Summary
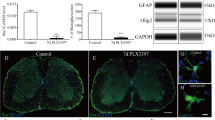
Emerging evidence indicates that microglia activation plays an important role in spinal cord injury (SCI) caused by trauma. Studies have found that inhibiting the Rho/Rho-associated protein kinase (ROCK) signaling pathway can reduce inflammatory cytokine production by microglia. In this study, Western blotting was conducted to detect ROCK2 expression after the SCI; the ROCK Activity Assay kit was used for assay of ROCK pathway activity; microglia morphology was examined using the CD11b antibody; electron microscopy was used to detect microglia phagocytosis; TUNEL was used to detect tissue cell apoptosis; myelin staining was performed using an antibody against myelin basic protein (MBP); behavioral outcomes were evaluated according to the methods of Basso, Beattie, and Bresnahan (BBB). We observed an increase in ROCK activity and microglial activation after SCI. The microglia became larger and rounder and contained myelin-like substances. Furthermore, treatment with fasudil inhibited neuronal cells apoptosis, alleviated demyelination and the formation of cavities, and improved motor recovery. The experimental evidence reveals that the ROCK inhibitor fasudil can regulate microglial activation, promote cell phagocytosis, and improve the SCI microenvironment to promote SCI repair. Thus, fasudil may be useful for the treatment of SCI.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ambrozaitis KV, Kontautas E, Spakauskas B, et al. Pathophysiology of acute spinal cord injury. Medicina (Kaunas), 2006,42(3):255–261
Qiao F, Atkinson C, Kindy MS, et al. The alternative and terminal pathways of complement mediate post-traumatic spinal cord inflammation and injury. Am J Pathol, 2010,177(6):3061–3070.
Beck KD, Nguyen HX, Galvan MD, et al. Quantitative analysis of cellular inflammation after traumatic spinal cord injury: evidence for a multiphasic inflammatory response in the acute to chronic environment. Brain, 2010, 133(Pt 2):433–447
Polazzi E, Monti B. Microglia and neuroprotection: from in vitro studies to therapeutic applications. Prog Neurobiol, 2010,92(3): 293–315
Napoli I, Neumann H, Microglial clearance function in health and disease. Neuroscience, 2009,158(3):1030–1038
Ishizaki T, Maekawa M, Fujisawa, K, et al. The small GTP-binding protein Rho binds to and activates a 160 kDa Ser/Thr protein kinase homologous to myotonic dystrophy kinase. EMBO J, 1996,15(8):1885–1093
Matsui T, Amano M, Yamamoto T, et al. Rho-associated kinase, a novel serine/threonine kinase, as a putative target for small GTP binding protein Rho. EMBO J, 1996,15(9):2208–2016
Gitik M, Reichert F, Rotshenker S. Cytoskeleton plays a dual role of activation and inhibition in myelin and zymosan phagocytosis by microglia. FASEB J, 2010,24(7): 2211–2221
Pellegrin S, Mellor H. Actin stress fibres. J Cell Sci, 2007, 120(20):3491–3499
Rousseau M, Gaugler MH, Rodallec A, et al. RhoA GTPase regulates radiation-induced alterations in endothelial cell adhesion and migration. Biochem Biophys Res Commun, 2011,414(4): 750–755
Anand RJ, Dai S, Gribar SC, et al. A role for connexin43 in macrophage phagocytosis and host survival after bacterial peritoneal infection. J Immunol, 2008,181(12):8534–8543
Boe DM, Richens TR, Horstmann SA, et al. Acute and chronic alcohol exposure impair the phagocytosis of apoptotic cells and enhance the pulmonary inflammatory response. Alcohol Clin Exp Res, 2010,34(10):1723–1732
Janssen WJ, McPhillips KA, Dickinson MG, et al. Surfactant proteins A and D suppress alveolar macrophage phagocytosis via interaction with SIRP alpha. Am J Respir Crit Care Med, 2008,178(2):158–167
Olson MF. Applications for ROCK kinase inhibition. Curr Opin Cell Biol, 2008,20(2):242–248
Miyata K, Shimokawa H, Kandabashi T, et al. Rho-kinase is involved in macrophage-mediated formation of coronary vascular lesions in pigs in vivo. Arterioscler Thromb Vasc Biol, 2000,20(11):2351–2358
Ding J, Li QY, Wang X, et al. Fasudil protects hippocampal neurons against hypoxia-reoxygenation injury by suppressing microglial inflammatory responses in mice. J Neurochem, 2010,114(6):1619–1629
Baba H, Tanoue Y, Maeda T, et al. Protective effects of cold spinoplegia with fasudil against ischemic spinal cord injury in rabbits. J Vasc Surg, 2010,51(2):445–452
Erschbamer M, Pernold K, Olson L. Inhibiting epidermal growth factor receptor improves structural, locomotor, sensory, and bladder recovery from experimental spinal cord injury. J Neurosci, 2007,27:6428–6435
Furuya T, Hashimoto M, Koda M, et al. Treatment of rat spinal cord injury with a Rho-kinase inhibitor and bone marrow stromal cell transplantation. Brain Res, 2009, 1295:192–202.
Wong JK, Sharp K, Steward O. A straight alley version of the BBB locomotor scale. Exp Neurol, 2009,217(2): 417–420
Streit WJ, Graeber MB, Kreutzberg GW. Functional plasticity of microglia: a review. Glia, 1988,1(5):301–307
Beattie MS. Inflammation and apoptosis: linked therapeutic targets in spinal cord injury. Trends Mol Med, 2004, 10(12):580–583
Morino T, Ogata T, Horiuchi H, et al. Delayed neuronal damage related to microglia proliferation after mild spinal cord compression injury. Neurosci Res, 2003,46(3):309–318
Zhao W, Xie W, Le W, et al. Activated microglia initiate motor neuron injury by a nitric oxide and glutamate-mediated mechanism. J Neuropathol Exp Neurol, 2004,63(9):964–977
Loane DJ, Byrnes KR. Role of microglia in neurotrauma. Neurotherapeutics, 2010,7(4):366–377
Schwab JM, Schluesener HJ. Microglia rules: insights into microglial-neuronal signaling. Cell Death Differ, 2004, 11(12):1245–1246
Neumann J, Sauerzweig JS, Ronicke R, et al. Microglia cells protect neurons by direct engulfment of invading neutrophil granulocytes: a new mechanism of CNS immune privilege. J Neurosci, 2008,28(23):5965–5975
Hou SW, Liu CY, Li YH, et al. Fasudil ameliorates disease progression in experimental autoimmune encephalomyelitis, acting possibly through antiinflammatory effect. CNS Neurosci Ther, 2012,18(11):909–917
Chan CC, Wong AK, Liu J, et al. ROCK inhibition with Y27632 activates astrocytes and increases their expression of neurite growth-inhibitory chondroitin sulfate proteoglycans. Glia, 2007,55(4):369–384
Ding J, Yu JZ, Li QY, et al. Rho kinase inhibitor Fasudil induces neuroprotection and neurogenesis partially through astrocyte-derived G-CSF. Brain Behav Immun. 2009,23(8):1083–1088
Boomkamp SD, Riehle MO, Wood J, et al. The development of a rat in vitro model of spinal cord injury demonstrating the additive effects of Rho and ROCK inhibitors on neurite outgrowth and myelination. Glia, 2012,60(3): 441–456
Wang QM, Liao JK. ROCKs as immunomodulators of stroke. Expert Opin Ther Targets, 2012,16(10):1013–1025
Nishio Y, Koda M, Kitajo K, et al. Delayed treatment with Rho-kinase inhibitor does not enhance axonal regeneration or functional recovery after spinal cord injury in rats. Exp Neurol, 2006,200(2):392–397
Watzlawick R, Sena ES, Dirnagl U, et al. Effect and reporting bias of RhoA/ROCK-blockade intervention on locomotor recovery after spinal cord injury: a systematic review and meta-analysis. JAMA Neurol, 2014,71(1): 91–99
Author information
Authors and Affiliations
Corresponding author
Additional information
This project was supported by grants from Natural Science Foundation of China (No. 81471200 and No. 81000521), National Basic Research Program of China (No. 2011CB504403) and the Young Medical Scientists Project of Wuhan (No. 2013-12).
Rights and permissions
About this article
Cite this article
Fu, Pc., Tang, Rh., Wan, Y. et al. ROCK inhibition with fasudil promotes early functional recovery of spinal cord injury in rats by enhancing microglia phagocytosis. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 36, 31–36 (2016). https://doi.org/10.1007/s11596-016-1537-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-016-1537-3