Abstract
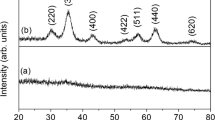
An investigation of Ag/(hematite-alumina) solid solutions (Ag/FexAl2−xO3, 0.5 ≤ x ≤ 2) was completed via x-ray diffraction (XRD), scanning electron microscopy (SEM), Fourier transform Infrared spectroscopy (FTIR) and Mössbauer spectroscopy. Results reveal that the materials obtained by combustion synthesis are multi-phase nanocomposites and silver metal is supported on hematite-rich and alumina-rich nanoparticles for x < 2. For the iron-rich hematite phase, the experimental results indicated a beginning transition to the superparamagnetic state. They also show that the distribution of iron and aluminum cations in the product phases strongly depends on the starting concentrations and that their compositions closely correspond to those of the equilibrium phase diagram at about 1250°C. Mössbauer signal intensities of samples with x = 0.5, 1.0 and 1.5 could consistently be explained by coexistence of phases of approximate composition Fe0.2Al1.8O3 and Fe1.8Al0.2O3 , which closely corresponds to the thermodynamic equilibrium phase diagram of Fe2O3-Al2O3 at about 1250°C. Finally, the Mössbauer spectra reveal line shape changes for the samples with x < 2 which are characteristic for the onset of superparamagnetic behavior.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. Bantsis, M. Betsiou, A. Bourliva, T. Yioultsis, and C. Sikalidis, Ceram. Int. 38, 721 (2012).
S. Bhagwat, S. Joshi, S.B. Ogale, G. Marest, A. Benyagoub, N. Mancoffre, and F. Thimon, J. Appl. Phys. 79, 4141 (1996).
W. Wang, W. Liu, X. Yang, and Z. Xie, Ceram. Int. 38, 2851 (2012).
L. Zhou, W. Zhou, J. Su, F. Luo, and D. Zhu, Ceram. Int. 38, 1077 (2012).
S. Bid, A. Banerjee, S. Kumar, S.K. Pradhan, U. De, and D. Banerjee, J. Alloys Compd. 326, 292 (2001).
R.J. Willey, H. Lai, and J.B. Peri, J. Catal. 130, 319 (1991).
M. Niwa, K. Yagi, and Y. Murakami, Bull. Chem. Soc. Jpn 54, 975 (1981).
A. Lycourghiotis and D. Vattis, React. Kinet. Catal. Lett. 18, 377 (1981).
V.I. Kuznetsov, E.N. Yurchenko, M.T. Protasova, E.A. Taraban, O.P. Krivoruchko, and R.A. Buyanov, Phys. Status Solidi (a) 113, 359 (1989).
M.C. Prieto, J.M.G. Amores, V.S. Escribano, and G. Busca, J. Mater. Chem. 4, 1123 (1994).
J.L. McArdle and G.L. Messing, J. Am. Ceram. Soc. 76, 214 (1993).
L.F. Cótica, A. Paesano Jr, S.C. Zanatta, S.N. de Medeiros, and J.B.M. da Cunha, J. Alloys Compd. 413, 265 (2006).
P. Tartaj and J. Tartaj, Acta Mater. 50, 5 (2002).
J. Kákoš, L. Bača, P. Veis, and L. Pach, J. Sol-Gel. Sci. Technol. 21, 167 (2001).
International Centre for Diffraction Data, PDF 10-173 (α-Al2O3) and PDF 13-534 (α-Fe2O3).
J.F. Bengoaa, A.M. Alvareza, A.E. Bianchib, G. Punteb, R.E. Vandenberghec, R.C. Mercaderb, and S.G. Marchettia, Mat. Chem. Phys. 123, 191 (2010).
N. Mimura and M. Saito, Catal. Today 55, 173 (2000).
G. Giecko, T. Borowiecki, W. Gac, and J. Kruk, Catal. Today 137, 403 (2008).
N.R.E. Radwan, E.A. El-Sharkawy, and A.M. Youssef, Appl. Catal. A 281, 93 (2005).
S. Kobayashi, S. Kaneko, M. Ohshima, H. Kurokawa, and H. Miura, Appl. Catal. A 417–418, 306 (2012).
B.M. Abu-Zied, Appl. Catal. A 334, 234 (2008).
G. Longworth: in Mössbauer Spectroscopy Applied to Inorganic Chemistry, G.J. Long (Ed.), Vol. I, p. 43. Plenum Press, New York (1984).
K. Lagarec, D.G. Rancourt, Recoil-Mössbauer spectral analysis software for windows version 1.02, Department of Physics, University of Ottawa, (1998).
W. Gessner, Anorg. Allgem Chemie 360, 247 (1968).
T. A. Taha, S., Elrabaie, M. T. Attia, J. Mater. Sci. Mater. Electron., 29(21), 18493 (2018)
M.H. Yao, R.J. Baird, F.W. Kunz, and T.E. Hoost, J. Catal. 166, 67 (1997).
S. Zhan, D. Chen, X. Jiao, and S. Liu, J. Colloid Interface Sci. 308, 265 (2007).
S. Krehula and S. Musić, J. Alloys Compd. 416, 284 (2006).
B.T. Poe, P.F. McMillan, C.A. Angell, and R.K. Sato, Chem. Geol. 96, 333 (1992).
M. Okuno, N. Zotov, M. Schmücker, H. Schneider, and J. Non-Cryst, Solids 351, 1032 (2005).
M. Ristić, E. De Grave, S. Musić, S. Popović, and Z. Orehovec, J. Mol. Struct. 834–836, 454 (2007).
S. Musić, I. Czakó-Nagy, I. Salaj-Obelić, and N. Ljubešić, Mater. Lett. 32, 301 (1997).
J.E. Iglesias and C.J. Serna, Miner. Petrogr. Acta 29A, 363 (1985).
M. Ristić and S. Musić, J. Alloys Compd. 425, 384 (2006).
Y. Wang, A. Muramatsu, and T. Sugimoto, Colloid Surf. A: Physicochem. Eng. Aspects 134, 281 (1998).
F. Menil, J. Phys. Chem. Solids 46, 763 (1985).
O.C. Kistner and A.W. Sunyar, Phys. Rev. Lett. 4, 412 (1960).
A. Muan and C.L. Gee, J. Am. Ceram. Soc. 39, 207 (1956).
F.A. Elrefaie and W.W. Smeltzer, Metall. Trans. B 14, 85 (1983).
R. Hansson, P.C. Hayes, and E. Jak, Metall. Mater. Trans. 35B, 633 (2004).
M.A. Rhamdhani, T. Hidayat, P.C. Hayes, and E. Jak, Metall. Mater. Trans. 40B, 25 (2009).
A.K. Ladavos and T.V. Bakas, React. Kinet. Catal. Lett. 73, 223 (2001).
A. Cordier, A. Peigney, E. De Grave, E. Flahaut, and C. Laurent, J. Eur. Ceram. Soc. 26, 3099 (2006).
L.F. Cotica, S.C. Zanatta, M.A. Rocha, I.A. Santos, A. Paesano Jr, J.B.M. da Cunha, and B. Hallouche, J. Appl. Phys. 95, 1307 (2004).
M. Liu, H. Li, L. Xiao, W. Yu, Y. Lu, and Z. Zhao, J. Magn. Magn. Mater. 294, 294 (2005).
W. Kündig, H. Bömmel, G. Constabaris, and R.H. Lindquist, Phys. Rev. 142, 327 (1966).
P. Fielitz, G. Borchardt, S. Ganschow, and R. Bertram, Defect Diffusion Forum 323–325, 75 (2012).
K. Hoshino and N.L. Peterson, J. Phys. Chem. Solids 46, 1247 (1985).
C. Wert and C. Zener, J. Appl. Phys. 21, 5 (1950).
R.J. Borg, G.J. Dienes, An Introduction to Solid State Diffusion, Academic Press Inc., Boston, Chap. XII, (1988).
Acknowledgments
We would like to thank Prof. Klaus-Dieter Becker, Institute of Physical and Theoretical Chemistry, Technische Universität Germany for providing the Mössbauer measurements.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mahmoud, M.H., Taha, T.A. FTIR and Mössbauer Spectroscopy Investigations of Ag/FexAl2−xO3 Nanocomposites. J. Electron. Mater. 48, 7396–7403 (2019). https://doi.org/10.1007/s11664-019-07568-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07568-x