Abstract
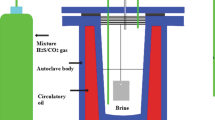
The corrosion behavior of 16Mn steel was studied in saturated H2S or H2S/CO2 solutions containing different Cl− concentrations at 80 °C. The microstructure and chemical composition of the corrosion products were investigated through scanning electron microscopy, energy-dispersive X-ray spectroscopy, EPMA, and X-ray diffraction. Results showed that the corrosion rate decreased with increasing Cl− concentration in saturated H2S or H2S/CO2 solution at pH 4. Conversely, the corrosion rate increased with increasing Cl− concentration in saturated H2S solution at pH 6. The relative H+ concentration decreased because of the increase of Cl− concentration at pH 4, and Cl− acted as a catalyst in the corrosive medium at pH 6 because the net H+ concentration decreased obviously compared with the condition at pH4. Cl− promoted the formation of Fe-deficient iron sulfide at pH 4, and the opposite effect was observed in the nearly neutral solution. The corrosion rate increased firstly with increasing Cl− concentration and then decreased in the saturated H2S/CO2 solution at pH 6. The corrosion products were mainly composed of two kinds of iron sulfide. Sulfide FeS1−x was a kind of tetragonal crystal, whereas the other was the hexagonal/monoclinic iron sulfide Fe1−xS. The corrosion film that was mainly composed of FeS1−x did not confer a protective effect on the base metal. The atomic ratio of Fe/S was more than 1 for FeS1−x. The appearance of sulfide FeS1−x resembled a square block or small, needle-like, flocculent particles. The atomic ratio of Fe/S was less than 1 for Fe1−xS, and the corrosion film mainly composed of Fe1−xS conferred some protective property on the base metal. The sulfide FeS1−x exhibited a long claviform morphology with a hexagonal or quadrilateral cross-sectional shape.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Saito N, Tsuchiya Y, Akai Y, et al. Corrosion Performance of Metals for Supercritical Water, Oxidation-utilized Organic Waste-processing Reactors[J]. Corrosion, 2006, 62(5): 383–394
Singer M, Camacho A, Brown B, et al. Sour Top-of-the-Line Corrosion in the Presence of Acetic Acid[J]. Corrosion, 2011,67 (8): B1–B16
Lashkari M, Arshadi M R. DFT Studies of Pyridine Corrosion Inhibitors in Electrical Double Layer: Solvent, Substrate and Electric Field Effects[J]. Chem. Phys., 2004, 299(1): 131–137
Hamzah R, Stephenson D J, Strutt J E. Erosion of Material used in Petroleum Production[J]. Wear, 1995, 186/187: 493–496
Stack M M, Abdulrahman G H. Mapping Erosion-corrosion of Carbon Steel in Oil Exploration Conditions: Some New Approaches to Characterizing Mechanisms and Synergies[J]. Tribol. Int., 2010, 43 (7): 1268–1277
Chiang K T, Dunn D S, Cragnolino G A. Effect of Simulated Groundwater Chemistry on Stress Corrosion Cracking of Alloy 22[J]. Corrosion, 2007,63(10): 940–950
Schroer C, Konys J, Novotny J, et al. Material Performance in Chlorinated Supercritical Water Systems[J]. Corrosion, 2007, 63(1): 46–62
Kappes M, Frankel G S, Sridhar N, et al. Corrosion Behavior of Carbon Steel in Acidified, Thiosulfate-Containing Brines[J]. Corrosion, 2012, (68): 872–884
Mendibide C, Michaud H, Pineau F, et al. Role of the Machining Residual Stress on the Sulfide Stress Cracking Resistance of Carbon Steel Evaluated According to NACE TM0177 Method A[J]. Corrosion, 2012, 68(10): 897–903
Yang Y F, Scantlebury J D, Koroleva F, et al. Underprotection of Mild Steel in Seawater and the Role of the Calcareous Film[J]. Corrosion, 2012, 68 (5): 432–440
Ma H Y, Cheng X L, Li G Q, et al. The Influence of Hydrogen Sulfide on Corrosion of Iron under Different Conditions[J]. Corros. Sci., 2000, 42(10): 1669–1683
Pessu F, Barker R, Neville A. The Influence of pH on Localized Corrosion Behavior of X65 Carbon Steel in CO2 Saturated Brines[J]. Corrosion, 2015, 71(12): 1452–1466
Choia Y S, Nesica S, Ling S. Effects of H2S on the CO2 Corrosion of Carbon Steel in Acidic Solutions[J]. Electrochim. Acta, 2011, 56(4): 1752–1760
W Sun. Kinetics of Iron Carbonate and Iron Sulfide Scale Formation in CO2/H2S Corrosion[D]. Ohio: Ohio University, 2006
Hu Y B, Dong C F, Sun M, et al. Effects of Solution pH and Cl-on Electrochemical Behavior of an Aermet100 Ultro-high Strength Steel in Acidic Environments[J]. Corros. Sci., 2011, 53(12): 4159–4165
Asahi H, Ueno M, Yonezawa T. Prediction of Sulfide Stress Cracking in high-strength Tubulars[J]. Corrosion, 1994, 50(7): 537–545
Hara T, Asahi H, Ogawa H. Conditions of Hydrogen-Induced Corrosion Occurrence of X65 Grade Line Pipe Steel in Sour Environments[J]. Corrosion, 2004, 60(12): 1113–1121
Tribollet B, Kittel J, Meroufel A, et al. Corrosion Mechanism in Aqueous Solutions Containing Dissolved H2S. Part 2: Model of the Cathodic Reactions on a 316L Stainless Steel Rotating Disc Electrode[J]. Electrochim. Acta, 2014, 124(1): 46–51
Linter B R, Burstein G T. Reactions of Pipeline Steels in Carbon Dioxide Solutions[J]. Corros. Sci., 1999, 41(1): 117–139
Teysseyre S, Was GS. Stress Corrosion Cracking of Austenitic Alloys in Supercritical Water[J]. Corrosion, 2006, 62(12): 1100–1116
Beerling D J, Lake J A, Berner R A, et al., Carbon Isotope Evidence Implying High O2/CO2 Ratios in the Permo-carboniferous Atmosphere[J]. Geochim. Cosmochim. Ac., 2002, 66(21): 3757–3767
David R, George W Luther H. Chemistry of Iron Sulfides[J]. J. Am. Chem. Soc., 2007, 107(2): 514–562
Sosa E, Cabrera-Sierra R, García I. The Role of Different Surface Damages in Corrosion Process in Alkaline Sour Media[J]. Corros. Sci., 2002, 44(7): 1515–1528
Yang L, Sridhar N, Pensado O, et al. An in-situ Galvanically Coupled Multielectrode Array Sensor for Localized Corrosion[J]. Corrosion, 2002, 58(12): 1004–1014
Fragiel A, Serna S, Pérez R. Electrochemical Study of Two Microalloyed Pipeline Steel in H2S Environments[J]. Int. J. Hydrogen Energ., 2005, 30(12): 1303–1309
Malo J M, Uruchurtu J, Corona O. Corrosion Detection of Mild Steel in a Two-phase Hydrocarbon/electrolyte System under Flow Conditions using Electrochemical Noise[J]. Corrosion, 2002, 58(11): 932–940
Colominas S, Mclafferty J, Macdonald D D. Electrochemical Studies of Sodium Borohydride in Alkaline Aqueous Solutions using a Gold Electrode[J]. Electrochim. Acta, 2009, 54(13): 3575–3579
Cao L, Frankel G S, Sridhar N. Effect of Chloride on Stress Corrosion Cracking Susceptibility of Carbon Steel in Simulated Fuel Grade Ethanol[J]. Electrochim. Acta, 2013, 104(1): 255–266
Yin Z F, Liu L, Zhang Y Q. Corrosion Behavior of SM 80SS Tube Steel in Stimulant Solution Containing H2S and CO2[J]. Electrochim. Acta, 2008, 53(10): 3690–3700
Ramanarayanan T A, Chun C M. Metal Dusting Corrosion of Metals and Alloys[M]. Developments in High Temperature Corrosion and Protection of Materials. Princeton: Woodhead Publishing, 2008
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by National Natural Science Foundation of China (No.51765035)
Rights and permissions
About this article
Cite this article
Song, W., Yang, G., Liao, B. et al. Effect of Cl− Concentration on the Corrosion Behavior of 16Mn Steel in Saturated H2S/CO2 Solution. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 33, 1205–1215 (2018). https://doi.org/10.1007/s11595-018-1954-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-018-1954-1