Abstract
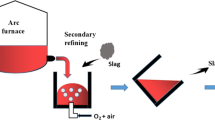
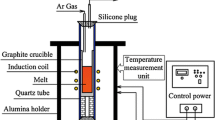
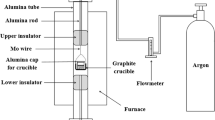
Phosphorus removal from metallurgical grade silicon by CaO-SiO2-CaCl2 slag treatment, HCl leaching, and vacuum refining was investigated. The effect of different compositions of slag was evaluated. The calcium concentration in slag-treated silicon increased with increasing CaO/SiO2 mass ratio of slag, decreasing the evaporation efficiency of phosphorus in molten silicon. The total phosphorus removal efficiency changed from 93.0% to 98.3% when the slag-treated silicon was treated with HCl before vacuum refining. The final concentration of phosphorus in silicon was 0.43 ppmw. This is because phosphorus was removed from metallurgical-grade silicon as follows: Phosphorus reacts with slag at the silicon/slag interface and forms Ca3(PO4)2 and Ca3P2, most of which diffuse from the interface to the slag phase. The remaining Ca3(PO4)2 and Ca3P2 reduce the phosphorus removal efficiency by altering the activity coefficient of phosphorus in molten silicon. HCl leaching enhanced the phosphorus removal efficiency by removing the remaining Ca3(PO4)2 and Ca3P2. Therefore, the mass transfer of phosphorus from metallurgical-grade silicon was accelerated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- MG-Si:
-
Metallurgical-grade silicon
- SoG-Si:
-
Solar-grade silicon
- Si-S:
-
45 wt.% CaO-45 wt.% SiO2-10 wt.% CaCl2 slag treated MG-Si sample
- Si-S-L:
-
HCl-leached Si-S sample
- Si-S-L-V:
-
Vacuum-refined Si-S-L sample
- Si-S-V:
-
Vacuum-refined Si-S sample
- ppmw:
-
Parts per million weight
References
B.N. Mukashev, Kh.A. Abdullin, M.F. Tamendarov, T.S. Turmagambetov, B.A. Beketov, M.R. Page, and D.M. Kline, Sol. Energy Mater. Sol. Cells 93, 1785 (2009).
J.K. Lee, J.S. Lee, B.Y. Jang, J.S. Kim, Y.S. Ahn, G.H. Kang, H.E. Song, M.G. Kang, and C.H. Cho, Sol. Energy 115, 322 (2015).
M.A. Martorano, J.B.F. Neto, T.S. Oliveira, and T.O. Tsubaki, Mater. Sci. Eng. B 176, 217 (2011).
T. Yoshikawa and K. Morita, ISIJ Int. 47, 582 (2007).
J. Safarian and M. Tangstad, Metall. Mater. Trans. B 43, 1427 (2012).
S.S. Zheng, T. Abel Engh, M. Tangstad, and X.T. Luo, Sep. Purif. Technol. 82, 128 (2011).
T. Ikeda and M. Maeda, ISIJ Int. 32, 635 (1992).
Y. Tan, X. Guo, S. Shi, W. Dong, and D.C. Jiang, Vacuum 93, 65 (2013).
S. Shi, W. Dong, X. Peng, D. Jiang, and Y. Tan, Appl. Surf. Sci. 266, 344 (2013).
L. Hu, Z. Wang, X. Gong, Z. Guo, and H. Zhang, Metall. Mater. Trans. B 44, 828 (2013).
D. Min and N. Sano, Metall. Mater. Trans. B 19, 433 (1988).
M.D. Johnston and M. Barati, Sep. Purif. Technol. 107, 129 (2013).
T.H. Ludwig, E. Schonhovd Dæhlen, P.L. Schaffer, and L. Arnberg, J. Alloys Compd. 586, 180 (2014).
L.A.V. Teixeira, Y. Tokuda, T. Yoko, and K. Morita, ISIJ Int. 49, 777 (2009).
J. Park, D. Min, and H. Song, Metall. Mater. Trans. B 33, 723 (2002).
L.A.V. Teixeira and K. Morita, ISIJ Int. 49, 783 (2009).
A. Tagaya, H. Chiba, F. Tsukihashi, and N. Sano, Metall. Mater. Trans. B 22, 499 (1991).
E.J. Jung, B.M. Moon, and D.J. Min, Sol. Energy Mater. Sol. Cells 95, 1779 (2011).
Y.V. Meteleva-Fischer, Y.X. Yang, R. Boom, B. Kraaijveld, and H. Kuntzel, JOM 64, 957 (2012).
K. Tang, E.J. Ovrelid, G. Tranell, and M. Tangstad, Mater. Trans. 50, 1978 (2009).
K. Morita, Modeling, Control and Optimization in Ferrous and Nonferrous Industry (Warrendale, PA, 2003).
T. Shimpo, T. Yoshikawa, and K. Morita, Metall. Mater. Trans. B 35, 277 (2004).
E.T. Turkdogan, Physical Chemistry of High Temperature Technology (New York: Academic Press, 1980).
K. Morita, K. Kume, and N. Sano, Scand. J. Metall. 31, 178 (2002).
T. Miki, K. Morita, and N. Sano, Metall. Mater. Trans. B 27B, 937 (1996).
E.T. Turkdogan, ISIJ Int. 40, 964 (2000).
I.H. Jung and Y. Zhang, JOM 64, 973 (2012).
S. Tabuchi and N. Sano, Metall. Mater. Trans. B 15, 351 (1984).
C.H. Lupis, Elsevier Science Publishing Co., Inc., 1983, p. 581.
M. Fang, C.H. Lu, L.Q. Huang, H.X. Lai, J. Chen, J.T. Li, W.H. Ma, P.F. Xing, and X.T. Luo, Ind. Eng. Chem. Res. 53, 972 (2014).
J.P. Martins and F. Margarido, Mater. Chem. Phys. 44, 156 (1996).
E. Machlin, Trans. Am. Inst. Min. Metall. Pet. Eng. Soc. Min. Eng. AIME 218, 314 (1960).
K. Tang, E.J. Øvrelid, G. Tranell, and M. Tangstad, JOM 61, 49 (2009).
J. Szekely, C. Chang, and W. Johnson, Metall. Mater. Trans. B 8, 514 (1977).
D.C. Jiang, S.Q. Ren, S. Shi, W. Dong, J.S. Qiu, Y. Tan, and J.Y. Li, J. Electron. Mater. 43, 314 (2014).
S.S. Zheng, T.A. Engh, M. Tangstad, and X.T. Luo, Metall. Mater. Trans. A 42, 2214 (2011).
Acknowledgements
We gratefully acknowledge the support of the National Science Foundation of China-Yunnan (U1137601), the National Natural Science Foundation of China Nos. 51334004 and 51204143, and the Scientific and Technological Innovation Platform of Fujian Province (2006L2003). Moreover, we thank Yanxu Lin for assistance with the samples.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Huang, L., Lai, H., Lu, C. et al. Evaporation Behavior of Phosphorus from Metallurgical Grade Silicon via Calcium-Based Slag Treatment and Hydrochloric Acid Leaching. J. Electron. Mater. 45, 541–552 (2016). https://doi.org/10.1007/s11664-015-4146-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-015-4146-1