Abstract
Objective
S100A11 is a member of the S100 calcium-binding protein family and has intracellular and extracellular regulatory activities. We previously reported that S100A11 was differentially expressed in the respiratory tracts of asthmatic rats as compared with normal controls. Here, we aimed to analyze the potential of S100A11 to regulate both allergen-induced airway hyperresponsiveness (AHR) as well as acetylcholine (ACh)-induced hypercontractility of airway smooth muscle (ASM) and contraction of ASM cells (ASMCs).
Methods
Purified recombinant rat S100A11 protein (rS100A11) was administered to OVA-sensitized and challenged rats and then the AHR of animals was measured. The relaxation effects of rS100A11 on ASM were detected using isolated tracheal rings and primary ASMCs. The expression levels of un-phosphorylated myosin light chain (MLC) and phosphorylated MLC in ASMCs were analyzed using Western blotting.
Results
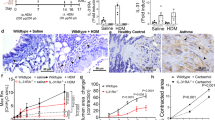
Treatment with rS100A11 attenuated AHR in the rats. ASM contraction assays showed that rS100A11 reduced the contractile responses of isolated tracheal rings and primary ASMCs treated with ACh. In addition, rS100A11 markedly decreased the ACh-induced phosphorylation of the myosin light chain in ASMCs. Moreover, rS100A11 also suppressed the contractile response of tracheal rings in calcium-free buffer medium.
Conclusion
These results indicate that S100A11 protein can relieve AHR by relaxing ASM independently of extracellular calcium. Our data support the idea that S100A11 is a potential therapeutic target for reducing airway resistance in asthma patients.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Oliver BG, Black JL. Airway Smooth Muscle and Asthma. Allergol Int, 2006,55(3):215–223
McGraw DW, Liggett SB. Molecular mechanisms of beta2-adrenergic receptor function and regulation. Proc Am Thorac Soc, 2005,2(4):292–296; discussion 311–292
Yim RP, Koumbourlis AC. Tolerance & resistance to beta(2)-agonist bronchodilators. Paediatr Respir Rev, 2013,14(3):195–198
Adhikari BB, Wang K. S100A1 modulates skeletal muscle contraction by desensitizing calcium activation of isometric tension, stiffness and ATPase. FEBS Lett, 2001,497(2-3):95–98
Wang W, Asp ML, Guerrero-Serna G, et al. Differential effects of S100 proteins A2 and A6 on cardiac Ca(2+) cycling and contractile performance. J Mol Cell Cardiol, 2014,72:117–125
Zhang W, Gunst SJ. S100A4 is activated by RhoA and catalyses the polymerization of non-muscle myosin, adhesion complex assembly and contraction in airway smooth muscle. J Physiol, 2020,598(20):4573–4590
Gorsler T, Murzik U, Ulbricht T, et al. DNA damage-induced translocation of S100A11 into the nucleus regulates cell proliferation. BMC Cell Biol, 2010,11:100
Wang C, Zhang Z, Li L, et al. S100A11 is a migration-related protein in laryngeal squamous cell carcinoma. Int J Med Sci, 2013,10(11):1552–1559
Xia Q, Li X, Zhou H, et al. S100A11 protects against neuronal cell apoptosis induced by cerebral ischemia via inhibiting the nuclear translocation of annexin A1. Cell Death Dis, 2018,9(6):657
Jaiswal JK, Lauritzen SP, Scheffer L, et al. S100A11 is required for efficient plasma membrane repair and survival of invasive cancer cells. Nat Commun, 2014, 5(1):3795
Andres Cerezo L, Sumova B, Prajzlerova K, et al. Calgizzarin (S100A11): a novel inflammatory mediator associated with disease activity of rheumatoid arthritis. Arthritis Res Ther, 2017,19(1):79
Zhao XQ, Naka M, Muneyuki M, et al. Ca2+-Dependent Inhibition of Actin-Activated Myosin ATPase Activity by S100C (S100A11), a Novel Member of the S100 Protein Family. Biochem Biophys Res Commun, 2000, 267(1):77–79
Fan C, Fu Z, Su Q, et al. S100A11 mediates hypoxia-induced mitogenic factor (HIMF)-induced smooth muscle cell migration, vesicular exocytosis, and nuclear activation. Mol Cell Proteomics, 2011,10(3):M110000901
Santamaria-Kisiel L, Rintala-Dempsey AC, Shaw GS. Calcium-dependent and -independent interactions of the S100 protein family. Biochem J, 2006,396(2):201–214
Xu YD, Cui JM, Wang Y, et al. The early asthmatic response is associated with glycolysis, calcium binding and mitochondria activity as revealed by proteomic analysis in rats. Respir Res, 2010,11:107
Xu Y, Yin L, Park GH, et al. Serum from asthmatic rat treated with acupuncture inhibits acetylcholine- induced contractile responses of airway smooth muscle cells. J Tradit Chin Med, 2017,37(1):101–107
Xu YD, Wang Y, Yin LM, et al. S100A8 protein attenuates airway hyperresponsiveness by suppressing the contraction of airway smooth muscle. Biochem Biophys Res Commun, 2017,484(1):184–188
Bresnick AR. S100 proteins as therapeutic targets. Biophys Rev, 2018,10(6):1617–1629
Aoki T, Matsumoto Y, Hirata K, et al. Expression profiling of genes related to asthma exacerbations. Clin Exp Allergy, 2009,39(2):213–221
Kang JH, Hwang SM, Chung IY. S100A8, S100A9 and S100A12 activate airway epithelial cells to produce MUC5AC via extracellular signal-regulated kinase and nuclear factor-kappaB pathways. Immunology, 2015, 144(1):79–90
Xu YD, Wei Y, Wang Y, et al. Exogenous S100A8 protein inhibits PDGF-induced migration of airway smooth muscle cells in a RAGE-dependent manner. Biochem Biophys Res Commun, 2016,472(1):243–249
Yin LM, Li HY, Zhang QH, et al. Effects of S100A9 in a rat model of asthma and in isolated tracheal spirals. Biochem Biophys Res Commun, 2010,398(3):547–552
Davey GE, Murmann P, Hoechli M, et al. Calcium-dependent translocation of S100A11 requires tubulin filaments. Biochim Biophys Acta, 2000,1498(2-3):220–232
Réty S, Osterloh D, Arié JP, et al. Structural basis of the Ca2+-dependent association between S100C (S100A11) and its target, the N-terminal part of annexin I. Structure, 2000,8(2):175–184
Chang N, Sutherland C, Hesse E, et al. Identification of a novel interaction between the Ca2+-binding protein S100A11 and the Ca2+- and phospholipid-binding protein annexin A6. Am J Physiol Cell Physiol, 2007, 292(4):C1417–C1430
Huang YK, Chou RH, Yu C. Tranilast Blocks the Interaction between the Protein S100A11 and Receptor for Advanced Glycation End Products (RAGE) V Domain and Inhibits Cell Proliferation. J Biol Chem, 2016,291(27):14300–14310
Sakaguchi M, Sonegawa H, Murata H, et al. S100A11, an Dual Mediator for Growth Regulation of Human Keratinocytes. Mol Biol Cell, 2008,19(1):78–85
Takamatsu H, Yamamoto KI, Tomonobu N, et al. Extracellular S100A11 Plays a Critical Role in Spread of the Fibroblast Population in Pancreatic Cancers. Oncol Res, 2019,27(6):713–727
Andres Cerezo L, Hulejova H, Sumova B, et al. Pro-inflammatory S100A11 is elevated in inflammatory myopathies and reflects disease activity and extra-muscular manifestations in myositis. Cytokine, 2019, 116:13–20
Cecil DL, Terkeltaub R. Transamidation by transglu-taminase 2 transforms S100A11 calgranulin into a procatabolic cytokine for chondrocytes. J Immunol, 2008,180(12):8378–8385
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
This work was supported by the National Natural Science Foundation of China (No. 81973952 and No. 81774429), the Natural Science Foundation of Shanghai (No. 19ZR1451500), and the Yangfan Innovation Project (No. 20YF1445300).
Rights and permissions
About this article
Cite this article
Cheng, M., Shi, Yl., Shang, Pp. et al. Inhibitory Effect of S100A11 on Airway Smooth Muscle Contraction and Airway Hyperresponsiveness. CURR MED SCI 42, 333–340 (2022). https://doi.org/10.1007/s11596-022-2559-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2559-7