Abstract
Objectives
To develop a new Chinese medicine (CM)-based drug and to evaluate its safety and effect for suppressing acute respiratory distress syndrome (ARDS) in COVID-19 patients.
Methods
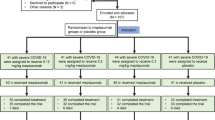
A putative ARDS-suppressing drug Keguan-1 was first developed and then evaluated by a randomized, controlled two-arm trial. The two arms of the trial consist of a control therapy (alpha interferon inhalation, 50 µg twice daily; and lopinavir/ritonavir, 400 and 100 mg twice daily, respectively) and a testing therapy (control therapy plus Keguan-1 19.4 g twice daily) by random number table at 1:1 ratio with 24 cases each group. After 2-week treatment, adverse events, time to fever resolution, ARDS development, and lung injury on newly diagnosed COVID-19 patients were assessed.
Results
An analysis of the data from the first 30 participants showed that the control arm and the testing arm did not exhibit any significant differences in terms of adverse events. Based on this result, the study was expanded to include a total of 48 participants (24 cases each arm). The results show that compared with the control arm, the testing arm exhibited a significant improvement in time to fever resolution (P=0.035), and a significant reduction in the development of ARDS (P=0.048).
Conclusions
Keguan-1-based integrative therapy was safe and superior to the standard therapy in suppressing the development of ARDS in COVID-19 patients. (Trial registration No. NCT 04251871 at www.clinicaltrials.gov)
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Wuhan Municipal Health Commission. Report of clustering pneumonia of unknown etiology in Wuhan City. Dec 31, 2019. Availabe at: http://wjw.wuhan.gov.cn/front/web/showDetail/2019123108989 Accessed Feb 29, 2020).
China National Emergency Office. Prevention and control of the pneumonia caused by a new coronavirus. Jan 21, 2020. Availabe at: http://www.nhc.gov.cn/yjb/s7860/202001/d9570f3a52614113ae0093df51509684.shtml (Accessed Feb 29, 2020).
WHO. Naming the coronavirus disease (COVID-19) and the virus that causes it. Availabe at: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it (Accessed Feb 29, 2020).
Anderson RM, Fraser C, Ghani AC, Donnelly CA, Riley S, Ferguson NM, et al. Epidemiology, transmission dynamics and control of SARS: the 2002–2003 epidemic. Philos Trans R Soc Lond B Biol Sci 2004;359:1091–1105.
Mackay IM, Arden KE. MERS coronavirus: diagnostics, epidemiology and transmission. Virol J 2015;22:222.
Al-Omari A, Rabaan AA, Salih S, Al-Tawfiq JA, Memish ZA. MERS coronavirus outbreak: implications for emerging viral infections. Diagn Microbiol Infect Dis 2019;93:265–285.
Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med 2000;342:1334–1349.
Bauer TT, Ewig S, Rodloff AC, Müller EE. Acute respiratory distress syndrome and pneumonia: a comprehensive review of clinical data. Clin Infect Dis 2006;43:748–756.
Labonté P, Seidah NG. Emerging viruses: risk of pandemic. Expert Rev Anti Infect Ther 2008;6: 581–583.
Adalja A, Inglesby T. Broad-spectrum antiviral agents: a crucial pandemic tool. Expert Rev Anti Infect Ther 2019;17:467–470.
Kil HR, Lee JH, Lee KY, Rhim JW, Youn YS, Kang JH. Early corticosteroid treatment for severe pneumonia caused by 2009 H1N1 influenza virus. Crit Care 2011;15:413.
Lee KY. Pneumonia, acute respiratory distress syndrome, and early immune-modulator therapy. Int J Mol Sci 2017;18:388.
Chan K. Progress in traditional Chinese medicine. Trends Pharmacol Sci 1995;16:182–187.
Chen Y, Liu Q, Xie H, Yin SM, Wu L, Yu XH, et al. Is Chinese medicine injection applicable for treating acute lung injury and acute respiratory distress syndrome? A systematic review and meta-analysis of randomized controlled trials. Chin J Integr Med 2019. [Epub ahead of print]
Luo H, Tang QL, Shang YX, Liang SB, Yang M, Robinson N, et al. Can Chinese medicine be used for prevention of corona virus disease 2019 (COVID-19)? A review of historical classics, research evidence and current prevention programs. Chin J Integr Med 2020;26:243–250.
Li N, Gao F. Viewpoint regarding “concise” of Jing-Fang from case. China J Chin Mater Med (Chin) 2019;44:3899–3902.
National Health Commission of the People’s Republic of China. Directive for the diagnosis and recommended treatments for COVID-19 (3rd ed). Jan 22, 2020. Available at: http://www.nhc.gov.cn/yzygj/s7653p/202001/f492c9153ea9437bb587ce2ffcbee1fa.shtml (Accessed Jan 23, 2020).
U.S. Department of Health and Human Services. Common terminology criteria for adverse events (CTCAE) Version 5.0. Available at: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_v5_Quick_Reference_5x7.pdf.
ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, Thompson BT, Furguson ND, Caldwell E, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA 2012;307:2526–2533.
Riviello ED, Kiviri W, Twagirumugabe T, Mueller A, Banner-Goodspeed VM, Officer L, et al. Hospital incidence and outcomes of the acute respiratory distress syndrome using the kigali modification of the Berlin definition. Am J Respir Crit Care Med 2016;193:52–59.
Habibzadeh P, Stoneman EK. The novel coronavirus: a bird’s eye view. Int J Occup Environ Med 2020;11:65–71.
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395:497–506.
Yang XB, Yuan Y, Xu JQ, Shu H, Xia J, Liu H, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med 2020:30079–5. [Epub ahead of print]
Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med 2020;382:1199–1207.
Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A trial of lopinavir-ritonavir in adults hospitalized with severe COVID-19. N Engl J Med 2020:NEJMoa2001282. [Epub ahead of print]
Peeri NC, Shrestha N, Rahman MS, Zaki R, Tan Z, Bibi S, et al. The SARS, MERS and novel coronavirus (COVID-19) epidemics, the newest and biggest global health threats: what lessons have we learned? Int J Epidemiol 2020:dyaa033. [Epub ahead of print]
Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020:NEJMoa2002032. [Epub ahead of print]
Poon PMK, Wong CK, Fung KP, Fong CYS, Wong ELY, Lau JTF, et al. Immunomodulatory effects of a traditional Chinese medicine with potential antiviral activity: a self-control study. Am J Chin Med 2006;34:13–21.
Cheng L, Li F, Ma R, Hu XP. Forsythiaside inhibits cigarette smoke-induced lung inflammation by activation of Nrf2 and inhibition of NF-κB. Int Immunopharmacol 2015;28:494–499.
Park SY, Jin ML, Yi EH, Kim Y, Park G. Neochlorogenic acid inhibits against LPS-activated inflammatory responses through up-regulation of Nrf2/HO-1 and involving AMPK pathway. Environ Toxicol Pharmacol 2018;62:1–10.
Čivljak R, Markotić A, Kuzman I. The third coronavirus epidemic in the third millennium: what’s next? Croat Med J 2020;61:1–4.
Acknowledgements
The authors acknowledge Academician WANG Fu-sheng for his advice to the trial.
Author information
Authors and Affiliations
Contributions
Wang JB, Wang ZX, and Jing J conducted the study and wrote the manuscript, they contributed equally to this work as co-first authors. Wang ZX, Jing J, Zhao P, Dong JH, Zhou YF, Yang G, Jiang TJ, Xu Z, Wu D, Sun YQ, Wang JB, He DC, and Chen Z collected data and provided technical assistance. Jing J, Yu SM, Niu M, Zhao X, Zhang P, Song XA, Bi JF, Bai ZF, Guo YM, Zhao X, Zhao PF, Tang JY, and Li PY analyzed data. Wang JB, Wang RL, Qin EQ, and Xiao XH initiated and oversaw the study.
Corresponding authors
Additional information
Conflict of interest
None declared.
Data Sharing Statements
See Appendix 10.
Supported by the National Administration of Traditional Chinese Medicine Project (No. 2020ZYLCYJ05-1), the National Natural Science Foundation of China (Nos. 81630100, 81721002, 81930110), China PLA Biosecurity Project (No. 19SWAQ13), China PLA Emergency Project (BWS20J006) and the Beijing Administration of Traditional Chinese Medicine Project (Nos. YJ2020-03, SYFY202011)
Supplementary Material
Rights and permissions
About this article
Cite this article
Wang, Jb., Wang, Zx., Jing, J. et al. Exploring an Integrative Therapy for Treating COVID-19: A Randomized Controlled Trial. Chin. J. Integr. Med. 26, 648–655 (2020). https://doi.org/10.1007/s11655-020-3426-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-020-3426-7