Abstract
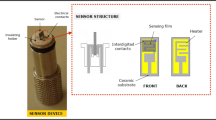
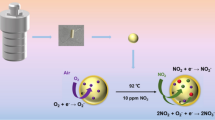
We have synthesized a chemiresistive sensor for chlorine (Cl2) gas in the range of 2–200 ppm based on nickel oxide (NiO) nanoparticles obtained by wet chemical synthesis. The nanoparticles were characterized by x-ray diffraction (XRD) analysis, field-emission scanning electron microscopy (FE-SEM), thermogravimetric analysis (TGA), transmission electron microscopy (TEM), Fourier-transform infrared (FTIR) spectroscopy, Raman spectroscopy, ultraviolet–visible (UV–Vis) spectroscopy, and photoluminescence (PL) spectroscopy. XRD spectra of the sensing layer revealed the cubic phase of NiO nanoparticles. The NiO nanoparticle size was calculated to be ∼ 21 nm using a Williamson-Hall plot. The bandgap of the NiO nanoparticles was found to be 3.13 eV using Tauc plots of the absorbance curve. Fast response time (12 s) and optimum recovery time (∼ 27 s) were observed for 10 ppm Cl2 gas at moderate temperature of 200°C. These results demonstrate the potential application of NiO nanoparticles for fabrication of highly sensitive and selective sensors for Cl2 gas.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
T.E. Graedel, J. Ind. Ecol. 13, 154 (2009).
T. Van Dang, N. Duc Hoa, N. Van Duy, and N. Van Hieu, ACS Appl. Mater. Interfaces 8, 4828 (2016).
C.W. White and J.G. Martin, Proc. Am. Thorac. Soc. 7, 257 (2010).
A. Sanger, A. Kumar, A. Kumar, J. Jaiswal, and R. Chandra, Sens. Actuators B 236, 16 (2016).
A. Kumar, A. Sanger, A. Kumar, and R. Chandra, RSC Adv. 6, 47178 (2016).
A. Sanger, A. Kumar, A. Kumar, P.K. Jain, Y.K. Mishra, and R. Chandra, Ind. Eng. Chem. Res. 55, 9452 (2016).
I. Hotovy, J. Huran, L. Spiess, S. Hascik, and V. Rehacek, Sens. Actuators B 57, 147 (1999).
A.C. Sonavane, A.I. Inamdar, P.S. Shinde, H.P. Deshmukh, R.S. Patil, and P.S. Patil, J. Alloys Compd. 489, 667 (2010).
B. Varghese, M.V. Reddy, Z. Yanwu, C.S. Lit, T.C. Hoong, G.V. Subba Rao, B.V.R. Chowdari, A.T.S. Wee, C.T. Lim, and C.-H. Sow, Chem. Mater. 20, 3360 (2008).
Y. Zhu, C. Cao, S. Tao, W. Chu, Z. Wu, and Y. Li, Sci. Rep. 4, 5787 (2014).
B. Vidhyadharan, N.K.M. Zain, I.I. Misnon, R.A. Aziz, J. Ismail, M.M. Yusoff, and R. Jose, J. Alloys Compd. 610, 143 (2014).
A. Cimino, M. Lo Jacono, and M. Schiavello, J. Phys. Chem. 75, 1044 (1971).
J. He, H. Lindström, A. Hagfeldt, and S.-E. Lindquist, J. Phys. Chem. B 103, 8940 (1999).
H. Steinebach, S. Kannan, L. Rieth, and F. Solzbacher, Sens. Actuators B 151, 162 (2010).
D.S. Dalavi, N.S. Harale, I.S. Mulla, V.K. Rao, V.B. Patil, I.Y. Kim, J.H. Kim, and P.S. Patil, Mater. Lett. 146, 103 (2015).
S.M. Kanan, O.M. El-Kadri, I.A. Abu-Yousef, and M.C. Kanan, Sensors 9, 8158 (2009).
A. Sanger, A. Kumar, S. Chauhan, Y.K. Gautam, and R. Chandra, Sens. Actuators B 213, 252 (2015).
S.J. Musevi, A. Aslani, H. Motahari, and H. Salimi, J. Saudi Chem. Soc. 20, 245 (2016).
D.S. Hall, D.J. Lockwood, C. Bock, and B.R. MacDougall, Proc. R. Soc. Math. Phys. Eng. Sci. 471, 20140792 (2015).
W. Guo, K.N. Hui, and K.S. Hui, Mater. Lett. 92, 291 (2013).
V. Mote, Y. Purushotham, and B. Dole, J. Theor. Appl. Phys. 6, 1 (2012).
N. Mironova-Ulmane, A. Kuzmin, I. Steins, J. Grabis, I. Sildos, and M. Pärs, J. Phys: Conf. Ser. 93, 012039 (2007).
M.-L. Lu, T.-Y. Lin, T.-M. Weng, and Y.-F. Chen, Opt. Express 19, 16266 (2011).
M. El-Kemary, N. Nagy, and I. El-Mehasseb, Mater. Sci. Semicond. Process. 16, 1747 (2013).
V. Biju and M. Abdul Khadar, Spectrochim. Acta Part A 59, 121 (2003).
M.A. Shah, Nanoscale Res. Lett. 3, 255 (2008).
P.V. Tong, N.D. Hoa, N.V. Duy, V.V. Quang, N.T. Lam, and N.V. Hieu, Int. J. Hydrogen Energy 38, 12090 (2013).
L. Liu, S. Li, J. Zhuang, L. Wang, J. Zhang, H. Li, Z. Liu, Y. Han, X. Jiang, and P. Zhang, Sens. Actuators B 155, 782 (2011).
W.X. Jin, S.Y. Ma, Z.Z. Tie, J.J. Wei, J. Luo, X.H. Jiang, T.T. Wang, W.Q. Li, L. Cheng, and Y.Z. Mao, Sens. Actuators B 213, 171 (2015).
J. Zhang, D. Zeng, Q. Zhu, J. Wu, Q. Huang, and C. Xie, J. Phys. Chem. C 120, 3936 (2016).
J. Zhang, D. Zeng, Q. Zhu, J. Wu, Q. Huang, W. Zhang, and C. Xie, Phys. Chem. Chem. Phys. 18, 5386 (2016).
S.-J. Choi, S. Chattopadhyay, J.J. Kim, S.-J. Kim, H.L. Tuller, G.C. Rutledge, and I.-D. Kim, Nanoscale 8, 9159 (2016).
A. Sanger, P.K. Jain, Y.K. Mishra, and R. Chandra, Sens. Actuators B 242, 694 (2017).
Y.K. Mishra and R. Adelung, Mater. Today (2017). https://doi.org/10.1016/j.mattod.2017.11.003.
S. Jain, A. Sanger, S. Chauhan, and R. Chandra, Mater. Res. Express 1, 35046 (2014).
A. Sanger, A. Kumar, A. Kumar, and R. Chandra, Sens. Actuators B 234, 8 (2016).
J. Tamaki, C. Naruo, Y. Yamamoto, and M. Matsuoka, Sens. Actuators B 83, 190 (2002).
C.V.G. Reddy, S.V. Manorama, and V.J. Rao, Sens. Actuators B 55, 90 (1999).
W. Ding, H. Ping, X. Jiaqiang, D. Xiaowen, and P. Qingyi, Sens. Actuators B 140, 383 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arif, M., Sanger, A. & Singh, A. Highly Sensitive NiO Nanoparticle based Chlorine Gas Sensor. J. Electron. Mater. 47, 3451–3458 (2018). https://doi.org/10.1007/s11664-018-6176-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-018-6176-y