Abstract
Pinus yunnanensis Franch. is a major component of coniferous forests in southwestern China. Little is known about its intraspecific variation. Morphological variations in needle and cone traits of P. yunnanensis were analyzed to quantify variability among and within natural populations. Seven traits were measured on 10 needles collected from 30 trees in each of the 18 sampled populations of P. yunnanensis. Four cone traits were measured in 221 individual trees from nine populations. The results showed that there were significant differences (p < 0.01) both among populations and within populations in each needle and cone trait. The proportion of phenotypic variation of nearly all needle and cone traits was over 50 % within populations, which showed trees within populations accounted for a majority of the total variation. The needle traits showed higher variability within population than cone traits. Variability in the needle traits was correlated with geo-climatic parameters (longitude, latitude, altitude, temperature, and precipitation). Needle length and the ratio of needle length to fascicle sheath length showed clinal variation in response to latitudinal and altitudinal gradients. A hierarchical classification of all populations based on needle traits led to the formation of four major groups. The findings provide important genetic information for the evaluation of variation. Moreover, it will assist in management of genetic diversity of P. yunnanensis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pinus yunnanensis Franch. is extensively used in reforestation efforts in Southwest China. Its range extends from 23° to 30°N and 96° to 108°E and it is a major component of coniferous forests in this region (Wu 1986; Chinese Academy of Sciences 1978; Jin and Peng 2004). It grows at altitudes ranging from 250 to 3500 m and in a variety of geological areas (Jin and Peng 2004). P. yunnanensis is shade-intolerant, deep-rooted, drought-resistant and tolerant of rocky soils with low fertility. It is a pioneer tree species that may be used for afforestation on barren hills (Chinese Academy of Sciences 1978). Its timber is used extensively for a variety of purposes. It plays a crucial role in regional economic development and ecological restoration (Wu 1986; Jin and Peng 2004). In southwest China, it occupies approximately 52 % of the forested area and produces 32 % of the timber volume (Jin and Peng 2004). Unfortunately, human overexploitation and natural disasters, such as fire and drought, have seriously damaged the P. yunnanensis resource.
Conservation and sustainable use of genetic resources depend upon knowledge of the extent and pattern of intra-specific variation (Loha et al. 2006). Characterization of natural forest resources is the necessary first step toward a better understanding of their genetics and implementation of in situ and ex situ conservation activities (Gil et al. 2002; Turna et al. 2006; Turna and Güney 2009). Sevık et al. (2010) emphasized the importance of characterizing similarities or differences among populations as an important stage of breeding. P. yunnanensis inhabits an extremely variable geographical range that encompasses great variation in geo-climatic conditions (Li 1984), possibly indicating the existence of ecotypes adapted to different environments (Wang et al. 2003, 2004; Ji et al. 2011). It has been demonstrated that the morphology and anatomy of leaves reflect adaptation to the habitat from which plants originate (Pham et al. 2010; Ali et al. 2011). Thus, variability in needle and cone morphology can be an important indicator of ecological specialization (Donahue and Upton 1996; Boulli et al. 2001; Dangasuk and Panetsos 2004; Wahid et al. 2006).
Genetic improvement of P. yunnanensis started in the 1980s. It was focused on identifying phenotypically superior trees from diverse regions and deploying them in seed orchards (Li et al. 2010). The genetic variation of P. yunnanensis has been characterized primarily in a few, small provenance tests that sampled a limited part of the species’ range (Li 1984; Chen and Wu 1987). There are few descriptions of the intra-specific variability of P. yunnanensis, but they have shown that morphological traits varied significantly among populations or provenances (Wang et al. 2003, 2004).
The objectives of this study were to: ascertain variability among and within populations of P. yunnanensis based on a range-wide sample of populations, evaluate population differentiation based on the phenotypic variation in needle and cone traits, determine the proportion of variation that exists among and within populations, and explore correlations between phenotypic traits and geographic or climatic factors.
Materials and methods
Sampling
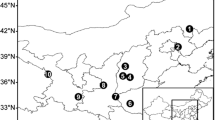
Fresh, mature needle samples were harvested from 18 widely separated populations covering almost all the P. yunnanensis range (Fig. 1; Table 1). Sampled trees (30 to 34 per population) were located at least 100 m apart in each population. All sampled trees were more than 25 years old (Dangasuk and Panetsos 2004). Needle traits were analyzed from 30 mother trees in each population. Cones were sampled from 221 individual trees in nine populations. Sample size for cone traits varied from 12 to 34 mother trees in each population (Table 1). Two-year-old needles were sampled randomly from three to five vigorous branches from each sampled tree. The needles and cones were collected from all sides of the canopy, from the upper and middle portion of the crown, and were immediately placed in individually labeled bags. The methods for sampling and trait measurement were derived from previously published studies of congeneric species, such as Scots pine (Bobowicz and Radziejewska 1989; Urbaniak et al. 2003; Boratyńska et al. 2008), slash and longleaf pines (De Vall 1940), and peat-bog pine (P. uliginosa) (Boratyńska et al. 2008).
Measurements
Ten fascicles were selected randomly from each sampled tree for the measurement of fascicle sheath length (FSL) and fascicle width (FW), and ten needles (one from each fascicle) were measured for needle length (NL) and needle width (NW) (Panetsos 1975; Urbaniak et al. 2003; Boratyńska et al. 2008). NL and FSL were measured with a metric ruler with an accuracy of 0.1 cm; NW and fascicle width (FW) were measured using digital vernier calipers with an accuracy of 0.1 mm. The ratio of length to width of needle (NL/NW), the ratio of needle length to fascicle sheath length (NL/FSL) and the ratio of fascicle width to needle width (FW/NW) were calculated separately. The mature cones (two to six per sampled tree) were harvested, weighed, and measured for length and width (Table 1). Cone length (CL) was measured in a straight line from the base to the top, cone curvature excluded, and cone diameter (across the broadest portion) (Donahue and Upton 1996) with digital vernier calipers, and then the ratio of cone length to cone diameter (CL/CD) was calculated. Cone weight (CW) was quantified with digital electronic balance.
Data analysis
The components of variance were determined from within populations and among populations to determine the percentage of variability due to these sources. Data from the measurement of needle and cone traits were subjected to analysis of variance (ANOVA, PROC MIXED) using a nested procedure (needles or cones in trees and trees in populations) (version 9.3, SAS Institute Inc., Cary, NC, USA). For all traits, comparison of means was conducted with Tukey’s multiple comparison test at 95 % confidence intervals to determine the differences among populations. The variability within populations was described using the coefficient of variation (CV) (López et al. 2013). The CVs were obtained from population means and standard deviations. The morphological divergence between natural population (Raeymaekers et al. 2007) or phenotypic differentiation (Leinonen et al. 2006) was assessed by calculating the P ST index; analogous to Q ST (Spitze 1993), P ST index was calculated using the formula \( P_{ST} = {{\sigma_{GB}^{2} } \mathord{\left/ {\vphantom {{\sigma_{GB}^{2} } {\left( {\sigma_{GB}^{2} + 2\sigma_{GW}^{2} } \right),}}} \right. \kern-0pt} {\left( {\sigma_{GB}^{2} + 2\sigma_{GW}^{2} } \right),}} \) where \( \sigma_{GB}^{2} \) and \( \sigma_{GW}^{2} \) are the among-population and within population variance components for a phenotypic trait, respectively (Raeymaekers et al. 2007).
The climatic data—that is the annual mean temperature (Tmean), maximum temperature of the warmest month (Tmax), minimum temperature of the coldest month (Tmin), and annual precipitation (prec)—were extracted from Worldclim using each populations’ longitude and latitude (Global Climate Data, http://www.worldclim.org/; Hijmans et al. 2005). Then mean values of particular features were used to characterize each population (Boratyńska et al. 2008). To investigate the associations among traits, a Spearman’s non-parametric correlation coefficient matrix was constructed between the traits studied and geo-climatic factors, including longitude, latitude, altitude, Tmean, Tmax, Tmin, and prec. This correlation coefficient is adequate for samples of small size and non-normal distribution (Steel et al. 1960; Wahid et al. 2006). A principal component analysis (PCA) was performed to display the relationships among populations based on a three-dimensional diagram using a PRINCOMP Procedure (version 9.3, SAS Institute Inc., Cary, NC, USA). SAS for Windows (version 9.3, SAS Institute Inc., Cary, NC, USA) and SPSS software (SPSS version 21, IBM Corporation, North Castle Drive, Armonk, NY, USA) were used in statistical calculations.
Results
High and significant levels of variation were found both among and within populations in all traits measured (p < 0.01). Mean needle length ranged from 16.7 cm (population EY) to 22.1 cm (population FN), and mean needle width ranged from 0.4 to 0.7 mm. Maximum fascicle width (1.5 mm) was observed in the KM and HQ populations, and minimum (1.1 mm) was observed in populations CX, GN, TL, YJ, and YR (Table 2). Fascicle-sheath length ranged from 1.2 to 1.8 cm with an overall average of 1.5 cm. Correspondingly, the ratio of needle length to width, needle length to fascicle sheath length, and fascicle width to needle width varied widely. The FN population presented the highest mean values for three of seven needle traits, i.e., needle length (22.1 ± 3.2 cm), fascicle sheath length (1.8 ± 0.3 cm), and the ratio of needle length to needle width (484.9 ± 87.9). No single population was consistently the lowest for any trait.
Differences in cone traits among populations were comparable to those for needle traits. Cones varied from 57.3 to 68.4 mm in length and from 30.6 to 38.8 mm in diameter (Table 3). Cones of HQ and BH were the largest, while those of population EY were the smallest. Cone weight was the trait with greatest variability. Cones of HQ were nearly twice as heavy as those of EY. Overall, values differed significantly among pair-wise populations (Tukey’s multiple comparisons tests significant at p = 0.05 for all traits).
The descriptive statistics showed that the coefficient of variation of needle traits within populations did not exceed 33 % or fall below 9 % (Fig. 2). The highest coefficient of variation was recorded for needle width in the XW population (32.13 %) followed by ratio of needle length to needle width in population GN (29.45 %). The trait with lowest intra-population variability was fascicle width (9.02 % in population SP, 9.38 % in population JS, and 9.75 % in population LL) and ratio of fascicle width to needle width (9.80 %). Cone weight presented the highest within population variability; the mean CV for this trait was 37.60 %. Mean CVs of the other cone traits were relatively low. Population TC showed the lowest coefficient of variance in all four cone traits.
The total variance for each trait could be divided into individual differences within populations, among population differences, and error. The results showed individual tree within population (intra-population) variability accounted for most of the total variation with the exception of the ratio of fascicle width to needle width (FW/NW) (Table 4). The morphological divergence between natural populations was further assessed by calculating the P ST index. Values of P ST varied considerably, from 0.18 for needle length to 0.69 for ratio of fascicle width to needle width, with an average value of 0.34 among all populations for needle traits, and from 0.03 for ratio of length to width of cone to 0.20 for cone diameter. Over all the traits, the morphological divergence was 0.25 among populations. The cone traits exhibited lower morphological divergence (i.e., P ST index) when compared to needle traits.
We observed significant correlations between the observed needle and cone traits and geo-climatic parameters (Table 5). Needle length (NL) and the ratio of needle length to fascicle-sheath length (NL/FSL) exhibited significant and negative correlations (p < 0.01) with latitude and altitude. Thus, shorter needles were associated with higher altitudes and latitudes. The same two traits were positively correlated with temperature (p < 0.01) and precipitation factors (p < 0.05), which implied that there were longer needles and higher ratio of needle length to fascicle sheath length in populations that grow in lower altitudes and latitudes and warmer climates with more rainfall.
Needle width (NW) displayed a positive correlation with longitude (p < 0.05). From east to west, the needles were narrower. However, the linkage between NW and latitude as well as the correlation between NW and altitude was weak (r = −0.103 and −0.119, respectively) and insignificant. Fascicle width, although different among populations, was not significantly correlated with any geo-climatic parameters we analyzed.
Correlations between cone traits and geo-climatic parameters were not significant except for the correlation between the ratio of length to diameter of cone (CL/CD) and longitude (α = 0.05) (Table 5). Cone shape was shorter and thinner with increasing longitude (r = −0.711, p < 0.05). The results indicated that the observed differences among populations in cone traits could not be associated clearly with any geo-climatic patterns.
Because cones were available from a limited group of populations, principal component analysis (PCA) was based on needle traits only (Fig. 3). The eigenvalues indicated that three components provided a good summary of the data, accounting for 87 % of the total variance. The first component (PC1) explained 35.15 % of the total variance. PC1 had high positive loadings on ratio of length to width of needle (NL/NW) and ratio of fascicle width to needle width (FW/NW), and high negative loadings on needle width (NW). The second component explained 30.27 % of the total variance. PC2 had high positive loadings on needle length (NL) and ratio of needle length to fascicle sheath length (NL/FSL). There was also a moderate positive loading on ratio of needle length to needle width (NL/NW). PC2 strongly separated population SJ from the others. Populations BH, CX, FN, GN, TL, and YR were separated from the others by PC3, which showed strong loading on fascicle width (FW) but strong negative loading on fascicle sheath length (FSL). The results of principal component analysis indicated that geographic proximity did not predict similarity based on needle traits.
Three-dimensional view of relationships among 18 populations of Pinus densata based on seven needle traits as separated by the first three principal components. The population abbreviations are the same as Table 1
Discussion
Significant differences in needle morphology were found both among and within populations. Similar patterns of variability in those traits were found in other species as well. Calamassi et al. (1988) showed that significant differences existed for all morphological and anatomical needle traits considered among 14 populations from different regions of the natural range of P. brutia. We also found high levels of variability in cone morphology among and within populations of P. yunnanensis. Similar findings were reported in conifer species such as P. canariensis (Gil et al. 2002), P. halepensis (Melzack et al. 1981), P. pinaster (Wahid et al. 2006), P. radiata (Forde 1964), P. strobus (Beaulieu and Simon 1995), P. sylvestris (Turna and Güney 2009), P. tecunumanii (Piedra 1984), and P. wallichiana (Singh and Thapliyal 2012).
Morphological traits of conifer species are known to vary adaptively with the geographic and climatic variables (Ji et al. 2011). Patterns of variation within tree species depend on several factors, including geographic distribution, breeding system, and historical events (Wahid et al. 2006). Long generation times, an outcrossing mating system (allogamy), wind pollination, high fecundity, and winged seed dispersal are associated with high levels of genetic and phenotypic variation (Hamrick et al. 1979). The level of genetic diversity detected in limited samples natural populations of P. yunnanensis was relatively high based on the analysis of allozymes (Yu et al. 2000).
Observed patterns of phenotypic variation may be the result of local adaptations to a variable environment as was observed for jack pine (P. banksiana) (Maley and Parker 1993). Although cone traits of P. yunnanensis showed high variability among populations coupled with sizeable variation within population, needle traits had higher variation than cones traits among populations. This may be because foliage size is easily influenced by local environmental conditions (Dangasuk and Panetsos 2004).
Conversely, reproductive traits are believed to be the most conserved plant traits and least affected by the environment (Vasudeva et al. 2004). Nevertheless, cone size and weight can be influenced by tree age, general health of the trees, and the macro- and micro- habitat of the parent trees (Dangasuk and Panetsos 2004). Long-term adaptation to diverse and changing environmental conditions (including fires, recurrent volcanic events, etc.) and the lack of competition with other pines may maintain high variability in cone morphology (Gil et al. 2002), which may explain the high levels of variability we observed for cone morphology in P. yunnanensis.
In the present study, within-population variation accounted for most of the total phenotypic variation for nine of eleven traits studied; the mean proportion of phenotypic variation among populations of P. yunnanensis for 11 needle and cone traits was about 38 % (data not shown). A similar conclusion was reached by Ji et al. (2011), who reported the mean proportion of phenotypic variation among populations of P. tabulaeformis in 12 measured traits was about 28 %. Piedra (1984) used analysis of variance (ANOVA) to demonstrate that almost two-thirds of the variation for the needle, cone, and seed characters was attributable to between-tree differences, based on 108 trees of P. tecunumanii from five geographic regions of Guatemala. Maley and Parker (1993) indicated that most of the variation was expressed between trees and within trees, with relatively little (1.6 to 18.9 % depending on the trait) expressed between sampled populations based on phenotypic variation of cone and needle morphology in jack pine (P. banksiana).
Rawat and Bakshi (2011) emphasized that genetic variation within and between populations was a substantial determinant of the adaptive abilities of populations. The high levels of variation in phenotypic traits we observed may reflect adaptation to competition or to a range of environments (Gil et al. 2002). An alternative for Q ST, which is reported widely in tree species (Leinonen et al. 2008; Alberto et al. 2013), is the mean phenotypic differentiation (P ST) (Spitze 1993). Genetic estimates of population differentiation in quantitative traits (Q ST) varied widely among tree species. A review by Alberto et al. (2013) showed that among 16 European conifers species with small or fragmented range, values of Q ST ranged from 0.028 to 0.616, with a mean of 0.192. For 11 European conifer species with a continuous range, Q ST varied from 0.075 to 0.830, with a mean of 0.463. The mean P ST among populations of P. yunnanensis for 11 needle and cone traits was 0.25, which probably indicates the adaptive differentiation of populations in phenotypic traits, but the values of P ST were moderate compared to published values for other species, probably indicating that the populations we sampled do not represent unusual patterns of variability.
In P. yunnanensis, needle length (NL) and the ratio of needle length to fascicle sheath length (NL/FSL) showed patterns of variation along latitudinal and altitudinal gradients. In warmer environments (low latitude and altitude), needles were longer, and the ratio of needle length to fascicle sheath length was higher. Other needle traits, such as needle width, fascicle width, and fascicle sheath length, however, showed only loose correlation with geo-climatic parameters. Therefore, particular needle traits, especially needle length and fascicle sheath length, were more sensitive to geographic and climatic gradients than the other measured traits. Similar findings were described in P. brutia by Dangasuk and Panetsos (2004), who reported that needle length and width, fascicle sheath length, number of teeth per cm, and the number of resin canals were the most useful traits for identifying altitudinal and longitudinal adaptation variations within and among populations. The relationship between needle length and width and geographic parameters like longitude and altitude for P. yunnanensis was in agreement with the findings of Wahid et al. (2006), who found needle length and width correlated negatively with altitude and positively with longitude in maritime pine (P. pinaster).
In spite of the overall absence of significant correlation between fascicle width and geo-climatic parameters (Table 5), we observed an association between thick fascicles and dry, cold environments. Populations HQ, EY, XW, and MD all came from high altitudes (Table 1) where precipitation was low and Tmax was generally low. These populations had relatively high fascicle width and short needle length. The population with highest fascicle width was KM, which was also high-altitude. Population EY had the shortest needles and was also from the highest elevation.
Although there was large variation in cone traits of P. yunnanensis among populations, no significant correlation was detected between cone traits and geo-climatic parameters except for the negative correlation between the ratio of length to diameter of cone (CL/CD) and longitude. Cone traits were significantly different (p < 0.01) among populations, but interpopulation differences were not associated with a climatic gradient. Similar results were observed in P. brutia (Dangasuk and Panetsos 2004) and Aleppo pine (P. halepensis Mill.) (Melzack et al. 1981). Singh and Thapliyal (2012) pointed out cone characteristics correlated negatively with latitude and positively with altitude in blue pine (P. wallichiana), but no particular trend was found between cone characteristics and geographic variables like latitude and altitude. Cone weight, length, and diameter were negatively correlated with precipitation in present study. This result was in agreement with findings in other species of the genus such as P. roxburghii (Roy et al. 2004). Cones may be larger in low-precipitation environments (Gil et al. 2002).
It was difficult to discern environmental patterns that explained the clustering of populations in the principal components space (Fig. 3). The most likely reason for this observation was that population-level trait measures concealed intra-collection-site environmental variation, which, as previously mentioned, can strongly affect needle morphology (Dangasuk and Panetsos 2004). Some traits with high P ST and high loading in PC1, such as NL/NW and NW, showed relatively high intra-population variability (Table 4; Fig. 2). This may indicate that these traits are developmentally plastic in response to environment. As described above, the correlation matrix between needle traits and environmental characteristics (Table 5) presents several strong and highly significant correlations: for example, between short needles and high altitude locations, which are reasonable and worth further investigation.
A lack of geographic correspondence between populations with similar phenotypes was also observed in P. canariensis (Gil et al. 2002), P. radiata (Forde 1964), and P. tecunumanii (Piedra 1984). Beaulieu and Simon (1995) showed that no general geographical trend was detected in the observed variation in P. strobus collected from ten natural populations in the Canadian province of Quebec.
References
Alberto FJ, Aitken SN, Alia R, González-Martínez SC, Hänninen H, Kremer A, Lefèvre F, Lenormand T, Yeaman S, Whetten R, Savolainen O (2013) Potential for evolutionary responses to climate change—evidence from tree populations. Glob Change Biol 19(6):1645–1661
Ali MA, Jabran K, Awan SI, Abbas A, Ehsanullah ZM, Acet T, Farooq J, Rehman A (2011) Morpho-physiological diversity and its implications for improving drought tolerance in grain sorghum at different growth stages. Aust J Crop Sci 5(3):311–320
Beaulieu J, Simon JP (1995) Variation in cone morphology and seed characters in Pinus strobus in Quebec. Can J Bot 73(2):262–271
Bobowicz MA, Radziejewska A (1989) The variability of Scots pine from Piekielna Gora as expressed by morphological and anatomical traits of needles. Acta Soc Bot Pol 58(3):375–384
Boratyńska K, Jasińska AK, Ciepłuch E (2008) Effect of tree age on needle morphology and anatomy of Pinus uliginosa and Pinus silvestris: species-specific character separation during ontogenesis. Flora 203(8):617–626
Boulli A, Baaziz M, M’Hirit O (2001) Polymorphism of natural populations of Pinus halepensis Mill. in Morocco as revealed by morphological characters. Euphytica 119:309–316
Calamassi R, Puglisi SR, Vendramin GG (1988) Genetic variation in morphological and anatomical needle characteristics in Pinus brutia Ten. Silv Genet 37(5–6):199–206
Chen SH, Wu JK (1987) A preliminary study on the seed division of Pinus yunnanensis. J Southwest For Coll 2:1–6
Chinese Academy of Sciences (1978) Flora of China, vol 7. Science Press, Beijing, pp 255–259
Dangasuk OG, Panetsos KP (2004) Altitudinal and longitudinal variations in Pinus brutia (Ten.) of Crete Island, Greece: some needle, cone and seed traits under natural habitats. New For 27:269–284
De Vall WB (1940) A diagnostic taxonomic constant for separating slash and longleaf pines. Proc Fla Acad Sci 4:113–115
Donahue JK, Upton JL (1996) Geographical variation in leaves, cone and seeds of Pinus gregii in native forests. For Ecol Manag 82:145–157
Forde MB (1964) Variation in natural populations of Pinus radiata in California. Part 3. Cone characters. NZ J Bot 2:459–485
Gil L, Climent J, Nanos N, Mutke S, Ortiz I, Schiller G (2002) Cone morphology variation in Pinus canariensis Sm. Plant Syst Evol 235:35–51
Hamrick JL, Linhart YB, Mitton JB (1979) Relationships between life history characteristics and electrophoretically detectable genetics variation in plants. Annu Rev Ecol Syst 1:173–200
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978
Ji M, Zhang Q, Deng J, Zhang X, Wang Z (2011) Intra-versus inter-population variation of cone and seed morphological traits of Pinus tabulaeformis Carr. in northern China: impact of climate-related conditions. Pol J Ecol 59(4):717–727
Jin ZZ, Peng J (2004) Yunnan Pine (Pinus yunnanensis Franch.). Yunnan Science and Technology Press, Kunming, pp 1–66
Leinonen T, Cano JM, Mäkinen H, Merilä J (2006) Contrasting patterns of body shape and neutral genetic divergence in marine and lake populations of three spine sticklebacks. J Evol Biol 19:1803–1812
Leinonen T, O’hara RB, Cano JM, Merilä J (2008) Comparative studies of quantitative trait and neutral marker divergence: a meta-analysis. J Evol Biol 21:1–17
Li BD (1984) The distribution of forest of Pinus yunnanensis and its areal areas. J Yunnan Univ 1:33–56
Li LF, Zheng W, Han MY, Su JW (2010) Progress of genetic improvement of Pinus yunnanensis and breeding strategy analysis. J West China For Sci 39(2):4–7
Loha A, Tigabu M, Teketay D, Lundkvist K, Fries A (2006) Provenance variation in seed morphometric traits, germination, and seedling growth of Cordia africana Lam. New For 32:71–86
López R, López de Heredia U, Collada C, Cano FJ, Emerson BC, Cochard H, Gil L (2013) Vulnerability to cavitation, hydraulic efficiency, growth and survival in an insular pine (Pinus canariensis). Ann Bot 111:1167–1179
Maley ML, Parker WH (1993) Phenotypic variation in cone and needle characters of Pinus banksiana (jack pine) in northwestern Ontario. Can J Bot 71(1):43–51
Melzack RN, Grunwald C, Schiller G (1981) Morphological variation in Aleppo pine (Pinus halepensis Mill.) in Israel. Isr J Bot 30(4):199–205
Panetsos CP (1975) Natural hybridization between Pinus halepensis and Pinus brutia in Greece. Silv Genet 24(5–6):163–168
Pham TD, Thi Nguyen TD, Carlsson AS, Bui TM (2010) Morphological evaluation of sesame (Sesamum indicum L.) varieties from different origins. Aust J Crop Sci 4(7):498–504
Piedra ET (1984) Geographic variation in needles, cones and seeds of Pinus tecunumanii in Guatemala. Silv Genet 33(2/3):72–79
Raeymaekers JAM, Van Houdt JKJ, Larmuseau MHD, Geldof S, Volckaert FAM (2007) Divergent selection as revealed by P ST and QTL-based F ST in three-spined stickleback (Gasterosteus aculeatus) populations along a coastal-inland gradient. Mol Ecol 16:891–905
Rawat K, Bakshi M (2011) Provenance variation in cone, seed and seedling characteristics in natural populations of Pinus wallichiana A.B. Jacks (Blue Pine) in India. Ann For Res 54(1):39–55
Roy SM, Thapliyal RC, Phartyal SS (2004) Seed source variation in cone, seed and seedling characteristics across the natural distribution of Himalayan low level pine Pinus roxburghii Sarg. Silv Genet 53(3):116–128
Sevık H, Ayan S, Turna I, Yahyaoglu Z (2010) Genetic diversity among populations in Scotch pine (Pinus silvestris L.) seed stands of Western Black Sea Region in Turkey. Afr J Biotechnol 9(43):7266–7272
Singh O, Thapliyal M (2012) Variation in cone and seed characters in blue pine (Pinus wallichiana) across natural distribution in western Himalayas. J For Res 23(2):235–239
Spitze K (1993) Population-structure in Daphnia obtusa: quantitative genetic and allozymic variation. Genetics 135:367–374
Steel RG, Torrie JH, Dickey DA (1960) Principles and procedures of statistics. McGrow-Hill Book Company Inc., New York, p 481
Turna I, Güney D (2009) Altitudinal variation of some morphological characters of Scots pine (Pinus sylvestris L.) in Turkey. Afr J Biotechnol 8(2):202–208
Turna I, Yahyaoglu Z, Yüksek F, Ayaz FA, Guney D (2006) Morphometric and electrophoretic analysis of 13 populations of Anatolian black pine in Turkey. J Environ Biol 27(3):491–497
Urbaniak L, Karliński L, Popielarz R (2003) Variation of morphological needle characters of Scots pine (Pinus silvestris L.) populations in different habitats. Acta Soc Bot Pol 72(1):37–44
Vasudeva R, Hanumantha M, Gunaga RP (2004) Genetic variation for floral traits among teak (Tectona grandis Linn.f.) clones: implications to seed orchard fertility. Curr Sci 87:358–362
Wahid N, González-Martínez SC, El Hadrami I, Boulli A (2006) Variation of morphological traits in natural populations of maritime pine (Pinus pinaster Ait.) in Morocco. Ann For Sci 63:83–92
Wang CM, Wang J, Jiang HQ (2003) A study on comparative morphology of Pinus yunnanensis needles under different habitats. J Southwest For Coll 23:4–7
Wang CM, Wang J, Jiang HQ (2004) A study on comparative anatomy of Pinus yunnanensis needles under different habitats. J Southwest For Coll 24:1–5
Wu ZY (1986) Flora Yunnanica (Tomus 4): Spermatophyta. Science Press, Beijing, pp 54–57
Yu H, Ge S, Huang RF, Jiang HQ (2000) A preliminary study on genetic variation and relationships of Pinus yunnanensis and its closely related species. Acta Bot Sin 42:107–110
Acknowledgments
The authors thank Jie ZENG and Cheng-Zhong HE for their technical suggestions. The use of trade names is for the information and convenience of the reader and does not imply official endorsement or approval by the USDA or the Forest Service of any product to the exclusion of others that may be suitable.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project funding: This study was financially supported by the National Natural Science Foundation of China (NSFC 31260191), Natural Science Foundation of Yunnan Province, China (2010CD065).
The online version is available at http://www.springerlink.com
Corresponding editor: Hu Yanbo
Rights and permissions
About this article
Cite this article
Xu, Y., Woeste, K., Cai, N. et al. Variation in needle and cone traits in natural populations of Pinus yunnanensis . J. For. Res. 27, 41–49 (2016). https://doi.org/10.1007/s11676-015-0153-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-015-0153-6