Abstract
The possible uniqueness of social stimuli constitutes a key topic for cognitive neuroscience. Growing evidence highlights graded contributions to their semantic processing by the anterior temporal lobe (ATL), where the omni-category response displayed by its ventrolateral sector might reflect the integration of information relayed from other regions. Among these, the superior polar ATL was specifically associated with representing social concepts. However, most previous studies neglected the close relationship between social and emotional semantic features, which might confound interpreting the degree of overlap vs. specificity of social and emotional conceptual processing. We addressed this issue via two activation-likelihood-estimation meta-analyses of neuroimaging studies reporting brain structures associated with processing social or emotional concepts. Alongside a common involvement of the ventromedial prefrontal cortex, we found social and emotional concepts to be specifically associated with lateral temporal areas (including the superior polar ATL) and the amygdala, respectively. These results support the specialization of distinct sectors of the fronto-temporo-limbic circuitry for processing social vs. emotional concepts, and the integration of their output in medial prefrontal regions underlying the regulation of social behavior. These results pave the way for further studies addressing the neural bases of conceptual knowledge, its impairment after fronto-temporal brain damage, and the effect of rehabilitative interventions targeting its main functional modules.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Some recent advancements in the neuroscience of conceptual knowledge reflect a wider paradigm change in cognitive neuroscience, from a modular view centered on cortical specialization to distributed models of neuro-cognitive functioning integrating across input modalities, stimuli content, and underlying processes (Taylor et al. 2011). A lively debate revolves around the putative neural specialization for the processing of social information (Insel and Fernald 2004) at increasing levels of complexity from selective stimuli such as faces to transmodal information detached from sensory experiences, i.e. a more generic “social knowledge” (Rice et al. 2018). The latter potentially includes any kind of information concerning both social entities and behaviors as well as the words referring to them, encapsulated in social concepts used to convey meanings in communicative exchanges and to interpret others’ behaviors (Olson et al. 2013). Whether affective information constitutes a core component of social concepts, or rather underpins an independent type of emotional concepts associated to specific neural correlates, is debated (Vigliocco et al. 2014; Zahn et al. 2007). Increasing data highlight the involvement of the anterior temporal lobe (ATL) in representing social (Olson et al. 2013), and possibly affective (Olson et al. 2007), concepts, but different interpretations of such role have been proposed.
The ATL has been considered an amodal convergence zone (Damasio et al. 2004; Tranel et al. 1997), or “hub”, supporting conceptual representations regardless of modality and semantic category (Patterson et al. 2007). However, the notion of a general-purpose semantic processor associating different aspects of conceptual knowledge has been challenged by considerable evidence of its prominent engagement in socio-affective processing (Ross and Olson 2010). The association between ATL damage and altered social semantic processing (Chan et al. 2009; Snowden et al. 2004, 2012; Thompson et al. 2004) led to suggest that this region might rather constitute a social-specific semantic hub (Wang et al. 2017, 2019), with its limbic and polar sectors possibly integrating affective and valence-related information (Rice et al. 2015). A distributed model of semantic knowledge indeed suggests that coherent conceptual representations are generated by “hub” transmodal regions merging modality- and content-specific contributions from other network nodes based on their connectivity patterns (Binney et al. 2016; Lambon-Ralph 2014).
Multifaceted evidence suggests that the ATL plays such a hub role in representing social concepts (Rice et al. 2018). The convergence of sensory pathways into the temporal pole (Olson et al. 2007), possibly supporting its role in multimodal perceptual analyses (Binney et al. 2012), fits with the effect of ATL damage on socio-affective processing (Kumfor et al. 2017) and social behavior (Mychack et al. 2001). Moreover, neuroimaging studies in healthy individuals have shown the distinct roles played by the ventrolateral ATL sector, associated with omni-category responses to different concept types, and its superior polar sector (sATL), showing a preferential response to social concepts (Binney et al. 2016). While this process also involves the main nodes of the mentalizing network, i.e. ventro- and dorso-medial prefrontal cortex (vmPFC and dmPFC), posterior cingulate cortex (PCC) and temporo-parietal junction (TPJ) (Frith and Frith 2006), sATL activity tracks the richness of conceptual knowledge (Zahn et al. 2007). Overall, increasing evidence suggests that the network traditionally associated with the broad notion of “mentalizing” can be parcellated into more specific and informative functions (Moll et al. 2005a, b; Zahn et al. 2020), including social conceptual processing within the ATL (Binney and Ramsey 2020; Ross and Olson 2010).
The available evidence on this topic might be confounded, however, by the close relationship between social and affective features (Wang et al. 2019), both of which have been associated with the fronto-limbic circuitry. Whether the ATL codes the affective features of social concepts, or rather integrates into coherent concepts emotional information processed in other structures such as amygdala or vmPFC (Kim et al. 2011), is still unclear. Preliminary neuroimaging findings suggested that the lateral ATL is not activated when processing emotional words (Beauregard 2007; Cato et al. 2004), nor its activity is modulated by emotional valence (Zahn et al. 2007). More recent evidence confirms the sATL involvement in social semantics, but with an additional modulation by emotional valence in the temporal poles (Wang et al. 2019). This evidence suggests that the neural representations of social and emotional concepts are not completely overlapping, and that the coding of social concepts might integrate affective features represented either in the ATL itself, or elsewhere. Testing these hypotheses requires to assess the neural bases of either concept types, regardless of the constraints inherent in specific studies. This goal can be pursued with coordinate-based meta-analyses, a quantitative unbiased approach which allows identifying the areas that are consistently activated in a particular class of paradigms (Laird et al. 2005; Turkeltaub et al. 2002).
We thus performed a coordinate-based meta-analysis to unveil the neural bases of social concepts, over and beyond the role of their affective facets that were separately addressed in a second meta-analysis on non-social emotional concepts. We predicted the engagement of ATL and amygdala when coding social and emotional concepts, respectively. We additionally assessed their overlap in the mPFC, in which the integrated processing of both concept types might support the regulation of social interactions (Binney and Ramsey 2020).
Materials and methods
See further details in Supplementary Methods.
Study design
Based on the current prescriptions for ALE meta-analyses (Eickhoff et al. 2016), we aimed at including at least 17 experiments (contrasts) in each dataset, to achieve sufficient power for moderate effects and ensure that results would not be driven by single experiments.
Social concepts
We first surveyed the relevant literature by searching for “social concept fMRI” on Pubmed (https://www.ncbi.nlm.nih.gov/pubmed/), and by constraining this search to studies on human subjects published in the last 15 years (see Fig.1 for a detailed flowchart of literature search and selection). From a pool of 319 studies, we retained only those fulfilling the following selection criteria:
-
1)
published studies written in English;
-
2)
empirical fMRI studies, while excluding review articles, behavioral studies and those employing other techniques, to ensure comparable spatio-temporal resolution;
-
3)
studies including non-clinical and drug-free participants, to prevent possible differences in brain activity associated with neuro-psychiatric diseases or pharmacological manipulations;
-
4)
studies with adult subjects (age range: 18–60 years);
-
5)
studies reporting whole-brain activation coordinates, rather than results limited to regions of interest (ROIs); moreover, we ensured that no partial brain coverage was reported in the Methods section of the retrieved papers.
-
6)
studies investigating brain activity related to the processing of social concepts, meant as linguistic stimuli (words or sentences) whose referents are meaningful in the context of sociality and/or interpersonal interactions (Lin et al. 2015; Wang et al. 2019). Following Desai et al. (2018), to control for orthographic and phonological processes we included only studies using verbal stimuli (written words or sentences), while excluding those involving nonverbal materials such as pictures. Namely, we selected contrasts requiring participants to attend to verbal stimuli aimed to elicit a representation of social interaction by contrasting this kind of stimulus with concepts devoid of social facets. This selection retained studies contrasting social and non-social emotions, while we excluded studies in which social concepts were contrasted with low-level baseline conditions such as rest or visual fixation.
We included studies fulfilling the above criteria regardless of: (a) stimulus valence; (b) experimental paradigm. This procedure led to include in the meta-analysis 22 previously published studies (Table S1), resulting from 23 experiments (individual comparisons reported) with 538 subjects and 302 foci. The inclusion of multiple contrasts/experiments from the same set of subjects can generate dependence across experiment maps and thus decrease the validity of meta-analytic results. To prevent this issue, we adjusted for within-group effects by pooling the coordinates from all the relevant contrasts of a paper into one experiment (Turkeltaub et al. 2002).
Emotional concepts
We searched for the coordinates associated with the neural processing of emotional concepts by applying additional inclusion criteria to the studies reported in meta-analyses by Desai et al. (2018) and Brooks et al. (2017). We then expanded our search for other relevant studies by carefully examining both the studies quoting, and those quoted by, those two meta-analyses.
We selected only studies reporting brain activations associated with the processing of emotional concepts compared with stimuli without emotional characterization (i.e. neutral stimuli), while excluding comparisons with rest, fixation, or similar low-level conditions. We included only studies using verbal stimuli (written sentences or words). Since we aimed to assess both the specific and overlapping neural representations of social and emotional concepts, we excluded from this ALE analysis the studies using mainly emotional stimuli with social connotation.
We included studies fulfilling these criteria regardless of stimulus valence and experimental task, to ensure the generalizability of results. This procedure led to include in this ALE meta-analysis 27 previously published studies (Table S2), resulting from 27 experiments (individual comparisons reported) with 453 subjects and 287 foci.
Activation likelihood estimation (ALE)
We used the GingerALE software, based on Eickhoff et al. (2009; 2012; see Supplementary Methods), to identify consistently activated regions associated with processing either social or emotional concepts, as well as those specifically or commonly associated with these types of stimuli via direct comparisons and a conjunction analysis, respectively. The statistical maps were thresholded at p < 0.05, corrected for cluster-level family-wise error (FWE) with voxel-level forming threshold at p < 0.001 uncorrected (Eickhoff et al. 2016; Muller et al. 2018).
Results
Neural representation of social concepts
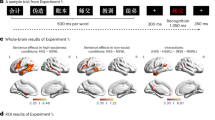
The neural processing of social concepts recruited the dmPFC and vmPFC, PCC and precuneus, alongside both the posterior and anterior sectors of the lateral temporal cortex. In the former case, activations encompassed the middle temporal gyrus (MTG), superior temporal sulcus (STS) and TPJ bilaterally (Fig. 2; Table 1). Anterior temporal activations involved the inferior and middle temporal gyri bilaterally, extending into the superior temporal sulcus.
The neural processing of social and emotional concepts. The figure reports the brain structures consistently associated with the processing of social (top, blue) and emotional (bottom, red) concepts. All the reported activations survived a statistical threshold of p < 0.05 corrected for multiple comparisons
Neural representation of emotional concepts
Processing emotional concepts was associated with consistent activity in the vmPFC, alongside the left amygdala and pSTS/TPJ (Fig. 2; Table 2).
Neural representation of social vs. emotional concepts
A conjunction analysis across social and emotional concepts highlighted a significant common activation in the vmPFC (Fig. 3; Table 3). Processing social, compared with emotional, concepts was associated with stronger activity in dmPFC, vmPFC and precuneus, alongside both posterior (MTG, pSTS and TPJ) and anterolateral temporal cortex, bilaterally (Fig. 3; Table 3). In the ATL, the social-related activity involved the inferior-middle temporal gyri bilaterally, extending into the superior temporal sulcus. The reverse comparison highlighted the left amygdala (Fig. 3; Table 3).
Common and specific brain activity for processing social and emotional concepts. The figure reports the structures showing significant common (top, pink) and specific (bottom) brain activations associated with processing social and emotional concepts. All the reported activations survived a statistical threshold of p < 0.05 corrected for multiple comparisons
Discussion
We confirmed that processing social, compared with emotional, concepts is more strongly associated with a set of regions which have been collectively ascribed to the mentalizing network, i.e. dmPFC, ATL, pSTS/TPJ and posterior cingulate cortex/precuneus (Ross and Olson 2010; Arioli et al. 2018b). Emotional concepts selectively recruited the left amygdala, while the vMPFC was associated with both concept types. These findings provide novel insights into the contribution of content-specific semantic encoding to superordinate processes such as mentalizing and social cognition (Binney and Ramsey 2020).
In keeping with several studies comparing social and non-social semantic processing, the superior polar ATL was specifically associated with social concepts (Fig. 2) (Binney et al. 2016; Lin et al. 2019, 2020; Skipper et al. 2011; Wang et al. 2019). This region is considered one of several “convergence zones” of a distributed network, centered in the ATL, in which the progressive integration of multiple features carried by upstream nodes generates coherent semantic representations (Olson et al. 2007; Rice et al. 2018; Visser et al. 2010). In particular, it has been suggested that the sATL role in social conceptual processing might reflect its sensitivity to the combination of social and either sensori-motor (Lin et al. 2019) or affective (Binney et al. 2015; Troche et al. 2014) information. Regardless of its specific interpretation, the functional role of the sATL is considered to reflect its connectivity within a larger network of areas, including frontal and limbic regions via the uncinate fasciculus (Bajada et al. 2017).
Indeed, processing social concepts was also associated with the mPFC, PCC and pSTS/TPJ, all of which are intrinsically connected to the ATL (Yeo et al. 2011; Wang et al. 2019). All these regions have been previously associated with tasks requiring the attribution of mental states (Brunet et al. 2000), particularly when they involve social interactions (Lahnakoski et al. 2012; Walter et al. 2004) and the associated affective responses (Harris et al. 2007). Unlike the sATL, however, none of these regions show a direct relationship with the depth of semantic retrieval (Zahn et al. 2007). This widespread pattern of activity is thus suggestive of multifaceted contributions to the construction of social meaning, which might in turn support mentalizing (Ross and Olson 2010). Processing social groups is more likely to promote the retrieval of personal knowledge and autobiographical memories compared with individuals (Holland et al. 2011), which might explain the involvement of precuneus (Johnson et al. 2006). In turn, the latter is strongly connected with the mPFC (Cavanna and Trimble 2006), whose activity reflects distinct variables of social processing such as the number of stimuli in one’s social network (Lewis et al. 2012). The larger complexity of social groups, compared with single individuals, might thus represent another factor promoting a stronger interplay between regions underlying social semantics and in-depth inferences on social actors’ intentional states, such as the dmPFC (Meyer and Lieberman 2012; Mitchell et al. 2005; Sliwa and Freiwald 2017; Van der Cruyssen et al. 2015). This region plays a key role in decoding intentions in terms of internal mental states - e.g. thoughts, desires, and beliefs - when visuomotor cues are unavailable or insufficient (Arioli et al. 2018a, Arioli and Canessa 2019; Canessa et al. 2011; Van Overwalle and Baetens 2009) and proportionally to the abstractness of information processing (Baetens et al. 2014). Overall, this evidence highlights the possible contribution of social semantics to mentalizing, in terms of social conceptual representations detached from perceptual aspects (Trope and Liberman 2010; Wang et al. 2019).
A closer connection with the sensory dimension of social processing might involve the vmPFC (Roy et al. 2012), which was recruited by both emotional and social concepts. This region underpins evaluative processes modulating both social understanding and decision-making (Hiser and Koenigs 2018). One such process concerns flexible evaluations of social cues and behaviors, in conjunction with some of the aforementioned regions including dmPFC and precuneus. Such a domain-general function of vmPFC reflect its involvement in several tasks revolving around affective processing, e.g. emotion recognition (Drolet et al. 2012), learning (Nashiro et al. 2012), interference (Fales et al. 2008, 2009) and regulation (Morawetz et al. 2017), often in association with social stimuli (Immordino-Yang and Singh 2013). The vmPFC activation for both concept types might thus underpin the integration of emotion-related information into the neural representations of social concepts, likely enriched by signals on its valence, provided by the amygdala.
Indeed, in keeping with neuroimaging (Bickart et al. 2011) and lesional (Bickart et al. 2014) evidence of its role in affective processing, the amygdala was the only structure showing a selective response to emotional concepts. This structure has been mostly associated with processing nonverbal stimuli such as emotional faces (Duerden et al. 2013; Lindquist et al. 2016; Sergerie et al. 2008; Wager et al. 2003), while the effects of emotional valence on word processing are less consistent (Citron 2012) and often associated to the temporal pole (Ethofer et al. 2006; Kuchinke et al. 2005). Among the ATL semantic nodes, however, the temporal pole shows the strongest intrinsic connectivity with the amygdala (Wang et al. 2019). In line with distributed accounts of semantic cognition, the selective amygdala activation for emotional concepts is thus consistent with the segregated processing of social and emotional features into distinct ATL sectors (Wang et al. 2019), and their subsequent integration in semantic hubs. Moreover, the left-hemispheric lateralization of amygdala activation fits with previous evidence of an automatic processing of emotional words in the left hemisphere, providing fast access to semantically-associated stimuli (Abbassi et al. 2015; Herbert et al. 2011a, b; Moseley et al. 2012; Ponz et al. 2014). Its engagement might reflect a domain-general function such as stimuli evaluation based on their salience and behavioral relevance in terms of affiliation/aversion (Bickart et al. 2014; Canessa et al. 2013). This consideration highlights a potential limitation of this study, due to the heterogeneity of the stimuli used in previous studies on emotional concepts (generally focusing on high arousal and negative emotions) vs. social concepts (typically controlling for valence but not arousal). The evidence of stronger amygdala response to emotional than social concepts might thus reflect an inherent bias, which future studies should address, towards the use of negative emotional stimuli.
Other limitations of this work, mainly due to the number of relevant studies, concern the lack of evidence on multiple factors which might drive brain responses to social concepts, such as the fine-grain in the definition of “social knowledge”, the level of linguistic processing (e.g. discourse, sentence, word), and/or the task employed to activate conceptual representations. Moreover, a cluster-based inference might favor large regions that are least likely to host content-specific representations. Finally, future studies might refine our findings by addressing subtler distinctions between the neural processing of exclusively-social, socio-emotional and exclusively-emotional concepts.
Conclusions
We provided novel meta-analytic evidence on both specific and overlapping neural bases of processing social and emotional concepts. Although the limited fine-grain of our findings prevents a parcellation of the “hub and spoke” functional modules (Binney et al. 2016), these results show that the neural correlates of processing social and affective semantic features are at least partially segregated in the lateral and medial ATL sectors, respectively. Once integrated into convergence zones, such as vmPFC, these features might underpin higher-order conceptual representations any kind of information concerning both social entities and behaviors as well as the words referring to them supporting communicative exchanges, as well as the evaluation and planning of social behaviors (Binney and Ramsey 2020). Observing distinct brain structures associated with processing either social or emotional concepts, or both, might suggest novel tools for assessing pathological conditions characterized by different degrees of semantic, socio-emotional or behavioral impairments, such as the variants of fronto-temporal dementia (Chan et al. 2009). These results highlight the potential translational implications of social neuroscience (Arioli et al. 2018a), and pave the way for future studies addressing the neural bases of conceptual knowledge, its impairment after ATL damage, and the effect of rehabilitative interventions targeting its functional modules.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Abbassi, E., Blanchette, I., Ansaldo, A. I., Ghassemzadeh, H., & Joanette, Y. (2015). Emotional words can be embodied or disembodied: the role of superficial vs. deep types of processing. Frontiers in Psychology, 6, 975. https://doi.org/10.3389/fpsyg.2015.00975
Arioli, M., & Canessa, N. (2019). Neural processing of social interaction: Coordinate-based meta-analytic evidence from human neuroimaging studies. Human Brain Mapping, 40(13), 3712–3737. https://doi.org/10.1002/hbm.24627
Arioli, M., Crespi, C., & Canessa, N. (2018a). Social Cognition through the Lens of Cognitive and Clinical Neuroscience. BioMed Research International, 4283427. 10.1155/2018a/4283427.
Arioli, M., Perani, D., Cappa, S., Proverbio, A. M., Zani, A., Falini, A., & Canessa, N. (2018). Affective and cooperative social interactions modulate effective connectivity within and between the mirror and mentalizing systems. Human Brain Mapping, 39(3), 1412–1427. https://doi.org/10.1002/hbm.23930
Baetens, K., Ma, N., Steen, J., & Van Overwalle, F. (2014). Involvement of the mentalizing network in social and non-social high construal. Social Cognitive and Affective Neuroscience, 9(6), 817–824. https://doi.org/10.1093/scan/nst048
Bajada, C. J., Jackson, R. L., Haroon, H. A., Azadbakht, H., Parker, G. J. M., Ralph, L. M. A., & Cloutman, L. L. (2017). A graded tractographic parcellation of the temporal lobe. Neuroimage, 155, 503–512. https://doi.org/10.1016/j.neuroimage.2017.04.016.
Beauregard, M. (2007). Mind does really matter: evidence from neuroimaging studies of emotional self-regulation, psychotherapy, and placebo effect. Progress in Neurobiology, 81(4), 218–236. https://doi.org/10.1016/j.pneurobio.2007.01.005
Bickart, K. C., Dickerson, B. C., & Barrett, L. F. (2014). The amygdala as a hub in brain networks that support social life. Neuropsychologia, 63, 235–248. https://doi.org/10.1016/j.neuropsychologia.2014.08.013.
Bickart, K. C., Wright, C. I., Dautoff, R. J., Dickerson, B. C., & Barrett, L. F. (2011). Amygdala volume and social network size in humans. Nature Neuroscience, 14(2), 163–164. https://doi.org/10.1038/nn.2724
Binney, R. J., Hoffman, P., & Lambon Ralph, M. A. (2016). Mapping the multiple graded contributions of the anterior temporal lobe representational hub to abstract and social concepts: Evidence from distortion-corrected fMRI. Cerebral Cortex. https://doi.org/10.1093/cercor/bhw260
Binney, R. J., Parker, G. J., & Lambon Ralph, M. A. (2012). Convergent connectivity and graded specialization in the rostral human temporal lobe as revealed by diffusion-weighted imaging probabilistic tractography. Journal of Cognitive Neuroscience, 24(10), 1998–2014. https://doi.org/10.1162/jocn_a_00263
Binney, R. J., & Ramsey, R. (2020). Social Semantics: The role of conceptual knowledge and cognitive control in a neurobiological model of the social brain. Neuroscience & Biobehavioral Reviews, 112, 28–38. https://doi.org/10.1016/j.neubiorev.2020.01.030
Brooks, J. A., Shablack, H., Gendron, M., Satpute, A. B., Parrish, M. H., & Lindquist, K. A. (2017). The role of language in the experience and perception of emotion: a neuroimaging meta-analysis. Social Cognitive and Affective Neuroscience, 12(2), 169–183. https://doi.org/10.1093/scan/nsw121
Brunet, E., Sarfati, Y., Hardy-Bayle, M. C., & Decety, J. (2000). A PET investigation of the attribution of intentions with a nonverbal task. Neuroimage, 11(2), 157–166. https://doi.org/10.1006/nimg.1999.0525.
Canessa, N., Crespi, C., Motterlini, M., Baud-Bovy, G., Chierchia, G., Pantaleo, G., & Cappa, S. F. (2013). The functional and structural neural basis of individual differences in loss aversion. The Journal of Neuroscience, 33(36), 14307–14317. https://doi.org/10.1523/JNEUROSCI.0497-13.2013
Canessa, N., Motterlini, M., Alemanno, F., Perani, D., & Cappa, S.F. (2011). Learning from other people’s experience: a neuroimaging study of decisional interactive-learning. Neuroimage, 55(1), 353–362. https://doi.org/10.1016/j.neuroimage.2010.11.065.
Cato, M. A., Crosson, B., Gokcay, D., Soltysik, D., Wierenga, C., Gopinath, K., & Briggs, R. W. (2004). Processing words with emotional connotation: an FMRI study of time course and laterality in rostral frontal and retrosplenial cortices. Journal of Cognitive Neuroscience, 16(2), 167–177. https://doi.org/10.1162/089892904322984481
Cavanna, A. E., & Trimble, M. R. (2006). The precuneus: a review of its functional anatomy and behavioural correlates. Brain, 129(Pt 3), 564–583. https://doi.org/10.1093/brain/awl004.
Chan, D., Anderson, V., Pijnenburg, Y., Whitwell, J., Barnes, J., Scahill, R.,.. . Fox, N. C. (2009). The clinical profile of right temporal lobe atrophy. Brain, 132(Pt 5), 1287–1298. https://doi.org/10.1093/brain/awp037.
Citron, F. M. (2012). Neural correlates of written emotion word processing: a review of recent electrophysiological and hemodynamic neuroimaging studies. Brain Lang, 122(3), 211–226. https://doi.org/10.1016/j.bandl.2011.12.007
Damasio, H., Tranel, D., Grabowski, T., Adolphs, R., & Damasio, A. (2004). Neural systems behind word and concept retrieval. Cognition, 92(1–2), 179–229. https://doi.org/10.1016/j.cognition.2002.07.001.
Desai, R. H., Reilly, M., & van Dam, W. (2018). The multifaceted abstract brain. Philos Philosophical Transactions of the Royal Society B: Biological Sciences, 373(1752). https://doi.org/10.1098/rstb.2017.0122.
Drolet, M., Schubotz, R. I., & Fischer, J. (2012). Authenticity affects the recognition of emotions in speech: behavioral and fMRI evidence. Cognitive, Affective, & Behavioral Neuroscience, 12(1), 140–150. https://doi.org/10.3758/s13415-011-0069-3
Duerden, E. G., Arsalidou, M., Lee, M., & Taylor, M. J. (2013). Lateralization of affective processing in the insula. Neuroimage, 78, 159–175. https://doi.org/10.1016/j.neuroimage.2013.04.014.
Eickhoff, S. B., Bzdok, D., Laird, A. R., Kurth, F., & Fox, P. T. (2012). Activation likelihood estimation meta-analysis revisited. Neuroimage, 59(3), 2349–2361. https://doi.org/10.1016/j.neuroimage.2011.09.017.
Eickhoff, S. B., Laird, A. R., Grefkes, C., Wang, L. E., Zilles, K., & Fox, P. T. (2009). Coordinate-based activation likelihood estimation meta-analysis of neuroimaging data: a random-effects approach based on empirical estimates of spatial uncertainty. Human Brain Mapping, 30(9), 2907–2926. https://doi.org/10.1002/hbm.20718
Eickhoff, S. B., Nichols, T. E., Laird, A. R., Hoffstaedter, F., Amunts, K., Fox, P. T.,.. . Eickhoff, C. R. (2016). Behavior, sensitivity, and power of activation likelihood estimation characterized by massive empirical simulation. Neuroimage, 137, 70–85. https://doi.org/10.1016/j.neuroimage.2016.04.072.
Ethofer, T., Anders, S., Erb, M., Herbert, C., Wiethoff, S., Kissler, J.,.. . Wildgruber, D. (2006). Cerebral pathways in processing of affective prosody: a dynamic causal modeling study. Neuroimage, 30(2), 580–587. https://doi.org/10.1016/j.neuroimage.2005.09.059.
Fales, C. L., Barch, D. M., Rundle, M. M., Mintun, M. A., Mathews, J., Snyder, A. Z., & Sheline, Y. I. (2009). Antidepressant treatment normalizes hypoactivity in dorsolateral prefrontal cortex during emotional interference processing in major depression. Journal of Affective Disorders, 112(1–3), 206–211. https://doi.org/10.1016/j.jad.2008.04.027
Fales, C. L., Barch, D. M., Rundle, M. M., Mintun, M. A., Snyder, A. Z., Cohen, J. D., & Sheline, Y. I. (2008). Altered emotional interference processing in affective and cognitive-control brain circuitry in major depression. Biological Psychiatry, 63(4), 377–384. https://doi.org/10.1016/j.biopsych.2007.06.012
Harris, L. T., McClure, S. M., van den Bos, W., Cohen, J. D., & Fiske, S. T. (2007). Regions of the MPFC differentially tuned to social and nonsocial affective evaluation. Cognitive, Affective, & Behavioral Neuroscience, 7(4), 309–316. https://doi.org/10.3758/cabn.7.4.309
Herbert, B. M., Herbert, C., & Pollatos, O. (2011a). On the relationship between interoceptive awareness and alexithymia: is interoceptive awareness related to emotional awareness? J Pers, 79(5), 1149–1175. https://doi.org/10.1111/j.1467-6494.2011.00717.x.
Herbert, C., Herbert, B. M., & Pauli, P. (2011b). Emotional self-reference: brain structures involved in the processing of words describing one’s own emotions. Neuropsychologia, 49(10), 2947–2956. https://doi.org/10.1016/j.neuropsychologia.2011.06.026.
Hiser, J., & Koenigs, M. (2018). The multifaceted role of the ventromedial prefrontal cortex in emotion, decision making, social cognition, and psychopathology. Biological Psychiatry, 83(8), 638–647. https://doi.org/10.1016/j.biopsych.2017.10.030
Holland, A. C., Addis, D. R., & Kensinger, E. A. (2011). The neural correlates of specific versus general autobiographical memory construction and elaboration. Neuropsychologia, 49(12), 3164–3177. https://doi.org/10.1016/j.neuropsychologia.2011.07.015.
Immordino-Yang, M. H., & Singh, V. (2013). Hippocampal contributions to the processing of social emotions. Human Brain Mapping, 34(4), 945–955. https://doi.org/10.1002/hbm.21485
Insel, T. R., & Fernald, R. D. (2004). How the brain processes social information: searching for the social brain. Annual Review of Neuroscience, 27, 697–722. https://doi.org/10.1146/annurev.neuro.27.070203.144148
Johnson, M. K., Raye, C. L., Mitchell, K. J., Touryan, S. R., Greene, E. J., & Nolen-Hoeksema, S. (2006). Dissociating medial frontal and posterior cingulate activity during self-reflection. Social Cognitive and Affective Neuroscience, 1(1), 56–64. https://doi.org/10.1093/scan/nsl004
Kim, M. J., Loucks, R. A., Palmer, A. L., Brown, A. C., Solomon, K. M., Marchante, A. N., & Whalen, P. J. (2011). The structural and functional connectivity of the amygdala: from normal emotion to pathological anxiety. Behavioural Brain Research, 223(2), 403–410. https://doi.org/10.1016/j.bbr.2011.04.025
Kuchinke, L., Jacobs, A. M., Grubich, C., Vo, M. L., Conrad, M., & Herrmann, M. (2005). Incidental effects of emotional valence in single word processing: an fMRI study. Neuroimage, 28(4), 1022–1032. https://doi.org/10.1016/j.neuroimage.2005.06.050.
Kumfor, F., Hazelton, J. L., De Winter, F. L., de Langavant, L. C., & Van den Stock, L. (2017). Clinical studies of social neuroscience: A lesion model approach. Neuroscience and Social Science:The Missing Link..
Lahnakoski, J. M., Glerean, E., Salmi, J., Jaaskelainen, I. P., Sams, M., Hari, R., & Nummenmaa, L. (2012). Naturalistic FMRI mapping reveals superior temporal sulcus as the hub for the distributed brain network for social perception. Frontiers in Human Neuroscience, 6, 233. https://doi.org/10.3389/fnhum.2012.00233
Laird, A. R., Fox, P. M., Price, C. J., Glahn, D. C., Uecker, A. M., Lancaster, J. L., & Fox, P. T. (2005). ALE meta-analysis: controlling the false discovery rate and performing statistical contrasts. Human Brain Mapping, 25(1), 155–164. https://doi.org/10.1002/hbm.20136
Lambon Ralph, M. A. (2014). Neurocognitive insights on conceptual knowledge and its breakdown. Philosophical Transactions of the Royal Society B: Biological Sciences, 369(1634), 20120392. https://doi.org/10.1098/rstb.2012.0392
Lewis, K., Gonzalez, M., & Kaufman, J. (2012). Social selection and peer influence in an online social network. Proceedings of the National Academy of Sciences of the United States of America, 109(1), 68–72. https://doi.org/10.1073/pnas.1109739109
Lin, N., Bi, Y., Zhao, Y., Luo, C., & Li, X. (2015). The theory-of-mind network in support of action verb comprehension: evidence from an fMRI study. Brain Lang, 141, 1–10. https://doi.org/10.1016/j.bandl.2014.11.004.
Lin, N., Xu, Y., Wang, X., Yang, H., Du, M., Hua, H., & Li, X. (2019). Coin, telephone, and handcuffs: Neural correlates of social knowledge of inanimate objects. Neuropsychologia, 133, 107187. https://doi.org/10.1016/j.neuropsychologia.2019.107187.
Lin, N., Xu, Y., Yang, H., Zhang, G., Zhang, M., Wang, S., & Li, X. (2020). Dissociating the neural correlates of the sociality and plausibility effects in simple conceptual combination. Brain Structure and Function, 225(3), 995–1008. https://doi.org/10.1007/s00429-020-02052-3
Lindquist, K. A., Satpute, A. B., Wager, T. D., Weber, J., & Barrett, L. F. (2016). The brain basis of positive and negative affect: Evidence from a meta-analysis of the human neuroimaging literature. Cerebral Cortex, 26(5), 1910–1922. https://doi.org/10.1093/cercor/bhv001
Meyer, M. L., & Lieberman, M. D. (2012). Social working memory: neurocognitive networks and directions for future research. Frontiers in Psychology, 3, 571. https://doi.org/10.3389/fpsyg.2012.00571
Mitchell, J. P., Banaji, M. R., & Macrae, C. N. (2005). The link between social cognition and self-referential thought in the medial prefrontal cortex. Journal of Cognitive Neuroscience, 17(8), 1306–1315. https://doi.org/10.1162/0898929055002418
Moll, J., de Oliveira-Souza, R., Moll, F. T., Ignacio, F. A., Bramati, I. E., Caparelli-Daquer, E. M., & Eslinger, P. J. (2005). The moral affiliations of disgust: a functional MRI study. Cognitive and Behavioral Neurology, 18(1), 68–78. https://doi.org/10.1097/01.wnn.0000152236.46475.a7
Moll, J., Zahn, R., de Oliveira-Souza, R., Krueger, F., & Grafman, J. (2005). Opinion: the neural basis of human moral cognition. Nature Reviews Neuroscience, 6(10), 799–809. https://doi.org/10.1038/nrn1768
Morawetz, C., Bode, S., Baudewig, J., & Heekeren, H. R. (2017). Effective amygdala-prefrontal connectivity predicts individual differences in successful emotion regulation. Social Cognitive and Affective Neuroscience, 12(4), 569–585. https://doi.org/10.1093/scan/nsw169
Moseley, R., Carota, F., Hauk, O., Mohr, B., & Pulvermuller, F. (2012). A role for the motor system in binding abstract emotional meaning. Cerebral Cortex, 22(7), 1634–1647. https://doi.org/10.1093/cercor/bhr238
Muller, V. I., Cieslik, E. C., Laird, A. R., Fox, P. T., Radua, J., Mataix-Cols, D., & Eickhoff, S. B. (2018). Ten simple rules for neuroimaging meta-analysis. Neuroscience & Biobehavioral Reviews, 84, 151–161. https://doi.org/10.1016/j.neubiorev.2017.11.012
Mychack, P., Kramer, J. H., Boone, K. B., & Miller, B. L. (2001). The influence of right frontotemporal dysfunction on social behavior in frontotemporal dementia. Neurology, 56(11 Suppl 4), S11-15. https://doi.org/10.1212/wnl.56.suppl_4.s11.
Nashiro, K., Sakaki, M., Nga, L., & Mather, M. (2012). Differential brain activity during emotional versus nonemotional reversal learning. Journal of Cognitive Neuroscience, 24(8), 1794–1805. https://doi.org/10.1162/jocn_a_00245
Olson, I. R., McCoy, D., Klobusicky, E., & Ross, L. A. (2013). Social cognition and the anterior temporal lobes: a review and theoretical framework. Social Cognitive and Affective Neuroscience, 8(2), 123–133. https://doi.org/10.1093/scan/nss119
Olson, I. R., Plotzker, A., & Ezzyat, Y. (2007). The Enigmatic temporal pole: a review of findings on social and emotional processing. Brain, 130(Pt 7), 1718–1731. https://doi.org/10.1093/brain/awm052.
Patterson, K., Nestor, P. J., & Rogers, T. T. (2007). Where do you know what you know? The representation of semantic knowledge in the human brain. Nature Reviews Neuroscience, 8(12), 976–987. https://doi.org/10.1038/nrn2277
Ponz, A., Montant, M., Liegeois-Chauvel, C., Silva, C., Braun, M., Jacobs, A. M., & Ziegler, J. C. (2014). Emotion processing in words: a test of the neural re-use hypothesis using surface and intracranial EEG. Social Cognitive and Affective Neuroscience, 9(5), 619–627. https://doi.org/10.1093/scan/nst034
Rice, G. E., Hoffman, P., Binney, R. J., & Lambon Ralph, M. A. (2018). Concrete versus abstract forms of social concept: an fMRI comparison of knowledge about people versus social terms. Philosophical Transactions of the Royal Society B: Biological Sciences, 373(1752). https://doi.org/10.1098/rstb.2017.0136.
Rice, G. E., Hoffman, P., & Lambon Ralph, M. A. (2015). Graded specialization within and between the anterior temporal lobes. Annals of the New York Academy of Sciences, 1359, 84–97. https://doi.org/10.1111/nyas.12951
Ross, L. A., & Olson, I. R. (2010). Social cognition and the anterior temporal lobes. Neuroimage, 49(4), 3452–3462. https://doi.org/10.1016/j.neuroimage.2009.11.012.
Roy, M., Shohamy, D., & Wager, T. D. (2012). Ventromedial prefrontal-subcortical systems and the generation of affective meaning. Trends in Cognitive Sciences, 16(3), 147–156. https://doi.org/10.1016/j.tics.2012.01.005
Sergerie, K., Chochol, C., & Armony, J. L. (2008). The role of the amygdala in emotional processing: a quantitative meta-analysis of functional neuroimaging studies. Neuroscience & Biobehavioral Reviews, 32(4), 811–830. https://doi.org/10.1016/j.neubiorev.2007.12.002
Skipper, L. M., Ross, L. A., & Olson, I. R. (2011). Sensory and semantic category subdivisions within the anterior temporal lobes. Neuropsychologia, 49(12), 3419–3429. https://doi.org/10.1016/j.neuropsychologia.2011.07.033.
Sliwa, J., & Freiwald, W. A. (2017). A dedicated network for social interaction processing in the primate brain. Science, 356(6339), 745–749. https://doi.org/10.1126/science.aam6383.
Snowden, J. S., Thompson, J. C., & Neary, D. (2004). Knowledge of famous faces and names in semantic dementia. Brain, 127(Pt 4), 860–872. https://doi.org/10.1093/brain/awh099.
Snowden, J. S., Thompson, J. C., & Neary, D. (2012). Famous people knowledge and the right and left temporal lobes. Behavioural Neurology, 25(1), 35–44. https://doi.org/10.3233/BEN-2012-0347
Taylor, K. I., Devereux, B. J., & Tyler, L. K. (2011). Conceptual structure: Towards an integrated neuro-cognitive account. Language and Cognitive Processes, 26(9), 1368–1401. https://doi.org/10.1080/01690965.2011.568227
Thompson, S. A., Graham, K. S., Williams, G., Patterson, K., Kapur, N., & Hodges, J. R. (2004). Dissociating person-specific from general semantic knowledge: roles of the left and right temporal lobes. Neuropsychologia, 42(3), 359–370. https://doi.org/10.1016/j.neuropsychologia.2003.08.004.
Tranel, D., Damasio, H., & Damasio, A. R. (1997). A neural basis for the retrieval of conceptual knowledge. Neuropsychologia, 35(10), 1319–1327. https://doi.org/10.1016/s0028-3932(97)00085-7.
Troche, J., Crutch, S., & Reilly, J. (2014). Clustering, hierarchical organization, and the topography of abstract and concrete nouns. Frontiers in Psychology, 5, 360. https://doi.org/10.3389/fpsyg.2014.00360
Trope, Y., & Liberman, N. (2010). Construal-level theory of psychological distance. Psychological Review, 117(2), 440–463. https://doi.org/10.1037/a0018963
Turkeltaub, P. E., Eden, G. F., Jones, K. M., & Zeffiro, T. A. (2002). Meta-analysis of the functional neuroanatomy of single-word reading: method and validation. Neuroimage, 16(3 Pt 1), 765–780.
Van der Cruyssen, L., Heleven, E., Ma, N., Vandekerckhove, M., & Van Overwalle, F. (2015). Distinct neural correlates of social categories and personality traits. Neuroimage, 104, 336–346. https://doi.org/10.1016/j.neuroimage.2014.09.022.
Van Overwalle, F., & Baetens, K. (2009). Understanding others’ actions and goals by mirror and mentalizing systems: a meta-analysis. Neuroimage, 48(3), 564–584. https://doi.org/10.1016/j.neuroimage.2009.06.009.
Vigliocco, G., Kousta, S. T., Della Rosa, P. A., Vinson, D. P., Tettamanti, M., Devlin, J. T., & Cappa, S. F. (2014). The neural representation of abstract words: the role of emotion. Cerebral Cortex, 24(7), 1767–1777. https://doi.org/10.1093/cercor/bht025
Visser, M., Jefferies, E., & Lambon Ralph, M. A. (2010). Semantic processing in the anterior temporal lobes: a meta-analysis of the functional neuroimaging literature. Journal of Cognitive Neuroscience, 22(6), 1083–1094. https://doi.org/10.1162/jocn.2009.21309
Wager, T. D., Phan, K. L., Liberzon, I., & Taylor, S. F. (2003). Valence, gender, and lateralization of functional brain anatomy in emotion: a meta-analysis of findings from neuroimaging. Neuroimage, 19(3), 513–531. https://doi.org/10.1016/s1053-8119(03)00078-8.
Walter, H., Adenzato, M., Ciaramidaro, A., Enrici, I., Pia, L., & Bara, B. G. (2004). Understanding intentions in social interaction: the role of the anterior paracingulate cortex. Journal of Cognitive Neuroscience, 16(10), 1854–1863. https://doi.org/10.1162/0898929042947838
Wang, X., Wang, B., & Bi, Y. (2019). Close yet independent: Dissociation of social from valence and abstract semantic dimensions in the left anterior temporal lobe. Human Brain Mapping, 40(16), 4759–4776. https://doi.org/10.1002/hbm.24735
Wang, Y., Collins, J. A., Koski, J., Nugiel, T., Metoki, A., & Olson, I. R. (2017). Dynamic neural architecture for social knowledge retrieval. Proceedings of the National Academy of Sciences of the United States of America, 114(16), E3305–E3314. https://doi.org/10.1073/pnas.1621234114
Yeo, B. T., Krienen, F. M., Sepulcre, J., Sabuncu, M. R., Lashkari, D., Hollinshead, M., & Buckner, R. L. (2011). The organization of the human cerebral cortex estimated by intrinsic functional connectivity. Journal of Neurophysiology, 106(3), 1125–1165. https://doi.org/10.1152/jn.00338.2011
Zahn, R., de Oliveira-Souza, R., & Moll, J. (2020). Moral motivation and the basal forebrain. Neuroscience & Biobehavioral Reviews, (108), 207–217.
Zahn, R., Moll, J., Krueger, F., Huey, E. D., Garrido, G., & Grafman, J. (2007). Social concepts are represented in the superior anterior temporal cortex. Proceedings of the National Academy of Sciences of the United States of America, 104(15), 6430–6435. https://doi.org/10.1073/pnas.0607061104
Funding
This work was supported by the University School for Advanced Studies IUSS (Pavia) and by ICS Maugeri IRCCS (Pavia). The funding sources had no role in any of the research stages.
Author information
Authors and Affiliations
Contributions
Author contributions included conception and study design (all authors), data collection (MA), statistical analysis (MA), interpretation of results (all authors), drafting the manuscript work (MA, NC) or revising it critically for important intellectual content (all authors), and approval of final version to be published and agreement to be accountable for the integrity and accuracy of all aspects of the work (all authors).
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have a conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 78.4 KB)
Rights and permissions
About this article
Cite this article
Arioli, M., Gianelli, C. & Canessa, N. Neural representation of social concepts: a coordinate-based meta-analysis of fMRI studies. Brain Imaging and Behavior 15, 1912–1921 (2021). https://doi.org/10.1007/s11682-020-00384-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-020-00384-6