Abstract
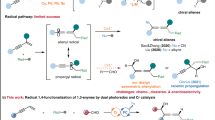
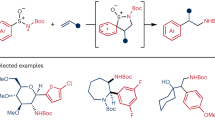
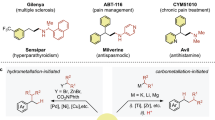
Difunctionalization of alkynes represents a powerful and straightforward approach to the synthesis of complex molecules. However, the radical difunctionalization of alkynes mediated by bifunctional reagents remains challenging and underexplored, despite significant progress having been made in alkene difunctionalization. Here, we report a novel arylsulfonylacetate skeleton in which aryl rings are attached to acetates through SO2, serving as a powerful bifunctional reagent for the alkylarylation of alkynes via vinyl-radical intermediate under photoredox conditions. This modular bifunctional reagent enables the simultaneous incorporation of a wide range of functional groups, including (hetero)aryl ring and alkyl carboxylate into alkynes, resulting in synthetically valuable all-carbon tetrasubstituted alkene derivatives. This transformation is distinguished by its redox-neutral nature, readily accessible starting materials, compatibility with diverse functional groups and its capacity to facilitate convergent synthesis. The utility of this approach was further demonstrated by the late-stage functionalization of complex molecules and the preparation of fluorescent molecules and anti-cancer drugs.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
McDonald RI, Liu G, Stahl SS. Chem Rev, 2011, 111: 2981–3019
Zhang C, Tang C, Jiao N. Chem Soc Rev, 2012, 41: 3464–3484
Wolfe JP. Angew Chem Int Ed, 2012, 51: 10224–10225
Tang S, Liu K, Liu C, Lei A. Chem Soc Rev, 2015, 44: 1070–1082
Huple DB, Ghorpade S, Liu R. Adv Synth Catal, 2016, 358: 1348–1367
Mei H, Yin Z, Liu J, Sun H, Han J. Chin J Chem, 2019, 37: 292–301
Li G, Huo X, Jiang X, Zhang W. Chem Soc Rev, 2020, 49: 2060–2118
Liu W, Kong W. Org Chem Front, 2020, 7: 3941–3955
Ding W, Chai J, Wang C, Wu J, Yoshikai N. J Am Chem Soc, 2020, 142: 8619–8624
Deng JR, Chan WC, Chun-Him Lai N, Yang B, Tsang CS, Chi-Bun Ko B, Lai-Fung Chan S, Wong MK. Chem Sci, 2017, 8: 7537–7544
You Y, Ge S. Angew Chem Int Ed, 2021, 60: 20684–20688
Murai M, Uemura E, Hori S, Takai K. Angew Chem Int Ed, 2017, 56: 5862–5866
Prier CK, Rankic DA, MacMillan DWC. Chem Rev, 2013, 113: 5322–5363
Egami H, Sodeoka M. Angew Chem Int Ed, 2014, 53: 8294–8308
Xie J, Li J, Weingand V, Rudolph M, Hashmi ASK. Chem Eur J, 2016, 22: 12646–12650
Chalotra N, Kumar J, Naqvi T, Shah BA. Chem Commun, 2021, 57: 11285–11300
Huang L, Rudolph M, Rominger F, Hashmi ASK. Angew Chem Int Ed, 2016, 55: 4808–4813
Zhu C, Yue H, Maity B, Atodiresei I, Cavallo L, Rueping M. Nat Catal, 2019, 2: 678–687
Hou J, Ee A, Feng W, Xu JH, Zhao Y, Wu J. J Am Chem Soc, 2018, 140: 5257–5263
Chakrasali P, Kim K, Jung YS, Kim H, Han SB. Org Lett, 2018, 20: 7509–7513
Ni B, Zhang B, Han J, Peng B, Shan Y, Niu T. Org Lett, 2020, 22: 670–674
Yasu Y, Koike T, Akita M. Angew Chem Int Ed, 2012, 51: 9567–9571
Yan M, Kawamata Y, Baran PS. Chem Rev, 2017, 117: 13230–13319
Siu JC, Fu N, Lin S. Acc Chem Res, 2020, 53: 547–560
Dong X, Klein M, Waldvogel SR, Morandi B. Angew Chem Int Ed, 2023, 62: e202213630
Yin G, Mu X, Liu G. Acc Chem Res, 2016, 49: 2413–2423
Sauer GS, Lin S. ACS Catal, 2018, 8: 5175–5187
Giri R, Kc S. J Org Chem, 2018, 83: 3013–3022
Li ZL, Fang GC, Gu QS, Liu XY. Chem Soc Rev, 2020, 49: 32–48
Chemler SR, Bovino MT. ACS Catal, 2013, 3: 1076–1091
Egami H, Sodeoka M. Angew Chem Int Ed, 2014, 53: 8294–8308
Merino E, Nevado C. Chem Soc Rev, 2014, 43: 6598–6608
Chen JR, Yu XY, Xiao WJ. Synthesis, 2015, 47: 604–629
Wu Y, Xiao Y, Yang Y, Song R, Li J. ChemCatChem, 2020, 12: 5312–5329
Piers E. Pure Appl Chem, 1988, 60: 107–114
Huang HM, Bellotti P, Ma J, Dalton T, Glorius F. Nat Rev Chem, 2021, 5: 301–321
Yu X, Meng QY, Daniliuc CG, Studer A. J Am Chem Soc, 2022, 144: 7072–7079
Li Z, Jiao L, Sun Y, He Z, Wei Z, Liao W. Angew Chem Int Ed, 2020, 59: 7266–7270
Link A, Fischer C, Sparr C. Angew Chem Int Ed, 2015, 54: 12163–12166
Erchinger JE, Hoogesteger R, Laskar R, Dutta S, Hümpel C, Rana D, Daniliuc CG, Glorius F. J Am Chem Soc, 2023, 145: 2364–2374
Liao LL, Cao GM, Jiang YX, Jin XH, Hu XL, Chruma JJ, Sun GQ, Gui YY, Yu DG. J Am Chem Soc, 2021, 143: 2812–2821
Li H, Neumann H, Beller M, Wu X. Angew Chem Int Ed, 2014, 53: 3183–3186
Farndon JJ, Young TA, Bower JF. J Am Chem Soc, 2018, 140: 17846–17850
Wu D, Fohn NA, Bode JW. Angew Chem Int Ed, 2019, 58: 11058–11062
Wu Z, Ren R, Zhu C. Angew Chem Int Ed, 2016, 55: 10821–10824
Wu Z, Wang D, Liu Y, Huan L, Zhu C. J Am Chem Soc, 2017, 139: 1388–1391
Wu X, Wang M, Huan L, Wang D, Wang J, Zhu C. Angew Chem Int Ed, 2018, 57: 1640–1644
Tang N, Shao X, Wang M, Wu X, Zhu C. Acta China Chem, 2019, 77: 922–926
Yu J, Wu Z, Zhu C. Angew Chem Int Ed, 2018, 57: 17156–17160
Liu J, Wu S, Yu J, Lu C, Wu Z, Wu X, Xue X, Zhu C. Angew Chem Int Ed, 2020, 59: 8195–8202
Zhang H, Wang M, Wu X, Zhu C. Angew Chem Int Ed, 2021, 60: 3714–3719
Yu J, Zhang X, Wu X, Liu T, Zhang ZQ, Wu J, Zhu C. Chem, 2023, 9: 472–482
Wu X, Zhu C. Acc Chem Res, 2020, 53: 1620–1636
Wu X, Ma Z, Feng T, Zhu C. Chem Soc Rev, 2021, 50: 11577–11613
Monos TM, McAtee RC, Stephenson CRJ. Science, 2018, 361: 1369–1373
Noten EA, McAtee RC, Stephenson CRJ. Chem Sci, 2022, 13: 6942–6949
Allen AR, Poon JF, McAtee RC, Watson NB, Pratt DA, Stephenson CRJ. ACS Catal, 2022, 12: 8511–8526
Hervieu C, Kiriia MS, Hu Y, Cuesta-Galisteo S, Merino E, Nevado C. Nat Chem, 2024, 16: doi: https://doi.org/10.1038/s41557-023-01414-8
Ren X, Lu Z. Chin J Catal, 2019, 40: 1003–1019
Wille U. Chem Rev, 2013, 113: 813–853
Wang Y, Shen S, He C, Zhou Y, Zhang K, Rao B, Han T, Su Y, Duan XH, Liu L. Chem Sci, 2023, 14: 6663–6668
Zhou Y, Zhao L, Hu M, Duan XH, Liu L. Org Lett, 2023, 25: 5268–5272
Zhang K, Wang Y, He C, Zhou Y, Wang D, Hu M, Duan XH, Liu L. Org Chem Front, 2022, 9: 5599–5605
He C, Zhang K, Wang DN, Wang M, Niu Y, Duan XH, Liu L. Org Lett, 2022, 24: 2767–2771
Negishi E, Huang Z, Wang G, Mohan S, Wang C, Hattori H. Acc Chem Res, 2008, 41: 1474–1485
Li G, Huo X, Jiang X, Zhang W. Chem Soc Rev, 2020, 49: 2060–2118
McCague R, Leclercq G, Legros N, Goodman J, Blackburn GM, Jarman M, Foster AB. J Med Chem, 1989, 32: 2527–2533
Connor CE, Norris JD, Broadwater G, Willson TM, Gottardis MM, Dewhirst MW, McDonnell DP. Cancer Res, 2001, 61: 2917–2922
Wang J, Wu XX, Cao Z, Zhang X, Wang X, Li J, Zhu C. Adv Sci, 2024, 20: e2309022
Deposition number CCDC 2280320 (for 3aa–acid) contains the supplementary crystallographic data for this paper. These data are provided free of charge by the joint Cambridge Crystallographic Data Centre and Fachinformationszentrum Karlsruhe Access Structures service
Metternich JB, Gilmour R. J Am Chem Soc, 2015, 137: 11254–11257
Metternich JB, Gilmour R. J Am Chem Soc, 2016, 138: 1040–1045
Neveselý T, Wienhold M, Molloy JJ, Gilmour R. Chem Rev, 2022, 122: 2650–2694
Utrilla RM, Sastre R, Catalina F, Mateo JL. J Photochem Photobiol A Chem, 1989, 46: 113–119
DFT calculations revealed that the energy barrier between the E/Z isomers of alkene 3bm was found to be extremely low (less than 0.1 kcal/mol). This finding aligns with the obtained poor E/Z ratios of the generated alkenes (for details, see Supporting Information)
Wu X, Fors BP, Buchwald SL. Angew Chem Int Ed, 2011, 50: 9943–9947
Vorogushin AV, Huang X, Buchwald SL. J Am Chem Soc, 2005, 127: 8146–8149
Danoun G, Tlili A, Monnier F, Taillefer M. Angew Chem Int Ed, 2012, 51: 12815–12819
Luo J, Zhang J. ACS Catal, 2016, 6: 873–877
Zheng G, Li Y, Han J, Xiong T, Zhang Q. Nat Commun, 2015, 6: 7011–7020
Hervieu C, Kirillova MS, Suárez T, Müller M, Merino E, Nevado C. Nat Chem, 2021, 13: 327–334
Whalley DM, Seayad J, Greaney MF. Angew Chem Int Ed, 2021, 60: 22219–22223
Chen Y, Chang L, Zuo Z. Acta Chim Sin, 2019, 77: 794–802
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21901199) and Xi’an Jiaotong University (7121192002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at chem.scichina.com and springerlink.bibliotecabuap.elogim.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
He, C., Wang, M., Wang, Y. et al. Photoredox catalytic alkylarylation of alkynes with arylsulfonylacetate as bifunctional reagent. Sci. China Chem. 67, 2022–2028 (2024). https://doi.org/10.1007/s11426-023-1930-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1930-6