Abstract
Background
Major vascular resection when necessary for margin control during pancreaticoduodenectomy is relatively universal with perioperative and oncological outcomes that are similar to those of patients undergoing a PD without venous involvement. The present study compares total laparoscopic pancreaticoduodenectomy (TLPD) versus open pancreaticoduodenectomy (OPD) with major vascular resection.
Methods
We reviewed data for all patients undergoing TLPD or OPD with vascular resection at Mayo Clinic Rochester, between the dates of July 2007 and July 2013.
Results
A total of 31 patients undergoing TLPD and 58 patients undergoing OPD with major vascular resection were identified. Mean operative blood loss was significantly less in the laparoscopic (842 cc) compared to the open group (1,452 cc) (p < 0.001), as was median hospital stay, 6 (4–118) versus 9 (6–73) days, respectively (p = 0.006). There was no significant difference in the total number of complications (lap 35 %, open 48 %) (p = 0.24) or severe complications (≥III) (lap 6.4 %, open 3.4 %) (p = 0.51) in the two groups. In-hospital mortality or 30-day mortality was not statistically different between the laparoscopic and open groups, 3.2 and 3.4 %, respectively (p = 0.96). Patency of the reconstructed vessels on postoperative imaging was not significantly different between the TLPD (93 %) and OPD groups (91 %) (p = 0.76). In patients with a diagnosis of adenocarcinoma, there was no significant difference in overall survival between the two groups (p = 0.22).
Conclusion
The present study clearly demonstrates that not only is TLPD with major vascular resection feasible and safe but that it can achieve results that are similar in morbidity and mortality as well as oncologic outcome compared to patients undergoing OPD with major vascular resection.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
For many gastrointestinal operations, the use of a minimally invasive approach has become a common practice, with many studies demonstrating advantages over open surgery.1 – 3 While laparoscopic distal pancreatectomy has become increasingly utilized, total laparoscopic pancreaticoduodenectomy (TLPD) is a technically advanced minimally invasive surgical procedure with a limited number of centers having published their results.4 – 9 Despite this, these initial publications have demonstrated the feasibility and safety of the procedure with several potential advantages over open pancreaticoduodenectomy (OPD) suggested including lower blood loss and shorter length of hospital stay.4 , 5 While there is no question that initial experience with TLPD involved highly selected cases, advances in operative technique and technology have allowed surgeons to push the envelope of the complexity of resection techniques that can be undertaken laparoscopically.
With more effective systemic chemotherapeutic options, patients who are potential candidates for pancreatectomy in combination with vascular resection are being seen with increasing frequency, requiring more complex resections for margin control. Major vascular resection with OPD has been shown in retrospective series to have perioperative and oncological outcomes that are similar to those of patients undergoing a pancreaticoduodenectomy (PD) without venous involvement.10 – 13 As such, pancreaticoduodenectomy (PD) with concomitant venous resection has become commonplace at many centers. We have previously shown in a small case series that major vascular resection with TLPD is feasible and can be performed safely.14 The present study was designed to compare TLPD and OPD with major vascular resection.
Materials and Methods
This study was performed with approval of the Mayo Clinic Rochester Institutional Review Board. Data was acquired from patient medical records, from outside medical records, and from a database maintained on all patients undergoing PD.
We reviewed data for all patients undergoing PD with vascular resection between the dates of July 2007 and July 2013. Clinical, operative, and pathologic information was reviewed on all patients. Patients were divided into those undergoing TLPD with major vascular resection and those undergoing OPD with major vascular resection. Patients were analyzed in an intention-to-treat fashion with patients who underwent conversion from a laparoscopic to open resection, being analyzed in the laparoscopic group. Complications were recorded using the Clavien-Dindo classification system.15 , 16
Borderline resectability was defined based on the National Comprehensive Cancer Network guidelines of abutment of the superior mesenteric artery, abutment with impingement of the superior mesenteric vein-portal vein, abutment or short encasement of the common hepatic artery, and no abutment of the celiac trunk.17 Locally advanced tumors were defined as those with more vascular involvement than the borderline group. Patients with known borderline or locally advanced disease on preoperative imaging underwent neoadjuvant therapy, followed by restaging and operative resection.
The technique of TLPD has been described previously.4 , 14 Briefly, the plane posterior to the pancreatic neck is developed early in the procedure to determine the extent of malignant mesoportal venous involvement demonstrated on preoperative imaging or to identify unsuspected involvement. Exposure of the PV, SMV, and distal splenic vein is accomplished for complete major venous control in anticipation of venous resection. To minimize mesoportal clamp time, the remainder of the pancreaticoduodenectomy dissection is completed reserving the venous transection as the final step in the resection.
In all patients undergoing TLPD with vascular resection, systemic intravenous unfractionated heparin (3,000 to 5,000 units) was given before clamping. Vascular clamping was performed with atraumatic endo vessel clamps (Aesculap Inc., Center Valley, PA, USA) using a dedicated applier that facilitates control and precise placement. Postoperatively, low-dose (81 mg) aspirin is prescribed and is continued for 3 months. In patients with malignancy and a history of thrombosis, full anticoagulation with warfarin is generally instituted.
In patients undergoing OPD with vascular resection, systemic anticoagulation was used at the discretion of the attending surgeon; however, a significant proportion of patients received the same regime of unfractionated heparin (3,000 to 5,000 units) before clamping. Postoperatively, low-dose (81 mg) aspirin was frequently used; however, this was again surgeon dependent. Postoperative imaging was obtained to assess patency of venous reconstruction and to rule out thrombosis.
All statistical analyses were performed using Stata 12 (Stata Corp., College Station, TX). Differences between groups were analyzed using the unpaired t test for continuous variables and by the χ 2 test or continuity correction method for categorical variables. Wilcoxon rank-sum was used for variables that did not display a normal distribution. Patient survival curves were generated using the Kaplan-Meier method and compared by the log-rank test. All statistical tests were two sided and differences were considered significant when p < 0.05.
Results
A total of 31 patients undergoing TLPD and 58 patients undergoing OPD with major vascular resection between the dates of July 2007 and July 2013 were identified. Patient demographics for the two groups can be seen in Table 1. Patients in the TLPD group were slightly older (69.5 ± 9.0 years) than those in the open group (63.6 ± 11.3 years, p = 0.01). The proportion of male patients was 54.8 % in the TLPD group and 56.9 % in the OPD group (p = 0.85). There was no significant difference in BMI in the two groups, 26.1 ± 4.7 and 26.2 ± 4.8, respectively (p = 0.90).
Adenocarcinoma was present in 80.6 % of patients in the TLPD group compared to 87.9 % in the OPD group (p = 0.35). IPMN was present in 6.5 % of patients in the laparoscopic group and 5.2 % in the open group (p = 0.8). Other diagnoses in the TLPD group included autoimmune pancreatitis, chronic pancreatitis with mass, neuroendocrine tumor, and metastatic acinar cell carcinoma. Other diagnoses in the OPD group included neuroendocrine tumor (n = 2), large serous cystadenoma, and ampullary adenocarcinoma. Tumor size was not significantly different between the TLPD and OPD groups, 3.6 ± 1.1 and 3.8 ± 1.4 cm, respectively (p = 0.59).
In two patients, robotic assistance was used for pancreaticobiliary reconstruction after pure laparoscopic resection and venous reconstruction. Four patients in the TLPD group underwent conversion to an open procedure for anticipated complex venous or pancreatic reconstruction.
Mean operative time for patients undergoing TLPD (465 ± 86 min) was not significantly different from patients in the OPD group (465 ± 98, p > 0.99). Mean operative blood loss was significantly less in the TLPD (841.8 ± 994.8 ml) compared to OPD group (1,452.1 ± 1,966.7 ml, p < 0.001). A longer vascular clamp time was seen in patients undergoing TLPD, 46.8 ± 30.8 and 25.1 ± 16.2 min, respectively (p < 0.001). Pylorus-preserving pancreaticoduodenectomy was performed in 90.3 % of patients in the TLPD group and 77.6 % of patients in the OPD group (p = 0.14). Tangential resection was used more commonly in the TLPD group compared to the OPD group (p < 0.001). One patient in the TLPD group had a tumor that involved a replaced right hepatic artery. The proximal right hepatic artery was ligated and a segmental resection performed; primary reconstruction of the distal right hepatic artery to the proximal gastroduodenal artery was performed laparoscopically. Operative variables can be seen in Table 2. The distribution of specific vessels involved by tumor was similar between groups. Vascular resection and reconstruction methods varied between groups based on surgeon preference of technique. Tangential resections with lateral venorrhaphy or patch reconstruction were more common in the TLPD group (Table 3).
A significantly higher rate of complete macroscopic and microscopic margin-negative (R0) resection was obtained in the TLPD (93.5 %) compared to the OPD group (75.9 %, p = 0.038). The median number of lymph nodes harvested was higher in the TLPD group (20.0 ± 8.6) compared to the OPD group (15.9 ± 6.6. p = 0.01).
There was no significant difference in the total rate of complications (lap 35 %, open 48 %) (p = 0.24) or severe complications (≥III) (lap 6.4 %, open 3.4 %, p = 0.51) in the two groups. In-hospital mortality or 30-day mortality was not statistically different between the TLPD and OPD groups, 3.2 and 3.4 %, respectively (p = 0.96) (Table 4).
Postoperative imaging was available for 28/31 patients in the TLPD group and 55/58 patients in the OPD group. Patency of the reconstructed vessels on postoperative imaging was not significantly different between the TLPD (93 %) and OPD groups (91 %) (p = 0.76). In the TLPD group, a non-occlusive thrombus was seen in one patient that was clinically asymptomatic and a complete SMV thrombosis was seen in one patient who had developed a pancreatic leak. The patients with no imaging at follow-up were alive and well 6 months postoperatively.
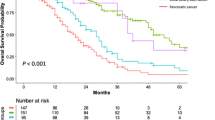
Median follow-up was 15.2 months in the TLPD group and 14.8 months in the OPD group. When an intention-to-treat analysis was performed in patients with a diagnosis of adenocarcinoma, there was no significant difference in overall survival between the two groups (p = 0.14; Fig. 1).
Discussion
TLPD is a technically challenging procedure because of the pancreas’s retroperitoneal location, intimate association with major vasculature, and difficult reconstruction of the biliary and pancreatic remnants. While initial series demonstrating the safety of TLPD undoubtedly had case selection bias, as the collective experience with this technique has evolved, so has the complexity of cases that can be undertaken laparoscopically. We had previously published our first 11 patient series demonstrating that the TLPD with major vascular resection is feasible and can be performed safely.14 Having established its feasibility and safety, the present study compares patients with concomitant major vascular resection undergoing TLPD with those patients undergoing OPD.
Patient demographics were similar between the two groups except for patients in the laparoscopic group being slightly older in age. There was also no significant difference in the number of patients with adenocarcinoma or in tumor size in the two groups. The distribution of vessels involved was similar between the two groups. In the TLPD group, a higher number of tangential resections were performed compared to the OPD group. This difference reflects surgeon-specific preference to perform a tangential resection with bovine pericardial patch reconstruction in patients with long-segment vein involvement and not a difference in the amount of vessel involved with the tumor.
Clamp time was significantly longer in the TLPD group. Clamp time for vein reconstruction ranges considerably from 25 to 45 min in the literature for OPD.10 – 13 Our shorter clamp times for open procedures likely demonstrate the vast experience of our surgeons with open vein resection rather than long clamp times with TLPD. Additionally, tangential patch venorrhaphy inherently takes longer than an end-to-end anastomosis and was more commonly employed in the TLPD group. To minimize clamp time, as much as possible, we always ensure that the remainder of the PD dissection is completed reserving the venous transection as the final step in the resection.
As experience with TLPD grows, authors have proposed that the ultimate goal should be to actually perform a better operation than using an open technique.5 Previous studies looking at open PD have postulated R0 margin status and increased lymph node retrieval/decreased lymph node ratio as markers of improved cancer outcomes.18 , 19 A significantly higher rate of complete macroscopic and microscopic margin-negative (R0) resection was obtained in the laparoscopic group (93.5 %) compared to the open group (75.9 %) as well as a higher mean number of lymph nodes in the specimen. We postulate that the superior views and magnification afforded by laparoscopy allow for a more detailed and precise lymphadenectomy and pancreatic resection.
To the best of our knowledge, four previous studies have compared laparoscopic (including robotic-assisted) PD to OPD and all have suggested equivalent or favorable outcomes for selected patients undergoing minimally invasive approaches.5 , 7 , 20 , 21 In the present study, we report a 30-day mortality that was not different between the laparoscopic and open groups, 3.2 and 3.4 %, respectively. This mortality rate is also comparable to that reported in the four comparative studies of lap/robotic PD without major vascular resection. Previous comparative studies of OPD have reported perioperative and oncologic outcomes that are similar between patients undergoing pancreaticoduodenectomy with and without major venous resection.10 – 13 The present study demonstrates that the same holds true for patients undergoing TLPD with venous involvement. With the current level of experience there are no exclusion criteria for TLPD; all patients undergo attempted laparoscopic approach with only a minority requiring conversion. The most challenging patients continue to be those with chronic pancreatitis as the primary indication or those who have undergone neoadjuvant chemoradiation with a long time interval between treatment and surgery. There has not been any specific scenario that has resulted in a higher conversion rate. Any decision to convert to an open procedure is made on a case by case basis. We would like to stress that conversions should not be viewed as failures particularly as surgeons progress through their learning curve.
When evaluating only those patients with adenocarcinoma, no significant difference was seen in overall survival between patients undergoing TLPD and OPD with major vascular resection. This clearly demonstrates that an excellent oncologic result can be achieved using TLPD, even in the setting of vascular involvement.
A caution must be stated that our center had considerable experience with TLPD before attempting major vascular resections laparoscopically. The acquisition of considerable experience in both OPD with and without vascular resection and TLPD without vascular resection before attempting laparoscopic major venous resection and reconstruction is strongly recommended.
The present study suggests that not only is TLPD with major vascular resection feasible and safe but that it can achieve results that are similar in morbidity and mortality as well as oncologic outcome compared to patients undergoing OPD with major vascular resection. Vascular involvement does not represent an absolute contraindication to TLPD in experienced centers.
Abbreviations
- TLPD:
-
Total laparoscopic pancreaticoduodenectomy
- OPD:
-
Open pancreaticoduodenectomy
- PD:
-
Pancreaticoduodenectomy
- CI:
-
Confidence interval
- CT:
-
Computed tomography
- SD:
-
Standard deviation
- PV:
-
Portal vein
- SMV:
-
Superior mesenteric vein
References
Lujan J, Valero G, Hernandez Q, Sanchez A, Frutos MD, Parrilla P. Randomized clinical trial comparing laparoscopic and open surgery in patients with rectal cancer. Br J Surg. 2009;96:982–989.
Venkat R, Edil BH, Schulick RD, Lidor AO, Makary MA, Wolfgang CL. Laparoscopic distal pancreatectomy is associated with significantly less overall morbidity compared to the open technique: a systematic review and meta-analysis. Ann Surg. 2012 Jun;255(6):1048-59.
Croome K, Yamashita, M. Laparoscopic vs Open Hepatic Resection for Benign and Malignant Tumors: An Updated Meta-Analysis. Archives of Surgery 2010;145(11):1109-1118.
Kendrick ML, Cusati D. Total laparoscopic pancreaticoduodenectomy: feasibility and outcome in an early experience. Arch Surg. 2010 Jan;145(1):19-23.
Asbun HJ, Stauffer JA. Laparoscopic vs open pancreaticoduodenectomy: overall outcomes and severity of complications using the Accordion Severity Grading System. J Am Coll Surg. 2012 Dec;215(6):810-9.
Palanivelu C, Rajan PS, Rangarajan M, Vaithiswaran V, Senthilnathan P, Parthasarathi R et al. (2009) Evolution in techniques of laparoscopic pancreaticoduodenectomy: a decade long experience from a tertiary center. J Hepatobiliary Pancreat Surg 16:731–740.
Buchs NC, Addeo P, Bianco FM, et al. Robotic versus open pancreaticoduodenectomy: a comparative study at a single institution. World J Surg 2011;35:2739e2746.
Zeh HJ, Zureikat AH, Secrest A, et al. Outcomes after robot-assisted pancreaticoduodenectomy for periampullary lesions. Ann Surg Oncol 2012;19:864e870.
Gagner M, Pomp A: Laparoscopic pylorus-preserving pancreatoduodenectomy. Surg Endosc 1994; 8:408-410.
Yekebas EF, Bogoevski D, Cataldegirmen G, Kunze C, Marx A, Vashist YK et al. (2008) En bloc vascular resection for locally advanced pancreatic malignancies infiltrating major blood vessels: perioperative outcome and long-term survival in 136 patients. Ann Surg 247:300– 309.
Muller SA, Hartel M, Mehrabi A, Welsh T, Martin DJ, Hinz U et al. (2009) Vascular resection in pancreatic cancer surgery: survival determinates. J Gastrointest Surg 13:784–792.
Kaneoka Y, Yamaguchi A, Isogai M. (2009) Portal or superior mesenteric vein resection for pancreatic head adenocarcinoma: prognostic value of the length of venous resection. Surgery 145:417–425.
Martin RCG, Scoggins CR, Egnatashvili V, Staley CA, McMasters KM, Kooby DA. (2009) Arterial and venous resection for pancreatic adenocarcinoma: operative and long-term outcomes. Arch Surg144:154–159.
Kendrick ML, Sclabas GM. Major venous resection during total laparoscopic pancreaticoduodenectomy. HPB (Oxford). 2011 Jul;13(7):454-8
Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 2009; 250(2):187-96.
Breitenstein S, DeOliveira ML, Raptis DA, et al. Novel and simple preoperative score predicting complications after liver resection in noncirrhotic patients. Ann Surg 2010; 252(5):726-34.
Tempero MA, Arnoletti JP, Behrman SW, Ben-Josef E, Benson 3rd AB, Casper ES, et al. Pancreatic adenocarcinoma, version 2.2012: featured updates to the NCCN Guidelines. JNCCN. 2012;10(6):703–13.
Howard TJ, Krug JE, Yu J, et al. A margin-negative R0 resection accomplished with minimal postoperative complications is the surgeon’s contribution to long-term survival in pancreatic cancer. J Gastrointest Surg 2006;10:1338e1345; discussion 1345_1336.
Pawlik TM, Gleisner AL, Cameron JL, et al. Prognostic relevance of lymph node ratio following pancreaticoduodenectomy for pancreatic cancer. Surgery 2007;141:610e618.
Kuroki T, Adachi T, Okamoto T, Kanematsu T. A non-randomized comparative study of laparoscopy-assisted pancreaticoduodenectomy and open pancreaticoduodenectomy. Hepatogastroenterology. 2012 Mar-Apr;59(114):570-3.
Chalikonda S, Aguilar-Saavedra JR, Walsh RM. Laparoscopic robotic-assisted pancreaticoduodenectomy: a case-matched comparison with open resection. Surg Endosc. 2012 Sep;26(9):2397-402.
Conflict of Interest
None of the authors have any conflict of interest to disclose. The manuscript did not receive any funding.
Author information
Authors and Affiliations
Corresponding author
Additional information
Discussant
Dr. R Matthew Walsh (Cleveland Heights, OH):
This study represents a great achievement in laparoscopic vein resection with laparoscopic Whipple procedure. It compares the results of one outstanding laparoscopic surgeon with his colleagues of equally outstanding success with open resections.
I have a few questions:
1. Can you readily tell that a vein resection needs to be done during laparoscopic dissection since imaging may be unreliable and some patients had neoadjuvant treatments? Did you compare pathologic vein involvement to be sure it is not over utilized?
2. Given the results of the NSQIP HPB demonstration project show nationally poor outcomes in adopting lap Whipple procedures, when in the learning curve should this be considered and what did you learn from the four patients you could not reconstruct as your own patient selection criteria?
3. The time to reconstruct in your expert hands was twice that of open. That did not change the outcomes here, but is that an important outcome measure for those embarking on this procedure?
Closing Discussant
Dr. Croome
Thank you Dr. Walsh for your comments and insightful questions.
It is our opinion that the magnification and fine dissection afforded by laparoscopic approaches enhances the intraoperative assessment of vascular involvement. Our pathologists do not consistently report confirmation of vascular invasion. Based on preoperative imaging and intraoperative assessment, we recommend vein resection for all patients with tumor adherence to avoid dissemination of tumor.
The learning curve for minimally invasive pancreaticoduodenectomy is quite long secondary to the advanced skills of resection and reconstruction required. The addition of a complex vascular resection and reconstruction certainly increases the difficulty. We recommend that laparoscopic major vascular resection be attempted only after considerable experience in open and laparoscopic pancreaticoduodenectomy as well as open major vascular resection and reconstruction. Conversion to an open approach should not be considered a complication, but rather sound clinical judgment to assure the best possible outcome for any given patient. In this study, the four patients were converted to open for either expected difficult reconstruction of the pancreatic anastomosis or vascular reconstruction and occurred at various stages of our learning curve.
The duration of reconstruction, and therefore clamp time, was longer for the laparoscopic group. This not only may be due to the learning curve of a new approach but also may be attributed to the greater time required to reconstruct using a long bovine pericardial patch venorrhaphy. We identified no complications that could be attributed to a longer clamp time.
Rights and permissions
About this article
Cite this article
Croome, K.P., Farnell, M.B., Que, F.G. et al. Pancreaticoduodenectomy with Major Vascular Resection: a Comparison of Laparoscopic Versus Open Approaches. J Gastrointest Surg 19, 189–194 (2015). https://doi.org/10.1007/s11605-014-2644-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-014-2644-8