Abstract
Summary
We investigated the effect of paravertebral muscle (PVM) on poor prognosis in osteoporotic vertebral fracture (OVF) and remaining lower back pain (LBP) in the thoracolumbar and lower lumbar regions. Additional OVF occurrence in the thoracolumbar and remaining LBP in the lumbar region was significantly related to PVM fat infiltration percentage.
Purpose
Paravertebral muscle (PVM) is an important component of the spinal column. However, its role in the healing process after osteoporotic vertebral fracture (OVF) is unclear. This study aimed to clarify the effect of PVM in thoracolumbar and lower lumbar regions on OVF clinical and radiological outcomes.
Methods
This was a multicenter prospective cohort study from 2012 to 2015. Patients ≥ 65 years old who presented within 2 weeks after fracture onset were followed up for 6 months. PVM was measured at the upper edge of the L1 and L5 vertebral body in the magnetic resonance imaging (MRI) T2-axial position at registration. The cross-sectional area (CSA), relative CSA (rCSA), and fat infiltration percentage (FI%) were measured. Severe vertebral compression, delayed union, new OVF, and remaining low back pain (LBP) were analyzed.
Results
Among 153 patients who were followed up for 6 months, 117 with measurable PVM were analyzed. Their average age was 79.1 ± 7.2 years, and 94 were women (80.3%). There were 48 cases of severe vertebral compression, 21 delayed unions, 11 new OVF, and 27 remaining LBP. Among all poor prognoses, only the FI% of the PVM was significantly associated with new OVF (p = 0.047) in the thoracolumbar region and remaining LBP (p = 0.042) in the lumbar region.
Conclusion
The occurrence of additional OVF in the thoracolumbar region and remaining LBP in the lumbar region was significantly related to the FI% of the PVM. Physicians should be aware that patients with such fatty degeneration shown in acute MRI may require stronger treatment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Paravertebral muscle (PVM) is an important component of the spinal column to consider in relation to a balanced spinal column, release from pain, and osteoporotic fractures [1,2,3,4]. Sarcopenia may be one of the main causes of several pathologies in the spinal column. Previous studies revealed that the cross-sectional area (CSA) and fat infiltration percentage (FI%) of the paraspinal muscle correlate with spinal stability and alignment [5].
Larsson et al. demonstrated that sarcopenia is an age-associated pathology; with an increase in age, the muscle mass reduces [6]. In our previous study, we reported the importance of trunk muscle mass in spinal balance, lumbar dysfunction, increased Oswestry Disability Index, visual analog scale (VAS), and EuroQol 5 Dimension [7]. Furthermore, it has been reported that the FI% of the paraspinal muscle is associated with lower back pain (LBP) and disability [8].
With an increase in age, the incidence of the osteoporotic vertebral fracture (OVF) increases. In Japan, the USA, and Europe, 18 to 26% of post-menopausal women suffer from vertebral deformity [9]. Due to the life expectancy rate in Japan, women over the age of 65 years account for approximately 28% of the entire population, and this percentage is the highest in the world [10]. The negative impact of OVF causes a patient to suffer from LBP, spinal deformity, altered daily life activity, and even mortality in developed counties [11,12,13,14,15]. Furthermore, it is important to diagnose delayed union, nonunion of vertebral fracture, various pathologies of the spine, and PVM appearance. Therefore, magnetic resonance imaging (MRI) is an important diagnostic tool for confirming the fracture scale [16, 17].
Several studies have validated the impact of the PVM on OVF and have revealed that PVM may play an important role in OVF incidence [18,19,20,21,22]. Our previous study demonstrated the natural course of PVM after the onset of OVF and showed that a reduction in PVM at the lumbar spine was significantly related to LBP and delayed union after OVF onset [23]. However, the impact of the PVM on OVF and remaining LBP in separate regions such as the thoracolumbar and lumbar regions has not been well studied. Therefore, this study aimed to clarify the impact of PVM on OVF and LBP in the thoracolumbar and lower lumbar regions.
Methods
This was a multicenter prospective cohort study involving 11 institutions in Japan (Osaka, Hyogo, and Nara). The details were described in our previous study [24], and 153 symptomatic consecutive patients completed a 6-month follow-up. The inclusion criteria of this study were symptomatic patients aged > 65 years with fresh fragile vertebral fracture, which had occurred within 2 weeks prior to presentation. The exclusion criteria were multiple fractures, malignancies, pathologic fracture, fracture due to high energy trauma, infection, and direct trauma. Patient demographic data such as age, sex, body mass index (BMI), smoking history, old OVF, level of fracture, and VAS score were analyzed. BMI was calculated as body weight in kilograms divided by the square of the body height in meters (kg/m2). VAS was used to assess back pain severity, which the patient complained about after the injury in the first 2 weeks during the 6-month follow-up.
The four poor prognosis outcomes used to determine the impact of the PVM at the 6-month follow-up were as follows: 1, severe vertebral compression (percentage of), defined as a decrease in vertebral body height of > 40% [25]; 2, delayed union (percentage of), defined by confirming the instability at fractured vertebra using dynamic X-ray at the 6-month follow-up; 3, new OVF, which comprised the detection of another fracture in addition to the previous fracture using MRI; and 4, remaining LBP, scored by patients as > 40 mm on the VAS at the 6-month follow-up.
The patients’ CSA, relative CSR (rCSA), and FI% in the thoracolumbar and lower lumbar regions were measured by MRI at enrollment and a the 6-month follow-up. X-ray was performed at enrollment and the 6-month follow-up.
Treatment by brace was continued for 2–3 months, and soft and hard braces were prescribed for 60% and 40% of the patients, respectively. The patients were allowed to be mobilized into an erect posture as the brace was applied. Additionally, patients were prescribed anti-osteoporotic and pain relief medication.
Imaging assessment
All patients were examined by plain X-rays and MRIs of the spine at the time of enrollment (during the first 2 weeks after the onset of fracture) and at the 6-month follow-up, and two authors (S.T and M. H, spine surgeons with 10 and 18 years of experience in spinal MRI, respectively) assessed the findings. Plain X-rays were taken in sagittal view in both the supine and weight-bearing positions. The relative height of the anterior wall (%) was calculated by the formula: {2 × affected vertebral height / (lower vertebral height + upper vertebral height)} × 100 [24].
When either the cranial or caudal adjacent vertebral body was deformed due to an old fracture, the vertical height of the anterior wall of the fractured vertebral body was divided by the vertical height of the anterior wall of the undeformed adjacent vertebral body. Delayed union was defined by a recognizable intravertebral cleft on plain X-rays at the 6-month follow-up. Dual-energy X-ray absorptiometry was used to measure the bone mineral density (BMD) of the mean femoral neck at the time of enrollment in all patients. This detailed setup method was not unified due to the multicenter study.
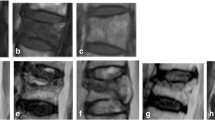
Our previous study demonstrated that MRI can provide better contrast compared to computed tomography; however, standard spin-echo T1-weighted sequences only provide a qualitative assessment of fat, which appears white, compared with muscle, which in this sequence is dark. The extent of larger agglomerations of adipose tissue can be measured, but the true fat content of muscle cannot be determined from T1-weighted images because the gray values of the muscle voxels do not scale in a known way with the fat content [26]. The reliability of MRI for measuring the CSA and FI% of the PVM has been reported to be acceptable [27]. In the present study, two institutions used a 1.5-T MRI scanner, while the remaining institutions used a 3.0-T MRI scanner. The following sequences were obtained with the MRI scanners: T1-weighted turbo spin echo with a slice thickness of 3 mm (repetition time [TR] 400–700 ms and time to echo [TE] minimum accessible, depending on the machine capability), T2-weighted turbo spin-echo with a slice thickness of 3 mm (TR 3000–4500 ms and TE 80–120 ms), and a fat saturation STIR sequence (TR 2000–4000 ms, TE 60–80 ms, and inversion time 120–170 ms) [28]. Patients’ CSA, rCSA, and FI% were measured at two different levels: thoracolumbar level (T12/L1) and lower lumbar level (L4/5). The multifidus (MF) and erector spinae (ES) were measured in the thoracolumbar and lower lumbar regions, which were chosen as the superior endplate of the L1 and L5 vertebra (Fig. 1). The CSA in cm2 was calculated as the average of the right and left PVM regions of interest of the axial T2-weighted MRI. The rCSA was calculated as the CSA of the PVM divided by the whole vertebral body area. The FI% was calculated as the ratio of the fat signal divided by the CSA of the muscle, multiplied by 100 (fat/CSA × 100). The CSAs of the muscles were outlined by measuring the borders of the muscles at two different levels.
Data analysis
The χ2 test or Fisher’s exact test was used for categorical variables and the t test was used for continuous variables. Analysis of covariance was used to compare the difference in PVM for each outcome. The models were adjusted for age, sex, and variables (severe vertebral compression, delayed union, new OVF, and remaining LBP) with a p value of < 0.10. Statistical test results were considered significant at p < 0.05. All p values were two-sided and all analyses were performed using SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA).
Results
Overall, 153 patients completed the 6-month follow-up, and 117 patients’ data were eligible for this study. The mean patient age was 79.1 ± 7.2 years, and 94 were female (80.3%). Forty-one patients had old OVF among the 117 patients (Table 1). OVFs were recorded in 10 patients at the thoracic level (T5–T9) (8.5%), 87 at the thoracolumbar level (T10–L2) (71.8%), and 24 at the lumbar level (19.7%). The average VAS at the first visit was 66.5 ± 12.5, and 23.1 ± 25.3 at the 6-month follow-up, and the patients’ BMD score was - 2.5 ± 0.4.
Severe vertebral compression was recorded in 63 patients (53.8%). There was no significant difference between the thoracolumbar region and lower lumbar region in terms of the CSA, rCSA, and FI% (Table 2). Delayed union occurred in 37 patients (32.5%). There was no significant difference in CSA, rCSA, and FI% of the PVM between the thoracolumbar (L1) and lumbar (L5) regions (Table 3). At the 6-month follow-up, 27 patients (23%) had remaining LBP. There were no significant differences in the CSA and rCSA between the thoracolumbar and lower lumbar regions. However, increased FI% of the PVM was significantly correlated with remaining LBP in the lumbar (L5) region (Table 4). Eleven patients (9.4%) had new OVF at the 6-month follow-up. The FI% of the PVM showed a significant correlation with new OVF in the thoracolumbar region (Table 5). Over 80% of both old and new OVF occurred in the thoracolumbar region.
Discussion
To the best of our knowledge, this is the first study to show the impact of the FI% in the PVM in two different regions. In the thoracolumbar region, an increase in PVM FI% was significantly related to the occurrence of new OVF and to the remaining LBP in the lower lumbar region. However, PVM had no effect on severe compression fracture or delayed union.
Regarding the CSA of the PVM, there was no significant difference as the CSA is unlikely to reflect early change in the PVMs compared with the FI% [23]. Shahidi et al. [29] demonstrated that there was no change in the CSA with age in either sex (p > 0.05), although there was an increase in the fat signal fraction with age in the ES and MF muscles in both sexes (p < 0.001). Moreover, in a cross-sectional study of 72 patients with LBP, Teichtahl et al. demonstrated that paraspinal FI%, but not muscle CSA, was associated with disability and structural abnormalities in the lumbar spine [8]. Similarly, we found that the FI% of the PVM, unlike the CSA and rCSA, showed a significant relationship with the occurrence of new OVF in the thoracolumbar region and with remaining LBP in the lower lumbar region. A previous study reported that the FI% of the muscles can lead to the hip fractures rather than fall-induced vertebral fracture [26], and this may explain our findings. However, we did not have access to the data regarding which participants were prone to direct trauma or hip fracture due to a fall.
Severe vertebral compression fracture and delayed union did not show any correlation with the CSA, rCSA, and FI% of the PVM in both regions. Severe vertebral compression fracture and delayed union might be affected by other causes rather than the CSA, rCSA, and FI% of the PVM. Intravertebral cleft, AO types A2 and A4 (AO Spine Thoracolumbar Spine Injury Classification: predictive for a progressive collapse in acute osteoporotic compression fractures), thoracolumbar level posterior wall injury, T1 or T2 diffuse low-signal change, and T2 diffuse low- or high-signal change on MRI have been previously reported as risk factors for vertebral compression [28, 30]. Additionally, Kim et al. and Hoshino et al. clearly demonstrated that the type of conservative treatment has no impact on vertebral compression and nonunion. Therefore, the morphological characteristics at injury are as important as severe compression and nonunion [31, 32]. Furthermore, spinal compression results from the interaction of the gravity force, ground reaction force, and force created by ligaments and muscles. Meanwhile, the thoracic compression force is greater due to body weight and kyphotic angle. Moreover, the gravity line falls anterior to the thoracic spine, causing flexion movement, which is counteracted by posterior extensor muscles and ligaments [33]. Harrison et al. reported that anterior translated posture, disc load, and stresses increase below the T9 level, and the posterior extensor muscle is required to maintain the static equilibrium balance [34].
Regarding the remaining LBP, previous studies have demonstrated the importance of the FI% of the PVM, which was related to the intensity of pain/disability and structural abnormality at the L3/L4 intervertebral disc level [8]. Furthermore, Paalanne et al. [35] previously reported the occurrence of back pain in patients with poor PVM mass due to an increase in the FI% regardless of the CSA of the PVM. The results of the present study are somewhat consistent with the findings of previous studies; however, our study differs from others in that separate regions were investigated (thoracolumbar and lumbar regions), with remaining LBP showing a significant correlation with the FI% of the PVM in the lumbar region (L4/5). In support of our results, the difference in the biomechanics of the PVM in the lumbar region has been previously demonstrated; powerful muscle must be the result of the continuity of a thick tendon, in order to transmit huge forces [36]. Given their large volume, lumbar PVM are considered powerful muscles [37, 38].
The occurrence of new OVF in the current study showed a significant relationship with the FI% of the PVM in the thoracolumbar region. Katsu et al. [18] reported that the FI% of the ES and MF muscles was related to the union of OVF at the L3 level. Similarly, Kim et al. [4] demonstrated that the increase in the FI% and decrease in the CSA at the L3/L4 level were associated with post-menopausal OVF. Additionally, Hori et al. [7] emphasized the importance of trunk muscle mass. Pogrund et al. [39] reported the importance of the decrease in the psoas at the L3 level related to osteoporosis. The current study demonstrated that the FI% of the PVM was related to remaining LBP in the lower lumbar region, and the FI% of the MF and ES at the upper endplate of L1, and absence of the psoas muscle, might be responsible for the occurrence of both old and new OVF.
The present study demonstrates the importance of MRI for accurate assessment of the FI% of the PVM in elderly patients. It is recommended that the clinician pays close attention to the follow-up of these patients and be aware of potentially new OVF, because the initiation of new treatment may decrease the risk of new OVF. The use of a brace or physical therapy may be beneficial, but requires further study.
There are several limitations of this study. First, the levels of vitamin D and parathyroid hormone were not checked in patients with low levels of anabolic hormones [8]. Second, the control patients without OVF were excluded from this study. It was unclear if the FI% of the PVM decreases due to age or other causes. Third, the prior cause of LBP was not assessed in the enrolled patients to ascertain whether the cause of the remaining LPB was due to sequels of OVF or other prior causes.
Regarding the clinical relevance of this study, the findings can serve as a guide for multi-field physicians to make earlier decisions regarding the treatment and prevention of OVF and LMP in elderly men and post-menopausal women upon detection of high FI% in axial T2-weighted MRI scanning.
In conclusion, this study demonstrates that the FI% of the PVM in the thoracolumbar region is highly correlated with the occurrence of new OVF, and the FI% of the PVM in the lumbar region is related to remaining LBP.
References
Hebert JJ, Kjaer P, Fritz JM, Walker BF (2014) The relationship of lumbar multifidus muscle morphology to previous, current, and future low back pain: a 9-year population-based prospective cohort study. Spine (Phila Pa 1976) 39:1417–1425. https://doi.org/10.1097/BRS.0000000000000424
Yagi M, Hosogane N, Watanabe K, Asazuma T, Matsumoto M, Keio Spine Research Group (2016) The paravertebral muscle and psoas for the maintenance of global spinal alignment in patient with degenerative lumbar scoliosis. Spine J 16:451–458. https://doi.org/10.1016/j.spinee.2015.07.001
Digirolamo DJ, Kiel DP, Esser KA (2013) Bone and skeletal muscle: neighbors with close ties. J Bone Miner Res 28:1509–1518. https://doi.org/10.1002/jbmr.1969
Kim JY, Chae SU, Kim GD, Cha MS (2013) Changes of paraspinal muscles in postmenopausal osteoporotic spinal compression fractures: magnetic resonance imaging study. 75–81
Choi MK, Kim SB, Park CK, Malla HP, Kim SM (2017) Cross-sectional area of the lumbar spine trunk muscle and posterior lumbar interbody fusion rate: a retrospective study. Clin Spine Surg 30:E798–E803. https://doi.org/10.1097/BSD.0000000000000424
Larsson L, Degens H, Li M, Salviati L, Lee Y, Thompson W, Kirkland JL, Sandri M (2019) Sarcopenia: aging-related loss of muscle mass and function. Physiol Rev 99:427–511. https://doi.org/10.1152/physrev.00061.2017
Hori Y, Hoshino M, Inage K, Miyagi M, Takahashi S, Ohyama S, Suzuki A, Tsujio T, Terai H, Dohzono S, Sasaoka R, Toyoda H, Kato M, Matsumura A, Namikawa T, Seki M, Yamada K, Habibi H, Salimi H, Yamashita M, Yamauchi T, Furuya T, Orita S, Maki S, Shiga Y, Inoue M, Inoue G, Fujimaki H, Murata K, Kawakubo A, Kabata D, Shintani A, Ohtori S, Takaso M, Nakamura H (2019) ISSLS PRIZE IN CLINICAL SCIENCE 2019: clinical importance of trunk muscle mass for low back pain, spinal balance, and quality of life—a multicenter cross-sectional study. Eur Spine J 28:914–921. https://doi.org/10.1007/s00586-019-05904-7
Teichtahl AJ, Urquhart DM, Wang Y, Wluka AE, Wijethilake P, O'Sullivan R, Cicuttini FM (2015) Fat infiltration of paraspinal muscles is associated with low back pain, disability, and structural abnormalities in community-based adults. Spine J 15:1593–1601. https://doi.org/10.1016/j.spinee.2015.03.039
Johnell O, Kanis J (2005) Epidemiology of osteoporotic fractures. In: Osteoporosis International. Springer, pp S3–S7
Statistics Bureau Home Page. http://www.stat.go.jp/english/. Accessed 6 Apr 2020
Pfirrmann CWA, Metzdorf A, Zanetti M, Hodler J, Boos N (2001) Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine (Phila Pa 1976) 26:1873–1878. https://doi.org/10.1097/00007632-200109010-00011
Chen P, Krege JH, Adachi JD, Prior JC, Tenenhouse A, Brown JP, Papadimitropoulos E, Kreiger N, Olszynski WP, Josse RG, Goltzman D, the CaMOS Research Group (2009) Vertebral fracture status and the World Health Organization risk factors for predicting osteoporotic fracture risk. J Bone Miner Res 24:495–502. https://doi.org/10.1359/jbmr.081103
Huang C (1996) Vertebral fracture and other predictors of physical impairment and health care utilization. Arch Intern Med 156:2469–2475. https://doi.org/10.1001/archinte.1996.00440200087011
Kado DM (1999) Vertebral fractures and mortality in older women. Arch Intern Med 159:1215–1220
Tsai AG, Bessesen DH (2019) Annals of internal medicine. Ann Intern Med 170:ITC33–ITC48. https://doi.org/10.7326/AITC201903050
Oner FC, Van Gils APG, Dhert WJA, Verbout AJ (1999) MRI findings of thoracolumbar spine fractures: a categorisation based on MRI examinations of 100 fractures. Skeletal Radiol 28:433–443. https://doi.org/10.1007/s002560050542
Cho T, Matsuda M, Sakurai M (1996) MRI findings on healing process of vertebral fracture in osteoporosis. J Orthop Sci 1:16–33. https://doi.org/10.1007/bf01234112
Katsu M, Ohba T, Ebata S, Haro H (2018) Comparative study of the paraspinal muscles after OVF between the insufficient union and sufficient union using MRI. BMC Musculoskelet Disord 19:143. https://doi.org/10.1186/s12891-018-2064-0
Huang CWC, Tseng IJ, Yang SW, Lin YK, Chan WP (2019) Lumbar muscle volume in postmenopausal women with osteoporotic compression fractures: quantitative measurement using MRI. Eur Radiol 29:4999–5006. https://doi.org/10.1007/s00330-019-06034-w
Briggs AM, Greig AM, Bennell KL, Hodges PW (2007) Paraspinal muscle control in people with osteoporotic vertebral fracture. Eur Spine J 16:1137–1144. https://doi.org/10.1007/s00586-006-0276-8
Cunha-Henriques S (2011) Postmenopausal women with osteoporosis and musculoskeletal status: a comparative cross-sectional study. J Clin Med Res. https://doi.org/10.4021/jocmr537w
Sinaki M, Khosla S, Limburg PJ, Rogers JW, Murtaugh PA (1993) Muscle strength in osteoporotic versus normal women. Osteoporos Int 3:8–12. https://doi.org/10.1007/BF01623170
Takahashi S, Hoshino M, Takayama K, Sasaoka R, Tsujio T, Yasuda H, Kanematsu F, Kono H, Toyoda H, Ohyama S, Hori Y, Nakamura H (2020) The natural course of the paravertebral muscles after the onset of osteoporotic vertebral fracture. Osteoporos Int 31:1089–1095. https://doi.org/10.1007/s00198-020-05338-8
Takahashi S, Hoshino M, Takayama K, Iseki K, Sasaoka R, Tsujio T, Yasuda H, Sasaki T, Kanematsu F, Kono H, Toyoda H, Nakamura H (2017) Time course of osteoporotic vertebral fractures by magnetic resonance imaging using a simple classification: a multicenter prospective cohort study. Osteoporos Int 28:473–482. https://doi.org/10.1007/s00198-016-3737-x
Genant HK, Wu CY, van Kuijk C, Nevitt MC (1993) Vertebral fracture assessment using a semiquantitative technique. J Bone Miner Res 8:1137–1148. https://doi.org/10.1002/jbmr.5650080915
Engelke K, Museyko O, Wang L, Laredo JD (2018) Quantitative analysis of skeletal muscle by computed tomography imaging—state of the art. J Orthop Transl 15:91–103. https://doi.org/10.1016/j.jot.2018.10.004
Hu ZJ, He J, Zhao FD, Fang XQ, Zhou LN, Fan SW (2011) An assessment of the intra- and inter-reliability of the lumbar paraspinal muscle parameters using CT scan and magnetic resonance imaging. Spine (Phila Pa 1976) 36:868–874. https://doi.org/10.1097/BRS.0b013e3181ef6b51
Takahashi S, Hoshino M, Takayama K, Iseki K, Sasaoka R, Tsujio T, Yasuda H, Sasaki T, Kanematsu F, Kono H, Toyoda H, Nakamura H (2016) Predicting delayed union in osteoporotic vertebral fractures with consecutive magnetic resonance imaging in the acute phase: a multicenter cohort study. Osteoporos Int 27:3567–3575. https://doi.org/10.1007/s00198-016-3687-3
Shahidi B, Parra CL, Berry DB, Hubbard JC, Gombatto S, Zlomislic V, Allen RT, Hughes-Austin J, Garfin S, Ward SR (2017) Contribution of lumbar spine pathology and age to paraspinal muscle size and fatty infiltration. Spine (Phila Pa 1976) 42:616–623. https://doi.org/10.1097/BRS.0000000000001848
Muratore M, Ferrera A, Masse A, Bistolfi A (2018) Osteoporotic vertebral fractures: predictive factors for conservative treatment failure. A systematic review. Eur Spine J 27:2565–2576
Kim HJ, Yi JM, Cho HG, Chang BS, Lee CK, Kim JH, Yeom JS (2014) Comparative study of the treatment outcomes of osteoporotic compression fractures without neurologic injury using a rigid brace, a soft brace, and no brace: a prospective randomized controlled non-inferiority trial. J Bone Jt Surg - Am Vol 96:1959–1966. https://doi.org/10.2106/JBJS.N.00187
Hoshino M, Tsujio T, Terai H, Namikawa T, Kato M, Matsumura A, Suzuki A, Takayama K, Takaoka K, Nakamura H (2013) Impact of initial conservative treatment interventions on the outcomes of patients with osteoporotic vertebral fractures. Spine (Phila Pa 1976) 38:E641–E648. https://doi.org/10.1097/BRS.0b013e31828ced9d
Rathore M, Sinha MB, Trivedi S, Sharma DK (2014) A focused review – thoracolumbar spine : Anatomy , Biomechanics and Clinical Significance. Indian J Clin Anat Physiol 1:41–46
Harrison DE, Colloca CJ, Harrison DD, Janik TJ, Haas JW, Keller TS (2005) Anterior thoracic posture increases thoracolumbar disc loading. Eur Spine J 14:234–242. https://doi.org/10.1007/s00586-004-0734-0
Paalanne N, Niinimäki J, Karppinen J, Taimela S, Mutanen P, Takatalo J, Korpelainen R, Tervonen O (2011) Assessment of association between low back pain and paraspinal muscle atrophy using opposed-phase magnetic resonance imaging: a population-based study among young adults. Spine (Phila Pa 1976) 36:1961–1968. https://doi.org/10.1097/BRS.0b013e3181fef890
Cutts A, Alexander RM, Ker RF (1991) Ratios of cross-sectional areas of muscles and their tendons in a healthy human forearm. J Anat 176:133–137
Hansen L, de Zee M, Rasmussen J, Andersen TB, Wong C, Simonsen EB (2006) Anatomy and biomechanics of the back muscles in the lumbar spine with reference to biomechanical modeling. Spine (Phila Pa 1976) 31:1888–1899. https://doi.org/10.1097/01.brs.0000229232.66090.58
Kalimo H, Rantanen J, Viljanen T, Einola S (1989) Lumbar muscles: structure and function. Ann Med 21:353–359. https://doi.org/10.3109/07853898909149220
Pogrund H, Bloom RA, Weinberg H, et al (2009) Relationship of psoas width to osteoporosis. 6470:8–11. https://doi.org/10.3109/17453678608994377
Funding
This study was funded by the Grant of Japan Orthopedics and Traumatology Research Foundation (grant no. 270).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Ethical approval
This study was approved by the Ethical Committee of Osaka City University. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Habibi, H., Takahashi, S., Hoshino, M. et al. Impact of paravertebral muscle in thoracolumbar and lower lumbar regions on outcomes following osteoporotic vertebral fracture: a multicenter cohort study. Arch Osteoporos 16, 2 (2021). https://doi.org/10.1007/s11657-020-00866-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-020-00866-6