Abstract
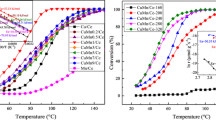
Metal-organic framework (MOF) material MIL-53(Al) with high thermal stability was prepared by a solvothermal method, serving as a support material of cerium doped copper catalyst (Ce-Cu)/MIL-53(Al) material for CO oxidation with high catalytic activity. The catalytic performance between the (Cu-Ce)/MIL-53(Al) and the Cu/MIL-53(Al) catalytic material was compared to understand the catalytic behavior of the catalysts. The catalysts were characterized by thermogravimetric-differential scanning calorimetry (TG-DSC), N2 adsorption- desorption, X-ray diffraction (XRD), and transmission electron microscopy (TEM). The characterization results showed that MIL-53(Al) had good stability and high surface areas, the (Ce-Cu) nanoparticles on the MIL-53(Al) support was uniform. Therefore, the heterogeneous catalytic composite materials (Ce-Cu)/MIL-53(Al) catalyst exhibited much higher activity than that of the Cu/MIL- 53(Al) catalyst in CO oxidation test, with 100% conversion at 80 °C. The results reveal that (Cu-Ce)/MIL- 53(Al) is the suitable candidate for achieving low temperature and higher activity CO oxidation catalyst of MOFs.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Tan H Y, Wu J P. Performance of a Metal-organic Framework MIL-53(Al)-Supported Cobalt Catalyst in the CO Catalytic Oxidation Reaction[J]. Acta Phys.-Chim. Sin., 2014, 30: 715–722
Jiang H L, Liu B, Akita T, et al. Au@ ZIF-8: CO Oxidation over Gold Nanoparticles Deposited to Metal-organic Framework[J]. J. Am. Chem. Soc., 2009, 131: 11302–11303
Liang Q, Zhao Z, Liu J, et al. Pd Nanoparticles Deposited on Metal-organic Framework of MIL-53(Al): an Active Catalyst for CO Oxidation[J]. Acta Phys.-Chim. Sin., 2014, 30: 129–134
Zhao X, Xiao B, Fletcher A J, et al. Hysteretic Adsorption and Desorption of Hydrogen by Nanoporous Metal-organic Frameworks[J]. Science, 2004, 306: 1012–1015
Chae H K, Siberio-Pérez D Y, Kim J, et al. A Route to High Surface Area, Porosity and Inclusion of Large Molecules in Crystals[J]. Nature, 2004, 427: 523–527
Feng P, Bu X, Stucky G D. Hydrothermal Syntheses and Structural Characterization of Zeolite Analogue Compounds Based on Cobalt Phosphate[J]. Nature, 1997, 388: 735–741
Lu Y, Tonigold M, Bredenkötter B, et al. A Cobalt (II)-containing Metal-organic Framework Showing Catalytic Activity in Oxidation Reactions[J]. Z. Anorg. Allg. Chem., 2008, 634: 2411–2417
Jiang D, Mallat T, Meier D M, et al. Copper Metal-organic Framework: Structure and Activity in the Allylic Oxidation of Cyclohexene with Molecular Oxygen[J]. J. Cater., 2010, 270: 26–33
Schlichte K, Kratzke T, Kaskel S. Improved Synthesis, Thermal Stability and Catalytic Properties of the Metal-organic Framework Compound Cu3(BTC)2[J]. Mirpopor. Mesopor. Mat., 2004, 73: 81–88
Huang Y, Liu S, Lin Z, et al. Facile Synthesis of Palladium Nanoparticles Encapsulated in Amine-functionalized Mesoporous Metal-organic Frameworks and Catalytic for Dehalogenation of Aryl Chlorides[J]. J. Cater., 2012, 292: 111–117
Yaghi O, Li H, Groy T. Construction of Porous Solids from Hydrogen-bonded Metal Complexes of 1, 3, 5-benzenetricarboxylic Acid[J]. J. Am. Chem. Soc., 1996, 118(38): 9096–9101
Jiang D, Mallat T, Krumeich F, et al. Polymer-assisted Synthesis of Nanocrystalline Copper-based Metal-organic Framework for Amine Oxidation[J]. Catal. Commun., 2011, 12: 602–605
Ishida T, Nagaoka M, Akita T, et al. Deposition of Gold Clusters on Porous Coordination Polymers by Solid Grinding and Their Catalytic Activity in Aerobic Oxidation of Alcohols[J]. Chem. Eur. J., 2008, 14: 8456–8460
Brown K, Zolezzi S, Aguirre P, et al. [Cu (H btec)(bipy)]∞: a Novel Metal Organic Framework (MOF) as Heterogeneous Catalyst for the Oxidation of Olefins[J]. Dalton Trans., 2009, 10: 1422–1427
Tonigold M, Lu Y, Bredenkötter B, et al. Heterogeneous Catalytic Oxidation by MFU-1: A Cobalt (II)-containing Metal-organic Framework[J]. Angew Chem. Int. Ed., 2009, 48: 7546–7550
Marx S, Kleist W, Baiker A. Synthesis, Structural Properties, and Catalytic Behavior of Cu-BTC and Mixed-linker Cu-BTC-PyDC in the Oxidation of Benzene Derivatives[J]. J. Cater., 2011, 281: 76–87
Sun C Y, Liu S X, Liang D D, et al. Highly Stable Crystalline Catalysts Based on a Microporous Metal-organic Framework and Polyoxometalates[J]. J. Am. Chem. Soc., 2009, 131: 1883–1888
Wee L H, Bajpe S R, Janssens N, et al. Convenient Synthesis of Cu3(BTC)2 Encapsulated Keggin Heteropolyacid Nanomaterial for Application in Catalysis[J]. Chem. Commun., 2010, 46: 8186–8188
Qiu W G, Wang Y, Li C Q, et al. Effect of Activation Temperature on Catalytic Performance of CuBTC for CO Oxidation[J]. Chin. J. Catal., 2012, 33: 986–992
Tan Z D, Tan H Y, Shi X Y, et al. Metal-organic Framework MIL-53 (Al)-supported Copper Catalyst for CO Catalytic Oxidation Reaction[J]. Inorg Chem. Commun., 2015, 61: 128–131
Kuo C H, Li W, Song W, et al. Facile Synthesis of Co3O4@ CNT with High Catalytic Activity for CO Oxidation under Moisture-rich Conditions[J]. ACS Appl. Mater. Inter., 2014, 6: 11311–11317
Lou Y, Cao X M, Lan J, et al. Ultralow-temperature CO oxidation on an In2O3-Co3O4 Catalyst: a Strategy to Tune CO Adsorption Strength and Oxygen Activation Simultaneously[J]. Chem. Commun., 2014, 50: 6835–6838
Song W, Poyraz A S, Meng Y, et al. Mesoporous Co3O4 with Controlled Porosity: Inverse Micelle Synthesis and High-Performance Catalytic CO Oxidation at-60 °C [J]. Chem. Mater., 2014, 26: 4629–4639
Xiao J, Wan L, Wang X, et al. Mesoporous Mn3O4-CoO Core-shell Spheres Wrapped by Carbon Nanotubes: a High Performance Catalyst for the Oxygen Reduction Reaction and CO Oxidation[J]. J. Mater. Chem. A, 2014, 2: 3794–3800
Díaz A A, Cecilia J, Santos L D, et al. Characterization and Performance in Preferential Oxidation of CO of CuO-CeO2 Catalysts Synthesized using Polymethyl Metacrylate (PMMA) as Template[J]. Int. J. Hydrogen Energ, 2015, 40: 11254–11260
Zeng S H, Zhang W L, Śliwa M, et al. Comparative Study of CeO2/CuO and CuO/CeO2 Catalysts on Catalytic Performance for Preferential CO Oxidation[J]. Int. J. Hydrogen Energ., 2013, 38: 3597–3605
Baneshi J, Haghighi M, Jodeiri N, et al. Homogeneous Precipitation Synthesis of CuO-ZrO2-CeO2-Al2O3 Nanocatalyst used in Hydrogen Production via Methanol Steam Reforming for Fuel Cell Applications[J]. Energy Convers. Manage., 2014, 87: 928–937
Qi L, Yu Q, Dai Y, et al. Influence of Cerium Precursors on the Structure and Reducibility of Mesoporous CuO-CeO2 Catalysts for CO Oxidation[J]. Appl. Catal. B., 2012, 119: 308–320
Reyes-Carmona Á, Arango-Díaz A, Moretti E, et al. CuO/CeO2 Supported on Zr doped SBA-15 as Catalysts for Preferential CO Oxidation (CO-PROX)[J]. J. Power Sources, 2011, 196: 4382–4387
Tang C, Sun J, Yao X, et al. Efficient Fabrication of Active CuO-CeO2/SBA-15 Catalysts for Preferential Oxidation of CO by Solid State Impregnation[J]. Appl. Catal. B, 2014, 146: 201–212
Ye J Y, Liu C J. Cu3(BTC)2: CO Oxidation over MOF Based Catalysts[J]. Chem. Commun., 2011, 47: 2167–2169
Tan H Y, Liu C, Yan Y F. Simple Preparation of Crystal Co3(BTC)2·12H2O and Its Catalytic Activity in CO Oxidation Reaction[J]. J. Wuhan University of Technology-Mater. Sci. Ed., 2015, 30: 71–75
Li T Y, Xiang G L, Zhuang J, et al. Enhanced Catalytic Performance of Assembled Ceria Necklace Nanowires by Ni Doping[J]. Chem. Commun., 2011, 47: 6060–6061
Yu Q, Wu X X, Tang C J, et al. Textural, Structural, and Morphological Characterizations and Catalytic Activity of Nanosized CeO2-MOx (M= Mg2+, Al3+, Si4+) Mixed Oxides for CO Oxidation[J]. J. Colloid Interface Sci., 2011, 354: 341–352
Acknowledgements
The authors thank the Key Laboratory of Catalysis and Materials Science of the State Ethnic Affairs Commission&Ministry of Education, Hubei Province, South-Central University for Nationalities for their measurement.
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the Guiding Research Project of Hubei Province Department of Education (No. B2016098)
Rights and permissions
About this article
Cite this article
Tan, H., Zhou, Y., Yan, Y. et al. Preparation of cerium doped Cu/MIL-53(Al) catalyst and its catalytic activity in CO oxidation reaction. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 32, 23–28 (2017). https://doi.org/10.1007/s11595-017-1551-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-017-1551-8