Summary
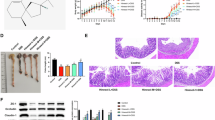
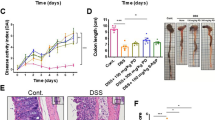
We aimed to explore the anti-inflammatory activity of mollugin extracted from Rubia cordifolia L, a traditional Chinese medicine, on dextran sulfate sodium (DSS)-induced ulcerative colitis (UC) in mice. Thirty C57BL/6 mice were divided into a control group (n=6), a model group (n=6), and three experimental groups (40, 20, 10 mg/kg of mollugin, n=6 each). DSS solution (3%) was given to mice in the model group and experimental groups from day 4 to day 10 to induce the mouse UC model. Mice in the experimental groups were intragastrically administrated mollugin from day 1 to day 10. Animals were orally given distilled water in the control group for the whole experiment time and in the model group from day 1 to day 3. The changes in colon pathology were detected by hematoxylin and eosin (HE) staining. Interleukin-1β (IL-1β) in the serum, and tumor necrosis factor-α (TNF-α) and interferon-γ (IFN) in the tissues were measured by enzyme linked immunosorbent assay. Expression levels of Toll-like receptor 4 (TLR4) and myeloid differentiation factor 88 in the colon tissues were detected by immunohistochemistry. Results showed that mollugin could significantly reduce weight loss and the disease activity index in the DSS-induced UC mouse model. HE examinations demonstrated that mollugin treatment effectively improved the histological damage (P<0.05). The overproduction of IL-1β and TNF-α was remarkably inhibited by mollugin treatment at doses of 20 and 40 mg/kg (P<0.05). Additionally, the levels of TLR4 in colon tissues were significantly reduced in mollugin-treated groups compared with the DSS group. Our findings demonstrated that mollugin ameliorates DSS-induced UC by inhibiting the production of pro-inflammatory chemocytokines.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Azuma K, Osaki T, Kurozumi S, et al.. Anti-inflammatory effects of orally administered glucosamine oligomer in an experimental model of inflammatory bowel disease. Carbohydr Polym, 2015,115:448–456
Ng SC, Tang W, Ching JY, et al. Incidence and phenotype of inflammatory bowel disease based on results from the Asia-Pacific Crohn’s and colitis epidemiology study. Gastroenterology, 2013,145(1):158–165
Al-Rejaie SS, Abuohashish HM, Al-Enazi MM, et al. Protective effect of naringenin on acetic acid-induced ulcerative colitis in rats. World J Gastroenterol, 2013,19(34):5633–5644
Xiao B, Laroui H, Ayyadurai, et al. Mannosylated bioreducible nanoparticle-mediated macrophage-specific TNF-α RNA interference for IBD therapy. Biomaterials, 2013,34(30):7471–7482
Cholapranee A, Hazlewood GS, Kaplan GG, et al. Systematic review with meta-analysis: comparative efficacy of biologics for induction and maintenance of mucosal healing in Crohn’s disease and ulcerative colitis controlled trials. Aliment Pharmacol Ther, 2017,45(10):1291–1302
Le Berre C, Roda G, Nedeljkovic Protic M, et al.. Modern use of 5-aminosalicylic acid compounds for ulcerative colitis. Expert Opin Biol Ther, 2019,18:1–16
Cruz-Topete D, Cidlowski JA. One hormone, two actions: anti-and pro-inflammatory effects of glucocorticoids. Neuroimmunomodulation, 2015, 22(1-2):20–32
Christophorou D, Funakoshi N, Duny Y, et al. Systematic review with meta-analysis: infliximab and immunosuppressant therapy vs infliximab alone for active ulcerative colitis. Aliment Pharmacol Ther, 2015,41(7):603–612
Gisbert JP, Marin AC, Chaparro M. The Risk of Relapse after Anti-TNF Discontinuation in Inflammatory Bowel Disease: Systematic Review and Meta-Analysis. AM J Gastroenterol, 2016,111(5):632–647
Jeon YD, Bang KS, Shin MK, et al. Regulatory effects of glycyrrhizae radix extract on DSS-induced ulcerative colitis. BMC Complement Altern Med, 2016, 16(1):459
Chen G, Yang Y, Liu M, et al.. Banxia xiexin decoction protects against dextran sulfate sodium-induced chronic ulcerative colitis in mice. J Ethnopharmacol, 2015,166:149–156
Chen P, Zhou X, Zhang L, et al.. Anti-inflammatory effects of Huangqin tang extract in mice on ulcerative colitis. J Ethnopharmacol, 2015,162:207–214
Chinese Pharmacopeia Commission. Phrmacopoeia of the People’s Republic of China, 2015 English ed. Chinese Medical Science Press: Beijing, China, 2015, 234–235
Brijesh S, Daswani P, Tetali P, et al. Studies on the antidiarrhoeal activity of Aegle marmelos unripe fruit: validating its traditional usage. BMC Complement Altern Med, 2009,9(47):1–12
Jun DY, Han CR, Lee JY, et al. Anti-adipogenic activity of 2-carbomethoxy-2,3-epoxy-3-prenyl-1,4-naphthoquinone from Rubia cordifolia L. J Med Food, 2011,14(5):454–461
Zhu ZG, Jin H, Yu PJ, et al. Mollugin inhibits the inflammatory response in lipopoly saccharide-stimulated RAW264.7macrophages by blocking the Janus kinase-signal transducers and activators of transcription signaling pathway. Biol Pharm Bull, 2013,36(03):339–406
Wang Z, Li MY, Mi C, et al. Mollugin Has an AntiCancer Therapeutic Effect by Inhibiting TNF-α-Induced NF-κB Activation. Int J Mol Sci, 2017,18(8):1–13
Idhayadhulla A, Xia L, Lee YR, et al.. Synthesis of novel and diverse mollugin analogues and their antibacterial and antioxidant activities. Bioorg Chem, 2014,52:77–82
Kim KJ, Lee JS, Kwak MK, et al. Anti-inflammatory action of mollugin and its synthetic derivatives in HT-29 human colonic epithelial cells is mediated through inhibition of NF-kappaB activation. Eur J Pharmacol, 2009,622(1-3):52–57
Zhang L, Wang H, Zhu J, et al. Mollugin induces tumor cell apoptosis and autophagy via the PI3K/AKT/mTOR/p70S6K and ERK signaling pathways. Biochem Biophys Res Commun, 2014,450(1):247–254
Zhang JL, Xiao M, Song YY, et al. Effect of the Aqueous Extract of Rubia Cordifolia’s Aerial Part on Ulcerative Colitis in Mice. Yi Yao Dao Bao Za Zhi (Chinese), 2019,38(10):1272–1277
Wang WQ, Dong K, Zhou L, et al. IL-37b gene transfer enhances the therapeutic efficacy of mesenchumal stromal cells in DSS-induced colitis mice. Acta Pharmacol Sin, 2015,36(11):1377–1387
Ma JM, Yin GH, Lu ZB, et al. Casticin prevents DSS induced ulcerative colitis in mice through inhibitions of NF-kB pathway and ROS signaling. Phytother Res, 2018,32(9):1770–1783
Pandurangan AK, Kumar SAS, Dharmalingam P, et al.. Luteolin, a bioflavonoid inhibits azoxymethane-induced colon carcinogenesis: Involvement of iNOS and COX-2. Pharmacogn Mag, 2014,10:S306–310
Rubin DC, Shaker A, Levin MS. Chronic intestinal inflammation: inflammatory bowel disease and colitis-associated colon cancer. Front Immunol, 2012,3(107):1–10
Ordás I, Eckmann L, Talamini M, et al.. Ulcerative colitis. Lancet, 2012,3:380 (9853):1606–1619
Vermeire BP. Treat to Target in Inflammatory Bowel Disease. Curr Treat Options Gastroenterol, 2016,14(1):61–72
Pineton de Chambrun G, Blanc P, Peyrin-Biroulet L. Current evidence supporting mucosal healing and deep remission as important treatment goals for inflammatory bowel disease. Expert Rev Gastroenterol Hepatol, 2016,10(8):915–927
Chi WN, Liu ZP. Research Progress of Therapeutic Drugs for Ulcerative Colitis. Med Recapitulate, 2019,25(04):742–747
Sun Y, Lin LJ, Lin Y, et al. Gingko biloba extract (Ginaton) ameliorates dextran sulfate sodium (DSS)-induced acute experimental colitis in mice via reducing IL-6/STAT3 and IL-23/IL-17. Int J Clin Exp Med, 2015,8(10):17235–17247
Wang K, Chen X, Shan M, et al. Study on intestinal absorption of mollugin and purpurin in rats. Zhong guo Zhong Yao Za Zhi (Chinese), 2012,37(12):1855–1858
Kim KJ, Lee JS, Kwak MK, et al. Anti-inflammatory action of mollugin and its synthetic derivatives in HT-29 human colonic epithelial cells is medicated through inhibition of NF-kappaB activation. Eur J Pharmacol, 2009,622(1-3):52–57
Magyari L, Kovesdi E, Sarlos P, et al. Interleukin and interleukinreceptor gene polymorphisms in inflammatory bowel diseases susceptibility. World J Gastroenterol, 2014,20(12):3208–3222
Neurath MF. Cytokines in inflammatory bowel disease. Nat Rev Immunol, 2014,14(5):329–342
Sawa Y, Oshitani N, Adachi K, et al. Comprehensive analysis of intestinal cytokine messenger RNA profile by real-time quantitative polymerase chain reaction in patients with inflammatory bowel disease. Int J Mol Med, 2003,11(2):175–179
Yang Y, He J, Suo Y, et al.. Anti-inflammatory effect of taurocholate on TNBS-induced ulcerative colitis in mice. Biomed Pharmacother, 2016,81:424–430
Baud V, Karin M. Signal transduction by tumor necrosis factor and its relatives. Trends Cell Biol, 2001,11(9):372–377
Leppkes M, Roulis M, Neurath MF, et al. Pleiotropic functions of TNF-α in the regulation of the intestinal epithelial response to inflammation. Int Immunol, 2014,26(9):509–515
Chen Q, Gou S, Ma P, et al.. Oral administration of colitis tissue-accumulating porous nanoparticles for ulcerative colitis therapy. Int J Pharm, 2019,557:135–144
Strober W, Zhang F, Kitani A, et al. Proinflammatory cytokines underlying the inflammtory of Crohn’s disease. Curr Opin Gastroenterol, 2010,26(4):310–317
Schroder K, Hertzog PJ, Ravasi T, et al. Interferon-gamma: an overview of signals, mechanisms and functions. J Leukoc Biol, 2004,75(2):163–189
Jung KH, Shin D, Kim S, et al.. Intratracheal Ovalbumin Administration Induces Colitis Through the IFN-gPathway in Mice. Front Immunol, 2019,10:530
Blasius AL, Beutler B. Intracellular toll-like receptors. Immunity, 2019,32(3):305–315
Chamanara M, Rashidian A, Mehr SE, et al. Melatonin ameliorates TNBS-induced colitis in rats through the melatonin receptors: involvement of TLR4/MyD88/NF-kB signalling pathway. Inflammopharmacology, 2019,27(2):361–371
Wang JP, Dong LN, Wang M, et al. MiR-146a regulates the development of ulcerative colitis via mediating the TLR4/MyD88/NF-kB signalling pathway. Eur Rev Med Pharmacol Sci, 2019,23(5):2151–2157
Li Y, Liu Q, Tang JH, et al.. Regulatory mechanism of mesalazine on TLR4 /MyD88 -dependent pathway in mouse ulcerative colitis model. Eur Rev Med Pharmacol Sci, 2019,10:530
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the National Natural Science Foundation of China (No. 81703380), the Natural Science Foundation of Hubei Province (No. 2017CFB782), the Scientific Research Project of Health and Family Planning Commission of Hubei Province (No. WJ2017M077), and the Applied Basic Research Project of Wuhan Science and Technology Bureau (No. 2017060201010215).
Conflict of Interest Statement
The authors declare that there is no conflict of interest with any financial organization or corporation or individual that can inappropriately influence this work.
Rights and permissions
About this article
Cite this article
Li, J., Zhang, Jl., Gong, Xp. et al. Anti-inflammatory Activity of Mollugin on DSS-induced Colitis in Mice. CURR MED SCI 40, 910–916 (2020). https://doi.org/10.1007/s11596-020-2262-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-020-2262-5