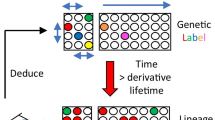
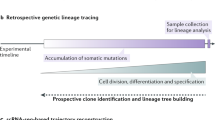
Abstract
Regeneration of many cell types found in adult organs relies upon the presence of relatively small pools of undifferentiated stem cells. Initial studies that attempted to isolate stem cells and propagate them in vitro have been complemented by analysis of stem cells in their endogenous tissues where they are subject to a variety of regulatory cues. This has been facilitated by the advent of new methods for lineage tracing and genetic manipulation of stem cells and their associated niche cells. The picture that is emerging is that different stem cell populations utilize diverse processes to ensure maintenance of the stem cell pool accompanied by production of cells committed to regenerate differentiated cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abud H E, Lock P, Heath J K (2004). Efficient gene transfer into the epithelial cell layer of embryonic mouse intestine using low-voltage electroporation. Gastroenterology, 126(7): 1779–1787
Abud H E, Watson N, Heath J K (2005). Growth of intestinal epithelium in organ culture is dependent on EGF signalling. Exp Cell Res, 303(2): 252–262
Amoyel M, Sanny J, Burel M, Bach E A (2013). Hedgehog is required for CySC self-renewal but does not contribute to the GSC niche in the Drosophila testis. Development, 140(1): 56–65
Barker N, van Es J H, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters P J, Clevers H (2007). Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature, 449(7165): 1003–1007
Becker A J, McCULLOCH E A, Till J E (1963). Cytological demonstration of the clonal nature of spleen colonies derived from transplanted mouse marrow cells. Nature, 197(4866): 452–454
Boyer L A, Plath K, Zeitlinger J, Brambrink T, Medeiros L A, Lee T I, Levine S S, Wernig M, Tajonar A, Ray M K, Bell G W, Otte A P, Vidal M, Gifford D K, Young R A, Jaenisch R (2006). Polycomb complexes repress developmental regulators in murine embryonic stem cells. Nature, 441(7091): 349–353
Boyle M, Wong C, Rocha M, Jones D L (2007). Decline in self-renewal factors contributes to aging of the stem cell niche in the Drosophila testis. Cell Stem Cell, 1(4): 470–478
Brand A H, Perrimon N (1993). Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development, 118(2): 401–415
Bunt S M, Hime G R (2004). Ectopic activation of Dpp signalling in the male Drosophila germline inhibits germ cell differentiation. Genesis, 39(2): 84–93
Bunt S M, Monk A C, Siddall N A, Johnston N L, Hime G R (2012). GAL4 enhancer traps that can be used to drive gene expression in developing Drosophila spermatocytes. Genesis, 50(12): 914–920
Carmon K S, Lin Q, Gong X, Thomas A, Liu Q (2012). LGR5 interacts and cointernalizes with Wnt receptors to modulate Wnt/β-catenin signaling. Mol Cell Biol, 32(11): 2054–2064
Cheasley D, Pereira L, Lightowler S, Vincan E, Malaterre J, Ramsay R G (2011). Myb controls intestinal stem cell genes and self-renewal. Stem Cells, 29(12): 2042–2050
Cheng H, Leblond C P (1974). Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine. V. Unitarian Theory of the origin of the four epithelial cell types. Am J Anat, 141(4): 537–561
Cheng J, Tiyaboonchai A, Yamashita Y M, Hunt A J (2011). Asymmetric division of cyst stem cells in Drosophila testis is ensured by anaphase spindle repositioning. Development, 138(5): 831–837
Clevers H (2013). The intestinal crypt, a prototype stem cell compartment. Cell, 154(2): 274–284
Conklin E G (1905). The organization and cell lineage of the ascidian egg. J Acad Nat Sci Phila, 12: 1–19
Coulombel L (2004). Identification of hematopoietic stem/progenitor cells: strength and drawbacks of functional assays. Oncogene, 23(43): 7210–7222
de Lau W, Barker N, Clevers H (2007). WNT signaling in the normal intestine and colorectal cancer. Front Biosci, 12(1): 471–491
Evans C J, Olson J M, Ngo K T, Kim E, Lee N E, Kuoy E, Patananan A N, Sitz D, Tran P, Do M T, Yackle K, Cespedes A, Hartenstein V, Call G B, Banerjee U (2009). G-TRACE: rapid Gal4-based cell lineage analysis in Drosophila. Nat Methods, 6(8): 603–605
Fre S, Huyghe M, Mourikis P, Robine S, Louvard D, Artavanis-Tsakonas S (2005). Notch signals control the fate of immature progenitor cells in the intestine. Nature, 435(7044): 964–968
Fuller M T (1993). Spermatogenesis. The Development of Drosophila melanogaster. C. S. Harbour. NY, Cold Spring Harbour Laboratory Press: 71–147
Haramis A P, Begthel H, van den Born M, van Es J, Jonkheer S, Offerhaus G J, Clevers H (2004). De novo crypt formation and juvenile polyposis on BMP inhibition in mouse intestine. Science, 303(5664): 1684–1686
Hardy R W, Tokuyasu K T, Lindsley D L, Garavito M (1979). The germinal proliferation center in the testis of Drosophila melanogaster. J Ultrastruct Res, 69(2): 180–190
He X C, Zhang J, Tong W G, Tawfik O, Ross J, Scoville D H, Tian Q, Zeng X, He X, Wiedemann L M, Mishina Y, Li L (2004). BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt-beta-catenin signaling. Nat Genet, 36(10): 1117–1121
Hime G R, Loveland K L, Abud H E (2007). Drosophila spermatogenesis: insights into testicular cancer. Int J Androl, 30(4): 265–274, discussion 274
Holmberg J, Genander M, Halford M M, Annerén C, Sondell M, Chumley M J, Silvany R E, Henkemeyer M, Frisén J (2006). EphB receptors coordinate migration and proliferation in the intestinal stem cell niche. Cell, 125(6): 1151–1163
Horvay K, Casagranda F, Gany A, Hime G R, Abud H E (2011). Wnt signaling regulates Snai1 expression and cellular localization in the mouse intestinal epithelial stem cell niche. Stem Cells Dev, 20(4): 737–745
Insco M L, Leon A, Tam C H, McKearin D M, Fuller M T (2009). Accumulation of a differentiation regulator specifies transit amplifying division number in an adult stem cell lineage. Proc Natl Acad Sci USA, 106(52): 22311–22316
Jaks V, Barker N, Kasper M, van Es J H, Snippert H J, Clevers H, Toftgård R (2008). Lgr5 marks cycling, yet long-lived, hair follicle stem cells. Nat Genet, 40(11): 1291–1299
Kaur P, Li A, Redvers R, Bertoncello I (2004). Keratinocyte stem cell assays: an evolving science. J Investig Dermatol Symp Proc, 9(3): 238–247
Kawase E, Wong M D, Ding B C, Xie T (2004). Gbb/Bmp signaling is essential for maintaining germline stem cells and for repressing bam transcription in the Drosophila testis. Development, 131(6): 1365–1375
Kiger A A, Jones D L, Schulz C, Rogers M B, Fuller M T (2001). Stem cell self-renewal specified by JAK-STAT activation in response to a support cell cue. Science, 294(5551): 2542–2545
Kiger A A, White-Cooper H, Fuller M T (2000). Somatic support cells restrict germline stem cell self-renewal and promote differentiation. Nature, 407(6805): 750–754
Kretzschmar K, Watt F M (2012). Lineage tracing. Cell, 148(1–2): 33–45
Leatherman J L, DiNardo S (2008). Zfh-1 controls somatic stem cell selfrenewal in the Drosophila testis, and non-autonomously influences germline stem cell self-renewal. Dev Biol, 319(2): 548
Leatherman J L, Dinardo S (2010). Germline self-renewal requires cyst stem cells and stat regulates niche adhesion in Drosophila testes. Nat Cell Biol, 12(8): 806–811
Lee T I, Jenner R G, Boyer L A, Guenther M G, Levine S S, Kumar RM, Chevalier B, Johnstone S E, Cole M F, Isono K, Koseki H, Fuchikami T, Abe K, Murray H L, Zucker J P, Yuan B, Bell G W, Herbolsheimer E, Hannett N M, Sun K, Odom D T, Otte A P, Volkert T L, Bartel D P, Melton D A, Gifford D K, Jaenisch R, Young R A (2006). Control of developmental regulators by Polycomb in human embryonic stem cells. Cell, 125(2): 301–313
Li Y, Ma Q, Cherry C M, Matunis E L (2014). Steroid signaling promotes stem cell maintenance in the Drosophila testis. Dev Biol, 394(1): 129–141
Lindsley D L, Tokuyasu K T (1980). Spermatogenesis. In: The genetics and biology of Drosophila. In: Ashburner M, Wright T R F. eds. London, Academic Press, 225–294
Livet J, Weissman T A, Kang H, Draft RW, Lu J, Bennis R A, Sanes J R, Lichtman J W (2007). Transgenic strategies for combinatorial expression of fluorescent proteins in the nervous system. Nature, 450(7166): 56–62
Matunis E, Tran J, Gönczy P, Caldwell K, DiNardo S (1997). punt and schnurri regulate a somatically derived signal that restricts proliferation of committed progenitors in the germline. Development, 124(21): 4383–4391
Maximow A (1909). The lymphocyte as a stem cell common to different blood elements in embryonic development and during the post-fetal life of mammals. Originally in German. Folia Haematol (Frankf), 8: 125–143 (English translation (2009) Cell Ther Transplant, 2001 (2003): 2014–2018)
Monk A C, Siddall N A, Volk T, Fraser B, Quinn LM, McLaughlin E A, Hime G R (2010). HOW is required for stem cell maintenance in the Drosophila testis and for the onset of transit-amplifying divisions. Cell Stem Cell, 6(4): 348–360
Montgomery R K, Carlone D L, Richmond C A, Farilla L, Kranendonk ME, Henderson D E, Baffour-Awuah N Y, Ambruzs DM, Fogli L K, Algra S, Breault D T (2011). Mouse telomerase reverse transcriptase (mTert) expression marks slowly cycling intestinal stem cells. Proc Natl Acad Sci USA, 108(1): 179–184
Morris R J, Liu Y, Marles L, Yang Z, Trempus C, Li S, Lin J S, Sawicki J A, Cotsarelis G (2004). Capturing and profiling adult hair follicle stem cells. Nat Biotechnol, 22(4): 411–417
O’Brien C A, Pollett A, Gallinger S, Dick J E (2007). A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature, 445(7123): 106–110
Oatley J M, Brinster R L (2012). The germline stem cell niche unit in mammalian testes. Physiol Rev, 92(2): 577–595
Ootani A, Li X, Sangiorgi E, Ho Q T, Ueno H, Toda S, Sugihara H, Fujimoto K, Weissman I L, Capecchi MR, Kuo C J (2009). Sustained in vitro intestinal epithelial culture within a Wnt-dependent stem cell niche. Nat Med, 15(6): 701–706
Osawa M, Hanada K, Hamada H, Nakauchi H (1996). Long-term lymphohematopoietic reconstitution by a single CD34-low/negative hematopoietic stem cell. Science, 273(5272): 242–245
Paoli P, Giannoni E, Chiarugi P (2013). Anoikis molecular pathways and its role in cancer progression. Biochim Biophys Acta, 1833(12): 3481–3498
Qian Y, Dominado N, Zoller R, Ng C, Kudyba K, Siddall N A, Hime G R, Schulz C (2014). Ecdysone signaling opposes epidermal growth factor signaling in regulating cyst differentiation in the male gonad of Drosophila melanogaster. Dev Biol, 394(2): 217–227
Reynolds B A, Weiss S (1992). Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science, 255(5052): 1707–1710
Ricci-Vitiani L, Lombardi D G, Pilozzi E, Biffoni M, Todaro M, Peschle C, De Maria R (2007). Identification and expansion of human coloncancer-initiating cells. Nature, 445(7123): 111–115
Rios A C, Fu N Y, Lindeman G J, Visvader J E (2014). In situ identification of bipotent stem cells in the mammary gland. Nature, 506(7488): 322–327
Rothenberg M E, Nusse Y, Kalisky T, Lee J J, Dalerba P, Scheeren F, Lobo N, Kulkarni S, Sim S, Qian D, Beachy P A, Pasricha P J, Quake S R, Clarke M F (2012). Identification of a cKit(+) colonic crypt base secretory cell that supports Lgr5(+) stem cells in mice. Gastroenterology, 142(5): 1195–1205 e1196
Sacco A, Doyonnas R, Kraft P, Vitorovic S, Blau H M (2008). Selfrenewal and expansion of single transplanted muscle stem cells. Nature, 456(7221): 502–506
Sancho E, Batlle E, Clevers H (2003). Live and let die in the intestinal epithelium. Curr Opin Cell Biol, 15(6): 763–770
Sangiorgi E, Capecchi M R (2008). Bmi1 is expressed in vivo in intestinal stem cells. Nat Genet, 40(7): 915–920
Sato T, Clevers H (2013). Growing self-organizing mini-guts from a single intestinal stem cell: mechanism and applications. Science, 340(6137): 1190–1194
Sato T, Stange D E, Ferrante M, Vries R G, Van Es J H, Van den Brink S, Van Houdt W J, Pronk A, Van Gorp J, Siersema P D, Clevers H (2011a). Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology, 141(5): 1762–1772
Sato T, van Es J H, Snippert H J, Stange D E, Vries R G, van den Born M, Barker N, Shroyer N F, van de Wetering M, Clevers H (2011b). Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature, 469(7330): 415–418
Sato T, Vries R G, Snippert H J, van de Wetering M, Barker N, Stange D E, van Es J H, Abo A, Kujala P, Peters P J, Clevers H (2009). Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature, 459(7244): 262–265
Schofield R (1978). The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells, 4(1–2): 7–25
Schulenburg A, Ulrich-Pur H, Thurnher D, Erovic B, Florian S, Sperr W R, Kalhs P, Marian B, Wrba F, Zielinski C C, Valent P (2006). Neoplastic stem cells: a novel therapeutic target in clinical oncology. Cancer, 107(10): 2512–2520
Schulz C, Kiger A A, Tazuke S I, Yamashita Y M, Pantalena-Filho L C, Jones D L, Wood C G, Fuller M T (2004). A misexpression screen reveals effects of bag-of-marbles and TGFβ class signaling on the Drosophila male germ-line stem cell lineage. Genetics, 167(2): 707–723
Schwank G, Andersson-Rolf A, Koo B K, Sasaki N, Clevers H (2013a). Generation of BAC transgenic epithelial organoids. PLoS ONE, 8(10): e76871
Schwank G, Koo B K, Sasselli V, Dekkers J F, Heo I, Demircan T, Sasaki N, Boymans S, Cuppen E, van der Ent C K, Nieuwenhuis E E, Beekman J M, Clevers H (2013b). Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell, 13(6): 653–658
Shackleton M, Vaillant F, Simpson K J, Stingl J, Smyth G K, Asselin-Labat M L, Wu L, Lindeman G J, Visvader J E (2006). Generation of a functional mammary gland from a single stem cell. Nature, 439(7072): 84–88
Shivdasani A A, Ingham P W (2003). Regulation of stem cell maintenance and transit amplifying cell proliferation by tgf-beta signaling in Drosophila spermatogenesis. Curr Biol, 13(23): 2065–2072
Siddall N A, McLaughlin E A, Marriner N L, Hime G R (2006). The RNA-binding protein Musashi is required intrinsically to maintain stem cell identity. Proc Natl Acad Sci USA, 103(22): 8402–8407
Singh S R, Zhen W, Zheng Z, Wang H, Oh SW, Liu W, Zbar B, Schmidt L S, Hou S X (2006). The Drosophila homolog of the human tumor suppressor gene BHD interacts with the JAK-STAT and Dpp signaling pathways in regulating male germline stem cell maintenance. Oncogene, 25(44): 5933–5941
Snippert H J, van der Flier L G, Sato T, van Es J H, van den Born M, Kroon-Veenboer C, Barker N, Klein A M, van Rheenen J, Simons B D, Clevers H (2010). Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell, 143(1): 134–144
Spradling A, Drummond-Barbosa D, Kai T (2001). Stem cells find their niche. Nature, 414(6859): 98–104
Tetteh P W, Farin H F, Clevers H (2014). Plasticity within stem cell hierarchies in mammalian epithelia. Trends Cell Biol, doi: 10.1016/j.tcb.2014.09.003
Tian H, Biehs B, Warming S, Leong K G, Rangell L, Klein O D, de Sauvage F J (2011). A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature, 478(7368): 255–259
Tran J, Brenner T J, DiNardo S (2000). Somatic control over the germline stem cell lineage during Drosophila spermatogenesis. Nature, 407(6805): 754–757
Tulina N, Matunis E (2001). Control of stem cell self-renewal in Drosophila spermatogenesis by JAK-STAT signaling. Science, 294(5551): 2546–2549
van Amerongen R, Bowman A N, Nusse R (2012). Developmental stage and time dictate the fate of Wnt/β-catenin-responsive stem cells in the mammary gland. Cell Stem Cell, 11(3): 387–400
van der Flier L G, Haegebarth A, Stange D E, van de Wetering M, Clevers H (2009). OLFM4 is a robust marker for stem cells in human intestine and marks a subset of colorectal cancer cells. Gastroenterology, 137(1): 15–17
van Es J H, van Gijn M E, Riccio O, van den Born M, Vooijs M, Begthel H, Cozijnsen M, Robine S, Winton D J, Radtke F, Clevers H (2005). Notch/gamma-secretase inhibition turns proliferative cells in intestinal crypts and adenomas into goblet cells. Nature, 435(7044): 959–963
Visvader J E, Stingl J (2014). Mammary stem cells and the differentiation hierarchy: current status and perspectives. Genes Dev, 28(11): 1143–1158
Xie T, Spradling A C (2000). A niche maintaining germ line stem cells in the Drosophila ovary. Science, 290(5490): 328–330
Xu T, Harrison S D (1994). Mosaic analysis using FLP recombinase. Methods Cell Biol, 44: 655–681
Yamashita Y M, Fuller M T (2005). Asymmetric stem cell division and function of the niche in the Drosophila male germ line. Int J Hematol, 82(5): 377–380
Yamashita Y M, Jones D L, Fuller M T (2003). Orientation of asymmetric stem cell division by the APC tumor suppressor and centrosome. Science, 301(5639): 1547–1550
Yui S, Nakamura T, Sato T, Nemoto Y, Mizutani T, Zheng X, Ichinose S, Nagaishi T, Okamoto R, Tsuchiya K, Clevers H, Watanabe M (2012). Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5+ stem cell. Nat Med, 18(4): 618–623
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hime, G.R., Siddall, N., Horvay, K. et al. Analyzing stem cell dynamics: use of cutting edge genetic approaches in model organisms. Front. Biol. 10, 1–10 (2015). https://doi.org/10.1007/s11515-015-1347-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11515-015-1347-1