor dyspraxia, visuo-integration delay, dysexecutive and attentional disorders (DSM IV classification of mental diseases).
Data brain MRI
The use of a standardized scoring system (Online Appendix 1) made it possible to semi-quantitatively evaluate the presence of lesions and its severity in the white and grey matter and the cerebellum.
The cerebral white matter scale (CWM) is the sum of six subscales:
-
(1)
Presence and severity of cystic lesions,
-
(2)
Signal abnormality,
-
(3)
Thinning of corpus callosum,
-
(4)
Lateral ventricular dilatation,
-
(5)
Periventricular white matter loss, and
-
(6)
Punctate lesions.
Each item was rated from 0 to 3, except for punctate lesions which were rated from 0 to 2. The CWM score was considered as normal if ≤ 5, slightly abnormal if the score was between 6 and 11 and moderately to severely abnormal if the score was between 12 and 17 (Woodward et al., 2006).
The cortical grey matter scale (CGM) was the sum of three subscales, with a score of 0 to 3 (Inder Wells et al., 2003).
-
(1)
Signal abnormality,
-
(2)
Delayed gyral maturation, and
-
(3)
Increased extra cerebral space.
The CGM scale was normal if ≤ 3, slightly abnormal if the score was between 4 and 6 and moderately to severely abnormal if between 7 and 9.
The cerebellum scale was considered normal in the absence of any hemorrhagic punctate abnormality, slightly abnormal if the bleeding lesion was unilateral, and moderately to severely abnormal if the abnormalities were bilateral, multiple or associated with a reduction in the volume of the cerebellum.
Four measurements were added to the analysis (Kidokoro et al., 2011; Nguyen et al., 2009) The measurements were taken from the coronal section and manually calculated:
-
(1)
The diameter of the lateral ventricles in order to assess ventricular dilatation;
-
(2)
The diameter biparietal (DBP) to evaluate any brain volume reduction;
-
(3)
The interhemispheric distance (IHD) to study the extra cerebral space;
-
(4)
The trans-cerebellar diameter (TCD) to study cerebellum volume.
Because the DBP and TCD diameters increase with GA, we calculated a modified score for each according to the following equations: modified DBP = measured DBP + [1.4 × (40-GA at the time of the MRI)]; and modified TCD = TCD measured + [0.83 × (40-GA at the time of the MRI)].
By using these measures, patterns of impaired brain growth were identified (Kidokoro et al., 2011):
-
1.
Biparietal corrected diameter: No anomaly ≥ 77 mm, Mild anomaly: ≥ 72 mm and < 77 mm, Moderate to severe anomaly < 72 mm.
-
2.
Corrected trans-cerebellar diameter: No anomaly: > 47 mm, moderate to severe anomaly: ≤ 47 mm.
-
3.
Ventricular dilatation: Normal or mild anomaly (Bilateral < 7.5 mm or unilateral ≤ 10 mm), moderate to severe anomaly (Bilateral or unilateral ≥ 10 mm).
-
4.
Interhemispheric distance: Normal or mild anomaly (< 5 mm), moderate to severe abnormality (≥ 5 mm).
Qualitative analysis and scoring were done by a radiologist (SC) and a neonatologist (GS). Both physicians were blinded to the clinical course quantitative analysis. Volumetry was performed manually by a neonatologist (TS) and supervised by the radiologist mentioned above. Interobserver concordance were between 0.7 and 0.8 for composite GMH score and composite grey score and measurements.
Data collection
Perinatal and pregnancy data collected from the medical records included maternal age, spontaneous or induced prematurity, antenatal corticosteroid therapy, multiple pregnancies; gestational age (weeks), birth weight, gender, and severe neonatal morbidities. Severe neonatal morbidities were defined as: severe bronchopulmonary dysplasia (defined as administration of oxygen for at least 28 days plus need for 30% or more oxygen and/or mechanical ventilatory support or continuous positive airway pressure at 36 weeks’ postmenstrual age); stage II and III necrotizing enterocolitis according to Bell’s staging; severe retinopathy of prematurity (ROP), defined as stage III or more and/or laser treatment; any of the following severe cerebral abnormalities on cranial ultrasonography (IVH III, IV white matter disease (periventricular leukomalacia), ventriculomegaly). The socio-demographic and family data, collected at the time of the assessment, included age, gender, parental education, parents’ employment, family’s material wealth, as reported by the child, using the Family Affluence Scale (FAS).
Statistical analysis
Qualitative variables were presented as numbers and percentages and quantitative data as means and standard deviations. Comparisons between pregnancy and perinatal data were performed between both participants and non-participant using chi-square test for qualitative data (when valid, Fisher test otherwise) and using Student’s t test for quantitative data (when valid, Mann–Whitney test otherwise).
Univariate analyses were performed to assess the relationship between the EPT’s MRI scores at term-equivalent age and their neuropsychological outcomes at school age. For neuropsychological quantitative outcomes a univariate linear regression model was used. This allowed us to estimate crude beta coefficients (expressing the average difference in neuropsychological results between two MRI score modalities), with their 95% confidence intervals. A univariate logistic regression model was used for neuropsychological results. These were expressed as binary qualitative data (attention deficit disorder, language delay, ideomotor dyspraxia, visuo-motor perception delay, dysexecutive disorders). This allowed us to estimate crude odds ratios (OR) (expressing the excess of risk to present an abnormal neuropsychological outcome between two MRI scoring modalities), with their 95% confidence interval.
A multivariate analysis completed this assessment in order to evaluate the independent effect of MRI abnormalities on neuropsychological outcomes. According to the literatures’ data, a forced adjustment was done on perinatal factors identified as being associated with outcomes: (corticosteroid exposure, GA, multiple birth, gender, birth weight, hypotrophy [defined by a birthweight < 10th percentile according to Epopé curves (Ego et al., 2016)], nosocomial infection, retinopathy grade III, severe bronchopulmonary dysplasia, necrotizing enterocolitis, and intraventricular haemorrhage III or IV, and FAS scores. All analyses were carried out using R software. All tests were performed two-sided and results were considered statistically significant if p < 0.05.
Results
Description of population
There were 114 children analyzed in this study (Table 1). The study’s population had a significantly greater neonatal morbidity (severe bronchopulmonary dysplasia, nosocomial infection) than those children not included in our analysis. The mean age at the evaluation was 8.47 years old (± 0.70). There were 42.11% of school-aged children born EPT who showed a mild to moderate disability. Additionally, 59% of children had at least one cognitive impairment and 53% had a dysexecutive disorder (Table 2).
MRIs at term-equivalent age data and assessment outcome at school age for the study’s EPT children
Sixteen MRIs could not be interpreted because of the child's movements (without sedation).The majority of the EPTs had none to mild anomalies on cerebral term corrected age MRIs. Cerebellar abnormalities were found in only six children (5.26%). The corrected biparietal diameter and corrected trans-cerebellar diameter abnormalities (mild, moderate to severe) were found respectively in 31.63 and 13.26% cases, respectively. Ventricular dilatation moderate to severe was found in 13.39% cases (Table 2).
MRI Correlations at term-equivalent age data and assessment outcomes at school age
We found no significant association between moderate to severe white or grey matter abnormalities on cerebral brain MRIs and FSIQ scores (Table 3). The existence of moderate to severe grey matter abnormalities with cerebral MRIs at term-equivalent age has a significant correlation to: the presence of at least two abnormal executive functions [OR 3.08 (95% CI 1.04–8.79), p = 0.04]; and with a language delay [OR 3.25 (95% CI 1.03–9.80), p = 0.04] at school age. After an adjustment for perinatal factors, the association between two abnormal executive functions remained significant in multivariate analyses [OR 5.09 (95% CI 1.35–20.82), p = 0.02], the association with language delay is close to significant [OR 3.44 (1.35–20.82), p = 0.06] (Table 3).
There was no significant association between moderate to severe abnormalities of the biometric parameters measured on the cerebral MRIs and the quantitative data of the FSIQ and these subtests. There was a significant association between ideomotor dyspraxia and white matter loss {ventricular dilatation [OR 7.49 (95% CI 1.48–35.95); p = 0.01], which remained significant in the multivariate analysis [OR 11.2 [95% CI 1.15–131.4), p = 0.02]}. There was a significant association between the visuo-motor integration delay and the biparietal corrected diameter [OR 4.13 (95% CI 1.23–13.64), p = 0.02], this association is close to significant in multivariate analysis [OR 4.19 (95%CI 0.81–27.63) p = 0.08] (Table 3).
Discussion
For the most part, those in our population who were free from severe neurosensory disorders had none to mild brain abnormalities but 42% of participants had a minor or moderate cognitive disability. Previously reported by Tich el al., a sub-normal term MRI was predictive of no cerebral palsy, but a lower predictive value for minor or moderate cognitive impairments (Tich et al., 2011). Our study confirms that WM lesions did not appear to be discriminating factors for correlating any occurrences of cognitive impairment at school age. However, several studies demonstrated the relationship between the brain’s WM abnormalities in neonatal MRIs and language abilities. (Anderson et al., 2015).
In our EP cohort, 8% had moderate to severe GM abnormalities versus 17% reported by Anderson et al. with school-aged children (Anderson et al., 2017). We found a correlation between GM abnormalities and the presence of executive dysfunction.
Concerning the cerebellum, MRIs detected nearly 20% of EPs with brain cerebellar lesions versus the 5.36% noted in our study. This is probably because due to the fact that our population’s initial selection was without major neurological disorders and our birth gestational age was lower than other studies (Anderson et al., 2017; Limperopoulos, 2016). Limperopoulos et al. correlated cerebellar involvement at term with language delay, poor cognitive function and behavioural disorders (Limpropoulos et al., 2007). Within our population, 13% of the cases had an abnormally sized cerebellum at term-equivalent age and no correlation with cognitive dysfunction at school age. In the literature, cerebellar atrophy is found in 50% of preterm infants < 30 weeks of gestation due to an interruption of the third trimester’s important in-utero growth activities (Volpe, 2009; Kidokoro et al., 2013).
Then, our study observed a statistically significant association between ventricular dilation and ideomotor dyspraxia and an association close to significant between decreased biparietal diameter and visuo-spatial integration. Tich et al. showed that the growth of biparietal diameter at term-equivalent age was predictive of cognitive and motor neurodevelopmental outcomes at two years in very preterm infants (Tich et al., 2011). More than half of our school-aged EPT children had at least one executive function impairment. The specific nature of those disorders and their underlying neuropathology remain poorly understood and with currently few predictors. From an anatomo-clinical point of view, executive function and motor disorders have been linked to the prefrontal region. However, they are also involved in a much larger brain network (Andrés, 2003) since the prefrontal cortex is the only cortical area where information from sensory regions converges and is also associated with the limbic system (support for emotional and motivational reactions). Independent of any perinatal factors, we found a significant correlation between subcortical lesions (grey nuclei) and dysexecutive syndromes and between cerebral volume and ideomotor dyspraxia during the school aged years. This confirms the importance of all these regions in the development of the prefrontal network.
Our study’s strengths include: a multicentric study with a homogeneous population (born between 24–27 GA), whose socio-economic levels were mostly high and without severe disability. These EPTs therefore had more discrete brain lesions, but were at a high risk of cognitive impairment because of their extreme prematurity; a large sample of EPTs with a corrected-term MRI with a long-term evaluation using the Wechsler Intelligence Scale for Children—Four Edition (WISC-IV) for school age children; the use of a standardized scoring system for reading brain MRIs allowing a semi-quantitative analysis of the MRI lesions; and a quantitative analysis by considering volumes with good correlation scores. A weakness of our study is that the population who had MRI scans represents only a small portion (1/3) of those premature babies who participated in the initial study. Moreover, participating preterm children had more neonatal morbidities than non-participants, which drive an indication bias of term MRI. Finally, the sizes of the subgroups of preterm children studied here were rather small, thus limiting its statistical power.
Conclusion
Cerebral MRIs at term-equivalent age could be predictive of minor or moderate impairment and could allow screening for early management of this population. Our metrics results (ventricular diameter and biparietal diameter) revealed a correlation between the measured surfaces and neurodevelopmental evolution. The use of software (specific algorithm to automatically obtain brain MRI metrics in preterm neonates revealed a significant correlation between reproducible metrics of the main brain structures with the neurodevelopment at two years of age (Morel et al., 2020). Automated brain MRI metrics could be now suitable in daily practice for neonatologists and radiologists. The combination of neuroimaging and longitudinal follow-up of EPT children provides a better long-term understanding of the neuro-anatomical abnormalities associated with EF deficits and subtypes of developmental coordination disorder.
References
Adams-Chapman, I., Heyne, R. J., DeMauro, S. B., Duncan, A. F., Hintz, S. R., Pappas, A., Vohr, B. R., McDonald, S. A., Das, A., Newman, J. E., & Higgins, R. D. (2018). Neurodevelopmental impairment among extremely preterm infants in the neonatal research network. Pediatrics, 141(5), e20173091.
Anderson, P. J., Cheong, J. L. Y., & Thompson, D. K. (2015). The predictive validity of neonatal MRI for neurodevelopmental outcome in very preterm children. Seminars in Perinatology, 39, 147–158.
Anderson, P. J., Treyvaud, K., Neil, J. J., Cheong, J. L. Y., Hunt, R. W., Thompson, D. K., Lee, K. J., Doyle, L. W., & Inder, T. E. (2017). Associations of newborn brain magnetic resonance imaging with long-term neurodevelopmental impairments in very preterm children. Journal of Pediatrics, 187, 58–65.
Andrés, P. (2003). Frontal cortex as the central executive of working memory: Time to revise our view. Cortex, 39, 871–895.
Bartlett, D. J., Galuppi, B., Palisano, R. J., & McCoy, S. W. (2016). Consensus classifications of gross motor, manual ability, and communication function classification systems between therapists and parents of children with cerebral palsy. Developmental Medicine and Child Neurology, 58(1), 98–99.
Bax, M. C. O., Flodmark, O., & Tydeman, C. (2007). Definition and classification of cerebral palsy. From syndrome toward disease. Developmental Medicine and Child Neurology, 109, 39–41.
Canivez, G. L. (2014). Construct validity of the WISC-IV with a referred sample: Direct versus indirect hierarchical structures. School Psychology Quarterly, 29(1), 38–51.
Dammann, O., Cesario, A., & Hallen, M. (2007). NEOBRAIN–an EU-funded project committed to protect the newborn brain. Neonatology, 92(4), 217–218.
De Vries, L. S., & Volpe, J. J. (2013). Value of sequential MRI in preterm infants. Neurology, 81(24), 2062–2063.
Ego, A., Prunet, C., Lebreton, E., & Zeitilin, J. (2016). Customized and non-customized French intrauterine growth curves. I—Methodology. J Gynecol Obstet Biol Reprod, 45(2), 155–64.
Farooqi, A., Adamsson, M., Serenius, F., & Hägglöf, B. (2016). Executive functioning and learning skills of adolescent children born at fewer than 26 weeks of gestation. PLoS ONE, 11(3), e0151819.
Gire, C., Resseguier, N., Brévaut-Malaty, V., Marret, S., Cambonie, G., Souksi-Medioni, I., Müller, J.-B., Garcia, P., Berbis, J., Tosello, B., & Auquier, P. (2018). Quality of life of extremly preterm school-age children without major handicap: A cross sectional observational study. Archives of Disease in Childhood. https://doi.org/10.1136/archdischild-2018-315046
Hadders-Algra, M., Heineman, K. R., Bos, A. F., & Middelburg, K. J. (2010). The assessment of minor neurological dysfunction in infancy using the Touwen infant neurological examination: Strengthsand limitations. Developmental Medicine and Child Neurology, 52, 87–92.
Hinojosa-Rodríguez, M., Harmony, T., Carrillo-Prado, C., Van Horn, J. D., Irimia, A., Torgerson, C., & Jacokes, Z. (2017). Clinical neuroimaging in the preterm infant: Diagnosis and prognosis. NeuroImage Clin, 16, 355–368.
Hintz, S. R., Vohr, B. R., Bann, C. M., Taylor, H. G., Das, A., Gustafson, K. E., Yolton, K., Watson, V. E., Lowe, J., DeAnda, M. E., Ball, M. B., Finer, N. N., Van Meurs, K. P., Shankaran, S., Pappas, A., Barnes, P. D., Bulas, D., Newman, J. E., Wilson-Costello, D. E.,...Higgins, R. D. (2018). Preterm neuroimaging and school-age cognitive outcomes. Pediatrics, 142(1), e20174058.
Hutchinson, E. A., De Luca, C. R., Doyle, L. W., Roberts, G., Anderson, P. J., & Victorian Infant Collaborative Study Group. (2013). School-age outcomes of extremely preterm or extremely low birth weight children. Pediatrics, 131, 1053–1061.
Inder, T. E., Wells, S. J., Mogridge, N. B., Spencer, C., & Volpe, J. J. (2003). Defining the nature of the cerebral abnormalities in the premature infant: A qualitative magnetic resonance imaging study. Journal of Pediatrics, 143, 171–179.
Janvier, A., & Barrington, K. (2012). Trying to predict the future of ex-preterm infants: Who benefits from a brain MRI at term? Acta Paediatrica, 101(10), 1016–1017.
Johnson, S., Strauss, V., Gilmore, C., Jaekel, J., Marlow, N., & Wolke, D. (2016). Learning disabilities among extremely preterm children without neurosensory impairment: Comorbidity, neuropsychological profiles and scholastic outcomes. Early Human Development, 103, 69–75.
Kidokoro, H., Neil, J. J., & Inder, T. E. (2011). New MR imaging assessment tool to define brain abnormalities in very preterm infants at term. AJNR Am J. Neuroradiol, 34, 2208–2214.
Korkman, M., Kemp, S. L., & Kirk, U. (2001). Effects of age on neurocognitive measures of children ages 5 to 12: A cross-sectional study on 800 children from the United States. Developmental Neuropsychology, 20, 331–354.
Limperopoulos, C. (2016). The vulnerable immature cerebellum. Seminars in Fetal and Neonatal Medicine, 21, 293–294.
Limperopoulos, C., Bassan, H., Gauvreau, K., Robertson, R. L., Sullivan, N. R., Benson, C. B., Avery, L., Stewart, J., MD, J. S. S., Ringer, S. A., Volpe, J. J., & duPlessis, A. J. (2007). Does cerebellar injury in premature infants contribute to the high prevalence of long-term cognitive, learning, and behavioral disability in survivors? Pediatrics, 120, 584–593.
Morel, B., Bertault, P., Favrais, G., Tavernier, E., Tosello, B., Proisy, M., Xu, Y., Bloch, I., Sirinelli, D., Adamsbaum, C., Tauber, C., Saliba, E., & EPIRMEX study group. (2020). Automated brain MRI metrics in the EPIRMEX cohort of preterm newborns: Correlation with the neurodevelopmental outcome at 2 years. Diagn Interv Imaging., 10, S2211-5684(20)30270–9.
Nguyen The Tich, S., Anderson, P. J., Shimony, J. S., Hunt, R. W., Doyle, L. W., & Inder, T. E. (2009). A novel quantitative simple brain metric using MR imaging for preterm infants. AJNR American Journal of Neuroradiology, 30(1), 125–131.
Nguyen, T. N., Spencer-Smith, M., Zannino, D., Burnett, A., Scratch, S. E., Pascoe, L., Ellis, R., Cheong, J., Thompson, D., Inder, T., Doyle, L. W., & Anderson, P. J. (2018). Developmental trajectory of language from 2 to 13 years in children born very preterm. Pediatrics, 141(5), e20172831.
Pierrat, V., Marchand-Martin, L., Arnaud, C., Kaminski, M., Resche-Rigon, M., Lebeaux, C., Bodeau-Livinec, F., Morgan, A. S., Goffinet, F., Marret, S., & Ancel, P. Y. (2014). Neurodevelopmental outcome at 2 years for preterm children born at 22 to 34 weeks’ gestation in France in 2011: EPIPAGE-2 cohort study. BMJ, 358, j3448.
Senese, V. P., De Lucia, N., & Consonb, M. (2015). Cognitive predictors of copying and drawing from memory of the Rey-Osterrieth complex figure in 7- to 10-year-old children. The Clinical Neuropsychologist, 29, 118–132.
Serenius, F., Ewald, U., Farooqi, A., Fellman, V., Hafström, M., Hellgren, K., Maršál, K., Ohlin, A., Olhager, E., Stjernqvist, K., Strömberg, B., Ådén, U., & Källén, K. (2016). Neurodevelopmental outcomes among extremely preterm infants 6.5 years after active perinatal care in Sweden. JAMA Pediatrics, 170(10), 954–963.
Shepherd, E., Salam, R. A., Middleton, P., Han, S., Makrides, M., McIntyre, S., Badawi, N., & Crowther, C. A. (2018). Neonatal interventions for preventing cerebral palsy: An overview of cochrane systematic reviews. Cochrane Database Syst Review, 6, CD012409.
Spittle, A. J., Cheong, J., Doyle, L. W., Roberts, G., Lee, K. J., Lim, J., Hunt, R. W., Inder, T. E., & Anderson, P. J. (2011). Neonatal white matter abnormality predicts childhood motor impairment in very preterm children. Developmental Medicine and Child Neurology, 53, 1000–1006.
Tich, S. N. T., Anderson, P. J., Hunt, R. W., Lee, K. J., Doyle, L. W., & Inder, T. E. (2011). Neurodevelopmental and perinatal correlates of simple brain metrics in very preterm infants. Archives of Pediatrics and Adolescent Medicine, 165, 216–222.
Vaivre-Douret, L., Lalanne, C., Ingster-Moati, I., Boddaert, N., Cabrol, D., Dufier, J. L., Golse, B., & Falissard, B. (2011). Subtypes of developmental coordination disorder: Research on their nature and etiology. Developmental Neuropsychology, 36(5), 614–43.
Volpe, J. J. (2009). Brain injury in premature infants: A complex amalgam of destructive and developmental disturbances. Lancet Neurology, 8, 110–124.
Wolke, D., Strauss, V. Y. C., Johnson, S., Gilmore, C., Marlow, N., & Jaekel, J. (2015). Universal gestational age effects on cognitive and basic mathematic processing: 2 cohorts in 2 countries. Journal of Pediatrics, 166, 1410–1416.
Wood, N. S., Costeloe, K., Gibson, A. T., Hennessy, E. M., Marlow, N., Wilkinson, A. R., & EPICure Study Group. (2005). The EPICure study: associations and antecedents of neurological and developmental disability at 30 months of age following extremely preterm birth. Archives of Disease Child Fetal Neonatal Edition, 90(2), F134–F140.
Woodward, L. J., Anderson, P. J., Austin, N. C., Howard, K., & Inder, T. E. (2006). Neonatal MRI to predict neurodevelopmental outcomes in preterm infants. New England Journal of Medicine, 55(7), 685–694.
Woodward, L. J., Clark, C. A. C., Pritchard, V. E., Anderson, P. J., & Inder, T. E. (2011). Neonatal white matter abnormalities predict global executive function impairment in children born very preterm. Developmental Neuropsychology, 36, 22–41.
Acknowledgements
We are grateful for the participation of all families of preterm infants in the GPQoL study and for the cooperation of the five neonatal units participating in the study. Group Information: The GPQOL Group members. The GPQOL Study Group: MC Lemarchand, Neuropsychologist, N Mestre, Scientific Research Manager (Department of Neonatal Medicine, Rouen University Hospital, Rouen), M Rebattel, Neuropsychologist (Department of Neonatal Medicine, Nimes University Hospital, Nimes), JC Rozé, MD, PhD, C Coudronnières, Neuropsychologist (Departement of neonatal medicine, Nantes University Hospital, Nantes), G Menard, Neuropsychologist, M Pache, Neuropsychologist, C Morando, Scientific Research Manager (Department of Neonatology, North Hospital, APHM University Hospital, Marseille, France), MA Einaudi MD, PhD (UMR 7268 ADÉS, Aix-Marseille University-EFS-CNRS, Faculty of Medicine, Marseille, France).
Funding
This study was supported by the promoter APHM, University Hospital and its partners, The French Health Ministry, Grant PHRC Ref ANSM B120183-30, Ref CPP: 12.018, Ref promoter: 2012-02.
Author information
Authors and Affiliations
Consortia
Contributions
Conception and study design (CG and AG), data collection or acquisition (AG, GS and BT), statistical analysis (NR and CG), interpretation of results (AG, GS, SC, BT and CG), drafting the manuscript work or revising it critically for important intellectual content (GS, AG, SC, NR, BT and CG) approval of final version to be published agreement to be accountable for the integrity and accuracy of all aspects of the work (All authors).
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the CPP (Committee for the Protection of Persons) (18/12/2012 ref 12.018) and is registered on ClinicalTrials.gov, number NCT01675726.
Conflict of interest
Authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The members of the GPQOL study Group are listed in acknowledgements section.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Garbi, A., Sorin, G., Coze, S. et al. Predictive value of brain MRI at term-equivalent age in extremely preterm children on neurodevelopmental outcome at school-age. Brain Imaging and Behavior 16, 878–887 (2022). https://doi.org/10.1007/s11682-021-00559-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-021-00559-9