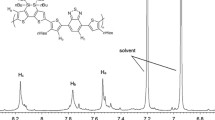
Abstract
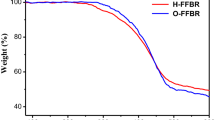
Two novel non-fullerene small molecule acceptors were prepared with the conjugated backbone of 5H-fluoreno[3, 2- b:6, 7- b’] dithiophene carrying the electron deficient unit of dicyanomethylene indanone (DICTFDT) and rhodanine (TFDTBR), respectively. The two acceptors exhibited excellent thermal stability and strong absorption in the visible region. The LUMO level is estimated to be at -3.89 eV for DICTFDT and -3.77 eV for TFDTBR. When utilized as the acceptor in bulk heterojunction polymer solar cells with the polymer donor of PBT7-Th, the optimized maximum power conversion efficiency of 5.12% and 3.95% was obtained for the device with DICTFDT and TFDTBR, respectively. The research demonstrates that 5H-fluoreno[3, 2- b:6, 7- b’] dithiophene can be an appealing candidate for constructing small molecular electron acceptor towards efficient polymer:non-fullerene bulk heterojunction solar cells.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Reference
Chen J, Cao Y. Development of Novel Conjugated Donor Polymers for High-Effciency Bulk-Heterojunction Photovoltaic Devices[J]. Acc. Chem. Res., 2009, 42(11): 1709–1718
Cheng Y-J, Yang S-H, Hsu C-S. Synthesis of Conjugated Polymers for Organic Solar Cell Applications[J]. Chem. Rev., 2009, 109(11): 5868–5923
Li G, Zhu R. Yang Y. Polymer Solar Cells[J]. Nat. Photon., 2012, 6(3): 153–161
Dou L, You J, Hong Z, et al. 25th Anniversary Article: A Decade of Organic/Polymeric Photovoltaic Research[J]. Adv. Mater., 2013, 25(46): 6642–6671
Li Y. Molecular Design of Photovoltaic Materials for Polymer Solar Cells: Toward Suitable Electronic Energy Levels and Broad Absorp-tion[J]. Acc. Chem. Res., 2012, 45(5): 723–733
Xiao S, Zhang Q, You W. Molecular Engineering of Conjugated Polymers for Solar Cells: An Updated Report[J]. Adv. Mater., 2017, 29(20): 1 601 391
Liu T, Troisi A. What Makes Fullerene Acceptors Special as Electron Acceptors in Organic Solar Cells and How to Replace Them[J]. Adv. Mater., 2013, 25(7): 1038–1041
He Y, Li Y. Fullerene Derivative Acceptors for High Performance Polymer Solar Cells[J]. Phys. Chem. Chem. Phys., 2011, 13(6): 1970–1983
Zhao J, Li Y, Yang G. et al. Efficient Organic Solar Cells Processed from Hydrocarbon Solvents[J]. Nat. Energy, 2016, 1: 15027
Lin Y, Li Y, Zhan X. Small Molecule Semiconductors for High-eff-ciency Organic Photovoltaics[J]. Chem. Soc. Rev., 2012, 41(11): 4245–4272
Anthony J E. Small-Molecule, Nonfullerene Acceptors for Polymer Bulk Heterojunction Organic Photovoltaics[J]. Chem. Mater., 2011, 23(3): 583–590
Lin Y, Wang J, Zhang Z-G et al. An Electron Acceptor Challenging Fullerenes for Efficient Polymer Solar Cells[J]. Adv. Mater., 2015, 27(7): 1170–1174
Lin Y, He Q, F Zhao et al. A Facile Planar Fused-Ring Electron Acceptor for As-Cast Polymer Solar Cells with 8.71% Effciency[J]. J. Am. Chem. Soc., 2016, 138(9): 2973–2976
Lin Y, Zhao F, Wu Y. Mapping Polymer Donors toward High-Effcien-cy Fullerene Free Organic Solar Cells[J]. Adv. Mater., 2017, 29(3): 1 604 155
Cheng P, Zhang M, Lau T-K et al. Realizing Small Energy Loss of 0.55 eV, High Open-Circuit Voltage >1 V and High Efficiency >10% in Fullerene-Free Polymer Solar Cells via Energy Driver[J]. Adv. Mater., 2017, 29(11): 1 605 216
Bin H, Zhang Z-G, Gao L et al. Non-Fullerene Polymer Solar Cells Based on Alkylthio and Fluorine Substituted 2D-Conjugated Polymers Reach 9.5% Effciency[J]. J. Am. Chem. Soc., 2016, 138(13): 4657–4664
Yang Y, Zhang Z-G, Bin H et al. Side-Chain Isomerization on an n-type Organic Semiconductor ITIC Acceptor Makes 11.77% High Effciency Polymer Solar Cells[J]. J. Am. Chem. Soc., 2016, 138(45): 15011–15018
Yao H, Chen Y, Qin Y et al. Design and Synthesis of a Low Bandgap Small Molecule Acceptor for Efficient Polymer Solar Cells[J]. Adv. Mater., 2016, 28(37): 8283–8287
Baran D, Ashraf R S, Hanif D A, et al. Reducing the Effciency-stabil-ity-cost Gap of Organic Photovoltaics with highly Effcient and Stable Small Molecule Acceptor Ternary Solar Cells[J]. Nat. Mater., 2017, 16(3): 363–369
Zhao W, Qian D, Zhang S, et al. Fullerene-Free Polymer Solar Cells with over 11% Effciency and Excellent Thermal Stability[J]. Adv. Mater., 2016, 28(23): 4734–4739
Li S, Ye L, Zhao W, et al. Energy-Level Modulation of Small-Molecule Electron Acceptors to Achieve over 12% Effciency in Polymer Solar Cells[J]. Adv. Mater., 2016, 28(42): 9423–9429
Liu T, Guo Y, Yi Y, et al. Ternary Organic Solar Cells Based on Two Compatible Nonfullerene Acceptors with Power Conversion Effciency >10%[J]. Adv. Mater., 2016, 28(45): 10008–10015
Zhao W, Li S, Yao H, et al. Molecular Optimization Enables over 13% Effciency in Organic Solar Cells[J]. Journal of the American Chemical Society, 2017, 139(21): 7148–7151
Holliday S, Ashraf R S, Nielsen C B, et al. A Rhodanine Flanked Non-fullerene Acceptor for Solution-Processed Organic Photovoltaics[J]. J. Am. Chem. Soc., 2015, 137(2): 898–904
Gao J, Wang W, Zhang S, et al. Distinction between PTB7-Th Samples Prepared from Pd(PPh3)4 and Pd2(dba)3/P(o-tol)3 Catalysed Stille Coupling Polymerization and the Resultant Photovoltaic Performance[J]. J. Mater. Chem. A, 2018, 6(1): 179–188
Yang M, Lau T-K, Xiao S, et al. A Ladder-type Heteroheptacene 12H-Dithieno[2’,3’:4,5]thieno[3,2-b:2’,3’-h]fuorene Based D-A Co-polymer with Strong Intermolecular Interactions toward Effcient Polymer Solar Cells[J]. ACS Appl. Mater. Interfaces, 2017, 9(40): 35159–35168
Lin Y, Zhang Z-G, Bai H, et al. High-performance Fullerene-free Polymer Solar Cells with 6.31% Effciency[J]. Energy Environ. Sci., 2015, 8(2): 610–616
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National Natural Science Foundation of China (No. 21673170)
Rights and permissions
About this article
Cite this article
Wu, J., Wang, W., Zhan, C. et al. 5H-Fluoreno [3,2- b:6,7- b’] Dithiophene Based Non-fullerene Small Molecular Acceptors for Polymer Solar Cell Application. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 34, 1220–1227 (2019). https://doi.org/10.1007/s11595-019-2181-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-019-2181-0