Abstract
Litterfall, which is influenced by physical and biological factors, is a major pathway for carbon and nutrient cycling in forest ecosystems. The purpose of this study was to investigate monthly litterfall production in three forests in Jeju Island differentiated by forest composition and precipitation: Cheongsu (Quercus glauca as the dominant species; low precipitation), Seonheulb (Q. glauca as the dominant species; high precipitation), and Seonheulm (Q. glauca and Pinus thunbergii as the dominant species; high precipitation). Litterfall was collected monthly from April to December 2015 and divided into leaf litter, twig, bark, seeds, and unidentified materials. Seasonal patterns of litterfall production varied across stands according to their species composition. However, the amount of leaf litterfall and total litterfall were comparable among stands, ranging from 362 to 375 g m−2 for leaf litter and 524 g m−2 to 580 g m−2 for total litterfall. Oak leaf litter in May was the highest in all stands, while needle litter was the highest in December in Seonheulm. High twig litterfall in July may be attributable to high rainfall with strong winds and storms during the rainy season. Although forest type and climate factor had no influence on litterfall amounts in this study, the pattern of litterfall production was species dependent, suggesting diverse effects on carbon and nutrient cycling in these forests.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forest ecosystems play an important role in global carbon cycling as atmospheric CO2 and climate continuously change (Pan et al. 2011). A large part of the net primary productivity (NPP) of forests is directly or indirectly contributed by leaves that generally accumulate as litterfall on the forest floor. This litterfall is an important pathway of carbon and nutrient cycling in forest ecosystems (Bray and Gorham 1964; Berg and Laskowski 2005; Kim et al. 2005; Kang et al. 2010).
Litter acts as an input–output system for nutrients on the forest floor and maintains soil fertility in forest ecosystems through this nutrient cycling (Olson 1963; Singh et al. 1999; Fioretto et al. 2003; Onyekwelu et al. 2006; Pandey et al. 2007). However, it is not easy to determine the amount and pattern of litterfall in forest ecosystems (Zhou et al. 2007) because of the many influential factors (e.g., temperature, precipitation, site conditions, time, and forest types).
Litterfall production depends on species composition as well as forest type. Zhou et al. (2007) found the lowest litterfall in a pine forest at 356 g m−2 and the highest litterfall in a ravine rain forest at 1061 g m−2 along successional and altitudinal gradients of subtropical monsoon evergreen broadleaved forests in China in an 11-year study. They also reported that litterfall production was highest from April to September, the summer season. Bray and Gorham (1964) reported that evergreen coniferous forests produced higher leaf litter (by 8%) and total litterfall (by 17%) than deciduous broadleaves forests did. Similarly, An et al. (2017) studied five deciduous broadleaved forests and four evergreen coniferous forests at the Gwangneung Experimental Forest in South Korea and reported 556 g m−2 litterfall for coniferous forests, higher than the 436 g m−2 litterfall for deciduous forests. In India, Sundarapandian and Swamy (1999) also reported variation in the mean annual litterfall (ranging from 563 to 865 g m−2) across sites attributable to species composition. However, Yanai et al. (2012) observed that litterfall varied considerably year to year rather than across 13 successional northern hardwood forests in USA.
Litterfall production is also influenced by climate factors in forests, but studies have shown diverse results. Litterfall production peaks in the dry season in a tropical forest in India (Sundarapandian and Swamy 1999). However, Guadalupe and Tolome (1996) reported that litterfall production is related to phytogeographic origin in a lower montane forest in Mexico: Holarctic species showed a strong negative correlation with precipitation and minimum temperatures, but tropical species showed a significant positive correlation with maximum temperature. Liu et al. (2004) determined the relationships between climatic factors and litterfall in Eurasian forests at a continental scale and found that a relative unit change in temperature (annual mean temperature change from − 8 to 30 °C) had more effect on litterfall than that in precipitation (annual mean precipitation change from 350 to 4000 mm). Kouki and Hokkanen (1992) studied needle litterfall in a Scots pine (Pinus sylvestris L.) stand in Finland for over 24 years and found that litterfall amount varied from year to year ranging from 18 to 213 g m−2 because of July temperature: a 1 °C increase in July temperature increased needle litterfall by 36 g m−2.
Jeju Island, formed by four volcanic events, is located in the Korea Strait in the southernmost part of South Korea, and approximately 48% of the island is covered with forests, of which 34% are broadleaved forests (Korea Forest Service 2015). The Gotjawal Forest is one of the most important forest types in Jeju Island referring to a unique natural unmanaged forest with dense trees and understory shrub vegetation formed on widely and disorderly distributed lava blocks (Park 2010). Thus, this unique ecosystem makes highly diverse microclimate with severe topographic heterogeneity (Choi and Lee 2015). Recently, the Gotjawal Forest has received more attention because it plays an important role in the maintenance of ecosystem services (Jeon et al. 2012; Jeong 2012; Kang et al. 2013). However, relatively little is known about litterfall production and dynamics in this area.
The main purpose of our study was to investigate the monthly litterfall production in three forests in relation to climatic factors. We hypothesized that (1) litterfall production varies according to forest types and time, and (2) the pattern of seasonal litterfall production differs by forest types and climatic factors.
Materials and methods
Study sites and stand descriptions
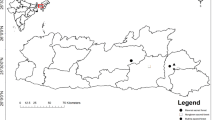
The experiment was conducted in 2015 in three forest stands at two sites in Jeju Island, Republic of Korea: Cheongsu Gotjawal (33°18′14.79′′N and 126°16′16.67′′E, about 120 m a.s.l.) and Seonheul Gotjawal (33°30′38.82′′N and 126°43′13.25′′E, about 110 m a.s.l.). Cheongsu Gotjawal (hearafter, Cheongsu) is distributed on lava plateau in the western part of Jeju Island, having locally distributed water holes on gentle slopes. Seonheul Gotjawal (hearafter, Seonheul) is located in the northeastern part of the island characterized by small lava caves, wetlands and a parasitic volcano terrain (Jeon et al. 2012).
Monthly temperature and precipitation data were provided by the meteorological center near each study site: Seongsan Station (33°23′N and 126°53′E, 20 m a.s.l.), 17 km from Cheongsu; Gosan Station (33°18′N and 126°10′E, 71 m a.s.l.), 9 km from Seonheul. Mean annual temperature did not differ between Cheongsu (15.6 °C) and Seonheul (15.7 °C); however, annual precipitation was about 61% higher at Seonheul (2496 mm) than Cheongsu (1546 mm). The temperature was the highest in August and the lowest in January; the winter temperature was mostly above 5 °C for the three stands (Fig. 1). The precipitation was greatest in July for Seonheul and in May for Cheongsu and lowest in October for both sites (Fig. 2). Monthly precipitation was always 1.2–2.8 times higher at Seonheul than Cheongsu except in June.
The stand in Cheongsu is an evergreen broadleaf forest dominated by Quercus glauca with 5.3 m2 ha−1 in mean basal area and 9.5 m in height (Table 1). Other canopy and subcanopy trees such as Actinodaphne lancifolia, Ficus erecta, Cinnamomum camphora, Cinnamomum yabunikkei, Celtis sinensis, Acer palmatum and Picrasma quassioides account for 34% of total basal area. As mentioned, annual mean temperature was similar among the stands, but the annual precipitation was the lowest in Cheongsu (Figs. 1, 2). The stand in Seonheul is also an evergreen broadleaf forest dominated by Q. glauca with 12.5 m in tree height, but the mean basal area and precipitation are higher than those in Cheongsu (hereafter, Seonheulb; Table 1, Fig. 2). Other canopy and subcanopy tree species such as Quercus salicina, Prunus pendula, and Styrax japonica, account for 15% of total basal area in Seonheulb. The other stand in Seonheul Gotjawal is a mixed evergreen forest dominated by Q. glauca and Pinus thunbergii with mean basal area and precipitation similar to those in Seonheulb (hereafter, Seonheulm; Table 1, Fig. 2). In Seonheulm, canopy tree height was 10.8 m for Q. glauca and 13.1 m for P. thunbergii. Here, subcanopy species such as Camellia japonica, Distylium racemosum, and Acer palmatum, etc. accounted for 8% of total basal area, and tree height ranged from 3.0 to 6.4 m.
Litterfall collection
Litterfall was collected using a circular plastic litter trap 67 cm in diameter and installed 30 cm from the ground in March 2015. To minimize the loss of litter from the litter traps, we installed an iron mesh (mesh size: 2 mm) at the bottom of the litter trap. Rocks were placed in each trap to prevent overflow and loss of litter.
Each stand was divided into four plots with four litter traps in each plot. The distance between plots was at least 20 m, and the four litter traps within each plot were spaced 12 m apart in the shape of a quadrangle. Thus, 48 litter traps were installed for the three stands.
Litterfall was collected monthly from April to December 2015. The collected litter was dried immediately at room temperature to prevent decomposition. Air-dried litterfall was separated into leaves, twigs, bark, seeds, and unidentified materials. Particularly, leaf litter was further classified into evergreen oak (Q. glauca, Q. gilva, and Q. saliciana), P. thunbergii, and others. The separated samples were oven-dried at 65 °C for 48 h to a constant mass and then weighed. Annual litterfall production was estimated by summing the monthly litterfall mass during the study.
Statistical analyses
Two-way analysis of variance (ANOVA) was performed to detect the effects of stands and time on leaf litter, other litterfall, and total litterfall production. When there was no interaction, one-way ANOVA was performed to test for significant differences in litterfall production. Multiple comparisons of means were completed using Duncan’s multiple range test at α = 0.05. All statistical analyses were performed using SAS 9.4 software (SAS Institute, Cary, NC, USA).
Results
Annual litterfall variation by stand
Total litterfall was not significantly different across three forests (Table 2) and the value was 524 g m−2 in Cheongsu, 580 g m−2 in Seonheulb, and 531 g m−2 in Seonheulm. However, the composition of litterfall showed different patterns by stand (Figs. 3, 4). Leaf litterfall of all tree species accounted for 86% on average of total litterfall (Fig. 5). As expected by species composition in basal area (Table 1), oak leaf litter was higher in Cheongsu and Seonheulb than in Seonheulm (p < 0.01), and needle litter was much higher in Seonheulm than in Cheongsu and Seonheulb (p < 0.01). The litter of other leaves was higher in Cheongsu and Seonheulb than in Seonheulm by 3.8 times (p < 0.01). Twig litter was the highest in Seonheulb and seed litter was the lowest in Cheongsu. Even though leaf litter by species varied by stand, total leaf litter was similar among the stands (p = 0.95) ranging from 362 to 375 g m−2 (Fig. 5).
Monthly litterfall variation
Litterfall production varied across months from May to December (Figs. 3, 4). Oak leaf litter was the highest in May, which was 99 g m−2 in Cheongsu, 144 g m−2 in Seonheulb, and 60 g m−2 in Seonheulm, but it was very low during the other months, ranging from 9.3 to 14.7 g m−2. On the other hand, needle litter in Seonheulm was the highest in December followed by November (Fig. 3). Although we did not classify flower litter separately, most of the unknown materials in May consisted of flower litter. There was a significant effect of season on twig and seed litter production (Table 2). Twig litter production was the highest in July, while seed litter production was higher in October and November than in other months in all stands. Unidentified materials were the highest in May.
The proportion of leaf litter in May was the highest, but the proportion varied significantly by stand: 33% in Cheongsu, 35% in Seonheulb, and 21% in Seonheulm. The proportion of leaf litter in December was 50% lower in Cheongsu and Seonheulb than in Seonheulm (Fig. 5).
Discussion
Leaf litter and total litterfall were comparable to those observed in the same subtropical forest zone in Korea from 2009 to 2012 (Jung 2015). The average leaf litter across three stands in this study was 369 g m−2, which was somewhat lower than that for broadleaf forests, 424 g m−2 (from 84 to 910 g m−2), but a little higher than that for needle leaf forests, 331 g m−2 (from 94 to 704 g m−2) in Asian subtropical zones (Liu et al. 2004). The total litterfall was 545 g m−2 on average within the range of the reported values for broadleaf and needle leaf forests in Korea (An et al. 2017).
Leaf litter and total litterfall were similar among three different forests even though the basal area in Cheongsu was only 56% of that in Seonheulb and Seonheulm. This result did not correspond with the results of other studies. For example, Starr et al. (2005) reported that both needle leaf litter and total leaf litter were significantly correlated with the basal area of Scots pine stands in Finland. Kunhamu et al. (2009) reported that basal area has a linear relationship with litterfall production in Acacia mangium stands subjected to various thinning intensities. However, litterfall mass differed according to the age and stand basal area of the forest in a northern hardwood forest in the United States; after a certain age (approximately 30 years), the amount of litterfall tended to be constant without a large increase (Yanai et al. 2006, 2012; Park et al. 2007). Comparable leaf litter and total litterfall among stands in this study may be attributed to canopy closure; that is, the crown was already closed in all three stands so that annual partitioning of carbon was greater to the bole or roots rather than to the leaves.
Liu et al. (2004) explored the litterfall data for broadleaf and coniferous forests in Eurasia to examine the relationship between climatic factors and litterfall. They found that the relationship between litterfall and climatic factors has significantly higher coefficients in broadleaf stands than in coniferous forests. They concluded that temperature change had a greater effect on litterfall than precipitation change did across the Eurasian forests. In contrast, we found no differences in leaf litter and total litterfall between Cheongsu and Seonheul forests even though the precipitation in Cheongsu was 40% lower than in Seonheulb and Seonheulm (Table 2, Fig. 2). It might be attributable to increased carbon allocation to the bole or coarse roots rather than to the leaf or fine root after crown closure as mentioned above. Another reason might be no water shortage in all stands: the annual precipitation of 1546 mm in Cheongsu during the study would provide enough water for tree growth in this area. One more possible explanation can be raised that precipitation or water availability is not a limiting factor for leaf production in all stands because trees adapted to rocky areas with high rainfall percolation.
In this study, the variation in litter production was more prominent for seasonal pattern. The timing of leaf fall varies depending on tree species, and this difference may be attributable to plant life histories or micro-environmental factors (Boojh and Ramakrishnan 1981; Sundriyal 1990; Kikim and Yadava 2001; Kikuzawa and Lechowicz 2011). As leaves get older, net primary production of trees declines because of decreased photosynthetic capacity and increased respiration cost in leaves (Reich 1995; Westoby et al. 2002; Kikuzawa 2003; Miyazawa and Kikuzawa 2004). Therefore, leaf shedding is cost effective to maintain the aged leaves and dry matter production. In the present study, total litterfall peaked in May for all stands, but the composition of litterfall varied between stands. The highest oak leaf litter was observed in May in all stands, and this pattern was comparable to a previous study in a similar climatic zone in northeast Spain, where evergreen oak leaf litter was the highest during May and June (Caritat et al. 2006). According to Ralhan et al. (1985), leaf drop for most evergreen broadleaved species is concentrated leaf drop in spring and summer, but leaves are not all shed until new leaves are fully expanded; old leaves of Quercus spp. start turning yellow and falling soon after appearing young shoots. Nitta and Ohsawa (1997) also reported peak leaf fall in May to June in a study of leaf dynamics in 11 warm-temperate evergreen broadleaved trees in Japan. In spring when the weather starts to warm, longer daylight time and increased soil water availability, the plants seem to prepare for the most favorable growth by replacing a majority of old leaves with new leaves early in the season, which in turn maximizes carbon gain (Ralhan et al. 1985; Devi and Garkoti 2013). On the other hand, in our study, pine needle litter peaked during fall and winter in November and December as found by Liu et al. (2001) who reported that pine litterfall peaked in December in North China. Also, needle litter production in P. sylvestris forests peaks in October in the northeastern region of the Iberian Peninsula (Pausas 1997), needle litterfall rate of P. densiflora is the highest between October and December in Japan (Miura 2000), and needle litterfall production reaches its maximum values between October and November in Korea (Kim et al. 2009). Compared with other studies on pine needle litterfall, we found slightly delayed needle litter production from late fall to winter, with the highest production in December. In the study area, monthly temperature decreased greatly from 14.6 °C in November to 9.3 °C in December, triggering high leaf drop (Bhat 1992). The oak trees, Q. glauca in this study, generally have a flowering peak in April, the average duration of flowering continues about 4 weeks; thereafter, flowers drop as litterfall (Ralhan et al. 1985). Likewise, we found a high proportion of flower litter in May in all stands although we included flower litter as unidentified materials. Seed production was much higher in November as a result of acorn production. The production of twig litter peaked in summer from the combined effects of rainfall, strong winds and storms (Caritat et al. 2006). Environmental factors such as strong winds and storms may have distinct effects on twig and branch litterfall (Christensen 1975; Park et al. 2008).
The different timing of litterfall production may affect the decomposition process on the forest floor. According to previous studies, the decomposition rate of oak leaf litter is much higher in summer due to high rainfall, air temperature, and relative humidity (Pandey et al. 2007). On the other hand, needle litter decomposition is much slower during winter because of low soil moisture and temperature (Tripathi and Singh 1992).
Conclusions
Litterfall production was similar among stands even though there were remarkable differences in basal area and annual precipitation between stands. However, the litterfall composition differed distinctly according to time and forest structure. The differences in average mean diameter at breast height after crown closure or climatic factors such as precipitation had no effect on the litterfall production. However, the genetic and ecological characteristics of tree species such as evergreen coniferous versus evergreen broadleaf species are considered to influence on the fluctuation in litterfall production. The difference in seasonal leaf and litterfall production among stands will also affect the decomposition process of litter, resulting in varying effects on carbon and nutrient cycling in these forests.
References
An JY, Park BB, Chun JH, Osawa A (2017) Litterfall production and fine root dynamics in cool-temperate forests. PLoS ONE 12:e0180126. https://doi.org/10.1371/journal.pone.0180126
Berg B, Laskowski R (2005) Litter Fall. In: Cragg JB (ed) Advances in ecological research. Litter decomposition: a guide to carbon and nutrient turnover, vol 38. Elsevier Academic Press, San Diego, pp 19–71
Bhat DM (1992) Phenology of tree species of tropical moist forest of Uttara Kannada district, Karnataka, India. J Biosci 17(3):325–352
Boojh R, Ramakrishnan PS (1981) Phenology of trees in a sub-tropical evergreen montain forest in north-east India. Geol Ecol Trop 5:189–209
Bray JR, Gorham E (1964) Litter production in forests of the world. In: Cragg JB (ed) Advances in ecological research, vol 2. Academic Press, Cambridge, pp 101–157
Caritat A, García-Berthou E, Lapeña R, Vilar L (2006) Litter production in a Quercus suber forest of Montseny (NE Spain) and its relationship to meteorological conditions. Ann For Sci 63:791–800. https://doi.org/10.1051/forest:2006061
Choi BK, Lee CB (2015) A study on the synecological values of the Torreya nucifera forest (Natural Monument No. 374) at Pyeongdae-ri in Jeju Island. J Korean Inst Tradit Landsc Archit 33:87–98
Christensen O (1975) Wood litter fall in relation to abscission, environmental factors, and the decomposition cycle in a Danish oak forest. Oikos 26:187–195
Devi AF, Garkoti SC (2013) Variation in evergreen and deciduous species leaf phenology in Assam, India. Trees 27:985–997. https://doi.org/10.1007/s00468-013-0850-8
Fioretto A, Papa S, Fuggi A (2003) Litter-fall and litter decomposition in a low Mediterranean shrubland. Biol Fertil Soils 39:37–44. https://doi.org/10.1007/s00374-003-0675-5
Guadalupe WL, Tolome J (1996) Litterfall, temperate and tropical dominant trees, and climate in a Mexican lower montane forest. Biotropica 28:649–656
Jeon YM, Ahn US, Ryu CG, Kang SS, Song ST (2012) A review of geological characteristics of Gotjawal terrain in Jeju Island: preliminary study. J Geol Soc Korea 48:425–434
Jeong KJ (2012) A study on perception and use of Gotjawal in Jeju Island. J Photo-Geogr 22:11–28
Jung HM (2015) Carbon distribution and budget of Quercus glauca community in warm-temperate forest and Abies koreana community in sub-alpine forest in Jeju island. Kongju National University, Gongju
Kang KN, Park GS, Lee SJ, Lee HG, Kim JS, Kim YT (2010) Carbon storages in aboveground and root of Pinus koraiensis and Larix leptolepis stands in Gongju, Chungnam province. Korean J Agric Sci 37:45–52
Kang HG, Kim CS, Kim ES (2013) Human influence, regeneration, and conservation of the Gotjawal forests in Jeju Island, Korea. J Mar Island Cult 2:85–92. https://doi.org/10.1016/J.IMIC.2013.11.003
Kikim A, Yadava PS (2001) Phenology of tree species in subtropical forests of Manipur in north eastern India. Trop Ecol 3:105–110
Kikuzawa K (2003) Phenological and morphological adaptations to the light environment in two woody and two herbaceous plant species. Funct Ecol 17:29–38. https://doi.org/10.1046/j.1365-2435.2003.00707.x
Kikuzawa K, Lechowicz MJ (2011) Ecology of leaf longevity, 1st edn. Springer Japan, Tokyo
Kim CS, Koo KS, Byung JK (2005) Litterfall and nutrient dynamics in pine (Pinus rigida) and larch (Larix leptolepis) plantations. J Korean For Soc 94:302–306
Kim C, An HC, Cho HS, Choo GC (2009) A comparison of litterfall dynamics in three coniferous plantations of identical age under similar site conditions. J Ecol Field Biol 32:97–102
Korea Forest Service (2015) Statistical yearbook of forestry. Korea Forest Science, Seoul, p 440
Kouki J, Hokkanen T (1992) Long-term needle litterfall of a Scots pine Pinus sylvestris stand: relation to temperature factors. Oecologia 89:176–181
Kunhamu TK, Kumar BM, Viswanath S (2009) Does thinning affect litterfall, litter decomposition, and associated nutrient release in Acacia mangium stands of Kerala in peninsular India? Can J For Res 39:792–801. https://doi.org/10.1139/X09-008
Liu CJ, Westman CJ, Ilvesniemi H (2001) Matter and nutrient dynamics of pine (Pinus tabulaeformis) and oak (Quercus variabilis) litter in north China. Silva Fenn 35:3–13. https://doi.org/10.14214/sf.599
Liu CJ, Westman CJ, Berg B, Kutsch W, Wang GZ, Man R, Ilvesniemi H (2004) Variation in litterfall-climate relationships between coniferous and broadleaf forests in Eurasia. Glob Ecol Biogeogr 13:105–114. https://doi.org/10.1111/j.1466-882X.2004.00072.x
Miura S (2000) Proposal for a new definition to evaluate the status of forest floor cover and floor cover percentage (FCP) from the viewpoint of the protection against raindrop splash. J Jpn Soc 82:132–140
Miyazawa Y, Kikuzawa K (2004) Phenology and photosynthetic traits of short shoots and long shoots in Betula grossa. Tree Physiol 24:631–637
Nitta I, Ohsawa M (1997) Leaf dynamics and shoot phenology of eleven warm-temperate evergreen broad-leaved trees near their northern limit in central Japan. Plant Ecol 130:71–88. https://doi.org/10.1023/A:1009735709258
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331. https://doi.org/10.2307/1932179
Onyekwelu JC, Mosandl R, Stimm B (2006) Productivity, site evaluation and state of nutrition of Gmelina arborea plantations in Oluwa and Omo forest reserves, Nigeria. For Ecol Manag 229:214–227. https://doi.org/10.1016/J.FORECO.2006.04.002
Pan YD, Birdsey RA, Fang J, Houghton R, Kauppi PE, Kurz WA, Phillips OL, Shvidenko A, Lewis SL, Canadell JG, Ciais P, Jackson RB, Pacala SW, McGuire AD, Piao SL, Rautiainen A, Sitch S, Hayes D (2011) A large and persistent carbon sink in the world’s forests. Science 333:988–993. https://doi.org/10.1126/science.1201609
Pandey RR, Sharma G, Tripathi SK, Singh AK (2007) Litterfall, litter decomposition and nutrient dynamics in a subtropical natural oak forest and managed plantation in northeastern India. For Ecol Manag 240:96–104. https://doi.org/10.1016/J.FORECO.2006.12.013
Park JB (2010) Characterization of lava-formed petrology and petrochemistry. Institute of Environmental Resource Research, Jeju Special Self-Governing Province, Jeju
Park BB, Yanai RD, Vadeboncoeur MA, Hamburg SP (2007) Estimating root biomass in rocky soils using pits, cores, and allometric equations. Soil Sci Soc Am J 71:206–213. https://doi.org/10.2136/sssaj2005.0329
Park J, Kim C, Jeong J, Byun JK, Son Y, Yi MJ (2008) Effect of fertilization on litterfall amounts in a Quercus acutissima stand. J Korean For Soc 97:582–588
Pausas JG (1997) Litter fall and litter decomposition in Pinus sylvestris forests of the eastern Pyrenees. J Veg Sci 8:643–650
Ralhan PK, Khanna RK, Singh SP, Singh JS (1985) Phenological characteristics of the tree layer of Kumaun Himalayan forests. Vegetation 60:91–101. https://doi.org/10.1007/BF00040351
Reich PB (1995) Phenology of tropical forests: patterns, causes, and consequences. Can J Bot 73:164–174
Singh KP, Singh PK, Tripathi SK (1999) Litterfall, litter decomposition and nutrient release patterns in four native tree species raised on coal mine spoil at Singrauli, India. Biol Fertil Soils 29:371–378
Starr M, Saarsalmi A, Hokkanen T, Meriaux P, Helmisaari H-S (2005) Models of litterfall production for Scots pine (Pinus sylvestris L.) in Finland using stand, site and climate factors. For Ecol Manag 205:215–225
Sundarapandian SM, Swamy PS (1999) Litter production and leaf-litter decomposition of selected trees species in tropical forests at Kodayar in the Western Ghats, India. For Ecol Manag 123:321–344
Sundriyal RC (1990) Phenology of some temperate woody species of the Garhwal Himalaya. Int J Ecol Environ Sci 6:107–117
Tripathi SK, Singh KP (1992) Nutrient immobilization and release patterns during plant decomposition in a dry tropical bamboo savana, India. Biol Fertil Soils 14:191–199
Westoby M, Falster DS, Moles AT, Vesk PA, Wright IJ (2002) Plant ecological strategies: some leading dimensions of variation between species. Annu Rev Ecol Syst 33:125–159
Yanai RD, Park BB, Hamburg SP (2006) The vertical and horizontal distribution of roots in northern hardwood stands of varying age. Can J For Res 36:450–459. https://doi.org/10.1139/x05-254
Yanai RD, Arthur MA, Acker M, Levine CR, Park BB (2012) Variation in mass and nutrient concentration of leaf litter across years and sites in a northern hardwood forest. Can J For Res 42:1597–1610. https://doi.org/10.1139/x2012-084
Zhou GY, Guan LL, Wei XH, Zhang DQ, Zhang QM, Yan JH, Wen DZ, Liu JX, Liu SG, Huang ZL, Kong GH, Mo JM, Yu QF (2007) Litterfall production along successional and altitudinal gradients of subtropical monsoon evergreen broadleaved forests in Guangdong, China. Plant Ecol 188:77–89
Acknowledgements
Field collection and sorting litters were accomplished by Loth Meng and Ji Hye Song.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2018R1D1A3B07045079).
The online version is available at http://www.springerlink.com
Corresponding editor: Tao Xu.
Rights and permissions
About this article
Cite this article
An, J.Y., Han, S.H., Youn, W.B. et al. Comparison of litterfall production in three forest types in Jeju Island, South Korea. J. For. Res. 31, 945–952 (2020). https://doi.org/10.1007/s11676-019-00929-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-019-00929-6