Abstract
Objective
To test the in vitro antimicrobial efficacy of water and methanol extracts of 23 plant species that are commonly used in Libyan folk medicine.
Methods
The antimicrobial activity was determined using the well-diffusion method. Four test microorganisms were used namely, Escherichia coli, Salmonella species, Staphylococcus aureus and Bacillus subtilis. The minimum inhibitory concentration (MIC) was determined for the high biologically active crude plant extracts.
Results
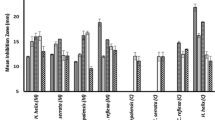
Among 23 medicinal plants used in the study, only 5 methanolic extracts [Rosmarinus offcinalis L., Carduus marianium L., Lantana camara L., Rhus tripartite (ueria) Grande, and Thymus capitatus (L.) Hoffm (link)] showed the highest antimicrobial activity against Staphylococcus aureus, Bacillus subtilis and Salmonella species, while 22 methanolic and aqueous extracts showed moderate to weak antimicrobial activity on all tested organisms. However 19 of the extracts showed no activity at all against Gram–ve and Gram +ve microorganisms. MIC was found to be 1.25 mg/mL (Thymus capitatus), 3 mg/mL (Rhus tripartite), 4 mg/mL (Carduus marianium), 5 mg/mL (Rosamarinus officinalis) and 5 mg/mL (Lantana camara), respectively.
Conclusions
The present results revealed that, crude methanolic extracts of the investigated Libyan folk medicinal plants exhibited mild to high in vitro antibacterial activities against Gram-positive and Gram-negative microorganisms.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Doit C, Barre J, Cohen R, Bonacorsi S, Bourrillon A, Bingen E. Bactericidal activity against intermediately cephalosporinresistant Streptococcus pneumoniae in cerebrospinal fluid of children with bacterial meningitis treated with high doses of cefotaxime and vancomycin. Antimicrob Agents Chemother 1997;41:2050–2052.
Farnsworth NR, Morris RW. Higher plants: the sleeping giant of drug development. Am J Pharm 1976;148:46–50.
Farnsworth NR. The role of medicinal plants in drug development. In: Natural products and drug developments. Krogsraard-Larsen P, Christensen SB, Kofod H, eds. [London: Ballier, Tindall and Cox; 1948:9–98.
Mayer BN, Ferrigni NR, Putnam JE, Nicolas PE, Mcaughin JL. Brine shrimp: a convenient bioassay for active plant constituents. Planta Med 1982;45:31–34.
Kivack B, Mert T, Tansel H. Antimicrobial and cytotoxic activities of Ceratonia siliqua L. extracts. Turkish J Biol 2001;26:197–200.
Essawi T, Srour M. Screening of some Palestinian medicinal plants for antibacterial activity. J Ethnopharmacol 2000;70:343–349.
Mehtab P, Ali MM, Mahbbob A, Faheem A, Pedro SPS, Manuela RS. Two new phenolic compounds from Ficus rumphii and their antiproliferative activity. J Nat Prod Res 2014;28:646–652.
Iwu MM, Duncan AR, Okunji CO. New antimicrobials of plant origin. In: Janick J, ed. Perspectives on new crops and new uses. Alexandria: ASHS Press; 1999:457–462.
Okeke IN, Laxmaninarayan R, Bhutta ZA, Duse AG, Jenkins P, O'Brien TF, et al. Antimicrobial resistance in developing countries. Part 1: Recent trends and current status. Lancet Infect Dis 2005;5:481–493.
Melendez PA, Capriles VA. Antibacterial properties of tropical plants from Puerto Rico. Phytomedicine 2006;13:272–276.
Janovska D, Kubikova K, Kokoska L. Screening for antimicrobial activity of some medicinal plants of traditional Chinese medicine. Czech J Food Sci 2003;21:107–110.
Carro RA, Maran JM, Fernandozalgo LM, Dominguez AA. Antimicrobial properties of some extracts obtained from some mediterranean plants of medicinal value. Plant Med Phyth 1987;2:195–202.
Kotb THF. Medicinal plants in Libya, part II, 1st ed. South Mount-Lebanon: Arab Encyclopedia House; 1985:142–822.
Ezoubeiri A, Gadhi CA, Fdil N, Benharref A, Jana M, Vanhaelen, M. Isolation and antimicrobial activity of two phenolic compounds from Pulicaria Odora L. J Ethnopharmacol 2005;287–292.
National Committee for Clinical Laboratory Standards. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard Document M7-A4, 4th ed. Wayne; Pa. 1997.
Remmal A, Bouchikhi T, Tantaoui-Elaraki A, Ettayebi M. Inhibition of antibacterial activity of essential oils by tween 80 and ethanol in liquid medium. J Pharm Belg 1993;48:352–356.
Van der Watt E, Pretorius JC. Purification and identification of active antibacterial components, In: Carpobrotus Edulis L. J Ethnopharmacol 2001;76:87–91.
Sateesh K, Marimuthua R, Thayumanavanb R, Nandakumara S. Antimicrobial activity and induction of systemic resistance in rice by leaf extract of Datura Metel against Rhizoctonia Solani and Xanthomonas Oryzae pv. Oryzae. Physiol Mol Plant Pathol 2004;65: 91–100.
Ramzi AM, Lindequisth, U. Antimicrobial activity of some medicinal plants of Island Soqotra. J Ethnopharmacol 2005;96:177–181.
Adebolu TT, Salau AO. Antimicrobial activity of leaf extracts of Ocimum gratissimum on selected diarrhea causing bacteria in South Western Nigeria. Afr J Biotechnol 2005;4:682–684.
Diǧrak M, Alma MH, İlçim A, Şen S. Antibacterial and antifungal effects of various commercial plants extract. Pharmaceut Biol 1999;37:216–220.
Buruk K, Sokmen A, Aydin F, Erturk M. Antimicrobial activity of someendemic plants growing in the Eastern Black Sea Region. Turkey: Fitoterapia; 2006;77: 388–391.
Ekwenye UN, Elegalam NN. Antimicrobial activity of ginger (Zingiber officinale) Roscoe and Garlic (Allium sativum L.) extracts on Ecscherichia Coli and Salmonella Typhi. Internat J Mol Med Adv Sci 2005;1:411–416.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muhaisen, H.M., Ab–Mous, M.M., Ddeeb, F.A. et al. Antimicrobial agents from selected medicinal plants in Libya. Chin. J. Integr. Med. 22, 177–184 (2016). https://doi.org/10.1007/s11655-015-2172-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11655-015-2172-8