Abstract
The ionic liquid polymer electrolyte (IL-PE) membrane is prepared by ultraviolet (UV) cross-linking technology with polyurethane acrylate (PUA), methyl methacrylate (MMA), ionic liquid (Py13TFSI), lithium salt (LiTFSI), ethylene glycol dimethacrylate (EGDMA), and benzoyl peroxide (BPO). N-methyl-N-propyl pyrrolidinium bis(trifluoromethanesulfonyl)imide (Py13TFSI) ionic liquid is synthesized by mixing N-methyl-N-propyl pyrrolidinium bromide (Py13Br) and lithium bis(trifluoromethanesulfonyl)imide (LiTFSI). The addition of Py13TFSI to polymer electrolyte membranes leads to network structures by the chain cross-linking. The resultant electrolyte membranes display the room temperature ionic conductivity of 1.37 × 10−3 S cm−1 and the lithium ions transference number of 0.22. The electrochemical stability window of IL-PE is about 4.8 V (vs. Li+/Li), indicating sufficient electrochemical stability. The interfacial resistances between the IL-PE and the electrodes have the less change after 10 cycles than before 10 cycles. IL-PE has better compatibility with the LiFePO4 electrode and the Li electrode after 10 cycles. The first discharge performance of Li/IL-PE/LiFePO4 half-cell shows a capacity of 151.9 mAh g−1 and coulombic efficiency of 87.9%. The discharge capacity is 131.9 mAh g−1 with 95.5% coulombic efficiency after 80 cycles. Therefore, the battery using the IL-PE exhibits a good cycle and rate performance.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The establishment of a sustainable energy and low-carbon society is a worldwide topic [1,2,3,4,5,6]. In recent years, lithium-ion batteries with high energy density have been proved to be one of the sustainable power sources and extensively studied because of their specific energy, long cycle life, and no memory effect [7,8,9,10]. However, the new lithium batteries of a high-energy and power density are necessary with increasing safety and low cost in order to meet the market of hybrid electric vehicles, portable devices, and so on [11].
At present, ionic liquid electrolyte has developed into gel polymer electrolyte and whole solid polymer electrolyte [12, 13]. Gel polymer electrolyte with higher conductivity and better security has attracted more attention [14,15,16]. Ionic liquids realize the cleaner produce, environment protection, and cycle economics due to its advantages including non-flammable and non-toxic property, low vapor pressure, high conductivity, high ion transference number, wide electrochemistry window, and good stability performance [17,18,19]. Therefore, ionic liquids are one of most important components in the ionic liquid polymer electrolyte (IL-PE) membrane since it is one of main conduct mediums. N-methyl-N-alkyl (alkyl(a)=methyl, ethyl, propyl and butyl) pyrrolidinium bis(trifluoromethanesulfonyl)imide (Py1aTFSI) ionic liquid has good electrochemistry, especially propyl and butyl as side chain [20]. Jae-Kwang Kim prepares the gel polymer electrolyte with N-methyl-N-butyl pyrrolidinium bis(trifluoromethanesulfonyl)imide (Py14TFSI) based on poly(vinylidene fluoride-co-hexafluoropropylene) (P(VdF-HFP)) by the electrostatic spinning method which shows the conductivity of 1.1 × 10−4 S cm−1 at 0 °C [21]. Mumin Rao uses electrostatic spinning technique to prepare PYR14TFSI/poly(acrylonictrile-co-mechyl mechacrylate) (PAN/PMMA) electrolyte film with the conductivity of 3.6 × 10−3 S cm−1 [22].
Polymer matrix is one of the indispensable materials as supporting framework of gel polymer electrolytes [23]. So far, there are mainly four systems as polymer matrix for polymer lithium-ion battery, including polyvinylidene fluoride (PVDF) system [24], polyether (PEO) system [25,26,27], polymethyl methacrylate (PMMA) [28], and polyacrylonictrile (PAN) system [29]. Wei Zhai prepares [BMIM] BF4/PVDF-HFP/PMMA gel polymer electrolyte by the phase inversion method which has the conductivity of 1.4 × 10−3 S cm−1 and the electrochemistry stability window of 4.5 V.
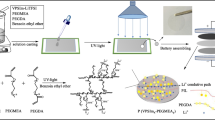
In this work, ultraviolet cross-linking technique is adapted to create a photochemical reaction for the bonds linking one polymer chain to another as a speed curing process. The IL-PE membrane contains the polymethyl methacrylate (PMMA) and light-sensitive urethane acrylate (PUA) as the electrolyte matrix, the lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) as the lithium salt and the N-methyl-N-propyl pyrrolidinium bis(trifluoromethanesulfonyl)imide (Py13TFSI) ionic liquid as improving agent of the conductivity. Meanwhile, the performances of the IL-PE membrane are measured.
Experimental
Preparation of ionic liquid
N-methyl-N-propyl pyrrolidinium bis(trifluoromethanesulfonyl)imide (Py13TFSI) was obtained by the substitution reaction between Py13Br and LiTFSI. The anion substitution reaction from bis(trifluoromethane sulfonyl)imide (TFSI−) to bromide ion (Br−) was performed as following chemical formula Eq. (1).
The proper Py13Br and LiTFSI (molar ratio of 1:1) was dissolved in deionized water and drastically mixed for 24 h. The solution was divided into two layers after 2-h standing, including the upper hydrophilic layer and the bottom hydrophobic layer of the ionic liquid. The bottom layer was repeatedly washed with deionized water to completely remove the Br− until the precipitate did not appear as adding of Ag+. Then, the bottom layer solution was heated for 12 h at 80 °C in vacuum for the evaporation of water. At last, the ionic liquid was quickly moved to an argon-filled glove box.
Preparation of the ionic liquid polymer electrolyte membrane
Py13TFSI ionic liquid, LiTFSI, PUA, and 5 wt% ethylene glycol dimethacrylate (EGDMA) as cross-linking agent to promote the formation of covalent bonds during polymer chains were mixed with a certain mass ratio according to the each experiment. Then, the solution A was formed by heating for 1 h at 60~70 °C. Another solution B was prepared by mixing proper MMA and 0.6 wt% benzoyl peroxide (BPO) for 20 min. The prodromic solution was obtained by mixing the above A and B solutions for 20-min stirring in the DMF solvent. Finally, IL-PE membranes were achieved by UV 6-min cross-linking of the curable solution on the glass template.
Characterization of electrolyte membranes
The scanning electron microscope (SEM, FEI Sirion, Philips) and the atomic force microscope (AFM, Dimension Icon, Bruker) were used to characterize the surface morphology of the ionic liquid polymer electrolyte membrane.
The stainless steel/electrolyte/stainless steel system (The electrodes area was 0.785 cm2 of SS/IL-PE/SS.) was assembled to test the conductivity of the electrolytes by electrochemical impedance spectroscopy (EIS) using CHI760E electrochemical workstation with a frequency range of 0.01 Hz–100 kHz between 20 and 80 °C. The conductivity was evaluated by using the following equation.
Where σ is the conductivity of electrolytes, R b is the bulk resistance, and S and d are the area and thickness of the electrolyte films, respectively.
In order to examine the ionic conductivities of the electrolytes, the Li/electrolyte membrane/Li was assembled and tested by electrochemical impedance spectroscopy and chronoamperometry using CHI760E electrochemical workstation for electrochemical cells. Li transference number (t Li+) of the electrolytes was estimated according to a Bruce-Vincent-Evans method [7]. The t Li+ was evaluated by using the following equation.
Where I o is the initial current, I ss is the steady-state current, △V is the polarization voltage (10 mV), and R 0 and R ss are the electrode/electrolyte interfacial resistances measured before and after the direct current polarization, respectively.
Linear sweep voltammetry (LSV) was used to test the electrochemical stability window of the ionic liquid polymer electrolytes. SS/electrolyte/Li cell was assembled and tested using CHI760E electrochemical workstation at 20 °C and a scan rate of 2 mV s−1 with the range of − 1~7 V.
Battery test
The LiFePO4/electrolyte/Li cells were assembled in an Ar-filled dry glove box. For preparing LiFePO4 electrode, LiFePO4 as active material, acetylene black (AB) as conductive agent, and polyvinylidene fluoride (PVDF) as binding agent with a weight ratio of 85:10:5 were mixed in N-methyl-2-pylorridone (NMP) as the organic solvent. The homogeneous solution after stirring 24 h was cast on an aluminum foil and heated for 12 h at 120 °C in a vacuum drying oven.
The cyclic voltammetry (CV) tests were performed by using CHI760E electrochemical workstation at 20 °C and the scanning rate of 0.1 mV s−1 of voltage range of 2.5~4.2 V. The charge-discharge cycling performances of the cells were measured on a Land battery test system (CT20001A) at room temperature.
Results and discussion
Morphology of electrolyte films
Figure 1 shows two SEM images of different samples for the microstructures. Figure 1a is the structure of the Py13TFSI/LiTIFSI/PUA ionic liquid polymer electrolyte membrane. It can be observed in Fig. 1a that the Py13TFSI/LiTIFSI/PUA ionic liquid polymer electrolyte membrane is aligned to be dense and tight chains without the hole, so the Py13TFSI/LiTIFSI/PUA ionic liquid polymer electrolyte membrane is not conductive to the intercalation/deintercalation behavior of Li ion. As shown in Fig. 1b, the structure of the membrane changes obviously after MMA is added. The segments of PMMA, PUA, and Py13TFSI are mutual cross-linking and finally present a plicated state. The path of Li+ is a circulation process of complexation and complex dissociation on the amorphous fraction of polymer. It is good for the intercalation/deintercalation behavior of Li ion. The mechanical properties of membranes are promoted because of PMMA supporting. Therefore, Py 13TFSI/PUA/PMMA ionic liquid polymer electrolyte is better for the membrane.
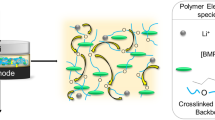
The chemical structure of Py13TFSI ionic liquid is given in Fig. 2a. A graphic image of the Py13TFSI + LiTFSI + PUA + PMMA complex ionic liquid polymer membrane is given in Fig. 2b. The IL-PE is highly transparent, uniform, and soft even when is 180° bended. As shown in Fig. 2c of AFM, a lot of pores of nanometer size homogeneously and densely emerge on the surface of the complex polymer membrane. The pores can provide excellent condition for intercalation/deintercalation behavior of Li ion in membrane.
Ionic transport properties
The conductivity of the gel polymer electrolytes is calculated according to Eq. (2), and the results are listed in Table 1. The conductivity of 1.37 × 10−3 S cm−1 at room temperature can completely satisfy the service condition of the lithium-ion batteries. The influence of temperature on the ionic conductivity of electrolytes with different mass fraction of Py13TFSI at the temperature of 20, 40, 60, and 80 °C is shown in Fig. 3. The relationship of logσ and 1000/T conforms to typical Arrhenius conductive behavior. The activation energy of the IL-PE membranes with 15, 18, and 21 wt% Py13TFSI according to the Vogel-Fulcher-Tammann (VFT) are 19.647, 18.201, and 20.329 kJ mol−1, respectively. The activation energy decreases with the increase of Py13TFSI content presumably due to –F of the Py13TFSI chains as an electron donor coupled with Li+ ion which benefits to transfer Li+. However, the activation energy decreases when the Py13TFSI content is more than 18 wt%. The reason is probably the increase of the space steric hindrance due to the Py13TFSI volume.
Lithium-ion transference number (t Li+) is obtained by electrochemical impedance spectroscopy and chronoamperometry method. Figure 4 shows impedance behavior of the ionic liquid and gel polymer electrolyte membrane with 18 wt% Py13TFSI ionic liquid at 20 °C. The impedance spectra are composed of semi-circles at the highest frequency and a straight line of Warburg impedance at the low frequency. The bulk electrolyte resistance (R b) as the resistance of gel polymer electrolyte membrane and electrode/electrolyte interfacial resistance (R f) as the resistance of the passive film on the lithium make up a semi-circle [30]. The initial electrode/electrolyte interfacial resistance R o and the electrode/electrolyte interfacial resistance R ss after polarization are obtained in Fig. 4a, the initial polarization current I o (1.009 × 10−5 A) and the steady-state current I ss (2.171 × 10−6 A) are observed in Fig. 4b, and △V is the applied potential difference (10 mV). The ion transference number is 0.22 according to Eq. (3).
Electrochemical stability
The electrochemical stability of electrolyte is very important for the practical application of batteries, especially for the high voltage system of the lithium battery because the decomposition of the electrolyte may lead to security issues. Linear sweep voltammetry (LSV) has been used in this study in order to characterize the stability of the IL-PE membrane. The linear sweep voltammetry of Li/IL-PE/SS (the stainless steel) at ambient temperature is shown in Fig. 5. Obviously, IL-PE based on Py13TFSI/PUA/PMMA manifests an anodic stability about 4.8 V (vs. Li+/Li) and a reduction potential about 0 V (vs. Li+/Li). Therefore, Py13TFSI PUA/PMMA polymer electrolyte membrane has electrochemical stability window of about 4.8 V (vs. Li+/Li) at room temperature. The Py13TFSI/LiTFSI/PUA/PMMA polymer electrolyte membrane has enough electrochemical stability as an electrolyte material in Li/IL-PE/LiFePO4 half-cells.
Interface impedance
When the anode or the cathode is contacted with the electrolyte, a passive film that is the solid electrolyte interphase (SEI) film will produce in the charge-discharge process of lithium-ion batteries. SEI film is not conductive, but it can allow lithium-ion through. During intercalation/deintercalation of the lithium ion, some lithium ions remain in the SEI film to produce lithium dendrites. This will not only prevent the transferring of lithium ions to increase the interface impedance of the electrode but also lead to microshort circuit within the battery if the lithium dendrite continues to increase, and seriously, even explosion is raised from the direct contact of the positive and the negative electrodes by piercing the membrane. Therefore, interface impedance characteristic plays an important role in lithium-ion battery. EIS spectra after the different cycles of Li/IL-PE/LiFePO4 cell are given in Fig. 6. The resistance of the battery after 0, 10, and 20 cycles is 1985, 8956, and 13,648 Ω, respectively, indicating the increase with the number of cycles increasing. The change in resistance corresponds to the formation of the passivation film (SEI) at the electrolyte/electrode interface. The interfacial resistance is less increased in 10–20 cycles than 0–10 cycles, suggesting that the formation and decomposition of SEI film are in tendency of dynamic equilibrium to reach a stable state. The ionic liquid polymer electrolyte membrane has better compatibility with LiFePO4 electrode and Li electrode. Therefore, the ionic liquid polymer batteries are expected to exhibit good cycle and rate performance.
Cycle performance for the Li/LiFePO4 cell
Figue 7 shows the cyclic voltammetry of Li/IL-PE/LiFePO4 cells. The voltage of the first-time intercalation/deintercalation of lithium ions occurs at 3.82 and 3.21 V, respectively, while the voltages in the latter cycles are 3.75 and 3.12 V, respectively. The SEI film is being formed at the first cycle and tread to steady at the third cycle and the fourth cycle. Thus, the ionic liquid polymer electrolyte membrane has a good compatibility with the LiFePO4 cathode and the Li anode.
Figure 8a shows voltage vs. specific capacity curves of the Li/IL-PE/LiFePO4 cells during charge-discharge process at room temperature. The initial charging platform of Li/IL-PE/LiFePO4 half-cell is about 3.60 V, and the discharging platform is about 3.28 V. After the fifth cycle, the charge-discharge potential polarization gradually stabilizes with the number of cycles increasing, which leads to the steady state of the cell internal resistance. The first charge and discharge capacity of the cell is 172.7 and 151.9 mAh g−1, respectively. However, the coulombic efficiency is only 87.9%, probably because the passivation film has not been completely formed.
The charge and discharge capacities and coulombic efficiency of Li/IL-PE/LiFePO4 half-cells at 0.5 C in the range of 2.6~4.2 V are shown in Fig. 8b. The discharge capacity of the Li/IL-PE/LiFePO4 half-cell is 151.9 mAh g−1, and the discharge capacity is maintained at 131.9 mAh g−1 after 80 cycles, illustrating better the charge and discharge stability. With the increase of the cycle number, the coulombic efficiency is also increased to 95.5% of 80 cycles although the first coulombic efficiency is only 87.9% since the SEI film may not be well formed. The good cycle performance of the battery including the ionic liquid electrolyte excellent thermal stability, electrochemical stability, and electrical conductivity is promising.
Conclusions
In this paper, a series of tests on the Py13TFSI/LiTFSI/PUA/PMMA ionic liquid polymer electrolyte membrane and the Li/IL-PE/LiFePO4 half-cell were carried out and analyzed. The structure and surface morphology of the membrane can be seen from the scanning electron microscopy (SEM) and atomic force microscopy (AFM), from which they can provide favorable conditions for Li+ intercalation/deintercalation. An electrochemical stabilization window of about 4.8 V was obtained, and the electrolyte membrane had sufficient electrochemical stability to act as an electrolyte material in the Li/IL-PE/LiFePO4 half-cell. The Li/IL-PE/Li cell’s transference number was calculated to be 0.22 according to the electrochemical impedance spectroscopy (EIS) and the polarization current. It was found that the ionic liquid polymer electrolyte membrane had a good compatibility with LiFePO4 electrode and Li electrode; the charge and discharge performance of Li-ion battery was studied. It was found that Li/IL-PE/LiFePO4 half-cells had good electrochemical stability and cycling performance. Therefore, the Py13TFSI/LiTFSI/PUA/PMMA type ionic liquid polymer electrolyte membranes were promising in lithium-ion battery.
References
Safa M, Chamaani A, Chawla N, El-Zahab B (2016) Polymeric ionic liquid gel electrolyte for room temperature lithium battery applications. Electrochim Acta 213:587–593
Bielejewski M, Lapinski A (2017) Molecular interactions in high conductive gel electrolytes based on low molecular weight gelator. J Colloid Interface Sci 490:279–286
Zhu Y, Yang Y, Fu L, Wu Y (2017) A porous gel-type composite membrane reinforced by nonwoven: promising polymer electrolyte with high performance for sodium ion batteries. Electrochim Acta 224:405–411
Gong S, Huang Y, Cao H, Lin YH, Li Y, Tang SH, Wang MS, Li X (2016) A green and environment-friendly gel polymer electrolyte with higher performances based on the natural matrix of lignin. J Power Sources 307:624–633
Köse H, Dombaycıoğlu S, Aydın AO, Akbulut H (2016) Production and characterization of free-standing ZnO/SnO2/MWCNT ternary nanocomposite Li-ion battery anode. Int J Hydrogen Energ 41:9924–9932
Pişkin B, Aydinol MK (2016) Development and characterization of layered li(NixMnyCo1−x−y)O2 cathode materials for lithium ion batteries, Int. J. Hydrogen Energ 41:9952–9955
Kang DW, Kim JK (2016) Characterization of fibrous gel polymer electrolyte for lithium polymer batteries with enhanced electrochemical properties. J Electroanal Chem 775:37–42
Karuppasamy K, Reddy PA, Srinivas G, Tewari A, Sharm R, Shajan XS, Gupta D (2016) Electrochemical and cycling performances of novel nonafluorobutanesulfonate (nonaflate) ionic liquid based ternary gel polymer electrolyte membranes for rechargeable lithium ion batteries. J Membrane Sci 514:350–357
Chen T, Liao Y, Wang X, Luo X, Li X, Li W (2016) Investigation on high-safety lithium ion battery using polyethylene supported poly(methyl methacrylate-acrylonitrile-butyl acrylate) copolymer based gel electrolyte. Electrochim Acta 191:923–932
Fasciani C, Panero S, Hassoun J, Scrosati B (2015) Novel configuration of poly(vinylidenedifluoride)-based gel polymer electrolyte for application in lithium-ion batteries. J Power Sources 294:180–186
Li H, Lin CE, Shi JL, Zhu LP (2014) Preparation and characterization of safety PVDF/P (MMA-co-PEGMA) active separators by studying the liquid electrolyte distribution in this kind of membrane. Electrochim Acta 115:317–325
Kovalska E, Kocabas C (2016) Organic electrolytes for graphene-based supercapacitor: liquid, gel or solid. Mater Today Comm 7:155–160
Fasciani C, Panero S, Hassoun J, Scrosati B (2015) Novel configuration of poly(vinylidenedifluoride)-based gel polymer electrolyte for application in lithium-ion batteries. J Power Sources 294:180–186
Zhu YS, Xiao SY, Li MX, Chang Z, Wang FX, Gao J, Wu YP (2015) Natural macromolecule based carboxymethyl cellulose as a gel polymer electrolyte with adjustable porosity for lithium ion batteries. J Power Sources 288:368–375
Choi JA, Yoo JH, Yoon WY, Kim DW (2014) Cycling characteristics of lithium powder polymer cells assembled with cross-linked gel polymer electrolyte. Electrochim Acta 132:1–6
Lim DH, Manuel J, Ahn JH, Kim JK, Jacobsson P, Matic A, Ha JK, Cho KK, Kim KW (2012) Polymer electrolytes based on poly(vinylidene fluoride-co-hexafluoropropylene) nanofibrous membranes containing polymer plasticizers for lithium batteries. Solid State Ionics 225:631–635
Kuo PL, Tsao CH, Hsu CH, Chen ST, Hsu HM (2016) A new strategy for preparing oligomeric ionic liquid gel polymer electrolytes for high-performance and nonflammable lithium ion batteries. J Membrane Sci 499:462–469
Nunes-Pereira J, Lopes AC, Costa CM, Leones R, Silva MM, Lanceros-Méndez S (2012) Porous membrane of montmorillonite/poly(vinylidene fluoride-trifluorethylene) for Li-ion battery separators. Electroanalysis 24:2147–2156
Padmaraj O, Venkateswarlu M, Satyanarayana N (2014) Characterization and electrochemical properties of p(VdF-co-HFP) based electrospun nanocomposite fibrous polymer electrolyte membrane forLithium battery applications. Electroanalysis 26:2373–2379
Appetecchi GB, Montanino M, Zane D, Carewska M, Alessandrini F, Passerini S (2009) Effect of the alkyl group on the synthesis and the electrochemical properties of N-alkyl-N-methyl-pyrrolidinium bis(trifluoromethanesulfonyl)imide ionic liquids. Electrochim Acta 54:1325–1332
Kim JK, Niedzicki L, Scheers J, Shin CR, Lim DH, Wieczorek W, Johansson P, Ahn JH, Matic A, Jacobsson P (2013) Characterization of N-butyl-N-methyl-pyrrolidinium bis(trifluoromethanesulfonyl)imide-based polymer electrolytes for high safety lithium batteries. J Power Sources 224:93–98
Rao M, Geng X, Liao Y, Hu S, Li W (2012) Preparation and performance of gel polymer electrolyte based on electrospun polymer membrane and ionic liquid for lithium ion battery. J Membrane Sci 399-400:37–42
Kimura K, Matsumoto H, Hassoun J, Panero S, Scrosati B, Tominaga Y (2015) A Quaternary poly(ethylene carbonate)-lithium bis(trifluoromethanesulfonyl)imide-ionic liquid-silica fiber composite polymer electrolyte for lithium batteries. Electrochim Acta 175:134–140
Arof AK, Aziz MF, Noor MM, Careem MA, Bandara LRAK, Thotawatthage CA, Rupasinghe WNS, Dissanayake MAKL (2014) Efficiency enhancement by mixed cation effect in dye-sensitized solar cells with a PVdF based gel polymer electrolyte. Int J Hydrogen Energ 39:2929–2935
Kang Y, Cho N, Noh KA, Kim J, Lee C (2005) Improvement on cycling efficiency of lithium by PEO-based surfactants in cross-linked gel polymer electrolyte. J Power Sources 146:171–175
Samad YA, Asghar A, Hashaikeh R (2013) Electrospun cellulose/PEO fiber mats as a solid polymer electrolytes for Li ion batteries. Renew Energy 56:90–95
Polu AR, Rhee HW (2017) Ionic liquid doped PEO-based solid polymer electrolytes for lithium-ion polymer batteries. Int J Hydrogen Energ 42:7212–7219
Osman Z, Ghazali MIM, Othman L, Md Isa KB (2012) AC ionic conductivity and DC polarization method of lithium ion transport in PMMA–LiBF4 gel polymerelectrolytes. Results Phys 2:1–4
Krishna Jyothi N, Ratnam KKV, Murthy PN, Kumar KV (2016) Electrical studies of gel polymer electrolyte based on PAN for electrochemical cell applications. Mater Today: Proc 3:21–30
Isken P, Winter M, Passerini S, Lex-Balducci A (2013) Methacrylate based gel polymer electrolyte for lithium-ion batteries. J Power Sources 225:157–162
Acknowledgements
This work has been supported financially by the Harbin Application Technology Research and Development Projects [grant number 2015RAQXJ030].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, L., Yang, X., Li, J. et al. A novel and shortcut method to prepare ionic liquid gel polymer electrolyte membranes for lithium-ion battery. Ionics 24, 735–741 (2018). https://doi.org/10.1007/s11581-017-2254-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-017-2254-z