Abstract
Himalayan glaciers are retreating, and glacial lakes are evolving and proliferating as a result of climate change. Glacier retreat marks in the formation and expansion, and sometimes outburst of moraine-dammed lakes. Lato Lake is one of the high-altitude and unexplored glacial lakes upstream of Gya-Miru village in the Leh-Ladakh region. This study is the first of its kind to assess hydrogeochemistry (HCO3−, SO42−, NO3−, Cl−, F−, Ca2+, Mg2+, Na+, and K+) and studying the dynamics of a moraine-dammed lake in the Ladakh Himalayas. We observed substantial expansion of Lato Lake over the past 50 years between 1969 and 2019, and the areas of the lake have increased while the glacier area is reduced by 16.4% and 4.15%, respectively. The pH values ranging from 7.6 to 8.1 show slightly alkaline. HCO3−, Ca2+, and SO42− were the most dominant ions during the study period 2018–19. The high (Ca2+ + Mg2+) and a low (Na+ + K+) ratio to the total cations show that Lato Lake receives ions from rock weathering, primarily from carbonate rocks. Gibbs and Na-Mixing plot also support the hydrogeochemistry of lake water was primarily controlled by rock weathering. HYSPILT backward trajectory model suggested that atmospheric input mainly originated from the seawater vapor transported by the summer monsoonal and westerly circulation systems. Results show that the lake has a substantial impact on the long-range transport of ocean water relative to local interferences.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The Himalayan region has gained significant attention due to its large frozen freshwater reserve, some of its large Asian rivers and lakes and the associated glacio-hydrological hazards (Schild 2008; Prakash 2020; Azam et al. 2021; Shugar et al. 2021). Most studies in the Himalayan region showed a glacier mass loss in the past decades and which is predicted to continue with higher rates in the coming decades (Bolch et al. 2019; Rounce et al. 2020; Hugonnet et al. 2021). This will result in the shrinking of the glaciated areas and consequently the water availability in the region. Ladakh region holds many standing fresh and saline water lakes in their glaciated areas among the Himalayan regions. The glacier decline in the Ladakh region will adversely affect the food security and socioeconomic development in the region (Hock et al. 2019; Nüsser et al. 2019) as most of the villages, e.g., Stok, Lato, Gya and Mathoo (Nusser 2012; Schmidt et al. 2020), are considerably dependent on glacier meltwater during pre-monsoon and summer months especially in low precipitation and drought years (Pritchard 2019). Under rapid warming conditions, down-wasting and terminus retreat of glaciers has aggravated the evolution of new pro-glacial lakes and the growth of pre-existing lakes (Thakuri et al. 2016; Bolch et al. 2019; Shugar et al. 2020). The failure of moraine-dammed lakes can turn into the Glacial Lake Outburst Floods (GLOFs) and pose a severe threat to the downstream community and village infrastructure, like one catastrophe in August 2014 in Gya village of Ladakh region (Schmidt et al. 2020; Majeed et al. 2021). Numerous factors are responsible for triggering the GLOFs such as heavy rainfall, earthquake, rock avalanche and landslide (Allen et al. 2016; Gurung et al. 2017; Hock et al. 2019; Nüsser et al. 2019). Such hazards related to GLOFs are most deceptive in the past few decades in the Himalayan region, where the number of GLOFs events has been increased significantly (Veh et al. 2019; Schmidt et al. 2020; Majeed et al. 2021; Shugar et al. 2021). Few studies have been conducted to account for the lake expansion in the Ladakh region regarding the impact of glacier recession and associated lake development due to climate change (Schmidt et al. 2020; Majeed et al. 2021). High-resolution satellite imagery is beneficial to account and monitor the evolution of glacial lakes and glacier changes in the Himalayan region (Schmidt et al. 2020; Pandey et al. 2021; Majeed et al. 2021; Allen et al. 2021) and gained considerable attention in the recent past decades.
Changing climate has also altered the general hydrogeochemistry of glacial lake water and shift in the biophysiochemical exchanges in the catchment area (Roberts et al. 2017; Woelders et al. 2018; Steingruber et al. 2020). It is predicted that the high-altitude lakes in the Himalayan region and Arctic are likely to respond to present climate instability, leading to variations in lake water chemical composition (Salerno et al. 2016; Kosek et al. 2021; Ruman et al. 2021). Lakes ecosystems are strongly coupled to the features of their different climatic regimes, neighboring landscapes, geomorphological characteristics, local vegetation, bedrock lithology, and accumulation of anthropogenic activity (Salerno et al. 2016; Gurung et al. 2018; Ruman et al. 2021; Kosek et al. 2021). Most of the glacial lakes are situated in a pristine environment high up in the Himalayan ranges and Arctic region, which are generally less impacted by the direct influence of anthropogenic activity (e.g., Samudra Tapu, Gepang Gath, Dehnasar, Satopanth Tal, Bhrigu Lakes in Himalayas and Hazen, Kongressvatn, Bungevatnet, and Revvatnet Lakes in high arctic regions). Climate change has also increased the moisture transport through the wind circulation from oceans to the high-mountains and polar regions and falling either as solid or liquid precipitation and brings a variety of pollutants (Kosek et al. 2021; Ruman et al. 2021). Any addition of these pollutants in the form of sulfur, nitrate-containing compounds, persistent organic pollutants (POPs) and trace elements to the glacial lake ecosystem via atmospheric deposition, there is a negligible chance for removal or uptake of such pollutants due to the limited buffering capacity of the lake (Li et al. 2014; Ruman et al. 2021). Therefore, high-altitude lake is identified as a sensitive indicator of changing climate in the global and regional environment (Karlsson et al. 2015; Salerno et al. 2016; Roberts et al. 2017; Woelders et al. 2018). Several studies are conducted on the impact of climate change through hydrochemistry on the glacial lakes of the Himalayas and High Arctic region (Karlsson et al. 2015; Dean et al. 2016; Roberts et al. 2017; Gurung et al. 2018; Woelders et al. 2018; Shukla et al. 2018; Slukovskii et al. 2020; Ruman et al. 2021; Khadka and Ramanathan 2021; Kaphle et al. 2021; Majeed et al. 2021). However, only a few Himalayan glacial lakes have been studied due to their inaccessibility, harsh climatic conditions, and challenging landscape.
Here, in this work, we study the evolution of Lato glacier lake in the Ladakh region with consideration to ongoing climate change and its impact. The Lato Lake is situated at the junction of two atmospheric circulation systems (mid-latitude westerlies and the Indian summer monsoon); thus, Lato Lake is a suitable site to understand the various environmental changes contributed from these circulations (Kumar et al. 2020). The current study focuses (a) to understand the sources of solute and factors controlling the hydrogeochemistry of the Lato Lake (b) to investigate the area change of Lato Glacier and lake under changing climate based on remote sensing datasets and (c) to understand the effect of long-range transport and atmospheric deposition in the hydrochemistry of lake water. This study provides a baseline dataset for further research work on the impact of climate change on hydrochemistry and area changes of the moraine-dammed lakes in the Ladakh and adjoining regions. This study will also help in policy and strategies to reduce the impact of GLOFs and adaptation practices on the forecasted changes in the region.
Site description and geology of the study area
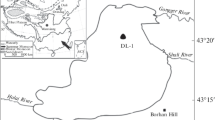
Lato Lake is one of the unexplored high-altitude lake near Lato and Gya-Miru Village in the Leh-Ladakh region (Fig. 1). The region is a core constituent of orthogenesis and granite, with late Cenozoic and Precambrian metasedimentary rocks surrounding it (Schlup et al. 2003). Meteorological data from Leh’s weather station are considered to reflect the semi-arid climate of Zanskar’s (Osmaston 2001). This lake is located 15 km away from Lato village and ~ 80 km from Leh city. The maximum length of the lake is about 900 m with 300 m width and located at an altitude of around 5200 m.a.s.l. Using a simple approach based on area-depth relationship, followed by several studies in the Himalayan region (Fujita et al. 2009; ICIMOD, 2011; Sakai, 2012; Fujita et al. 2013, Patel et al. 2017), the mean depth of Lato Lake is about 19 m. The present study focuses on the Lato Glacier and Lato village (altitude: ~ 3900 m.a.s.l) located in the Leh-Ladakh, Western Himalaya. At present, the glacier area is about 5.06 km2 extending ~ 3.8 km, with the glacier snout located at an elevation of 5300 m.a.s.l. Glacial meltwater contributes to the various agricultural and domestic requirements in the downstream village. Lato Lake is connecting with a series of small lakes, and these small lakes are continuously expanding (according to our field observation of 2 years). There are no documented works available on the glacier/lake area change and hydrochemistry of the lake. In this region, the meltwater from the glacier is terminating in Lato Lake and finally drains downstream. The stream from the Lato catchment fed the Lato and Miru village (∼100 households; ∼500 individuals) before the confluence with the Chabe Nama stream and further with the Indus River.
Materials and methods
Sampling preparation and analytical procedure
Samples were collected from Lato Lake in the month of July during 2018–2019. The samples (n = 20) were gathered from different locations around the lake (Fig. 1). Samples were taken in pre-cleaned high-density plastic bottles (100 ml) around 10–15 cm beneath the surface water. The bottles were washed thrice with the lake water before the sample collection. In situ measurements of pH and EC were carried out using a portable Multiparameter kit (Thermo-STARA3250 series). For chemical analysis, the collected water samples were transported to the School of Environmental Science laboratory, Jawaharlal Nehru University, New Delhi. They were then filtered using 0.45 μm pore size syringe filters (Axiva SFNY25X Nylon) and refrigerated at 4 °C before analysis. Cations (Ca2+, Mg2+, Na+, and K+) were analyzed by ICP-OES (Thermo iCAP 76,000 Series), whereas anions (F−, Cl−, NO3− and SO42−) were determined by ion chromatography (Metrhom 930 Compact IC Flex systems). HCO3− was analyzed by acid titration using the brucine-sulphanilic acid method (APHA 2005). In order to avoid contamination, masks and gloves were used during the sampling and analysis in the laboratory. Freshly prepared standards were used for the analysis, where blank standard and sample after analysis showed no cross-contamination. Limits of detection (LOD) and quantification (LOQ), precision and accuracy of major ions for 2018 and 2019 are presented in Table 1. Error and accuracy of the major ions data are calculated by using the formula {(TZ+–TZ−)/(TZ+–TZ)} × 100 which represent < 10% error.
Statistical software
Aquachem software (Version 5.1)) was used for plotting the Piper diagram, whereas for statistical analysis, SPSS software (Version 10.5) was used for principal component analysis, and R-Studio software was used for correlation matrix.
HYSPLIT backward trajectory model
The backward trajectory model is a valuable technique for tracing the sources of atmospheric input in the high-altitude glacial lake (Diéguez et al. 2019; Deka et al. 2015a). In order to trace the atmospheric input into the lake environment, trajectories were computed from the HYSPLIT model using the open-source available at the Air Resources Laboratory website (http://www.arl.noaa.gov/ready.html) (Stein et al. 2015; Rolph et al. 2017). Archive data set from Global Data Assimilation (GDS) was used as meteorological input. The trajectory’s height was evaluated at three different altitudes of 5000, 5100, and 5200 above sea level and run for 120 h from the lake location.
Satellite data
The study uses easily accessible satellite images to define the glacier and lake boundaries of different years (Fig. 2). We use images of the Landsat series from 1991, 1998, 2001, 2010, and 2019 (TM, ETM + , and OLI) to delineate glacier and lake boundaries. We use the semi-automatic method of shortwave infrared and red band ratio with other manual modifications (Racoviteanu et al. 2009). Followed by then, we also use declassified aerial corona images from 1969 to 1993 to find out the lake and glacier area (detailed in the paper of Soheb et al. 2020).
Results and discussion
Hydrochemistry of the lake
Hydrogeochemical characteristics of lake water provide vital information about the solutes sources and weathering processes in the glacial lake environment. The average chemical configuration of lake water is presented in Table 2. The lake is mostly alkaline, with pH ranging between 7.6 and 8.1, whereas the electrical conductivity ranges between 29 and 61 μS/cm during the study period. Lake water chemistry shows anionic abundance: HCO3− > SO42− > Cl− > NO3− > F− in 2018, while in 2019, HCO3− > SO42− > NO3− > Cl− > F−, whereas phosphorus was below detection limit in both the years. Cationic abundance varied as follows: Ca2+ > Mg2+ > Na+ > K+ in 2018, while in 2019, Ca2+ > Mg2+ > K+ > Na+. However, Na+ prevailing over K+ due to its greater mobility and weathering of silicate minerals from the metamorphic and igneous rocks. (Sharma et al. 2013). HCO3− is the most predominant ion followed by Ca2+ and SO42− as in other Himalayan glacial lakes presented in Table 3. Among the cations, Ca2+ and Mg2+ are the major constituents and produce 68 and 18% of the total cations, whereas for the anions, HCO3− and SO42− are the major constituent and produce 76 and 13% of the total anions.
Ionic Ratio and Effective CO2 pressure
Ionic ratios of Lato Lake during the period 2018–19 are presented in Table 2. Ionic ratios were Na+/Cl− > 1 in 2018 and > 2 in 2019, while K+/Cl− > 0.9 in 2018 and > 2 in 2019. All the ratios were greater than the resultant seawater ratios (K+/Cl− = 0.02 and Na+/Cl− = 0.86) (Pant et al. 2018). These ratios revealed the influence of atmospheric contributions in lake water (Singh et al. 2014b). The comparatively high (Mg2+ + Ca2+)/TZ+ (> 0.75) and the low of (Na+ + K+)/TZ + (< 0.15) ratio revealed that the hydrochemistry is mainly dominated by carbonate weathering afterward silicate weathering (Singh et al. 2016). The cationic ratio of [Mg2+ + Ca2+)/ Na+ + K+] is generally used to estimate the comparative influence of different rock types in the catchment (Ahmad et al. 1998; Zhang et al. 2007). The high molar ratio (> 6 in 2018 and > 15 in 2019) signifying the relative weakness of silicate dissolution and suggested the dominance of dolomite and calcite rich-weathering (Singh et al. 2014b).
C-ratio and pCO2 variations in Lato Lake were presented for two years of 2018–19. pCO2 was calculated using pH and HCO3− based on Wadham et al. (2001). The average pCO2 values for Lato Lake were 10–4.16 atm during 2018 and 10–4.26 atm during 2019, respectively. The lower value of atmospheric pCO2 during 2018–19 signified the dissolution of CO2 into the lake water. If the adequate pressure of CO2 in the solution is not equal to 10–3.5, then it is considered as an imbalance concerning the atmosphere. The low value of pCO2 in the lake supported the coupling of carbonate dissolution and sulfide oxidation (Wadham et al. 1998). High-altitude glacial lakes are extremely cold, while CO2 has a comparatively higher solubility rate associated with the release of surplus CO2 to the atmosphere (Stumm and Morgan 1981). The average value of C-ratios is 0.92 ± 0.03 and 0.81 ± 0.02 during 2018–19. According to the earlier study, C-ratio indicates a huge massive source of atmospheric CO2 increasing carbonate weathering when it is > 0.5 (Brown et al. 1996), the C-ratio in Lato Lake is quite high (0.81–0.92) hence suggests protons are resultant primarily from the dissociation and dissolution of atmospheric CO2.
Hydrogeochemical features
We used a piper diagram (Piper 1944) for representing the association between water and rock types present in the lake (Fig. 2). In the piper plot, the cation triangle showed that maximum samples lie on the bottom-left side, suggesting the domination of Ca2+ in the lake water, whereas anion triangle presented that most meltwater samples are located in the bottom-left side close to the HCO3-apex, indicating the domination of carbonate-rich lithology. The piper diagram showed the chemical facies of Lato Lake lied in the Ca–HCO3 type for both years. Lake water composition is mostly mainly influenced by the bedrock lithology of the catchment area (carbonates, sulfide minerals, and silicates), which play a crucial role in varying the hydrochemical facies in the formation of Ca–HCO3 type (Zang et al. 2020). The Piper diagram also suggested the dominance of alkaline earth metals (Ca2+ + Mg2+) over alkalis earth metals (Na+ + K+) and weak acid (HCO3−) dominance over strong acid (SO42− + Cl−). There were no significant changes seen in the hydrochemical facies in the lake in 2018–19, which suggested that major ions primarily originated from the chemical weathering of carbonate rock from the lake catchment area.
Gibbs plot
The climatic parameters, mainly precipitation and temperature, have a large impact on the concentration of ions in the lake and glacial catchments. Lake hydrochemistry and its governing mechanism in a glacial environment can be qualitatively studied with Gibb’s semi-logarithmic coordinates (Gibbs 1970). The ratios of Cl−/(Cl− + HCO3−) and Na+/(Na+ + Ca2+) in lake are < 0.5 and the distribution of water samples lies in the middle of the left side (Fig. 3). A lower concentration of Cl− and Na+ and a higher concentration of HCO3− and Ca2+ reflect that all the sites are mainly influenced by rock weathering of carbonate type. The results of these Gibbs plots revealed that the chemical characteristic of water is primarily governed by the geology of the area, which is consistent with the piper plot described above. Our outcomes are comparable with past studies of other Himalayan lakes and show rock weathering plays a significant role in glacial and river runoffs (Kumar et al. 2019; Singh et al. 2016).
Scatter diagram and mixing plot
SO42− is the most abundant anion in both years after HCO3− in the Lato Lake. The possible sources of SO42− in the Lato Lake could be from the natural origin, such as weathering of pyrite and gypsum (own mineralogy data, which are not shown here). In addition, anthropogenic (combustion of sulfur-containing fossil fuels and the smelting of nonferrous metals) and biological sources of SO42− can also be contributing through atmospheric transport (Potapowicz et al. 2020). Overall rock-water interactions, atmospheric deposition, biological activity, morphology, and human activities are likely to affect the hydrogeochemistry in glacial lake environments (Singh et al. 2012). Sulfur dioxide in the atmosphere can further be reacted with water vapor to form H2SO4, and affecting the pristine lake environment (Zhang et al. 2020). The relationship between Ca2+ and SO42− (Fig. 4) indicates that the samples in the Lato Lake for both years are found below the 1:1 gypsum dissolution line and below the 1:2 sulfide-carbonate weathering line. This implies that the majority of excess Ca2+ is coming from carbonate dissolution and silicate weathering. Hence, the contribution of sulfide dissolution from natural origin (like from evaporites sources) to the hydrochemistry in Lato Lake is insignificant. Therefore, the SO42− contribution to the total anions 8.79% in 2018 and 17.73% in 2019, respectively, may be attributable to a weak influence from atmospheric acid deposition. Glacier mass loss is also considered a key factor for the rise of sulfate in lake water attributed to chemical variations, primarily to the sulfide oxidation that occurs in the subglacial catchment (Salerno et al. 2016).
Mixing end members among weathering products such as carbonates, silicates, and evaporates are widely used to study the rock weathering contribution to the primary element compositions in various hydrological regimes (Cao et al. 2016; Ollivier et al. 2010). The Na-standardized molar ratio of the Lato Lake for 2018 and 2019 is shown in Fig. 5. The plots show a high contribution of Ca2+ and HCO3−, corresponding to the shift of the water samples toward the carbonate weathering end member. It shows that the Lato Lake’s major ion chemistry is contributed chiefly by the CO3− dissolution (~ 75%) and little contribution (~ 25%) from silicate and none from evaporates weathering. The dominance of carbonate dissolution over silicate weathering and the insignificant contribution from evaporates during the study suggest that Lato Lake functions predominantly as a temporary CO2 sink. This observation is significant for the global carbon cycle since carbonate dissolution consumes CO2 released into the atmosphere from oceans through carbonate precipitation over the time scale of thousands of years (Cao et al. 2015). The hydrochemistry in the lake is likely influenced by atmospheric deposition inputs, chemical and mineral weathering of rocks.
Major sources and controlling factors of the hydrochemistry
Correlations Analysis
Correlation analysis can be useful for establishing the interrelationship among two variables and prophesying the degree of dependency of inter-elemental variables. In this present study, the correlation matrix is represented in the hydrochemical variables in Lato Lake (Fig. 6). In both years, we observed a strong positive association among Ca2+–SO42−, Mg2+–SO42−, Na+–SO42−. These positive associations suggested a similar source of these ions, and they might be coming from the pyrite oxidation, as well as and dissolution of sulfide minerals (Salerno et al. 2016; Singh et al. 2016). On the contrary, the relationship among HCO3−–Ca2+, HCO3−–Mg2+, HCO3−–Na+ and HCO3−–K+ is not significant. The relationship between SO42− and HCO3− was not significant during the study period, whereas Na+ and K+ showed a strong positive association during 2019, indicating a similar origin of these two ions most possibly associated with the weathering silicate minerals. The negative correlation of Na+–Cl− in 2018–19 indicates halite’s interface with meltwater and supported common lithogenic and marine inputs of these solutes (Jiang et al. 2015).
Principal Component Analysis
PCA is a useful statistical technique applied to the hydrochemical parameter of lake water to reduce the dimensionality containing interrelated datasets (Singh et al. 2004). Factor analysis was applied to 12 parameters based on the 20 water samples. Factor analysis is categorized as strong and moderate loading equivalent to absolute values > 0.75 and 0.75–0.5 (Liu et al. 2003). It is indicated that three-factor loading with eigenvalues > 1, explained about 77.14% of the total variance in the dataset (Fig. 7). Factor 1 accounting for 35.54% of the total variance has strong positive loading of EC, NO3− and SO42−, whereas moderate negative loading of Cl−, K+ and Na+. The group of these hydrochemical parameters specifies diverse sources, for instance, atmospheric deposition, sulfide oxidation, and weathering of silicate mineral’s might be the governing factor of these ions (Pant et al. 2018). Factor 2 accounted for 26.62% of the total variance, with strong positive Ca2+ and Mg2+ and moderate loading with TDS. These attributes specify that CO3− weathering is the primary source of these ions. Factor 3 accounts for 15.07% of the total variance while pH and HCO3− show moderate positive loading which reveals that the potential influences of rock–water interaction, as well as atmospheric deposition (Sun et al. 2010).
Cluster analysis (CA)
Hierarchical CA is a useful statistical tool applied to the hydrogeochemical dataset to group similar sampling sites (Pacheco Castro et al. 2018). Hierarchical cluster analysis shows that lake water is divided into two principal cluster components presented through the dendrogram (Fig. 8). In both years, all sampling sites reflected low-to-moderate ionic strengths in the lake water. Meltwater samples from the similar sampling site reach were nearly classified into a similar cluster, indicating the similarity of geological background and hydrochemical characteristics with some minor changes over both years of the study. Present studies show a good agreement with previous studies conducted in other parts of the Himalayan Lake (Singh et al. 2016). Additionally, rock–water interactions, atmospheric deposition, biological activity, and morphology may also have been altered the hydrogeochemistry (Singh et al. 2012; Szopińska et al. 2018).
Tracing the atmospheric sources through HYSPLIT backward trajectory
Using the HYSPLIT model, the air mass is tracked during the study time to trace the sources of atmospheric input in the lake environment. The black star represents the lake location, and HYSPLIT backward trajectory modeled the sources of water vapor. Figure 9a and b shows the possible source of atmospheric input reaching Lato Lake during the sampling period. The air mass trajectories (Fig. 9a) suggested that air inward at the locality of the lake had originated from the westward direction from the Gulf of Persian/Arabian in 2018, while during 2019 (Fig. 9b), it shows the eastward direction from the Bay of Bengal. In the lake, the primary source of water vapor came from the Gulf of Persian/Arabian in July 2018 due to the westerly circulation system, while in July 2019, it came from the Bay of Bengal due to the Indian summer monsoon. Although the atmospheric sources mainly come from the seawater vapor transported by the monsoonal and westerly circulation systems, the results show that Lato Lake has substantial impacts on long-range transport rather than local interferences. Long-term monitoring can further verify the outcomes of this study.
Recession and expansion of Lato Lake and glacier
Lake, glacier, and snout changes between 1969 and 2019
Geospatial analysis carried out for Lato Lake shows substantial expansion in the lake area over the last five decades between 1969 and 2019. The glacier area is reduced by about 4%, while the lake area has increased by ~ 16%. The result shows that increased melting of the feeding Lato Glacier contributes to lake area expansion. The magnitude of the glacier retreat rate is 0.08%, while glacial lake increase rate is of 0.33% per year between 1969 and 2019. The lake area has increased about six times since 1969 (Figs. 10 and 11), which is quite significant in terms of GLOF hazards potential. Nüsser (2012) and Soheb et al. (2020) have reported a significant increase in the Ladakh region’s air temperature and precipitation variability, which could be the primary cause of lake area expansion. Such expansion of lake area and water volume is a rising concern for the downstream villages, which could be vulnerable to catastrophic natural hazards like GLOF or avalanche break (Schmidt et al. 2020; Majeed et al. 2021). Such lake-related floods and catastrophic events are becoming more frequent in the Himalayan region, such as in 2014 adjacent to Gya village in the same catchment (Schmidt et al. 2020; Majeed et al. 2021).
Environmental implication and concern
The Himalayas has steadily laid more than 5,000 moraine-dammed glacial lakes that are possibly unstable (Kumar and Murugesh 2012). Thinning of glaciers due to retreating/shrinking and melting has aggravated the evolution of pro-glacial lakes and the amplification of present ones behind loosely consolidated moraines (Kumar and Murugesh 2012). These moraine-dammed glacial lakes are potentially unstable, and when it breaches, GLOF can cause geomorphic and disastrous societal impact (Veh et al. 2020). Several catastrophic in this region, Gya village of Ladakh, witnessed a GLOF in 2014 while a flash flood in Leh city in 2010 (Majeed et al. 2021). GLOF and debris flow catastrophic event in Kedarnath, Uttarakhand in June 2013 and a massive rock and ice avalanches in 7th February 2021 were the destructive disasters in the Chamoli district Uttarakhand (Dash and Punia. 2019; Shugar et al.2021). Hence, the monitoring of glacial lakes and preparedness for disaster risk reduction are some of the prime objectives of this study. If future projected glaciers mass loss remains, there is a possibility of forming more unstable moraine-dammed lakes; eventually, GLOF events in these regions may increase in the future. The gradual intensification and significant fluctuation of temperature and precipitation patterns can further result in frequent flash floods, cloudbursts, landslides, and GLOF in a climate-sensitive region like Ladakh with much higher intensity than reported earlier (Schmidt et al. 2020; Majeed et al. 2021).
Conclusions
The study presents two years’ primary dataset of the hydrogeochemistry and area change of moraine-dammed lake in the Ladakh region of western Himalaya. Our study covering 20 surface water samples reveals that the lake water is slightly alkaline with pH between 7.4 and 8.1. HCO3−, SO42− and Ca2+ were the most dominant ions, respectively. CO3− weathering is the primary source of major ions, showing that the hydrochemistry of the lake water is primarily governed by local lithology and the high concentration of sulfate in the samples that are predominantly controlled by wet atmospheric deposition. We also hypothesize that glacier mass loss is another important factor for higher sulfate concentration in the lake water through subglacial sulfite oxidation. Such phenomena were previously reported in 20 Nepalese glacial lakes (Salerno et al. 2016). This study also explores the link between Lato Lake’s area increase and Lato Glacier fluctuations under the influence of climate change. Between 1969 and 2019, Lato Lake expanded from 14,184 to 86,290 m2 at the rate of 0.33% per year. Glacier area has reduced by about 4%, while the area of the lake area has increased by 16%, which is six times increased since 1969. This observation is quite significant in terms of GLOF hazards potential in the future with immediate impacts on the people of Lato village. Hereafter, the current study results build the baseline dataset, and continuous monitoring programs are suggested for the expansion and assessment of the lakes to trace the impact of climate change.
References
Ahmad T, Khanna PP, Chakrapani GJ, Balakrishnan S (1998) Geochemical characteristics of water and sediment of the Indus River, Trans-Himalaya, India: constraints on weathering and erosion. J Asian Earth Sci 16(2–3):333–346. https://doi.org/10.1016/S0743-9547(98)00016-6
Allen SK, Linsbauer A, Randhawa SS, Huggel C, Rana P, Kumari A (2016) Glacial lake outburst flood risk in Himachal Pradesh, India: an integrative and anticipatory approach considering current and future threats. Nat Hazards 84(3):1741–1763. https://doi.org/10.1007/s11069-016-2511-x
Allen S, Sattar A, King O, Zhang G, Bhattacharya A, Yao T, Bolch T (2021) Glacial lake outburst flood hazard under current and future conditions: first insights from a transboundary Himalayan basin. Nat Hazards and Earth Syst Sci Discuss. https://doi.org/10.5194/nhess-2021-167
Anshumali RAL (2007) Seasonal variation in the major ion chemistry of Pandoh Lake, Mandi District, Himachal Pradesh, India. Appl Geochem 22:1736–1747. https://doi.org/10.1016/j.apgeochem.2007.03.045
APHA (2005) Standard methods for the examination of water and wastewater. APHA Washington DC, USA
Azam MF, Kargel JS, Shea JM, Nepal S, Haritashya UK, Srivastava S, Bahuguna IM (2021) Glaciohydrology of the Himalaya-Karakoram. Science. https://doi.org/10.1126/science.abf3668
Bolch T, Shea JM, Liu S, Azam FM, Gao Y, Gruber S, Zhang Y (2019) Status and change of the cryosphere in the extended Hindu Kush Himalaya region. The Hindu Kush Himalaya Assessment. Springer, Cham, pp 209–255
Brown G, Sharp M, Tranter M (1996) Subglacial chemical erosion: seasonal variations in solute provenance, Haut Glacier D’ArollaD’Arolla, Valais, Switzerland. Ann Glaciol 22:25–31. https://doi.org/10.3189/1996AoG22-1-25-31
Cao Y, Tang C, Song X, Liu C (2015) Major ion chemistry, chemical weathering, and CO2 consumption in the Songhua River basin, Northeast China. Environ Earth Sci 73(11):7505–7516. https://doi.org/10.1007/s12665-014-3921-2
Cao Y, Tang C, Cao G, Wang X (2016) Hydrochemical zoning: natural and anthropogenic origins of the major elements in the surface water of Taizi River Basin, Northeast China. Environ Earth Sci 75(9):811. https://doi.org/10.1007/s12665-016-5469-9
Dash P, Punia M (2019) Governance and disaster: analysis of land use policy with reference to Uttarakhand flood 2013, India. Int J Disaster Risk Reduct 36:101090. https://doi.org/10.1016/j.ijdrr.2019.101090
Dean JF, Billett MF, Baxter R, Dinsmore KJ, Lessels JS, Street LE, Wookey PA (2016) Biogeochemistry of “pristine” freshwater stream and lake systems in the western Canadian Arctic. Biogeochemistry 130(3):191–213. https://doi.org/10.1007/s10533-016-0252-2
Deka JP, Baruah B, Singh S, Chaudhury R, Prakash A, Bhattacharyya P, Kumar M (2015a) Tracing phosphorous distributions in the surficial sediments of two eastern Himalayan high altitude lakes through sequential extraction, multivariate and HYSPLIT back trajectory analyses. Environ Earth Sci 73(11):7617–7629. https://doi.org/10.1007/s12665-014-3931-0
Deka JP, Tayeng G, Singh S, Hoque RR, Prakash A, Kumar M (2015b) Source and seasonal variation in the major ion chemistry of two eastern Himalayan high altitude lakes. India. Arab J Geosci 8(12):10597–10610. https://doi.org/10.1007/s12517-015-1964-7
Diéguez MC, Bencardino M, García PE, D’Amore F, Castagna J, De Simone F, Sprovieri F (2019) A multi-year record of atmospheric mercury species at a background mountain station in Andean Patagonia (Argentina): temporal trends and meteorological influence. Atmos Environ 214:116819. https://doi.org/10.1016/j.atmosenv.2019.116819
Fujita K, Sakai A, Nuimura T, Yamaguchi S, Sharma RR (2009) Recent changes in Imja Glacial Lake and its damming moraine in the Nepal Himalaya revealed by in situ surveys and multi-temporal ASTER imagery. Environ Res Lett 4(4):045205. https://doi.org/10.1088/17489326/4/4/045205/meta
Fujita K, Sakai A, Takenaka S, Nuimura T, Surazakov AB, Sawagaki T, Yamanokuchi T (2013) Potential flood volume of Himalayan glacial lakes. Nat Hazard 13(7):1827–1839. https://doi.org/10.5194/nhess-13-1827-2013
Gibbs RJ (1970) Mechanisms controlling world water chemistry. Science 170(3962):1088–1090. https://doi.org/10.1126/science.170.3962.1088
Gurung DR, Khanal NR, Bajracharya SR, Tsering K, Joshi S, Tshering P, Penjor T (2017) Lemthang Tsho glacial Lake outburst flood (GLOF) in Bhutan: cause and impact. Geoenviron Disasters 4(1):1–13. https://doi.org/10.1186/s40677-017-0080-2
Gurung S, Gurung A, Sharma CM, Jüttner I, Tripathee L, Bajracharya RM, Guo J (2018) Hydrochemistry of Lake Rara: a high mountain lake in western Nepal. Lakes Reserv Res Manag 23(2):87–97. https://doi.org/10.1111/lre.12218
Hock R, Rasul G, Adler C, Cáceres B, Gruber S, Hirabayashi Y, & Zhang Y (2019) High mountain areas, 131–202.
Hugonnet R, McNabb R, Berthier E, Menounos B, Nuth C, Girod L, Kääb A (2021) Accelerated global glacier mass loss in the early twenty-first century. Nature 592(7856):726–731. https://doi.org/10.1038/s41586-021-03436-z
International Centre for Integrated Mountain Development (2011) Glacial lakes and glacial lake outburst floods in Nepal. ICIMOD, Kathmandu, Nepal, p 99
Jiang L, Yao Z, Liu Z, Wang R, Wu S (2015) Hydrochemistry and its controlling factors of rivers in the source region of the Yangtze River on the Tibetan Plateau. J Geochem Explor 155:76–83. https://doi.org/10.1016/j.gexplo.2015.04.009
Kaphle B, Wang JB, Kai JL, Lyu XM, Paudayal KN, Adhikari S (2021) Hydrochemistry of Rara Lake: a Ramsar lake from the southern slope of the central Himalayas, Nepal. J Mt Sci 18(1):141–158. https://doi.org/10.1007/s11629-019-5910-0
Karlsson JM, Jaramillo F, Destouni G (2015) Hydro-climatic and lake change patterns in Arctic permafrost and non-permafrost areas. J Hydrol 529:134–145. https://doi.org/10.1016/j.jhydrol.2015.07.005
Khadka UR, Ramanathan AL (2013) Major ion composition and seasonal variation in the Lesser Himalayan lake: case of Begnas Lake of the Pokhara Valley, Nepal. Arab J Geosci 6(11):4191–4206. https://doi.org/10.1007/s12517-012-0677-4
Khadka UR, Ramanathan AL (2020) Hydrogeochemical analysis of Phewa lake: a lesser Himalayan lake in the Pokhara Valley, Nepal. Environ Nat Resour J 19(1):68–83. https://doi.org/10.32526/ennrj/19/2020083
Kosek K, Ruman M (2021) Arctic freshwater environment altered by the accumulation of commonly determined and potentially new POPs. Water 13(13):1739. https://doi.org/10.3390/w13131739
Kumar R, Kumar R, Singh S, Singh A, Bhardwaj A, Chaudhary H (2019) Hydro-geochemical characteristics of glacial meltwater from Naradu Glacier catchment, Western Himalaya. Environ Earth Sci 78(24):1–12. https://doi.org/10.1007/s12665-019-8687-0
Kumar O, Ramanathan AL, Bakke J, Kotlia BS, Shrivastava JP, Kumar P, Kumar P (2020) Role of Indian summer monsoon and Westerlies on glacier variability in the Himalaya and East Africa during late quaternary: review and new data. Earth Sci Rev. https://doi.org/10.1016/j.earscirev.2020.103431
Kumar B, Murugesh Prabhu TS (2012) Impacts of climate change: Glacial Lake outburst floods (GLOFs). Climate Change in Sikkim Patterns, Impacts and Initiatives. Inf Public Relat Dep, Gov Sikkim, Gangtok, 81–102.
Li Z, Xu J, Shilpakar RL, Ma X (2014) Mapping wetland cover in the greater Himalayan region: a hybrid method combining multispectral and ecological characteristics. Environ Earth Sci 71(3):1083–1094. https://doi.org/10.1007/s12665-013-2512-y
Liu CW, Lin KH, Kuo YM (2003) Application of factor analysis in the assessment of groundwater quality in a blackfoot disease area in Taiwan. Sci Total Environ 313(1–3):77–89. https://doi.org/10.1016/s0048-9697(02)00683-6
Majeed U, Rashid I, Sattar A, Allen S, Stoffel M, Nüsser M, Schmidt S (2021) Recession of gya glacier and the 2014 glacial lake outburst flood in the Trans-Himalayan region of Ladakh, India. Sci Total Environ 756:144008. https://doi.org/10.1016/j.scitotenv.2020.144008
Nüsser M, Schmidt S, Dame J (2012) Irrigation and development in the Upper Indus Basin: characteristics and recent changes of a socio-hydrological system in Central Ladakh, India. Mt Res Dev 32(1):51–61. https://doi.org/10.1659/MRD-JOURNAL-D-11-00091.1
Nüsser M, Dame J, Parveen S, Kraus B, Baghel R, Schmidt S (2019) Cryosphere-fed irrigation networks in the northwestern Himalaya: precarious livelihoods and adaptation strategies under the impact of climate change. Mt Res Dev 39(2):R1–R11. https://doi.org/10.1659/MRD-JOURNAL-D-18-00072.1
Ollivier P, Hamelin B, Radakovitch O (2010) Seasonal variations of physical and chemical erosion: A three-year survey of the Rhone River (France). Geochimica et Cosmochimica Acta 74(3):907–927
Osmaston, H. (2001). The geology, geomorphology and Quaternary history of Zanskar. Himalayan Buddhist Villages, 1–36.
Pacheco Castro R, Pacheco Ávila J, Ye M, Cabrera Sansores A (2018) Groundwater quality: analysis of its temporal and spatial variability in a Karst Aquifer. Groundwater 56(1):62–72. https://doi.org/10.1111/gwat.12546
Pandey P, Ali SN, Ray PKC (2021) Glacier-glacial lake interactions and glacial lake development in the central Himalaya, India (1994–2017). J Earth Sci. https://doi.org/10.1007/s12583-020-1056-9
Pant RR, Zhang F, Rehman FU, Wang G, Ye M, Zeng C, Tang H (2018) Spatiotemporal variations of hydrogeochemistry and its controlling factors in the Gandaki River Basin, Central Himalaya Nepal. Sci Total Environ 622–623:770–782. https://doi.org/10.1016/j.scitotenv.2017.12.063
Patel LK, Sharma P, Laluraj CM, Thamban M, Singh A, Ravindra R (2017) A geospatial analysis of Samudra Tapu and Gepang Gath glacial lakes in the Chandra Basin, Western Himalaya. Nat Hazards 86(3):1275–1290. https://doi.org/10.1007/s11069-017-2743-4
Piper AM (1944) A graphic procedure in the geochemical interpretation of water-analyses. EOS Trans Am Geophys Union 25(6):914–928. https://doi.org/10.1029/TR025i006p00914
Potapowicz J, Szumińska D, Szopińska M, Bialik RJ, Machowiak K, Chmiel S, Polkowska Ż (2020) Seashore sediment and water chemistry at the Admiralty Bay (King George Island, Maritime Antarctica)–Geochemical analysis and correlations between the concentrations of chemical species. Mar Pollut Bull 152:110888. https://doi.org/10.1016/j.marpolbul.2020.110888
Prakash ANJAL (2020) Retreating glaciers and water flows in the Himalayas: implications for governance. Obs Res Found, New Delhi, India 400:1–14
Pritchard HD (2019) Asia’s shrinking glaciers protect large populations from drought stress. Nature 569(7758):649–654. https://doi.org/10.1038/s41586-019-1240-1
Racoviteanu A, Paul F, Raup B, Khalsa S, Armstrong R (2009) Challenges and recommendations in mapping of glacier parameters from space: results of the 2008 global land ice measurements from space (GLIMS) workshop, Boulder, Colorado, USA. Ann Glaciol 50(53):53–69. https://doi.org/10.3189/172756410790595804
Roberts KE, Lamoureux SF, Kyser TK, Muir DCG, Lafrenière MJ, Iqaluk D, Normandeau A (2017) Climate and permafrost effects on the chemistry and ecosystems of High Arctic Lakes. Sci Rep 7(1):1–8. https://doi.org/10.1038/s41598-017-13658-9
Rolph G, Stein A, Stunder B (2017) Real-time environmental applications and display sYstem: READY. Environ Model Softw 95:210–228. https://doi.org/10.1016/j.envsoft.2017.06.025
Rounce DR, Hock R, Shean DE (2020) Glacier mass change in high mountain Asia through 2100 using the open-source python glacier evolution model (PyGEM). Front Earth Sci 7:331. https://doi.org/10.3389/feart.2019.00331
Ruman M, Kosek K, Koziol K, Ciepły M, Kozak-Dylewska K, Polkowska Ż (2021) A High-Arctic flow-through lake system hydrochemical changes: revvatnet, southwestern Svalbard (years 2010–2018). Chemosphere 275:130046. https://doi.org/10.1016/j.chemosphere.2021.130046
Sakai A (2012) Glacial lakes in the Himalayas: a review on formation and expansion processes. Glob Environ Res 16(2011):23–30
Salerno F, Rogora M, Balestrini R, Lami A, Tartari GA, Thakuri S, Tartari G (2016) Glacier melting increases the solute concentrations of Himalayan glacial lakes. Environ Sci Technol 50(17):9150–9160. https://doi.org/10.1021/acs.est.6b02735
Schild A (2008) ICIMOD’s ICIMOD’s position on climate change and mountain systems. Mt Res Dev 28(3/4):328–331. https://doi.org/10.1659/mrd.mp009
Schlup M, Carter A, Cosca M, Steck A (2003) Exhumation history of eastern Ladakh revealed by 40 Ar/ 39 Ar and fission-track ages: the Indus River-Tso Morari transect, NW Himalaya. J Geol Soc 160(3):385–399. https://doi.org/10.1144/0016-764902-084
Schmidt S, Nüsser M, Baghel R, Dame J (2020) Cryosphere hazards in Ladakh: the 2014 Gya glacial lake outburst flood and its implications for risk assessment. Nat Hazards 104(3):2071–2095. https://doi.org/10.1007/s11069-020-04262-8
Sharma P, Ramanathan AL, Pottakkal J (2013) Study of solute sources and evolution of hydrogeochemical processes of the Chhota Shigri Glacier meltwaters, Himachal Himalaya, India. Hydrol Sci J 58(5):1128–1143. https://doi.org/10.1080/02626667.2013.802092
Shugar DH, Burr A, Haritashya UK, Kargel JS, Watson CS, Kennedy MC, Strattman K (2020) Rapid worldwide growth of glacial lakes since 1990. Nat Clim Chang 10(10):939–945. https://doi.org/10.1038/s41558-020-0855-4
Shugar DH, Jacquemart M, Shean D, Bhushan S, Upadhyay K, Sattar A, Westoby MJ (2021) A massive rock and ice avalanche caused the 2021 disaster at Chamoli, Indian Himalaya. Science. https://doi.org/10.1126/science.abh4455
Shukla A, Garg PK, Srivastava S (2018) Evolution of glacial and high-altitude lakes in the Sikkim, Eastern Himalaya over the past four decades (1975–2017). Front Environ Sci 6:81. https://doi.org/10.3389/fenvs.2018.00081
Singh KP, Malik A, Mohan D, Sinha S (2004) Multivariate statistical techniques for the evaluation of spatial and temporal variations in water quality of Gomti River (India)-A case study. Water Res 38(18):3980–3992. https://doi.org/10.1016/j.watres.2004.06.011
Singh VB, Ramanathan AL, Pottakkal JG, Sharma P, Linda A, Azam MF, Chatterjee C (2012) Chemical characterization of meltwater draining from Gangotri glacier, Garhwal Himalaya, India. J Earth Syst Sci 121(3):625–636. https://doi.org/10.1007/s12040-012-0177-7
Singh VB, Ramanathan AL, Pottakkal JG, Kumar M (2014a) Seasonal Variation of Solute and Suspended Sediment Load in Gangotri Glacier Meltwater, Central Himalaya, India. J Asian Earth Sci 79:224–234. https://doi.org/10.1016/j.jseaes.2013.09.010
Singh Y, Khattar JIS, Singh DP, Rahi P, Gulati A (2014) Limnology and cyanobacterial diversity of high altitude lakes of Lahaul-Spiti in Himachal Pradesh, India. J Biosci 39(4):643–657
Singh VB, Ramanathan AL, Mandal A (2016) Hydrogeochemistry of high-altitude lake: a case study of the Chandra Tal, Western Himalaya. India Arab J Geosci 9(4):308. https://doi.org/10.1007/s12517-016-2358-1
Slukovskii Z, Dauvalter V, Guzeva A, Denisov D, Cherepanov A, Siroezhko E (2020) The hydrochemistry and recent sediment geochemistry of small lakes of Murmansk, Arctic Zone of Russia. Water 12(4):1130. https://doi.org/10.3390/w12041130
Soheb M, Ramanathan A, Angchuk T, Mandal A, Kumar N, Lotus S (2020) Mass-balance observation, reconstruction, and sensitivity of Stok glacier, Ladakh region, India, between 1978 and 2019. J Glaciol 66(258):627–642. https://doi.org/10.1017/jog.2020.34
Stein AF, Draxler RR, Rolph GD, Stunder BJB, Cohen MD, Ngan F (2015) NOAA’s HYSPLIT atmospheric transport and dispersion modeling system. Bull Amer Meteor Soc 96:2059–2077. https://doi.org/10.1175/BAMS-D-14-00110.1
Steingruber SM, Bernasconi SM, Valenti G (2020) Climate change-induced changes in the chemistry of a high-altitude mountain lake in the central alps. Aquat Geochem. https://doi.org/10.1007/s10498-020-09388-6
Stumm W, Morgan JJ (1981) Aquatic Chemistry: An Introduction Emphasizing Chemical Equilibria in Natural Waters. Wiley-Interscience Publication, p 780
Sun H, Han J, Li D, Zhang S, Lu X (2010) Chemical weathering inferred from riverine water chemistry in the lower Xijiang basin, South China. Sci Total Environ 408(20):4749–4760. https://doi.org/10.1016/j.scitotenv.2010.06.007
Szopińska M, Szumińska D, Bialik RJ, Chmiel S, Plenzler J, Polkowska Ż (2018) Impact of a newly-formed periglacial environment and other factors on freshwater chemistry at the western shore of Admiralty Bay in the summer of 2016 (King George Island, Maritime Antarctica). Sci Total Environ 613:619–634. https://doi.org/10.1016/j.scitotenv.2017.09.060
Thakuri S, Salerno F, Bolch T, Guyennon N, Tartari G (2016) Factors controlling the accelerated expansion of Imja Lake, Mount Everest region, Nepal. Annals of Glaciol 57(71):245–257. https://doi.org/10.3189/2016AoG71A063
Veh G, Korup O, von Specht S, Roessner S, Walz A (2019) Unchanged frequency of moraine-dammed glacial lake outburst floods in the Himalaya. Nat Climate Change 9(5):379–383. https://doi.org/10.1038/s41558-019-0437-5
Veh G, Korup O, Walz A (2020) Hazard from Himalayan glacier lake outburst floods. Proc Natl Acad Sci 117(2):907–912. https://doi.org/10.1073/pnas.1914898117
Wadham JL, Hodson AJ, Tranter M, Dowdeswell JA (1998) The hydrochemistry of meltwaters draining a polythermal-based, high Arctic glacier, south Svalbard: i the ablation season. Hydrol Process 12(12):1825–1849. https://doi.org/10.1002/(SICI)10991085(19981015)12:12%3C1825:AIDHYP669%3E3.3.CO;2-I
Wadham JL, Cooper RJ, Tranter M, Hodgkins R (2001) Enhancement of glacial solute fluxes in the pro-glacial zone of a polythermal glacier. J Glaciol 47(158):378–386. https://doi.org/10.3189/172756501781832188
Woelders L, Lenaerts JT, Hagemans K, Akkerman K, van Hoof TB, Hoek WZ (2018) Recent climate warming drives ecological change in a remote high-Arctic lake. Sci Rep 8(1):1–8. https://doi.org/10.1038/s41598-018-25148-7
Zhang S-R, Lu XX, Higgitt DL, Chen C-TA, Sun H-G, Han J-T (2007) Water chemistry of the Zhujiang (Pearl River): Natural processes and anthropogenic influences. J Geophys Res 112(F1):F01011. https://doi.org/10.1029/2006JF000493
Zhang Z, Jia W, Zhu G, Ma X, Xu X, Yuan R, Xiong H (2020) Hydrochemical characteristics and ion sources of precipitation in the upper reaches of the shiyang river. China Water 12(5):1442. https://doi.org/10.3390/w12051442
Acknowledgements
We thank Professor A.L Ramanathan, Chandranath Chatterjee, and Jawaharlal Nehru University, New Delhi, for providing all the facilities to carrying out this work. The authors are grateful to Central Instruments Facility-School of Environmental Science, Jawaharlal Nehru University, New Delhi, for providing the facilities to analyze the samples.
Funding
This work was financially supported by National Centre for Polar and Ocean Research (NCPOR) under the HiCOM project (PAC-SES-ALR_NCPOR- (MoES) 03180321–1108/ Sanction Order no. 721/2018) and the Ministry of Earth Sciences (MoES) Government of India.
Author information
Authors and Affiliations
Contributions
HK, ALR, MS, and MSS conceptualized the study. ALR supervised the study. HK, KB, CS, and AM performed the analysis, developed the figures, and wrote the paper. All authors contributed significantly in fieldwork and to improve the draft manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Prof. Robert Bialik, (ASSOCIATE EDITOR), Dr. Michael Nones, (CO-EDITOR-IN-CHIEF).
Rights and permissions
About this article
Cite this article
Kaushik, H., Ramanathan, A., Soheb, M. et al. Climate change-induced high-altitude lake: Hydrochemistry and area changes of a moraine-dammed lake in Leh-Ladakh. Acta Geophys. 69, 2377–2391 (2021). https://doi.org/10.1007/s11600-021-00670-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11600-021-00670-x