Abstract
Objective
While the upregulation of cytochrome P450 family 24 subfamily A member 1 (CYP24A1) gene expression has been reported in colon cancer, its role in tumorigenesis remains largely unknown. In this study, we aimed to investigate the involvement of CYP24A1 in Wnt pathway regulation via the nuclear factor kappa B (NF-κB) pathway.
Methods
The human colon cancer cell lines HCT-116 and Caco-2 were subjected to stimulation with interleukin-6 (IL-6) as well as tumor necrosis factor alpha (TNF-α), with subsequent treatment using the NF-κB pathway-specific inhibitor ammonium pyrrolidinedithiocarbamate (PDTC). Furthermore, CYP24A1 expression was subjected to knockdown via the use of small interfering RNA (siRNA). Subsequently, NF-κB pathway activation was determined by an electrophoretic mobility shift assay, and the transcriptional activity of β-catenin was determined by a dual-luciferase reporter assay. A mouse ulcerative colitis (UC)-associated carcinogenesis model was established, wherein TNF-α and the NF-κB pathway were blocked by anti-TNF-α monoclonal antibody and NF-κB antisense oligonucleotides, respectively. Then the tumor size and protein level of CYP24A1 were determined.
Results
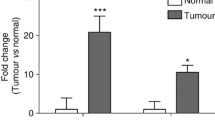
IL-6 and TNF-α upregulated CYP24A1 expression and activated the NF-κB pathway in colon cancer cells. PDTC significantly inhibited this increase in CYP24A1 expression. Additionally, knockdown of CYP24A1 expression by siRNA could partially antagonize Wnt pathway activation. Upregulated CYP24A1 expression was observed in the colonic epithelial cells of UC-associated carcinoma mouse models. Anti-TNF-α monoclonal antibody and NF-κB antisense oligonucleotides decreased the tumor size and suppressed CYP24A1 expression.
Conclusion
Taken together, this study suggests that inflammatory factors may increase CYP24A1 expression via NF-κB pathway activation, which in turn stimulates Wnt signaling.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Di Rosa M, Malaguarnera M, Zanghì A, et al. Vitamin D3 insufficiency and colorectal cancer. Crit Rev Oncol Hematol, 2013, 88(3): 594–612
Fleet JC, DeSmet M, Johnson R, et al. Vitamin D and Cancer: A review of molecular mechanisms. Biochem J, 2012, 441(1): 61–76
Dusso AS, Brown AJ, Slatopolsky E. Vitamin D. Am J Physiol Renal Physiol, 2005, 289(1): F8–28
Gandini S, Boniol M, Haukka J, et al. Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma. Int J Cancer, 2011, 128(6): 1414–1424
Ulitsky A, Ananthakrishnan AN, Naik A, et al. Vitamin D deficiency in patients with inflammatory bowel disease: association with disease activity and quality of life. JPEN J Parenter Enteral Nutr, 2011, 35(3): 308–316
Hummel DM, Thiem U, Hobaus J, et al. Prevention of preneoplastic lesions by dietary vitamin D in a mouse model of colorectal carcinogenesis. J Steroid Biochem Mol Biol, 2013, 136: 284–288
Chen XQ, Mao JY, Li WB, et al. Association between CYP24A1 polymorphisms and the risk of colonic polyps and colon cancer in a Chinese population. World J Gastroenterol, 2017, 23(28): 5179–5186
Feng YJ, Wang N, Fang LW, et al. CYP24A1 is a potential biomarker for the progression and prognosis of human colorectal cancer. Hum Pathol, 2016, 50: 101–108
Horvath HC, Lakatos P, Kosa JP, et al. The Candidate Oncogene CYP24A1: A Potential Biomarker for Colorectal Tumorigenesis. J Histochem Cytochem, 2010, 58(3): 277–285
Noyola-Martínez N, Díaz L, Zaga-Clavellina V, et al. Regulation of CYP27B1 and CYP24A1 gene expression by recombinant pro-inflammatory cytokines in cultured human trophoblasts. J Steroid Biochem Mol Biol, 2014, 144(A): 106–9
Wang L, Gao Z, Wang L, et al. Upregulation of nuclear factor- κ B activity mediates CYP24 expression and reactive oxygen species production in indoxyl sulfate-induced chronic kidney disease. Nephrology (Carlton), 2016, 21: 774–781
Larriba MJ, González-Sancho JM, Barbáchano A, et al. Vitamin D is a multilevel repressor of Wnt/b-Catenin signaling in cancer cells. Cancers (Basel), 2013, 5(4): 1242–1260
Mao J, Chen X, Wang C, et al. Effects and mechanism of the bile acid (farnesoid X) receptor on the Wnt/β-catenin signaling pathway in colon cancer. Oncol Lett, 2020, 20(1): 337–345
Gröschel C, Tennakoon S, Kállay E. Cytochrome P450 Vitamin D Hydroxylases in Inflammation and Cancer. Adv Pharmacol, 2015, 74: 413–58
Ullman TA, Itzkowitz SH. Intestinal inflammation and cancer. Gastroenterology, 2011, 140(6): 1807–1816
Ben-neriah Y, Karin M. Inflammation meets cancer, with NF-κB as the matchmaker. Nat Immunol, 2011, 12(8): 715–723
Li Y, de Haar C, Chen M, et al. Disease-related expression of the IL6/STAT3/SOCS3 signalling pathway in ulcerative colitis and ulcerative colitis-related carcinogenesis. Gut, 2010, 59(2): 227–235
Oshima H, Oshima M. The inflammatory network in the gastrointestinal tumor microenvironment: lessons from mouse models. J Gastroenterol, 2012, 47(2): 97–106
Grivennikov S, Karin E, Terzic J, et al. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell, 2009, 15(2): 103–113
Deng L, Zhou J-F, Sellers RS, et al. A novel mouse model of inflammatory bowel disease links mammalian Target of Rapamycin-Dependent hyperproliferation of colonic epithelium to inflammation-associated tumorigenesis. Am J Pathol, 2010, 176(2): 952–967
Kawada M, Seno H, Uenoyama Y, et al. Signal transducers and activators of transcription 3 activation is involved in nuclear accumulation of beta-catenin in colorectal cancer. Cancer Res, 2006, 66(6): 2913–2917
Popivanova BK, Kitamura K, Wu Y, et al. Blocking TNF-alpha in mice reduces colorectal carcinogenesis associated with chronic colitis. J Clin Invest, 2008, 118(2): 560–570
Hummel DM, Fetahu IS, Gröschel C, et al. Role of proinflammatory cytokines on expression of vitamin D metabolism and target genes in colon cancer cells. J Steroid Biochem Mol Biol, 2014,144Pt A:91–95
Murano M, Maemura K, Hirata I, et al. Therapeutic effect of intracolonically administered nuclear factor kappa B (p65) antisense oligonucleotide on mouse dextran sulphate sodium (DSS)-induced colitis. Clin Exp Immunol, 2000, 120(1): 51–58
Wang L, Walia B, Evans J, et al. IL-6 induces NF-kappaB activation in the intestinal epithelia. J Immunol, 2003, 171(6): 3194–3201
He F, Peng J, Deng X, et al. Cytokine mechanisms of tumor necrosis factor-alpha-induced leaks in intestine epithelial barrier. Cytokine, 2012, 59(2): 264–272
Tanaka T, Kohno H, Suzuki R, et al. A novel inflammation-related mouse colon carcinogenesis model induced by azoxymethane and dextran sodium sulfate. Cancer Sci, 2003, 94(11): 965–973
Naugler WE, Karin M. NF-kappaB and cancer-identifying targets and mechanisms. Curr Opin Genet Dev, 2008, 18(1): 19–26
Greten FR, Eckmann L, Greten TF, et al. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell, 2004, 118(3): 285–296
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
The present study was supported by grants from the National Natural Science Foundation of China (No. 81370500 and No. 81770559), CAMS Innovation Fund for Medical Sciences (No. CIFMS2021-I2M-C&T-A-001 and No. 2016-I2M-3-005) and the CAMS Initiative for Innovative Medicine (No. CAMS-a12M 2016-I2M-1-007).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Xq., Mao, Jy., Wang, Cs. et al. CYP24A1 Involvement in Inflammatory Factor Regulation Occurs via the Wnt Signaling Pathway. CURR MED SCI 42, 1022–1032 (2022). https://doi.org/10.1007/s11596-022-2564-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2564-x