Abstract
Introduction
C-reactive protein may predict anastomotic complications after colorectal surgery, but its predictive ability may differ between laparoscopic and open resection due to differences in stress response. Therefore, the objective of this study was to perform a systematic review and meta-analysis on the diagnostic characteristics of C-reactive protein to detect anastomotic leaks and infectious complications after laparoscopic and open colorectal surgery.
Methods
A systematic review was performed according to PRISMA. Studies were included if they reported on the diagnostic characteristics of postoperative day 3–5 values of serum C-reactive protein to diagnose anastomotic leak or infectious complications specifically in patients undergoing elective laparoscopic and open colorectal surgery. The main outcome was a composite of anastomotic leak and infectious complications. A random-effects model was used to perform a meta-analysis of diagnostic accuracy.
Results
A total of 13 studies were included (9 for laparoscopic surgery, 8 for open surgery). The pooled incidence of the composite outcome was 14.8% (95% CI 10.2–19.3) in laparoscopic studies and 21.0% (95% CI 11.9–30.0) for open. The pooled diagnostic accuracy characteristics were similar for open and laparoscopic studies. However, the C-reactive protein threshold cutoffs were lower in laparoscopic studies for postoperative days 3 and 4, but similar on day 5.
Conclusions
The diagnostic characteristics of C-reactive protein in the early postoperative period to detect infectious complications and leaks are similar after laparoscopic and open colorectal surgery. However, thresholds are lower for laparoscopic surgery, suggesting that the interpretation of serum CRP values needs to be tailored based on operative approach.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Anastomotic and infectious complications after colorectal surgery are common and result in significant morbidity and healthcare resource utilization.1, 2 Furthermore, these complications also affect cancer outcomes, as patients who experience such postoperative morbidity are more likely to have worse long-term oncologic outcomes.3 Patients with severe infections, which can include organ-space surgical site infections or sepsis, are at particularly high risk.4
Early recognition of these complications may result in improved outcomes.2 Serum inflammatory markers, most notably C-reactive protein (CRP) and procalcitonin, may be helpful to detect presence of surgical site infections prior to the development of clinical symptoms.5,6,7 Previous studies have reported different CRP thresholds to detect anastomotic leak or infectious complications in the first few postoperative days with acceptable diagnostic accuracy.6, 7 However, it is unclear whether previously described CRP thresholds and diagnostic accuracy are altered based on surgical approach. Laparoscopic surgery is associated with less surgical trauma than the open approach, which subsequently results in a lower systemic inflammatory response.8, 9 Thus, levels of acute phase reactants such as serum CRP are lower after laparoscopic compared with open surgery.10, 11 Therefore, the objective of this study is to perform a meta-analysis of the diagnostic accuracy of serum CRP to detect infectious complications and anastomotic leakage in patients undergoing laparoscopic versus open colorectal surgery.
Materials and Methods
Search Strategy
A systematic literature search of all English- and French-language articles published up to May 15, 2018, was conducted according to the PRISMA guidelines.12 The Medline, Embase, CENTRAL, DARE, PubMed, Scopus, ClinicalTrials.gov, and WHO International Clinical Trials Registry Platform databases were queried. The systematic search terms and strategy are shown in Table 1. In adherence with the PRISMA guidelines, two independent reviewers (TP, AZ) performed a primary screen of title and abstracts. Disagreements were addressed by discussion between the two reviewers. Residual disagreements were resolved by a third author (LL). Following primary screening, the remaining studies underwent full-text analysis and data extraction using a pre-determined datasheet. Full-text articles were included if they reported on the diagnostic characteristics (CRP threshold, sensitivity, and specificity) of postoperative day 3–5 values of serum CRP to diagnose anastomotic leak or infectious complications, specifically in patients undergoing elective laparoscopic and/or open colorectal surgery. Studies were excluded if CRP diagnostic characteristics could not be differentiated between laparoscopic and open cases, if CRP was used to diagnose non-infectious complications, or if there were insufficient data to calculate diagnostic characteristics. The protocol was registered a priori with PROSPERO (CRD42018097270). The EndNote 8 software (Clarivate Analytics, Philadelphia, PA) was used to manage references, duplication removal, and facilitate primary and secondary screening analysis.
Data Extraction and Synthesis
Details recorded included study design, study population and operative characteristics, CRP threshold and diagnostic characteristics (sensitivity, specificity, positive and negative likelihood ratios) for the main outcomes on postoperative days 3 to 5 based on surgical approach, and incidence of anastomotic leak and/or infectious complications. Study quality was assessed using the Methodological Index for Non-Randomized Studies (MINORS) instrument for observational studies.13 The MINORS instrument includes 12 items that are each scored as 0 (not reported), 1 (reported but inadequate), or 2 (reported and adequate), and is scored over a range of 0 to 24. Further quality assessment was determined if the appropriate statistical analyses were performed to determine serum CRP cutoffs for study outcome, and categorized as “yes” if a receiver operating characteristics (ROC) curve with Youden’s index or the graphical method was done for each postoperative day and subsequent diagnostic accuracy characteristics calculated based on this cutoff.
The main outcome for meta-analysis was a composite of anastomotic leak and infectious complications. CRP thresholds on postoperative days 3 to 5 were pooled using geometric means. Serum CRP is an acute phase reactant and is expected to peak on postoperative days (POD) 2–3, suggesting it would be most clinically useful at POD 3 and beyond.14 A random-effects model was used to perform a meta-analysis of diagnostic accuracy characteristics (area under the curve (AUC), sensitivity, specificity, and positive and negative likelihood ratios). This method was chosen because we anticipated important heterogeneity in study population and surgical characteristics (i.e., studies would include a subject population with mixed indications for surgery such as colorectal malignancy, inflammatory bowel disease, and other benign conditions, as well as report results for both colon and rectal procedures).15 Heterogeneity was assessed using the I2 statistic. The AUC calculates the area under the receiver operative characteristics curve (plot of sensitivity versus 1-specificity) and represents a global measure of diagnostic accuracy, with a value of 1 signifying a perfect test and 0.5 representing a non-discriminatory test.16 Summary ROC (SROC) plots were created to visually graph the summary point showing summary sensitivity and specificity, confidence contour showing the 95% confidence region for the summary point, and the hierarchical model SROC curve. The number needed to diagnose (NND) was calculated as 1 / (sensitivity + specificity − 1) and number needed to predict (NNP) as 1 / (positive predictive value (PPV) + negative predictive value (NPV) − 1), using the pooled sensitivity, specificity, PPV, and NPV.17 Statistical analysis was performed using the METANDI (meta-analysis for diagnostic criteria) and MIDAS (meta-analytical integration of diagnostic accuracy studies) software packages in STATA 15.1 (StataCorp, College Station, TX).
Results
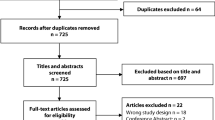
A total of 1070 unique citations were identified in the systematic literature search, of which 166 studies underwent full-text review and 13 were included for meta-analysis (Fig. 1). There were 9 studies reporting diagnostic characteristics after laparoscopic surgery, and 8 for open surgery (Table 2). There were five prospective trials.19,20,21, 24, 27 Routine serum CRP measurements were performed in nine of the included studies. The median sample size in the laparoscopic studies was 160 (Q1 134, Q3 253) and 367 (Q1 225, Q3 589) in open studies. The pooled incidence of the composite outcome was 14.4% (95% CI 10.0, 18.8) in laparoscopic studies and 15.7% (95% CI 8.3, 23.2) for open. Only one study reported diagnostic characteristics specifically for proctectomy.28 The overall incidence of inflammatory bowel disease in the included studies was low (Table 2). The median quality score was 12 (Q1 10, Q3 14) and seven studies did not perform or adequately describe the appropriate statistical analysis in determining serum CRP cutoffs for the main outcome (Table 2).
Diagnostic characteristics for serum CRP to detect the composite outcome were reported more frequently for POD 3 and 4 in both laparoscopic and open groups (Table 3). The pooled serum CRP thresholds were lower in the laparoscopic group for all POD 3–5 (Table 4). The pooled diagnostic characteristics were similar for both groups regardless of the POD. The summary ROC curves for both laparoscopic and open groups on POD 3–5 are shown in Figs. 2, 3, and 4.
Discussion
Infectious complications and anastomotic leak after colorectal surgery are morbid and can significantly affect both short- and long-term outcomes in patients with colorectal cancer.2, 3 Past studies and reviews have suggested that serum CRP may be used to screen for these complications.6 However, the baseline systematic inflammatory response may be affected by surgical approaches, laparoscopic versus open, thus affecting interpretation of serum CRP.31 This systematic review and meta-analysis pooled 13 studies reporting diagnostic accuracy of serum CRP after laparoscopic and open colorectal surgery.
The diagnostic accuracy characteristics of serum CRP were comparable between laparoscopic and open colorectal surgery, but serum CRP thresholds were lower for laparoscopic surgery. Pooled AUC and sensitivity were largely similar across all three postoperative days between the two approaches, but pooled specificity was lower in the open group. This may be reflective of the more pronounced systemic inflammatory response of a laparotomy, resulting in higher baseline serum CRP even in patients without complications.31 This was further demonstrated by the lower serum CRP thresholds for all three postoperative days in patients undergoing laparoscopic surgery. The main outcome measure in this study was a composite of infectious complications and/or anastomotic leakage. We did not have enough data to perform a subgroup analysis on the diagnostic characteristics for anastomotic leak only. However, the diagnostic accuracy characteristics of serum CRP specifically for anastomotic leak may not be very useful given that serum CRP would be higher in patients with complications other than anastomotic leak.
These results suggest that the interpretation of serum CRP should be tailored based on surgical approach. Clinicians should therefore be suspicious of infectious complications or anastomotic leakage with lower absolute serum CRP levels in patients who underwent laparoscopic surgery compared with open (see cutoffs in Table 4). The ideal management of these patients has not yet been defined. One of the important elements that are lacking in the current body of literature is whether clinical signs are apparent with elevated serum CRP levels. In other words, the timeline of symptom manifestation was not reported in published studies. It is thus unclear whether elevations in serum CRP can detect complications prior to the development of clinical signs and symptoms. To date, there are no prospective trials that have answered this important question. Studies in other patient populations (such as thoracic surgery) have suggested that other serum inflammatory markers can detect complications prior to the onset of related clinical signs.32 It would be useful to determine whether routine measurement of serum CRP and potential early detection of morbidity can improve outcomes after colorectal surgery. One study reported earlier time to imaging and reintervention in patients who had elevated serum CRP on postoperative day 4.33 In another study, CT scans performed based on increased serum CRP above 125 mg/L on postoperative day 4, without clinical signs and symptoms, reported a sensitivity of 76.7% for intra-abdominal infections.34 In that same study, 12.7% of patients who did not undergo a CT in the presence of a high serum CRP later developed an intra-abdominal collection. Earlier detection of complications may allow for earlier intervention, but it is unclear whether this will translate to better outcomes. It is also important to remember that the pooled diagnostic accuracy characteristics are relatively poor and would usually not qualify for routine screening test. Despite these poor overall characteristics, there are few other early detection markers. Given the morbidity of anastomotic leakage, interest in serum CRP is such that several studies are currently ongoing to better define the clinical impact of routine postoperative serum CRP measurements.35, 36
While the present study only investigated the diagnostic accuracy of serum CRP for laparoscopic and open surgery based on single-day values, the trend of serum CRP should also be taken into account. Studies have shown that increasing values of serum CRP over time is more accurate than single-day values for the detection of the infectious complications.37 Given the differences in the inflammatory response for laparoscopic and open surgery, it would be interesting to know if the serum CRP trend would differ between the surgical approaches and how this might affect diagnostic accuracy. Routine preoperative serum CRP may also be useful to identify patients that may already have elevated serum CRP before surgery, as this may affect interpretation of postoperative values and trend. Furthermore, future studies should investigate the cost implications of routine serum CRP measurements and the additional investigations that are performed to investigate patients with elevated CRP levels. In this study, the NND and NNP suggest that at least 2 to 4 tests are required to correctly diagnose or predict a true positive or a true negative. Whether this is cost-effective as a routine measurement has yet to be determined. Lastly, there are no studies that have used serum CRP measurements as part of a multifactorial risk index. In higher risk patients, serum CRP may have greater utility in prompting earlier investigation and potential treatments. The effect of the surgical pathology on serum CRP should also be investigated, especially given that it may be affected by neoadjuvant therapy, inflammatory bowel disease, and other inflammatory comorbidities. The overall proportion of patients with inflammatory bowel disease (who may have elevated serum CRP levels at baseline) was low, suggesting that this likely did not affect the results significantly.
This study should be interpreted with several other limitations in mind. First, there was a heterogenous definition of infectious complications between studies. Certain studies only included intra-abdominal infectious complications, whereas other studies included all infectious complications. This may have biased the diagnostic accuracy characteristics, in particular the CRP threshold, as there may be misclassification bias. Misclassification bias based on other postoperative complications may have also affected threshold values. The way in which the diagnostic characteristics were calculated in each study should be taken into account, as there was some variability in the method used to calculate the threshold value. Most studies used Youden’s index, whereas others used graphical methods. There was also some variability in terms of whether serum CRP was drawn routinely, or only based on the physicians’ discretion. In the latter case, risk of selection bias cannot be excluded if CRP was tested only if clinical signs of complications were present. There was also significant statistical heterogeneity in the pooled estimates, reflecting the various indications for surgery, operative details (extraction site, length of incision, etc.), and perioperative management strategies. These factors may affect systemic inflammation10, 38, 39 and serum CRP levels but, due to poor study reporting, they could not be taken into account in this meta-analysis.
Conclusion
The diagnostic accuracy of CRP on POD 3 to 5 to detect infectious postoperative complications and anastomotic leak after laparoscopic and open colorectal surgery is similar. However, serum CRP thresholds are lower for laparoscopic surgery, suggesting the interpretation of serum CRP values should be tailored based on the operative approach. These findings can be used to develop standardized investigation protocols to allow for earlier detection of these highly morbid complications in patients undergoing colorectal surgery.
References
Midura EF, Hanseman D, Davis BR, Atkinson SJ, Abbott DE, Shah SA et al. Risk factors and consequences of anastomotic leak after colectomy: a national analysis. Dis Colon Rectum. 2015;58(3):333-8. https://doi.org/10.1097/DCR.0000000000000249.
Tevis SE, Carchman EH, Foley EF, Heise CP, Harms BA, Kennedy GD. Does Anastomotic Leak Contribute to High Failure-to-rescue Rates? Ann Surg. 2016;263(6):1148-51. https://doi.org/10.1097/SLA.0000000000001409.
Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P. Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg. 2011;253(5):890-9. https://doi.org/10.1097/SLA.0b013e3182128929.
Artinyan A, Orcutt ST, Anaya DA, Richardson P, Chen GJ, Berger DH. Infectious postoperative complications decrease long-term survival in patients undergoing curative surgery for colorectal cancer: a study of 12,075 patients. Ann Surg. 2015;261(3):497-505. https://doi.org/10.1097/SLA.0000000000000854.
Tan WJ, Ng WQ, Sultana R, de Souza NN, Chew MH, Foo FJ et al. Systematic review and meta-analysis of the use of serum procalcitonin levels to predict intra-abdominal infections after colorectal surgery. Int J Colorectal Dis. 2018;33(2):171-80. https://doi.org/10.1007/s00384-017-2956-8.
Singh PP, Zeng IS, Srinivasa S, Lemanu DP, Connolly AB, Hill AG. Systematic review and meta-analysis of use of serum C-reactive protein levels to predict anastomotic leak after colorectal surgery. Br J Surg. 2014;101(4):339-46. https://doi.org/10.1002/bjs.9354.
Gans SL, Atema JJ, van Dieren S, Groot Koerkamp B, Boermeester MA. Diagnostic value of C-reactive protein to rule out infectious complications after major abdominal surgery: a systematic review and meta-analysis. Int J Colorectal Dis. 2015;30(7):861-73. https://doi.org/10.1007/s00384-015-2205-y.
Madbouly KM, Senagore AJ, Delaney CP. Endogenous morphine levels after laparoscopic versus open colectomy. Br J Surg. 2010;97(5):759-64. https://doi.org/10.1002/bjs.6987.
Kuntz C, Wunsch A, Bay F, Windeler J, Glaser F, Herfarth C. Prospective randomized study of stress and immune response after laparoscopic vs conventional colonic resection. Surg Endosc. 1998;12(7):963-7. https://doi.org/10.1007/s004649900757.
Veenhof AA, Vlug MS, van der Pas MH, Sietses C, van der Peet DL, de Lange-de Klerk ES et al. Surgical stress response and postoperative immune function after laparoscopy or open surgery with fast track or standard perioperative care: a randomized trial. Ann Surg. 2012;255(2):216-21. https://doi.org/10.1097/SLA.0b013e31824336e2.
Tsimogiannis KE, Tellis CC, Tselepis AD, Pappas-Gogos GK, Tsimoyiannis EC, Basdanis G. Toll-like receptors in the inflammatory response during open and laparoscopic colectomy for colorectal cancer. Surg Endosc. 2012;26(2):330-6. https://doi.org/10.1007/s00464-011-1871-2.
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS medicine. 2009;6(7):e1000097.
Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J. Methodological index for non-randomized studies (minors): development and validation of a new instrument. ANZ journal of surgery. 2003;73(9):712-6.
Cole DS, Watts A, Scott-Coombes D, Avades T. Clinical utility of peri-operative C-reactive protein testing in general surgery. Ann R Coll Surg Engl. 2008;90(4):317-21. https://doi.org/10.1308/003588408X285865.
Riley RD, Higgins JP, Deeks JJ. Interpretation of random effects meta-analyses. BMJ. 2011;342:d549. https://doi.org/10.1136/bmj.d549.
Simundic AM. Measures of Diagnostic Accuracy: Basic Definitions. EJIFCC. 2009;19(4):203-11.
Linn S, Grunau PD. New patient-oriented summary measure of net total gain in certainty for dichotomous diagnostic tests. Epidemiol Perspect Innov. 2006;3:11. https://doi.org/10.1186/1742-5573-3-11.
Adamina M, Warschkow R, Naf F, Hummel B, Rduch T, Lange J et al. Monitoring c-reactive protein after laparoscopic colorectal surgery excludes infectious complications and allows for safe and early discharge. Surg Endosc. 2014;28(10):2939-48. https://doi.org/10.1007/s00464-014-3556-0.
Facy O, Paquette B, Orry D, Santucci N, Rat P, Rat P et al. Inflammatory markers as early predictors of infection after colorectal surgery: the same cut-off values in laparoscopy and laparotomy? Int J Colorectal Dis. 2017;32(6):857-63. https://doi.org/10.1007/s00384-017-2805-9.
Mik M, Dziki L, Berut M, Trzcinski R, Dziki A. Neutrophil to Lymphocyte Ratio and C-Reactive Protein as Two Predictive Tools of Anastomotic Leak in Colorectal Cancer Open Surgery. Dig Surg. 2018;35(1):77-84. https://doi.org/10.1159/000456081.
Munoz JL, Alvarez MO, Cuquerella V, Miranda E, Pico C, Flores R et al. Procalcitonin and C-reactive protein as early markers of anastomotic leak after laparoscopic colorectal surgery within an enhanced recovery after surgery (ERAS) program. Surg Endosc. 2018;32(9):4003-10. https://doi.org/10.1007/s00464-018-6144-x.
Nason GJ, Barry BD, Obinwa O, McMacken E, Rajaretnam NS, Neary PC. Early rise in C-reactive protein is a marker for infective complications in laparoscopic colorectal surgery. Surg Laparosc Endosc Percutan Tech. 2014;24(1):57-61. https://doi.org/10.1097/SLE.0b013e31828fa03e.
Pedersen T, Roikjaer O, Jess P. Increased levels of C-reactive protein and leukocyte count are poor predictors of anastomotic leakage following laparoscopic colorectal resection. Dan Med J. 2012;59(12):A4552.
Pedrazzani C, Moro M, Mantovani G, Lazzarini E, Conci S, Ruzzenente A et al. C-reactive protein as early predictor of complications after minimally invasive colorectal resection. J Surg Res. 2017;210:261-8. https://doi.org/10.1016/j.jss.2016.11.047.
Platt JJ, Ramanathan ML, Crosbie RA, Anderson JH, McKee RF, Horgan PG et al. C-reactive protein as a predictor of postoperative infective complications after curative resection in patients with colorectal cancer. Ann Surg Oncol. 2012;19(13):4168-77. https://doi.org/10.1245/s10434-012-2498-9.
Ramanathan ML, MacKay G, Platt J, Horgan PG, McMillan DC. The impact of open versus laparoscopic resection for colon cancer on C-reactive protein concentrations as a predictor of postoperative infective complications. Ann Surg Oncol. 2015;22(3):938-43. https://doi.org/10.1245/s10434-014-4065-z.
Ramos Fernandez M, Rivas Ruiz F, Fernandez Lopez A, Loinaz Segurola C, Fernandez Cabrian JM, de la Portilla de Juan F. C Reactive Protein as a Predictor of Anastomotic Leakage in Colorectal Surgery. Comparison Between Open and Laparoscopic Surgery. Cir Esp. 2017;95:529-35.
Welsch T, Muller SA, Ulrich A, Kischlat A, Hinz U, Kienle P et al. C-reactive protein as early predictor for infectious postoperative complications in rectal surgery. Int J Colorectal Dis. 2007;22(12):1499-507. https://doi.org/10.1007/s00384-007-0354-3.
Waterland P, Ng J, Jones A, Broadley G, Nicol D, Patel H et al. Using CRP to predict anastomotic leakage after open and laparoscopic colorectal surgery: is there a difference? Int J Colorectal Dis. 2016;31(4):861-8. https://doi.org/10.1007/s00384-016-2547-0.
Warschkow R, Tarantino I, Torzewski M, Naf F, Lange J, Steffen T. Diagnostic accuracy of C-reactive protein and white blood cell counts in the early detection of inflammatory complications after open resection of colorectal cancer: a retrospective study of 1,187 patients. Int J Colorectal Dis. 2011;26(11):1405-13. https://doi.org/10.1007/s00384-011-1262-0.
Watt DG, Horgan PG, McMillan DC. Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: a systematic review. Surgery. 2015;157(2):362-80. https://doi.org/10.1016/j.surg.2014.09.009.
Fink-Neuboeck N, Lindenmann J, Bajric S, Maier A, Riedl R, Weinberg AM et al. Clinical impact of interleukin 6 as a predictive biomarker in the early diagnosis of postoperative systemic inflammatory response syndrome after major thoracic surgery: A prospective clinical trial. Surgery. 2016;160(2):443-53. https://doi.org/10.1016/j.surg.2016.04.004.
McSorley ST, Khor BY, MacKay GJ, Horgan PG, McMillan DC. Examination of a CRP first approach for the detection of postoperative complications in patients undergoing surgery for colorectal cancer: A pragmatic study. Medicine (Baltimore). 2017;96(7):e6133. https://doi.org/10.1097/MD.0000000000006133.
Holl S, Fournel I, Orry D, Facy O, Cheynel N, Rat P et al. Should CT scan be performed when CRP is elevated after colorectal surgery? Results from the inflammatory markers after colorectal surgery study. Journal of visceral surgery. 2017;154(1):5-9. https://doi.org/10.1016/j.jviscsurg.2016.07.003.
Benedetti M, Ciano P, Pergolini I, Ciotti S, Guercioni G, Ruffo G et al. Early diagnosis of anastomotic leakage after colorectal surgery by the Dutch leakage score, serum procalcitonin and serum C-reactive protein: study protocol of a prospective multicentre observational study by the Italian ColoRectal Anastomotic Leakage (iC). G Chir. 2019;40(1):20-5.
Kornmann V, van Ramshorst B, van Dieren S, van Geloven N, Boermeester M, Boerma D. Early complication detection after colorectal surgery (CONDOR): study protocol for a prospective clinical diagnostic study. Int J Colorectal Dis. 2016;31(2):459-64. https://doi.org/10.1007/s00384-015-2468-3.
Barbic J, Ivic D, Alkhamis T, Drenjancevic D, Ivic J, Harsanji-Drenjancevic I et al. Kinetics of changes in serum concentrations of procalcitonin, interleukin-6, and C- reactive protein after elective abdominal surgery. Can it be used to detect postoperative complications? Coll Antropol. 2013;37(1):195-201.
Persec J, Persec Z, Husedzinovic I. Postoperative pain and systemic inflammatory stress response after preoperative analgesia with clonidine or levobupivacaine: a randomized controlled trial. Wien Klin Wochenschr. 2009;121(17-18):558-63. https://doi.org/10.1007/s00508-009-1221-8.
Cabellos Olivares M, Labalde Martinez M, Torralba M, Rodriguez Fraile JR, Atance Martinez JC. C-reactive protein as a marker of the surgical stress reduction within an ERAS protocol (Enhanced Recovery After Surgery) in colorectal surgery: A prospective cohort study. J Surg Oncol. 2018;117(4):717-24. https://doi.org/10.1002/jso.24909.
Author information
Authors and Affiliations
Contributions
Conception or design: JF, GMF, LSF, LL
Data acquisition, analysis, or interpretation of data: TP, AZ, MT, JF, GMF, LSF, LL
Manuscript drafting: TP, AZ, MT, LL
Critical revision: JF, GMF, LSF, LL
Final approval of the version to be published: TP, AZ, MT, JF, GMF, LSF, LL
Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: TP, AZ, MT, JF, GMF, LSF, LL
Corresponding author
Ethics declarations
Conflict of Interest
LL is the recipient of an investigator-initiated grant from Johnson & Johnson. JF has received investigator-initiated grants from Merck and personal fees for consulting from Shionogi. LSF receives consulting fees from Merck and Abbott. TP, AZ, MT, and GMF have no conflicts of interests to disclose.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Paradis, T., Zorigtbaatar, A., Trepanier, M. et al. Meta-analysis of the Diagnostic Accuracy of C-Reactive Protein for Infectious Complications in Laparoscopic Versus Open Colorectal Surgery. J Gastrointest Surg 24, 1392–1401 (2020). https://doi.org/10.1007/s11605-020-04599-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-020-04599-2