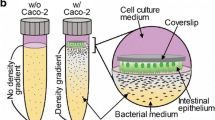
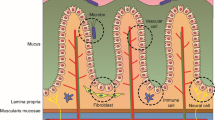
Abstract
Background
Organoid is an in vitro three-dimensional organ-bud that shows realistic microanatomy and physiological relevance. The progress in generating organoids that faithfully recapitulate human in vivo tissue composition has extended organoid applications from being just a basic research tool to a translational platform with a wide range of uses. Study of hostmicrobial interactions relies on model systems to mimic the in vivo infection. Researchers have developed various experimental models in vitro and in vivo to examine the dynamic host-microbial interactions. For some infectious pathogens, model systems are lacking whereas some of the used systems are far from optimal.
Objective
In the present work, we will review the brief history and recent findings using organoids for studying hostmicrobial interactions.
Methods
A systematic literature search was performed using the PubMed search engine. We also shared our data and research contribution to the field.
Results
we summarize the brief history of 3D organoids. We discuss the feasibility of using organoids in studying hostmicrobial interactions, focusing on the development of intestinal organoids and gastric organoids. We highlight the advantage and challenges of the new experimental models. Further, we discuss the future direction in using organoids in studying hostmicrobial interactions and its potential application in biomedical studies.
Conclusion
In combination with genetic, transcriptome and proteomic profiling, both murine- and human-derived organoids have revealed crucial aspects of development, homeostasis and diseases. Specifically, human organoids from susceptible host will be used to test their responses to pathogens, probiotics, and drugs. Organoid system is an exciting tool for studying infectious disease, microbiome, and therapy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aoki-Yoshida A, Saito S, Fukiya S, Aoki R, Takayama Y, Suzuki C, Sonoyama K (2016). Lactobacillus rhamnosus GG increases Toll-like receptor 3 gene expression in murine small intestine ex vivo and in vivo. Benef Microbes, 7(3): 421–429
Arnold J W, Roach J, Azcarate-Peril M A (2016). Emerging technologies for gut microbiome research. Trends Microbiol, 24(11): 887–901
Barrandon Y, Green H (1987). Three clonal types of keratinocyte with different capacities for multiplication. Proc Natl Acad Sci USA, 84(8): 2302–2306
Bartfeld S, Bayram T, van de Wetering M, Huch M, Begthel H, Kujala P, Vries R, Peters P J, Clevers H (2015). In vitro expansion of human gastric epithelial stem cells and their responses to bacterial infection. Gastroenterology, 148(1): 126–136.e6
Bartfeld S, Clevers H (2015). Organoids as model for infectious diseases: Culture of human and murine stomach organoids and microinjection of Helicobacter pylori. J Vis Exp, 43(105):816–818
Bertaux-Skeirik N, Feng R, Schumacher MA, Li J, MaheMM, Engevik A C, Javier J E, Peek RMJr, Ottemann K, Orian-Rousseau V, Boivin G P, Helmrath M A, Zavros Y (2015). CD44 plays a functional role in Helicobacter pylori-induced epithelial cell proliferation. PLoS Pathog, 11(2): e1004663
Crosnier C, Stamataki D, Lewis J (2006). Organizing cell renewal in the intestine: stem cells, signals and combinatorial control. Nat Rev Genet, 7(5): 349–359
D’Aiuto L, Di Maio R, Heath B, Raimondi G, Milosevic J, Watson A M, Bamne M, Parks WT, Yang L, Lin B, Miki T, Mich-Basso J D, Arav-Boger R, Sibille E, Sabunciyan S, Yolken R, Nimgaonkar V (2012). Human induced pluripotent stem cell-derived models to investigate human cytomegalovirus infection in neural cells. PLoS One, 7(11): e49700
Dedhia P H, Bertaux-Skeirik N, Zavros Y, Spence J R (2016). Organoid models of human gastrointestinal development and disease. Gastroenterology, 150(5): 1098–1112
Dingli D, Nowak M A (2006). Cancer biology: infectious tumour cells. Nature, 443(7107): 35–36
Engevik M A, Aihara E, Montrose M H, Shull G E, Hassett D J, Worrell R T (2013). Loss of NHE3 alters gut microbiota composition and influences Bacteroides thetaiotaomicron growth. Am J Physiol Gastrointest Liver Physiol, 305(10): G697–G711
Ettayebi K, Crawford S E, Murakami K, Broughman J R, Karandikar U, Tenge V R, Neill F H, Blutt S E, Zeng X L, Qu L, Kou B, Opekun A R, Burrin D, Graham D Y, Ramani S, Atmar R L, Estes M K (2016). Replication of human noroviruses in stem cell-derived human enteroids. Science, 353(6306): 1387–1393
Fang S B, Schüller S, Phillips A D (2013). Human intestinal in vitro organ culture as a model for investigation of Bacteriae-host interactions. J Exp Clin Med, 5(2): 43–50
Fatehullah A, Tan S H, Barker N (2016). Organoids as an in vitro model of human development and disease. Nat Cell Biol, 18(3): 246–254
Finkbeiner S R, Zeng X L, Utama B, Atmar R L, Shroyer N F, Estes MK (2012). Stem cell-derived human intestinal organoids as an infection model for rotaviruses. MBio, 3(4): e00159–e12
Forbester J L, Goulding D, et al (2014). Intestinal organoids are a novel system to study Salmonella enterica Serovar Typhimurium interaction with the intestinal epithelial barrier. Immunology, 143: 111–112
Forbester J L, Goulding D, Vallier L, Hannan N, Hale C, Pickard D, Mukhopadhyay S, Dougan G (2015). Interaction of Salmonella enterica Serovar Typhimurium with intestinal organoids derived from human induced pluripotent stem cells. Infect Immun, 83(7): 2926–2934
Foulke-Abel J, In J, Kovbasnjuk O, Zachos N C, Ettayebi K, Blutt S E, Hyser J M, Zeng X L, Crawford S E, Broughman J R, Estes M K, Donowitz M (2014). Human enteroids as an ex-vivo model of hostpathogen interactions in the gastrointestinal tract. Exp Biol Med (Maywood), 239(9): 1124–1134
Garcez P P, Loiola E C, Madeiro da Costa R, Higa L M, Trindade P, Delvecchio R, Nascimento J M, Brindeiro R, Tanuri A, Rehen S K (2016). Zika virus impairs growth in human neurospheres and brain organoids. Science, 352(6287): 816–818
Gjorevski N, Sachs N, Manfrin A, Giger S, Bragina M E, Ordóñez-Morán P, Clevers H, Lutolf M P (2016). Designer matrices for intestinal stem cell and organoid culture. Nature, 539(7630): 560–564
Harrison R G (1907). Observations on the living developing fiber. Proc Soc Exp Biol Med, 4(1): 140–143
Heuberger J, Kosel F, Qi J, Grossmann K S, Rajewsky K, Birchmeier W (2014). Shp2/MAPK signaling controls goblet/paneth cell fate decisions in the intestine. Proc Natl Acad Sci USA, 111(9): 3472–3477
Hilleman M R (1990). History, precedent, and progress in the development of mammalian cell culture systems for preparing vaccines: safety considerations revisited. J Med Virol, 31(1): 5–12
Huang G, Ye S, Zhou X, Liu D, Ying Q L (2015a). Molecular basis of embryonic stem cell self-renewal: from signaling pathways to pluripotency network. Cell Mol Life Sci, 72(9): 1741–1757
Huang J Y, Sweeney E G, Sigal M, Zhang H C, Remington S J, Cantrell M A, Kuo C J, Guillemin K, Amieva M R (2015b). Chemodetection and destruction of host urea allows Helicobacter pylori to locate the epithelium. Cell Host Microbe, 18(2): 147–156
Huch M, Koo B K (2015). Modeling mouse and human development using organoid cultures. Development, 142(18): 3113–3125
In J G, Foulke-Abel J, Estes MK, Zachos N C, Kovbasnjuk O, Donowitz M (2016). Human mini-guts: new insights into intestinal physiology and host-pathogen interactions. Nat Rev Gastroenterol Hepatol, 13(11): 633–642
Jung P, Sato T, Merlos-Suárez A, Barriga F M, Iglesias M, Rossell D, Auer H, Gallardo M, Blasco M A, Sancho E, Clevers H, Batlle E (2011). Isolation and in vitro expansion of human colonic stem cells. Nat Med, 17(10): 1225–1227
Klotz C, Aebischer T, Seeber F (2012). Stem cell-derived cell cultures and organoids for protozoan parasite propagation and studying hostparasite interaction. Int J Med Microbiol, 302(4–5): 203–209
Kristin W, Weitz J, et al (2016). Organoids as model systems for gastrointestinal diseases: tissue engineering meets. Curr Pathobiol Rep, 4(1): 1–9
Leslie J L, Huang S, Opp J S, Nagy M S, Kobayashi M, Young V B, Spence J R (2015). Persistence and toxin production by Clostridium difficile within human intestinal organoids result in disruption of epithelial paracellular barrier function. Infect Immun, 83(1): 138–145
Mahe M M, Aihara E, Schumacher M A, Zavros Y, Montrose M H, Helmrath M A, Sato T, Shroyer N F (2013). Establishment of gastrointestinal epithelial organoids. Curr Protoc Mouse Biol, 3(4): 217–240
Mahe M M, Sundaram N, Watson C L, Shroyer N F, Helmrath M A (2015). Establishment of human epithelial enteroids and colonoids from whole tissue and biopsy. J Vis Exp, (97): e52483-e52483
McCracken K W, Catá E M, Crawford C M, Sinagoga K L, Schumacher M, Rockich B E, Tsai Y H, Mayhew C N, Spence J R, Zavros Y, Wells J M (2014). Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature, 516(7531): 400–404
Miyoshi H, Stappenbeck T S (2013). In vitro expansion and genetic modification of gastrointestinal stem cells in spheroid culture. Nat Protoc, 8(12): 2471–2482
Ng S, Schwartz R E, March S, Galstian A, Gural N, Shan J, Prabhu M, Mota M M, Bhatia S N (2015). Human iPSC-derived hepatocyte-like cells support Plasmodium liver-stage infection in vitro. Stem Cell Rep, 4(3): 348–359
Ootani A, Li X, Sangiorgi E, Ho Q T, Ueno H, Toda S, Sugihara H, Fujimoto K, Weissman I L, Capecchi MR, Kuo C J (2009). Sustained in vitro intestinal epithelial culture within a Wnt-dependent stem cell niche. Nat Med, 15(6): 701–706
Penkert R R, Kalejta R F (2013). Human embryonic stem cell lines model experimental human cytomegalovirus latency. MBio, 4(3): e00298–e13
Roelandt P, Obeid S, Paeshuyse J, Vanhove J, Van Lommel A, Nahmias Y, Nevens F, Neyts J, Verfaillie CM (2012). Human pluripotent stem cell-derived hepatocytes support complete replication of hepatitis C virus. J Hepatol, 57(2): 246–251
Salama N R, Hartung ML, Müller A (2013). Life in the human stomach: persistence strategies of the bacterial pathogen Helicobacter pylori. Nat Rev Microbiol, 11(6): 385–399
Sato T, Stange D E, Ferrante M, Vries R G, Van Es J H, Van den Brink S, Van Houdt W J, Pronk A, Van Gorp J, Siersema P D, Clevers H (2011a). Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology, 141(5): 1762–1772
Sato T, van Es J H, Snippert H J, Stange D E, Vries R G, van den Born M, Barker N, Shroyer N F, van de Wetering M, Clevers H (2011b). Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature, 469(7330): 415–418
Sato T, Vries R G, Snippert H J, van de Wetering M, Barker N, Stange D E, van Es J H, Abo A, Kujala P, Peters P J, Clevers H (2009). Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature, 459(7244): 262–265
Saxena K, Blutt S E, Ettayebi K, Zeng X L, Broughman J R, Crawford S E, Karandikar U C, Sastri N P, Conner M E, Opekun A R, Graham D Y, Qureshi W, Sherman V, Foulke-Abel J, In J, Kovbasnjuk O, Zachos N C, Donowitz M, Estes M K (2015). Human intestinal enteroids: a new model to study human rotavirus infection, host restriction, and pathophysiology. J Virol, 90(1): 43–56
Schlaermann P, Toelle B, Berger H, Schmidt S C, Glanemann M, Ordemann J, Bartfeld S, Mollenkopf H J, Meyer T F (2016). A novel human gastric primary cell culture system for modelling Helicobacter pylori infection in vitro. Gut, 65(2): 202–213
Schumacher M A, Feng R, Aihara E, Engevik A C, Montrose M H, Ottemann K M, Zavros Y (2015). Helicobacter pylori-induced Sonic Hedgehog expression is regulated by NFkB pathway activation: the use of a novel in vitro model to study epithelial response to infection. Helicobacter, 20(1): 19–28
Schwank G, Koo B K, Sasselli V, Dekkers J F, Heo I, Demircan T, Sasaki N, Boymans S, Cuppen E, van der Ent C K, Nieuwenhuis E E, Beekman J M, Clevers H (2013). Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell, 13(6): 653–658
Schwartz R E, Trehan K, Andrus L, Sheahan T P, Ploss A, Duncan S A, Rice C M, Bhatia S N (2012). Modeling hepatitis C virus infection using human induced pluripotent stem cells. Proc Natl Acad Sci USA, 109(7): 2544–2548
Shlomai A, Schwartz R E, Ramanan V, Bhatta A, de Jong Y P, Bhatia S N, Rice C M (2014). Modeling host interactions with hepatitis B virus using primary and induced pluripotent stem cell-derived hepatocellular systems. Proc Natl Acad Sci USA, 111(33): 12193–12198
Sigal M, RothenbergME, Logan C Y, Lee J Y, Honaker R W, Cooper R L, Passarelli B, Camorlinga M, Bouley D M, Alvarez G, Nusse R, Torres J, Amieva M R (2015). Helicobacter pylori activates and expands Lgr5(+) stem cells through direct colonization of the gastric glands. Gastroenterology, 148(7): 1392–404.e21
Spence J R, Mayhew C N, Rankin S A, Kuhar M F, Vallance J E, Tolle K, Hoskins E E, Kalinichenko V V, Wells S I, Zorn A M, Shroyer N F, Wells J M (2011). Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature, 470(7332): 105–109
Unsworth B R, Lelkes P I (1998). Growing tissues in microgravity. Nat Med, 4(8): 901–907
Van Dussen K L, Marinshaw J M, Shaikh N, Miyoshi H, Moon C, Tarr P I, Ciorba M A, Stappenbeck T S (2015). Development of an enhanced human gastrointestinal epithelial culture system to facilitate patient-based assays. Gut, 64(6): 911–920
Wang X, Yamamoto Y, Wilson L H, Zhang T, Howitt B E, Farrow M A, Kern F, Ning G, Hong Y, Khor C C, Chevalier B, Bertrand D, Wu L, Nagarajan N, Sylvester F A, Hyams J S, Devers T, Bronson R, Lacy D B, Ho K Y, Crum C P, McKeon F, Xian W (2015). Cloning and variation of ground state intestinal stem cells. Nature, 522(7555): 173–178
Wilson S S, Tocchi A, Holly MK, Parks WC, Smith J G (2015). A small intestinal organoid model of non-invasive enteric pathogen-epithelial cell interactions. Mucosal Immunol, 8(2): 352–361
Wroblewski L E, Peek R M Jr, Wilson K T (2010). Helicobacter pylori and gastric cancer: factors that modulate disease risk. Clin Microbiol Rev, 23(4): 713–739
Wu X, Robotham J M, Lee E, Dalton S, Kneteman N M, Gilbert D M, Tang H (2012). Productive hepatitis C virus infection of stem cellderived hepatocytes reveals a critical transition to viral permissiveness during differentiation. PLoS Pathog, 8(4): e1002617
Yin Y, Bijvelds M, Dang W, Xu L, van der Eijk A A, Knipping K, Tuysuz N, Dekkers J F, Wang Y, de Jonge J, Sprengers D, van der Laan L J, Beekman J M, Ten Berge D, Metselaar H J, de Jonge H, Koopmans M P, Peppelenbosch M P, Pan Q (2015). Modeling rotavirus infection and antiviral therapy using primary intestinal organoids. Antiviral Res, 123: 120–131
Yoshida T, Takayama K, Kondoh M, Sakurai F, Tani H, Sakamoto N, Matsuura Y, Mizuguchi H, Yagi K (2011). Use of human hepatocytelike cells derived from induced pluripotent stem cells as a model for hepatocytes in hepatitis C virus infection. Biochem Biophys Res Commun, 416(1–2): 119–124
Yui S, Nakamura T, Sato T, Nemoto Y, Mizutani T, Zheng X, Ichinose S, Nagaishi T, Okamoto R, Tsuchiya K, Clevers H, Watanabe M (2012). Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5+ stem cell. Nat Med, 18(4): 618–623
Zhang Y G, Wu S, Xia Y, Sun J (2014). Salmonella-infected cryptderived intestinal organoid culture system for host-bacterial interactions. Physiol Rep, 2(9): e12147
Acknowledgements
This work was supported by the NIDDK 1R01DK105118-01 and the UIC Cancer Center to Jun Sun.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, J. Intestinal organoid as an in vitro model in studying host-microbial interactions. Front. Biol. 12, 94–102 (2017). https://doi.org/10.1007/s11515-017-1444-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-017-1444-4