Summary
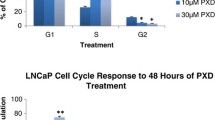
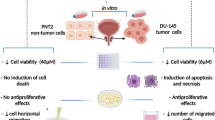
In this study, one immortalized human normal prostatic epithelial cell line (BPH) and four human prostate cancer cell lines (LNCaP, 22Rv1, PC-3, and DU-145) were treated with Ganoderma Lucidum triterpenoids (GLT) at different doses and for different time periods. Cell viability, apoptosis, and cell cycle were analyzed using flow cytometry and chemical assays. Gene expression and binding to DNA were assessed using real-time PCR and Western blotting. It was found that GLT dose-dependently inhibited prostate cancer cell growth through induction of apoptosis and cell cycle arrest at G1 phase. GLT-induced apoptosis was due to activation of Caspases-9 and -3 and turning on the downstream apoptotic events. GLT-induced cell cycle arrest (mainly G1 arrest) was due to up-regulation of p21 expression at the early time and down-regulation of cyclin-dependent kinase 4 (CDK4) and E2F1 expression at the late time. These findings demonstrate that GLT suppresses prostate cancer cell growth by inducing growth arrest and apoptosis, which might suggest that GLT or Ganoderma Lucidum could be used as a potential therapeutic drug for prostate cancer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cohen N, Cohen J, Asatiani MD, et al. Chemical composition and nutritional and medicinal value of fruit bodies and submerged cultured mycelia of culinary-medicinal higher Basidiomycetes mushrooms. Int J Med Mushrooms, 2014,16(3):273–291
Cilerdžic J, Vukojevic J, Stajic M, et al. Biological activity of Ganoderma lucidum basidiocarps cultivated on alternative and commercial substrate. J Ethnopharmacol, 2014,155(1):312–319
Zhang W, Tao J, Yang X, et al. Antiviral effects of two Ganoderma lucidum triterpenoids against enterovirus 71 infection. Biochem Biophys Res Commun, 2014,449(3):307–312
Wang J, Yuan Y, Yue T. Immunostimulatory activities of ß-d-glucan from Ganoderma Lucidum. Carbohydr Polym, 2014,102:47–54
Tello I, Campos-Pena V, Montiel E, et al. Anticonvulsant and neuroprotective effects of oligosaccharides from Lingzhi or Reishi medicinal mushroom, Ganoderma lucidum (Higher Basidiomycetes). Int J Med Mushrooms, 2013,15(6):555–568
Zhao ZH, Zheng XW, Fang F. Ganoderma lucidum polysaccharides supplementation attenuates exercise-induced oxidative stress in skeletal muscle of mice. Saudi J Biol Sci, 2014,21(2):119–123
Choi S, Nguyen VT, Tae N, et al. Anti-inflammatory and heme oxygenase-1 inducing activities of lanostane triterpenes isolated from mushroom Ganoderma lucidum in RAW264.7 cells. Toxicol Appl Pharmacol, 2014,280(3): 434–442
Deepalakshmi K, Mirunalini S, Krishnaveni M, et al. In vitro and in vivo antioxidant potentials of an ethanolic extract of Ganoderma lucidum in rat mammary carcinogenesis. Chin J Nat Med, 2013,11(6):621–627
Chen J, Xiao HJ, Huang ZS, et al. MicroRNA124 regulate cell growth of prostate cancer cells targeting iASPP. Int J Clin Exp Pathol, 2014,7(5):2283–2290
Da J, Cheng CR, Yao S, et al. A reproducible analytical system based on the multi-component analysis of triterpene acids in Ganoderma lucidum. Phytochemistry, 2014,pii:S0031–9422(14)00322–7
Zhang B, Xiao HJ, Chen J, et al. Inhibitory member of the apoptosis-stimulating protein of p53 (ASPP) family promotes growth and tumorigenesis in human p53-deficient prostate cancer cells. Prostate Cancer Prostatic Dis, 2011,14(3):219–224
Zárate-Chaves CA, Romero-Rodríguez MC, Niño-Arias FC, et al. Optimizing a culture medium for biomass and phenolic compounds production using Ganoderma lucidum. Braz J Microbiol, 2013,44(1):215–223
Zhao ZZ, Yin RH, Chen HP, et al. Two new triterpenoids from fruiting bodies of fungus Ganoderma lucidum. J Asian Nat Prod Res, 2015,29:1–6
Xia Q, Zhang H, Sun X, et al. A comprehensive review of the structure elucidation and biological activity of triterpenoids from Ganoderma spp. Molecules, 2014,19(11): 17 478–17 535
Qiu JX, Zhou ZW, He ZX, et al. Plumbagin elicits differential proteomic responses mainly involving cell cycle, apoptosis, autophagy, and epithelial-to-mesenchymal transition pathways in human prostate cancer PC-3 and DU145 cells. Drug Des Devel Ther, 2015,9:349–417
Sardana G, Jung K, Stephan C, et al. Proteomic analysis of conditioned media from the PC3, LNCaP, and 22Rv1 prostate cancer cell lines: discovery and validation of candidate prostate cancer biomarkers. J Proteome Res, 2008,7(8):3329–3338
Xiong H, Xu Y, Tan G, et al. Glycyrrhizin ameliorates imiquimod-induced psoriasis-like skin lesions in BALB/c mice and inhibits TNF-α-induced ICAM-1 expression via NF-?B/MAPK in HaCaT cells. Cell Physiol Biochem, 2015,35(4):1335–1346
Wen X, Zhou J, Zhang D, et al. Denatonium inhibits growth and induces apoptosis of airway epithelial cells through mitochondrial signaling pathways. Respir Res, 2015,16(1):13
Zhang J, Tang Q, Zhou C, et al. GLIS, a bioactive proteoglycan fraction from Ganoderma lucidum, displays anti-tumour activity by increasing both humoral and cellular immune response. Life Sci, 2010,87(19–22):628–637
Hsieh TC, Wu JM. Regulation of cell cycle transition and induction of apoptosis in HL-60 leukemia cells by the combination of Coriolus versicolor and Ganoderma lucidum. Int J Mol Med, 2013,32(1):251–257
Guo Y, Luo X, Yu M, et al. Active ingredients and efficacies of Ganoderma lucidum cultivated on non-medicinal parts of Chinese medicinal herbs. Wei Sheng Wu Xue Bao (Chinese), 2011,51:764–768
Dudhgaonkar S, Thyagarajan A, Sliva D. Suppression of the inflammatory response by triterpenes isolated from the mushroom Ganoderma lucidum. Int Immunopharmacol, 2009,9(11):1272–1280
Yao X, Li G, Xu H, et al. Inhibition of the JAK-STAT3 signaling pathway by ganoderic acid A enhances chemosensitivity of HepG2 cells to cisplatin. Planta Med, 2012,78:1740–1748
Chen NH, Liu JW, Zhong JJ. Ganoderic acid T inhibits tumor invasion in vitro and in vivo through inhibition of MMP expression. Pharmacol Rep, 2010,62: 150–163
Chang UM, Li CH, Lin LI, et al. Ganoderiol F, a ganoderma triterpene, induces senescence in hepatoma HepG2 cells. Life Sci, 2006,79(12):1129–1139
Lin SB, Li CH, Lee SS, et al. Triterpene-enriched extracts from Ganoderma lucidum inhibit growth of hepatoma cells via suppressing protein kinase C, activating mitogen-activated protein kinases and G2-phase cell cycle arrest. Life Sci, 2003,72(21):2381–2390
Yue QX, Song XY, Ma C, et al. Effects of triterpenes from Ganoderma lucidum on protein expression profile of HeLa cells. Phytomedicine, 2010,17(8–9):606–613
Thyagarajan A, Jedinak A, Nguyen H, et al. Triterpenes from Ganoderma Lucidum induce autophagy in colon cancer through the inhibition of p38 mitogen- activated kinase (p38 MAPK). Nutr Cancer, 2010,62(5):630–640
Feng L, Yuan L, Du M, et al. Anti-lung cancer activity through enhancement of immunomodulation and induction of cell apoptosis of total triterpenes extracted from Ganoderma luncidum (Leyss. ex Fr.) Karst. Molecules, 2013,18(8):9966–9981
Martinez-Montemayor MM, Acevedo RR, Otero-Franqui E, et al. Ganoderma lucidum (Reishi) inhibits cancer cell growth and expression of key molecules in inflammatory breast cancer. Nutr Cancer, 2011,63(7):1085–1094
Parajuli KR, Zhang Q, Liu S, et al. Methoxyacetic acid suppresses prostate cancer cell growth by inducing growth arrest and apoptosis. Am J Clin Exp Urol, 2014,2(4):300–312
Author information
Authors and Affiliations
Corresponding authors
Additional information
The authors contributed equally to this work.
This project was supported by Science and Technology Planning Project of Guangdong Province, China (No. 2011B080701075).
Rights and permissions
About this article
Cite this article
Wang, T., Xie, Zp., Huang, Zs. et al. Total triterpenoids from Ganoderma Lucidum suppresses prostate cancer cell growth by inducing growth arrest and apoptosis. J. Huazhong Univ. Sci. Technol. [Med. Sci.] 35, 736–741 (2015). https://doi.org/10.1007/s11596-015-1499-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-015-1499-x