Abstract
Gastric cancer (GC) is a commonly occurring neoplasm worldwide. The occurrence of multidrug resistance (MDR) in GC cells is the main obstacle to effective GC chemotherapy. The aim of the present study was to reveal the functional role and the underlying mechanisms of basic leucine zipper ATF-like transcription factor 2 (BATF2), a novel tumor suppressor, on MDR in GC cells. Here, we first found that SGC7901/VCR and SGC7901/ADR cells had higher drug resistance than SGC7901 cells using methylthiazol tetrazolium (MTT) and flow cytometry analysis. Moreover, MDR-related proteins and Wnt/β-catenin pathway markers were all upregulated in SGC7901/VCR cells compared to SGC7901 cells by quantitative reverse transcription-PCR (qRT-PCR) and western blot analyses. Subsequently, we observed BATF2 was downregulated in SGC7901/VCR cells and BATF2 overexpression significantly induced cell cycle G0/G1 phase arrest and apoptosis. Furthermore, overexpression of BATF2 could suppress Wnt/β-catenin signaling and increase drug susceptibility by downregulating Wnt/β-catenin pathway markers. In addition, knockdown of β-catenin imitated the effects of BATF2 overexpression on drug susceptibility. Importantly, enhancing the Wnt/β-catenin pathway could reverse the inhibitory effects of BATF2 on MDR. In conclusion, BATF2 was downregulated in MDR GC cells and overexpression of BATF2 could reverse the MDR of GC cells by inactivating the Wnt/β-catenin pathway.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Worldwide, gastric cancer (GC) was the most prevalent type of cancer in the past, and today it is still the fourth most common type of cancer globally (Bray et al. 2013; Van Cutsem et al. 2016), as well as the second most prevalent cancer in China after lung cancer. In 2015, there were an estimated 498,000 deaths associated with GC in China (Chen et al. 2016). Despite crucial improvements in GC diagnosis strategies, the burden of GC is still high (Zhang and Ge 2013; Ferro et al. 2014; Zheng et al. 2017). Currently, adequate surgical removal of lesions at the early stages is still the only curative treatment for GC, while most patients who are diagnosed at advanced stages still have poor prognosis after operative treatment (Van Cutsem et al. 2011; Lutz et al. 2012; Zhang et al. 2018). Chemotherapy is another therapeutic option that can help eradicate malignant cells. However, the 5-yr survival rate of GC patients remains low and does not reach 30% as a result of the gradually developing resistance of GC cells to chemotherapy that occurs during treatment (Rugge et al. 2017; Eom et al. 2018; Wang et al. 2018). Multidrug resistance (MDR) in GC cells is deemed to be the primary cause of chemotherapy failure, accounting for the stubbornly high death rate observed in GC patients (Jemal et al. 2010; Fodale et al. 2011).
Despite a great number of studies involving MDR in neoplastic cells, GC MDR is still not fully understood. Therefore, it would be helpful to elucidate the molecular mechanisms underlying the initiation of MDR in GC cells and then solve the problem of refractory tumors. The ATP binding cassette (ABC) transporter is a critical mediator of drug efflux (Gillet et al. 2007). As an ABC transporter, P-glycoprotein (P-gp) is one of the most studied molecules in MDR and it can transport chemotherapeutic drugs out of cancer cells by means of ATP hydrolysis (Yuan et al. 2017). P-gp has been reported to usually be upregulated in cancer cells and is a key factor for the development of MDR (Sun et al. 2012).
The Wnt/β-catenin pathway is highly conserved throughout evolution. The proteins belonging to the Wnt family are important regulators of cell proliferation, differentiation, and adhesion (He et al. 1998; Tetsu and McCormick 1999; MacDonald et al. 2009). The Wnt signaling pathway plays a crucial role in regulating tumor initiation and over activation of the Wnt signaling pathway is common in several cancers (Klaus and Birchmeier 2008). Hence, research focusing on the Wnt signaling pathway has become a primary factor in studies of drug design. The β-catenin molecule is the key effector responsible for transduction of the signal to the nucleus in the Wnt pathway. When β-catenin accumulates in the cytoplasm, it is translocated to the nucleus, where it associates with DNA binding transcription factors of the T cell factor/lymphoid enhancer factor (TCF/LEF) and then activates the transcription of target genes (Valenta et al. 2012). Moreover, inactivation of the Wnt/β-catenin pathway can downregulate expression of P-gp in a variety of human cancers and can reverse MDR (Lim et al. 2008; Shen et al. 2013; Kageji et al. 2016).
Basic leucine zipper ATF-like transcription factor 2 (BATF2), a novel tumor suppressor, has been found to inhibit cancer growth and migration (Su et al. 2008) and to be a potential marker for diagnosis and prognosis of several cancers (Liu et al. 2015; Dai et al. 2016; Huang et al. 2017). Moreover, BATF2 was also suggested as a possible treatment for a wide variety of cancers (Guler et al. 2015). The mechanism responsible for the inhibitory effect of BATF2 on neoplasms is complex, involving the suppression of AP-1 in the network of transcriptional regulation (Tussiwand et al. 2012), inactivating Wnt/β-catenin pathway to downregulate P-gp (Huang et al. 2017).
Given the effect of BATF2 to inactivate the Wnt/β-catenin pathway, which could regulate the expression of P-gp, we hypothesized that overexpression of BATF2 may have the potential to reverse the MDR of GC by targeting the Wnt/β-catenin signaling pathway and suppressing the expression of P-gp. Therefore, in the present study, we investigated the effects of BATF2 on the Wnt/β-catenin pathway, P-gp, and the MDR of GC.
Materials and Methods
Cell cultures and treatment
The human gastric adenocarcinoma cell line SGC7901 and the MDR gastric adenocarcinoma cell lines SGC7901/VCR and SGC7901/ADR were obtained from the Shanghai Institute of Biochemistry and Cell Biology, Chinese Academy of Sciences (Shanghai, China). The cells were cultivated in RPMI 1640 medium (Gibco, Anaheim, CA) supplemented with 10% bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin in a humidified atmosphere with 5% CO2 at 37°C. For the SGC7901/VCR and SGC7901/ADR cell lines, an additional 0.8 μg/mL of vincristine and Adriamycin (Shenzhen Main Luck Pharmaceuticals, Guangdong, China) were added into the medium, to maintain drug resistance. Cisplatin (DDP), 5-fluorouracil (5-FU), and vincristine sulfate (VCR) were purchased from Sigma (St. Louis, MO).
Cell transfection
To obtain stable BATF2 overexpression, cells were transduced with the indicated lentiviral particles, followed by puromycin (Sigma). The BATF2 overexpression plasmid was created by cloning BATF2 cDNA into pcDNA3.1 (Invitrogen, Carlsbad, CA). BATF2 ectopic expression was achieved through pcDNA3.1-BATF2 transfection using Lipofectamine 2000 (Invitrogen). An empty pCDNA3.1 vector was used as a control. For β-catenin knockdown, Signal Silence β-catenin siRNA (siβ-catenin, #6225), purchased from Cell Signaling Technology (Boston, MA), transfected into SGC7901/VCR cells in a final concentration of 50 nM using Lipofectamine 2000 (Invitrogen).
Quantitative real-time transcription-PCR (qRT-PCR) analysis
Total RNA was isolated from two cell lines using Trizol reagent (Invitrogen) and reverse transcribed into cDNA using a Superscript First-Strand Synthesis System (Invitrogen) according to the manufacturer’s instructions. The primer sequences used in the present study are listed in Table 1. The relative expression of the mRNA level was analyzed based on the 2–ΔΔCt method. All the reactions were carried out in triplicate.
Methylthiazol tetrazolium assay
Cells were planted in a 96-well plate at a density of 2 × 103 cells per well for 72 h. Methylthiazol tetrazolium (MTT) (20 μL per well, Sigma) was added into each well (pH = 7.4) and then incubated at 37°C for 4 h. Then 100 μL acidic isopropanol (0.01 mol/L HCl, 10% SDS, and 5% isopropanol) was added to terminate the reaction. The absorbency of test sample was detected with an ELISA reader (BioRad, Hercules, CA) at a wavelength of 595 nm.
Cell cycle and apoptosis analysis
For the cell cycle assay, cells were fixed with 70% ethanol at 4°C overnight, resuspended in 100 μg/mL RNase A (KeyGen Biotech, Nanjing, China) at 37°C for 30 min, and then stained with 50 μg/mL propidium iodide at 4°C for 30 min in the dark. The cells were analyzed by flow cytometry at a wavelength of 488 nm, and the data were analyzed using the CellFit software (BD Bioscience, San Jose, CA). Apoptosis was detected by an Annexin V-APC/7-AAD apoptosis detection kit (KeyGen Biotech) following the manufacturer’s instructions.
Western blotting
The protein used for western blot analysis was extracted using RIPA lysis buffer (Beyotime Biotechnology, Shanghai, China) supplemented with protease inhibitors (Roche, Basel, Switzerland). The proteins were quantified using BCA Protein Assay Kits (Pierce, Appleton, WI). The western blot system was established using a Bio-Rad Bis-Tris Gel System (Bio-Rad) according to the manufacturer’s instructions. The following antibodies were used: anti-BATF2 (diluted 1:1000), anti-TCF (diluted 1:500), anti-P-Glycoprotein antibody produced in mouse (diluted 1:1000), anti-β-catenin antibody produced in mouse (diluted 1:1000), anti-c-myc antibody produced in rabbit (diluted 1:1000), anti-cyclin D1 antibody produced in rabbit (diluted 1:1000), anti-GAPDH antibody produced in rabbit (0.1–0.2 μg/mL), and anti-Survivin antibody produced in rabbit (diluted 1:500). All of the antibodies were purchased from Sigma and were diluted 1:1000.
Statistical analysis
Statistical analyses were conducted using the SPSS 22.0 software (IBM Corp, Armonk, NY) and GraphPad Prism 7 (GraphPad, La Jolla, CA). All data were presented as mean ± SEM, and the Student’s t test was used to determine statistical significance. Comparisons between two groups were performed using paired t tests. A P value < 0.05 was considered statistically significant.
Results
Biological characteristics of SGC7901/VCR and SGC7901/ADR cells
The results of the MTT assay showed that the SGC7901/VCR and SGC7901/ADR cells grew much faster than the SGC7901 cells (Fig. 1A) in 5 d. The results of cell cycle analysis revealed that the SGC7901/VCR and SGC7901/ADR cells had significantly decreased G0/G1 phases and increased S and G2/M phases compared with the SGC7901 cells (Fig. 1B), indicating that the SGC7901/VCR and SGC7901/ADR cells had shorter cell cycles. In addition, after treatment with three common chemotherapeutics (DDP, VCR, and 5-FU), SGC7901/VCR and SGC7901/ADR cells had lower cell apoptotic rates than the SGC7901 cells (Fig. 1C). Thus, the SGC7901/VCR cells had higher drug resistance than the SGC7901/ADR cells by combining these above data and thus were selected for further in vitro experiments.
Biological characteristics in SGC7901/VCR and SGC7901/ADR cells. (A) Growth curve assayed by MTT. (B) Cell cycle distribution determined by flow cytometry. (C) Effects of three common chemotherapeutics (60 μM, 24 h) on the apoptosis of SGC7901/VCR and SGC7901/ADR cells by flow cytometry analysis. *P < 0.05, **P < 0.01, ***P < 0.001. DDP, cisplatin; VCR, vincristine sulfate; 5-FU, 5-fluorouracil.
Upregulation of the Wnt/β-catenin pathway contributed to MDR
Lung resistance protein (LRP), multidrug resistance protein 1 (MDR1), and multidrug resistance-associated protein 1 (MRP1) are three important MDR-related proteins. The mRNA expression levels of these proteins were significantly increased in the SGC7901/VCR cells compared to the SGC7901 cells (Fig. 2A). Meanwhile, the results of western blotting indicated that P-gp, β-catenin, c-myc, cyclin D1, and Survivin in the SGC7901/VCR and SGC7901/ADR cultures were overexpressed in the SGC7901/VCR cells (Fig. 2B).
MDR and Wnt/β-catenin pathway-related protein expression is upregulated in SGC7901/VCR and SGC7901/ADR cells. (A) Expression of MDR-associated genes detected by qRT-PCR. **P < 0.01. (B) Protein expression of P-gp, β-catenin, c-myc, cyclin D1, and Survivin measured by western blotting. MDR, multidrug resistance.
BATF2 overexpression increased and its silence decreased drug susceptibility
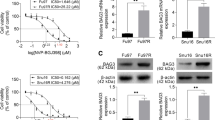
The mRNA expression of BATF2 in SGC7901/VCR cells was determined through qRT-PCR analysis and the BATF2 was downregulated in SGC7901/VCR cells compared to SGC7901 cells (Fig. 3A). After transfecting with the BATF2 overexpression plasmid, the transfection efficiency (shown in Fig. 3B) and BATF2 mRNA expression were significantly increased in the SGC7901/VCR cells (Fig. 3C). Furthermore, overexpression of BATF2 in the SGC7901/VCR cells led to a higher proportion of cells in G0/1 phase and cell cycle was arrested at S and G2/M phases (Fig. 3D). Overexpression of BATF2 also greatly enhanced cell apoptosis of the SGC7901/VCR cells with the different drug treatments (Fig. 3E). By transfecting with specific siRNA targeting BATF2 in the drug-sensitive SGC7901 cells, its expression was successfully significantly knocked down by three siRNAs (designated as siBATF2#1, siBATF2#2, and siBATF2#3), among which, siBATF2#3 showed the highest knockdown efficiency (Fig. 3F). Through MTT assay, we found that BATF2 silence by siBATF2#3 was able to enhance cell viability of SGC7901 cells. Compared with the cells treated with siNC+VCR, the cells treated with siBATF2#3+VCR showed higher viability, indicating that BATF2 knockdown is capable of enhancing drug resistance (Fig. 3G).
BATF2 overexpression reverses MDR of SGC7901/VCR cells, and BATF2 silence causes enhanced cell viability of SGC7901 cells. (A) Expression of BATF2 detected by qRT-PCR; ***P < 0.001, compared with SGC7901. (B) The transfection efficiency was shown in SGC7901/VCR cells after BATF2 overexpressing vector transfection. (C) Expression of BATF2 detected by qRT-PCR after transfection. (D) Cell cycle distribution determined by flow cytometry. (E) Effects of three common chemotherapeutic agents (60 μM, 24 h) on the apoptosis of SGC7901/VCR cell assayed by flow cytometry; **P < 0.01, ***P < 0.001, compared with vector group. (F) Relative mRNA level of BATF2 in SGC7901 cells after transfection with BATF2 specific siRNA. (G) Cell viability of SGC7901 cells after treatment with VCR in the absence or presence of BATF2 siRNA; *P < 0.05, **P < 0.01, compared with siNC; ##P < 0.01, compared with siNC+VCR; ^^P < 0.01, compared with siBATF2#3. DDP, cisplatin; VCR, vincristine sulfate; 5-FU, 5-fluorouracil
BATF2 overexpression suppressed Wnt/β-catenin pathway and its silence enhanced Wnt/β-catenin signaling
To determine how BATF2 weakens the drug susceptibility of SGC7901/VCR cells, we measured the mRNA and protein levels of the P-gp and Wnt/β-catenin pathways after overexpression of BATF2. We found that BATF2 overexpression in SGC7901/VCR cells could suppress P-gp, β-catenin, c-myc, cyclin D1, and Survivin protein levels (Fig. 4A), and in SGC7901 cells, BATF2 knockdown promoted the level of the above proteins (Fig. 4B), indicating that BATF2 might impair the drug susceptibility through downregulating P-gp and inactivating the Wnt/β-catenin pathway.
BATF2 overexpression downregulates P-gp and Wnt/β-catenin pathway in SGC7901/VCR cells and BATF2 silence upregulates P-gp and Wnt/β-catenin pathway in SGC7901 cells. Protein expression of P-gp, β-catenin, c-myc, cyclin D1, and Survivin in SGC7901/VCR cells (A) and SGC7901 cells (B) was measured by western blotting.
Knockdown of β-catenin imitated the effects of BATF2 overexpression on drug susceptibility
To confirm that BATF2 enhanced drug susceptibility occurred via the downregulation of β-catenin, we utilized β-catenin knockdown SGC7901/VCR cells. As shown in Fig. 5A, β-catenin knockdown attenuated cell proliferation compared with siNC transfection. In addition, β-catenin knockdown induced cell cycle G0/G1 phase arrest (Fig. 5B) and apoptosis (Fig. 5C) by flow cytometry analysis in SGC7901/VCR cells. Importantly, β-catenin siRNA transfection could inhibit P-gp, cyclin D1, and Survivin in SGC7901/VCR cells (Fig. 5D). These results suggested that the increased drug susceptibility to BATF2 seen in the SGC7901/VCR cells might be dependent on β-catenin.
Knockdown of β-catenin imitated the effects of BATF2 overexpression on drug susceptibility. (A) Growth curve assayed by MTT. (B) Cell cycle distribution determined by flow cytometry. (C) Effects of three common chemotherapeutics (60 μM, 24 h) on apoptosis of SGC7901/VCR cells by flow cytometry. (D) Protein expressions of P-gp, cyclin D1 and Survivin measured by western blotting. *P < 0.05, **P < 0.01, ***P < 0.001. DDP, cisplatin; VCR, vincristine sulfate; 5-FU, 5-fluorouracil.
Enhanced Wnt signaling reversed the inhibition of BATF2 on MDR
To determine whether BATF2 impaired drug susceptibility through suppressing the Wnt/β-catenin pathway, biological characteristics of SGC7901/VCR cells were detected after Wnt3a was added to enhance Wnt/β-catenin pathway. Cell cycle analysis results showed that Wnt3a could reverse the cell cycle blocking effects of BATF2 (Fig. 6A). Enhancing the Wnt/β-catenin pathway could also reverse the impairment in drug susceptibility (Fig. 6B). Downregulation of BATF2, TCF, P-gp, β-catenin, and Survivin were recovered by activation of the Wnt/β-catenin pathway (Fig. 6C).
Enhanced Wnt/β-catenin pathway reverses the inhibitory effect of BATF2 on MDR in SGC7901/VCR cells. (A) Cell cycle distribution determined by flow cytometry. (B) Effects of three common chemotherapeutics (60 μM, 24 h) on apoptosis of SGC7901/VCR cell by flow cytometry. (C) Protein expressions of BATF2, TCF, P-gp, β-catenin, and Survivin measured by western blotting. *P < 0.05, **P < 0.01, #P < 0.05. MDR, multidrug resistance; DDP, cisplatin; VCR, vincristine sulfate; 5-FU, 5-fluorouracil.
Discussion
GC is one of the most common digestive system malignancies worldwide. Surgical intervention combined with chemotherapy is the primary treatment for GC and the only feasible management of GC diagnosed at an advanced stage is chemotherapy. The occurrence of MDR in tumor cells is the main obstacle to chemotherapy and it is attributable to the presence of multiple intricate mechanisms. Therefore, understanding the underlying mechanisms of MDR is important for improving the outcomes following chemotherapy.
In the present study, SGC7901/VCR was used to investigate the mechanisms of MDR in GC. We found that the SGC7901/VCR cells grew faster and had a shorter cell cycle and increased resistance to common chemotherapy drugs compared to SGC7901 cells.
There were four main mechanisms of MDR in tumor cells: (i) drug resistance proteins, such as LRP, P-gp, and MRP1, tend be highly expressed in MDR cancer cell lines and these factors prevent drugs from accessing the cell nucleus, promote drug release, and lead to drug resistance (Borst et al. 2000); (ii) p53 mutation suppresses apoptosis (Yeh et al. 1999); (iii) glutathione that can detoxify chemotherapy drugs in tumor cells (Komiya et al. 1998); and (iv) tumor micro-environment changes (Straussman et al. 2012). P-gp, LRP, and MRP1 were significantly upregulated in the SGC7901/VCR cells and the data suggested that increased expression of these drug resistance proteins might contribute to the occurrence of MDR in GC cells.
Upregulation of P-gp is known to increase drug resistance in cancers. However, the molecular mechanisms of P-gp overexpression in GC cells are still largely unknown. Previous studies have shown that the Wnt/β-catenin pathway was closely correlated to the development of tumors and played a critical role in the induction of MDR (Zhang et al. 2012). Moreover, the Wnt/β-catenin pathway has proven to be an important inhibitor of the Wnt/β-catenin pathway that can decrease the expression of P-gp and reverse the MDR of cholangiocarcinoma (Shen et al. 2013). Here, we found evidence that Wnt/β-catenin pathway was over activated in the SGC7901/VCR cells and the over activation of Wnt/β-catenin pathway might contribute to upregulation of P-gp.
Recently, BATF2 has been reported to be a potential tumor suppressor that could block the growth of various types of cancer (Su et al. 2008; Dash et al. 2010). It was found to directly inhibit the activity of AP-1, which is an essential transcription factor for cell proliferation and oncogenic transformation (Su et al. 2008). Moreover, BATF2 can suppress migration by lung adenocarcinoma cells by inhibiting the Wnt/β-catenin pathway (Wang et al. 2012). This raised the possibility that BATF2 could reverse MDR via inactivating the Wnt/β-catenin pathway to downregulate P-gp in GC. We found that BATF2 was downregulated in SGC7901/VCR cells. Overexpression of BATF2 could arrest the cell cycle and increase the susceptibility of the SGC7901/VCR cells to drugs. Moreover, after BATF2 was overexpressed, the expression of P-gp was retrained and the Wnt/β-catenin pathway was inactivated, indicating that BATF2 might be able to reverse MDR by suppressing the Wnt/β-catenin pathway in GC. To test our hypothesis, SGC7901/VCR cells were transfected with siβ-catenin and we found that knockdown of β-catenin imitated the effects of BATF2 overexpression on drug susceptibility. On the other hand, when SGC7901/VCR cells were further treated with Wnt3a, an activator of Wnt/β-catenin pathway, the inhibitory effect of BATF2 on MDR of SGC7901/VCR cells was abolished after the Wnt/β-catenin pathway had been enhanced. This emphasized a potential application of BATF2 to increase susceptibility of GC cells to chemotherapeutic agents.
Conclusion
The present study demonstrates that BATF2 can reverse MDR of GC cells by inhibiting the Wnt/β-catenin pathway. BATF2 is a potential target for overcoming the resistance of GC to chemotherapeutic agents.
References
Borst P, Evers R, Kool M, Wijnholds J (2000) A family of drug transporters: the multidrug resistance-associated proteins. J Natl Cancer Inst 92:1295–1302
Bray F, Ren JS, Masuyer E, Ferlay J (2013) Global estimates of cancer prevalence for 27 sites in the adult population in 2008. Int J Cancer 132:1133–1145
Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J (2016) Cancer statistics in China, 2015. CA Cancer J Clin 66:115–132
Dai L, Cui X, Zhang X, Cheng L, Liu Y, Yang Y, Fan P, Wang Q, Lin Y, Zhang J, Li C, Mao Y, Wang Q, Su X, Zhang S, Peng Y, Yang H, Hu X, Yang J, Huang M, Xiang R, Yu D, Zhou Z, Wei Y, Deng H (2016) SARI inhibits angiogenesis and tumour growth of human colon cancer through directly targeting ceruloplasmin. Nat Commun 7:11996
Dash R, Su ZZ, Lee SG, Azab B, Boukerche H, Sarkar D, Fisher PB (2010) Inhibition of AP-1 by SARI negatively regulates transformation progression mediated by CCN1. Oncogene 29:4412–4423
Eom BW, Kim S, Kim JY, Yoon HM, Kim MJ, Nam BH, Kim YW, Park YI, Park SR, Ryu KW (2018) Survival benefit of perioperative chemotherapy in patients with locally advanced gastric cancer: a propensity score matched analysis. J Gastric Cancer 18:69–81
Ferro A, Peleteiro B, Malvezzi M, Bosetti C, Bertuccio P, Levi F, Negri E, La Vecchia C, Lunet N (2014) Worldwide trends in gastric cancer mortality (1980-2011), with predictions to 2015, and incidence by subtype. Eur J Cancer 50:1330–1344
Fodale V, Pierobon M, Liotta L, Petricoin E (2011) Mechanism of cell adaptation: when and how do cancer cells develop chemoresistance? Cancer J 17:89–95
Gillet JP, Efferth T, Remacle J (2007) Chemotherapy-induced resistance by ATP-binding cassette transporter genes. Biochim Biophys Acta 1775:237–262
Guler R, Roy S, Suzuki H, Brombacher F (2015) Targeting Batf2 for infectious diseases and cancer. Oncotarget 6:26575–26582
He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT, Morin PJ, Vogelstein B, Kinzler KW (1998) Identification of c-MYC as a target of the APC pathway. Science 281:1509–1512
Huang W, Zhang C, Cui M, Niu J, Ding W (2017) Inhibition of bevacizumab-induced epithelial-mesenchymal transition by BATF2 overexpression involves the suppression of Wnt/beta-catenin signaling in glioblastoma cells. Anticancer Res 37:4285–4294
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60:277–300
Kageji T, Obata F, Oka H, Kanematsu Y, Tabata R, Tani K, Bando H, Nagahiro S (2016) Drip-and-ship thrombolytic therapy supported by the telestroke system for acute ischemic stroke patients living in medically under-served areas. Neurol Med Chir 56:753–758
Klaus A, Birchmeier W (2008) Wnt signalling and its impact on development and cancer. Nat Rev Cancer 8:387–398
Komiya S, Gebhardt MC, Mangham DC, Inoue A (1998) Role of glutathione in cisplatin resistance in osteosarcoma cell lines. J Orthop Res 16:15–22
Lim JC, Kania KD, Wijesuriya H, Chawla S, Sethi JK, Pulaski L, Romero IA, Couraud PO, Weksler BB, Hladky SB, Barrand MA (2008) Activation of beta-catenin signalling by GSK-3 inhibition increases p-glycoprotein expression in brain endothelial cells. J Neurochem 106:1855–1865
Liu Z, Wei P, Yang Y, Cui W, Cao B, Tan C, Yu B, Bi R, Xia K, Chen W, Wang Y, Zhang Y, Du X, Zhou X (2015) BATF2 deficiency promotes progression in human colorectal cancer via activation of HGF/MET signaling: a potential rationale for combining MET inhibitors with IFNs. Clin Cancer Res 21:1752–1763
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods 25:402–408
Lutz MP, Zalcberg JR, Ducreux M, Ajani JA, Allum W, Aust D, Bang YJ, Cascinu S, Holscher A, Jankowski J, Jansen EP, Kisslich R, Lordick F, Mariette C, Moehler M, Oyama T, Roth A, Rueschoff J, Ruhstaller T, Seruca R, Stahl M, Sterzing F, van Cutsem E, van der Gaast A, van Lanschot J, Ychou M, Otto F, First St Gallen EGCCEP (2012) Highlights of the EORTC St. Gallen International Expert Consensus on the primary therapy of gastric, gastroesophageal and oesophageal cancer—differential treatment strategies for subtypes of early gastroesophageal cancer. Eur J Cancer 48:2941–2953
MacDonald BT, Tamai K, He X (2009) Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17:9–26
Rugge M, Genta RM, Di Mario F, El-Omar EM, El-Serag HB, Fassan M, Hunt RH, Kuipers EJ, Malfertheiner P, Sugano K, Graham DY (2017) Gastric cancer as preventable disease. Clin Gastroenterol Hepatol 15:1833–1843
Shen DY, Zhang W, Zeng X, Liu CQ (2013) Inhibition of Wnt/beta-catenin signaling downregulates P-glycoprotein and reverses multi-drug resistance of cholangiocarcinoma. Cancer Sci 104:1303–1308
Straussman R, Morikawa T, Shee K, Barzily-Rokni M, Qian ZR, Du J, Davis A, Mongare MM, Gould J, Frederick DT, Cooper ZA, Chapman PB, Solit DB, Ribas A, Lo RS, Flaherty KT, Ogino S, Wargo JA, Golub TR (2012) Tumour micro-environment elicits innate resistance to RAF inhibitors through HGF secretion. Nature 487:500–504
Su ZZ, Lee SG, Emdad L, Lebdeva IV, Gupta P, Valerie K, Sarkar D, Fisher PB (2008) Cloning and characterization of SARI (suppressor of AP-1, regulated by IFN). Proc Natl Acad Sci U S A 105:20906–20911
Sun YL, Patel A, Kumar P, Chen ZS (2012) Role of ABC transporters in cancer chemotherapy. Chin J Cancer 31:51–57
Tetsu O, McCormick F (1999) Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature 398:422–426
Tussiwand R, Lee WL, Murphy TL, Mashayekhi M, Kc W, Albring JC, Satpathy AT, Rotondo JA, Edelson BT, Kretzer NM, Wu X, Weiss LA, Glasmacher E, Li P, Liao W, Behnke M, Lam SS, Aurthur CT, Leonard WJ, Singh H, Stallings CL, Sibley LD, Schreiber RD, Murphy KM (2012) Compensatory dendritic cell development mediated by BATF-IRF interactions. Nature 490:502–507
Valenta T, Hausmann G, Basler K (2012) The many faces and functions of beta-catenin. EMBO J 31:2714–2736
Van Cutsem E, Dicato M, Geva R, Arber N, Bang Y, Benson A, Cervantes A, Diaz-Rubio E, Ducreux M, Glynne-Jones R, Grothey A, Haller D, Haustermans K, Kerr D, Nordlinger B, Marshall J, Minsky BD, Kang YK, Labianca R, Lordick F, Ohtsu A, Pavlidis N, Roth A, Rougier P, Schmoll HJ, Sobrero A, Tabernero J, Van de Velde C, Zalcberg J (2011) The diagnosis and management of gastric cancer: expert discussion and recommendations from the 12th ESMO/World Congress on Gastrointestinal Cancer, Barcelona, 2010. Ann Oncol 22(Suppl 5):v1–v9
Van Cutsem E, Sagaert X, Topal B, Haustermans K, Prenen H (2016) Gastric cancer. Lancet 388:2654–2664
Wang C, Su Y, Zhang L, Wang M, You J, Zhao X, Zhang Z, Liu J, Hao X (2012) The function of SARI in modulating epithelial-mesenchymal transition and lung adenocarcinoma metastasis. PLoS One 7:e38046
Wang QW, Zhang XT, Lu M, Shen L (2018) Impact of duration of adjuvant chemotherapy in radically resected patients with T4bN1-3M0/TxN3bM0 gastric cancer. World J Gastrointestin Oncol 10:31–39
Yeh KH, Shun CT, Chen CL, Lin JT, Lee WJ, Lee PH, Chen YC, Cheng AL (1999) Overexpression of p53 is not associated with drug resistance of gastric cancers to 5-fluorouracil-based systemic chemotherapy. Hepato-gastroenterology 46:610–615
Yuan Z, Shi X, Qiu Y, Jia T, Yuan X, Zou Y, Liu C, Yu H, Yuan Y, He X, Xu K, Yin P (2017) Reversal of P-gp-mediated multidrug resistance in colon cancer by cinobufagin. Oncol Rep 37:1815–1825
Zhang ZY, Ge HY (2013) Micrometastasis in gastric cancer. Cancer Lett 336:34–45
Zhang H, Zhang X, Wu X, Li W, Su P, Cheng H, Xiang L, Gao P, Zhou G (2012) Interference of Frizzled 1 (FZD1) reverses multidrug resistance in breast cancer cells through the Wnt/beta-catenin pathway. Cancer Lett 323:106–113
Zhang WH, Song XH, Chen XZ, Yang K, Liu K, Chen ZX, Zhou ZG, Hu JK (2018) Characteristics and survival outcomes related to the infra-pyloric lymph node status of gastric cancer patients. World J Surg Oncol 16:116
Zheng R, Zeng H, Zhang S, Chen W (2017) Estimates of cancer incidence and mortality in China, 2013. Chin J Cancer 36:66
Acknowledgements
This study was supported by the applied Basic Research Joint Fund Project of Yunnan Provincial Science and Technology Department-Kunming Medical University (Grant No. 2017FE468(-108), 2015FB099) and Scientific Research Fund Project of Yunnan Provincial Education Department (Grant No. 2016ZDX020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Editor: Tetsuji Okamoto
Rights and permissions
About this article
Cite this article
Yang, W., Wu, B., Ma, N. et al. BATF2 reverses multidrug resistance of human gastric cancer cells by suppressing Wnt/β-catenin signaling. In Vitro Cell.Dev.Biol.-Animal 55, 445–452 (2019). https://doi.org/10.1007/s11626-019-00360-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-019-00360-5