Abstract
Objective
The activation state of microglia is known to occupy a central position in the pathophysiological process of cerebral inflammation. Autophagy is a catabolic process responsible for maintaining cellular homeostasis. In recent years, autophagy has been demonstrated to play an important role in neuroinflammation. Resolvin D1 (RvD1) is a promising therapeutic mediator that has been shown to exert substantial anti-inflammatory and proresolving activities. However, whether RvD1-mediated resolution of inflammation in microglia is related to autophagy regulation needs further investigation. The present study aimed to explore the effect of RvD1 on microglial autophagy and its corresponding pathways.
Methods
Mouse microglial cells (BV-2) were cultured, treated with RvD1, and examined by Western blotting, confocal immunofluorescence microscopy, transmission electron microscopy, and flow cytometry.
Results
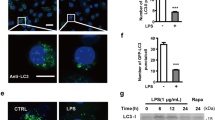
RvD1 promoted autophagy in both BV-2 cells and mouse primary microglia by favoring the maturation of autophagosomes and their fusion with lysosomes. Importantly, RvD1 had no significant effect on the activation of mammalian target of rapamycin (mTOR) signaling. Furthermore, RvD1-induced mTOR-independent autophagy was confirmed by observing reduced cytoplasmic calcium levels and suppressed calcium/calmodulin-dependent protein kinase II (CaMK II) activation. Moreover, by downregulating ATG5, the increased phagocytic activity induced by RvD1 was demonstrated to be tightly controlled by ATG5-dependent autophagy.
Conclusion
The present work identified a previously unreported mechanism responsible for the role of RvD1 in microglial autophagy, highlighting its therapeutic potential against neuroinflammation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Han KM, Ham BJ. How Inflammation Affects the Brain in Depression: A Review of Functional and Structural MRI Studies. J Clin Neurol, 2021,17(4):503–515
Lua J, Ekanayake K, Fangman M, et al. Potential Role of Soluble Toll-like Receptors 2 and 4 as Therapeutic Agents in Stroke and Brain Hemorrhage. Int J Mol Sci, 2021,22(18):9977
Muhammad S, Chaudhry SR, Kahlert UD, et al. Brain Immune Interactions-Novel Emerging Options to Treat Acute Ischemic Brain Injury. Cells, 2021,10(9):2429
Ogunmokun G, Dewanjee S, Chakraborty P, et al. The Potential Role of Cytokines and Growth Factors in the Pathogenesis of Alzheimer’s Disease. Cells, 2021,10(10):2790
Peng X, Luo Z, He S, et al. Blood-Brain Barrier Disruption by Lipopolysaccharide and Sepsis-Associated Encephalopathy. Front Cell Infect Microbiol, 2021,11:768108
Sampson TR, Debelius JW, Thron T, et al. Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson’s Disease. Cell, 2016,167(6):1469–1480
Wies Mancini VSB, Di Pietro AA, Pasquini LA. Microglia depletion as a therapeutic strategy: friend or foe in multiple sclerosis models? Neural Regen Res, 2023,18(2):267–272
Hanisch U-K, Kettenmann H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci, 2007,10(11):1387–1394
Smith JA, Das A, Ray SK, et al. Role of pro-inflammatory cytokines released from microglia in neurodegenerative diseases. Brain Res Bull, 2012,87(1):10–20
Serhan CN. Pro-resolving lipid mediators are leads for resolution physiology. Nature, 2014,510(7503):92–101
Li L, Wu Y, Wang Y, et al. Resolvin D1 promotes the interleukin-4-induced alternative activation in BV-2 microglial cells. J Neuroinflammation, 2014,11:72
Ponce J, Ulu A, Hanson C, et al. Role of Specialized Pro-resolving Mediators in Reducing Neuroinflammation in Neurodegenerative Disorders. Front Aging Neurosci, 2022,14:780811
Serhan CN. Discovery of specialized pro-resolving mediators marks the dawn of resolution physiology and pharmacology. Mol Aspects Med, 2017,58:1–11
Tiberi M, Chiurchiù V. Specialized Pro-resolving Lipid Mediators and Glial Cells: Emerging Candidates for Brain Homeostasis and Repair. Front Cell Neurosci, 2021,15:673549
Wang YP, Wu Y, Li LY, et al. Aspirin-triggered lipoxin A4 attenuates LPS-induced pro-inflammatory responses by inhibiting activation of NF-κB and MAPKs in BV-2 microglial cells. J Neuroinflammation, 2011,8:95
Wu Y, Zhai H, Wang Y, et al. Aspirin-triggered lipoxin A4 attenuates lipopolysaccharide-induced intracellular ROS in BV2 microglia cells by inhibiting the function of NADPH oxidase. Neurochem Res, 2012,37(8):1690–1696
Xia H, Chen L, Liu H, et al. Protectin DX increases survival in a mouse model of sepsis by ameliorating inflammation and modulating macrophage phenotype. Sci Rep, 2017,7(1):99
Xian W, Wu Y, Xiong W, et al. The pro-resolving lipid mediator Maresin 1 protects against cerebral ischemia/reperfusion injury by attenuating the pro-inflammatory response. Biochem Biophys Res Commun, 2016,472(1):175–181
Buckley CD, Gilroy DW, Serhan CN. Proresolving lipid mediators and mechanisms in the resolution of acute inflammation. Immunity, 2014,40(3):315–327
Chiang N, Serhan CN. Specialized pro-resolving mediator network: an update on production and actions. Essays Biochem, 2020,64(3):443–462
Zhu M, Wang X, Hjorth E, et al. Pro-Resolving Lipid Mediators Improve Neuronal Survival and Increase Aβ42 Phagocytosis. Mol Neurobiol, 2016,53(4):2733–2749
Bisicchia E, Sasso V, Catanzaro G, et al. Resolvin D1 Halts Remote Neuroinflammation and Improves Functional Recovery after Focal Brain Damage Via ALX/FPR2 Receptor-Regulated MicroRNAs. Mol Neurobiol, 2018,55(8):6894–6905
Rey C, Nadjar A, Buaud B, et al. Resolvin D1 and E1 promote resolution of inflammation in microglial cells in vitro. Brain Behav Immun, 2016,55:249–259
Das G, Shravage BV, Baehrecke EH. Regulation and function of autophagy during cell survival and cell death. Cold Spring Harb Perspect Biol, 2012,4(6):a008813
Levine B, Mizushima N, Virgin HW. Autophagy in immunity and inflammation. Nature, 2011,469(7330):323–335
Pyo JO, Nah J, Jung YK. Molecules and their functions in autophagy. Exp Mol Med, 2012,44(2):73–80
Bjørkøy G, Lamark T, Pankiv S, et al. Monitoring autophagic degradation of p62/SQSTM1. Methods Enzymol, 2009,452:181–197
Mizushima N, Yoshimori T. How to interpret LC3 immunoblotting. Autophagy, 2007,3(6):542–545
Gordon PB, Holen I, Fosse M, et al. Dependence of hepatocytic autophagy on intracellularly sequestered calcium. J Biol Chem, 1993,268(35):26107–26112
Saxton RA, Sabatini DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell, 2017,168(6):960–976
Williams A, Sarkar S, Cuddon P, et al. Novel targets for Huntington’s disease in an mTOR-independent autophagy pathway. Nat Chem Biol, 2008,4(5):295–305
Egan D, Kim J, Shaw RJ, et al. The autophagy initiating kinase ULK1 is regulated via opposing phosphorylation by AMPK and mTOR. Autophagy, 2011,7(6):643–644
Yip CK, Murata K, Walz T, et al. Structure of the human mTOR complex I and its implications for rapamycin inhibition. Mol Cell, 2010,38(5):768–774
Li G, Sherchan P, Tang Z, et al. Autophagy & Phagocytosis in Neurological Disorders and their Possible Cross-talk. Curr Neuropharmacol, 2021,19(11):1912–1924
Martinez J, Malireddi RKS, Lu Q, et al. Molecular characterization of LC3-associated phagocytosis reveals distinct roles for Rubicon, NOX2 and autophagy proteins. Nat Cell Biol, 2015,17(7):893–906
Su P, Zhang J, Wang D, et al. The role of autophagy in modulation of neuroinflammation in microglia. Neuroscience, 2016,319:155–167
Deretic V, Saitoh T, Akira S. Autophagy in infection, inflammation and immunity. Nat Rev Immunol, 2013,13(10):722–737
Prieto P, Rosales-Mendoza CE, Terrón V, et al. Activation of autophagy in macrophages by pro-resolving lipid mediators. Autophagy, 2015,11(10):1729–1744
Fredman G, Ozcan L, Spolitu S, et al. Resolvin D1 limits 5-lipoxygenase nuclear localization and leukotriene B4 synthesis by inhibiting a calcium-activated kinase pathway. Proc Natl Acad Sci USA, 2014,111(40):14530–14535
Graeber MB. Changing face of microglia. Science, 2010,330(6005):783–788
Boche D, Perry VH, Nicoll JAR. Review: activation patterns of microglia and their identification in the human brain. Neuropathol Appl Neurobiol, 2013,39(1):3–18
Krashia P, Cordella A, Nobili A, et al. Blunting neuroinflammation with resolvin D1 prevents early pathology in a rat model of Parkinson’s disease. Nat Commun, 2019,10(1):3945
Liu G-J, Tao T, Wang H, et al. Functions of resolvin D1-ALX/FPR2 receptor interaction in the hemoglobin-induced microglial inflammatory response and neuronal injury. J Neuroinflammation, 2020,17(1):239
Sun Q, Yan H, Chen F, et al. Restoration of Proresolution Pathway with Exogenous Resolvin D1 Prevents Sevoflurane-Induced Cognitive Decline by Attenuating Neuroinflammation in the Hippocampus in Rats with Type 2 Diabetes Mellitus. Front Pharmacol, 2021,12:720249
Plaza-Zabala A, Sierra-Torre V, Sierra A. Autophagy and Microglia: Novel Partners in Neurodegeneration and Aging. Int J Mol Sci, 2017,18(3):598
Olah M, Biber K, Vinet J, et al. Microglia phenotype diversity. CNS Neurol Disord Drug Targets, 2011,10(1):108–11
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
This study was supported by the National Natural Science Foundation of China (No. 81902016).
Rights and permissions
About this article
Cite this article
Pan, Sw., Hu, Ls., Wang, H. et al. Resolvin D1 Induces mTOR-independent and ATG5-dependent Autophagy in BV-2 Microglial Cells. CURR MED SCI 43, 1096–1106 (2023). https://doi.org/10.1007/s11596-023-2787-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-023-2787-5