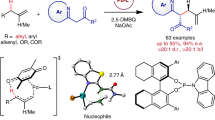
Abstract
Palladium-catalyzed allylic alkylation enabled by ketone umpolung via Pudovik addition/[1,2]-phospha-Brook rearrangement with phosphites has been developed. The protocol offers a straightforward method for the synthesis of potentially bioactive homoallylic alcohol phosphonates in an efficient and economical way. This cascade reaction proceeds under mild conditions with excellent functional group compatibility. Furthermore, the catalytic asymmetric version has also been explored.

Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Zhang MM, Qu BL, Shi B, Xiao WJ, Lu LQ. Chem Soc Rev, 2022, 51: 4146–4174
Cera G, Maestri G. ChemCatChem, 2022, 14: e202200295
Mohammadkhani L, Heravi MM. Chem Rec, 2021, 21: 29–68
Pàmies O, Margalef J, Cañellas S, James J, Judge E, Guiry PJ, Moberg C, Bäckvall JE, Pfaltz A, Pericas MA, Diéguez M. Chem Rev, 2021, 121: 43 73–45 05
Cheng Q, Tu HF, Zheng C, Qu JP, Helmchen G, You SL. Chem Rev, 2019, 119: 1855–1969
Tang H, Huo X, Meng Q, Zhang W. Acta Chim Sin, 2016, 74: 219–233
Butt NA, Zhang W. Chem Soc Rev, 2015, 44: 7929–7967
Trost BM. Tetrahedron, 2015, 71: 5708–5733
Dai XJ, Li CC, Li CJ. Chem Soc Rev, 2021, 50: 10733–10742
Wang S, König B. Angew Chem Int Ed, 2021, 60: 21624–21634
Chen XY, Gao ZH, Ye S. Acc Chem Res, 2020, 53: 690–702
Song X, Zhang J, Wu YX, Ouyang Q, Du W, Chen YC. J Am Chem Soc, 2022, 144: 9564–9569
Huang S, Zhang GP, Jiang YJ, Yu FL, Ding CH, Hou XL. Chem Commun, 2022, 58: 3513–3516
Wang TC, Zhu L, Luo S, Nong ZS, Wang PS, Gong LZ. J Am Chem Soc, 2021, 143: 20454–20461
Huang S, Tong FF, Bai DC, Zhang GP, Jiang YJ, Zhang B, Leng X, Guo YL, Wan XL, Zhang X, Ding CH, Hou XL. Nat Commun, 2021, 12: 6551
Shen C, Wang RQ, Wei L, Wang ZF, Tao HY, Wang CJ. Org Lett, 2019, 21: 6940–6945
Liu J, Cao CG, Sun HB, Zhang X, Niu D. J Am Chem Soc, 2016, 138: 13103–13106
Wu Y, Hu L, Li Z, Deng L. Nature, 2015, 523: 445–450
Yasuda S, Ishii T, Takemoto S, Haruki H, Ohmiya H. Angew Chem Int Ed, 2018, 57: 2938–2942
Haruki H, Yasuda S, Nagao K, Ohmiya H. Chem Eur J, 2019, 25: 724–727
Bai Y, Xiang S, Leow ML, Liu XW. Chem Commun, 2014, 50: 6168–6170
Bai Y, Leng WL, Li Y, Liu XW. Chem Commun, 2014, 50: 13391–13393
Liu H, Han YF, Gao ZH, Zhang CL, Wang C, Ye S. ACS Catal, 2022, 12: 1657–1663
Ueda Y, Masuda Y, Iwai T, Imaeda K, Takeuchi H, Ueno K, Gao M, Hasegawa J, Sawamura M. J Am Chem Soc, 2022, 144: 2218–2224
Yabushita K, Yuasa A, Nagao K, Ohmiya H. J Am Chem Soc, 2019, 141: 113–117
Tietze LF, Kinzel T, Brazel CC. Acc Chem Res, 2009, 42: 367–378
Wang PS, Shen ML, Gong LZ. Synthesis, 2018, 50: 956–967
Yus M, González-Gómez JC, Foubelo F. Chem Rev, 2013, 113: 5595–5698
Zhang X, Li Y, Miao Z. Daxue Huaxue, 2020, 0: 1–0
Kondoh A, Terada M. Asian J Org Chem, 2023, 12: e202300003
Kondoh A, Ozawa R, Terada M. Chem Lett, 2019, 48: 1164–1167
Kondoh A, Aita K, Ishikawa S, Terada M. Org Lett, 2020, 22: 2105–2110
Kondoh A, Ojima R, Terada M. Org Lett, 2021, 23: 7894–7899
Corbett MT, Uraguchi D, Ooi T, Johnson JS. Angew Chem Int Ed, 2012, 51: 4685–4689
Horwitz MA, Zavesky BP, Martinez-Alvarado JI, Johnson JS. Org Lett, 2016, 18: 36–39
Horwitz MA, Tanaka N, Yokosaka T, Uraguchi D, Johnson JS, Ooi T. Chem Sci, 2015, 6: 6086–6090
Lin Q, Zheng S, Chen L, Wu J, Li J, Liu P, Dong S, Liu X, Peng Q, Feng X. Angew Chem Int Ed, 2022, 61: e202203650
Tan Q, Guo N, Yang L, Wang F, Feng X, Liu X. J Org Chem, 2023, 88: 9332–9342
Cheibas C, Fincias N, Casaretto N, Garrec J, El Kaïm L. Angew Chem IntEd, 2022, 61: e202116249
Ali A, Jajoria R, Harit HK, Singh RP. J Org Chem, 2022, 87: 5213–5228
Kaur R, Singh D, Singh RP. J Org Chem, 2021, 86: 15702–15711
Zhang X, Gao Y, Liu Y, Miao Z. J Org Chem, 2021, 86: 8630–8640
Jiang J, Liu H, Lu CD, Xu YJ. Org Lett, 2016, 18: 880–883
Montchamp JL. Phosphorus Chemistry I: Asymmetric Synthesis and Bioactive Compounds. Cham: Springer, 2015
Montchamp JL. Phosphorus Chemistry II: Synthetic Methods. Cham: Springer, 2015
Corbridge DEC. Phosphorus Chemistry, Biochemistry and Technology. Sixth Edition. New York: Taylor & Francis Group, 2013
Zhao D, Wang R. Chem Soc Rev, 2012, 41: 2095–2108
Zhang L, Liu W, Zhao X. Eur J Org Chem, 2014, 2014: 6846–6849
Zhu J, Huang L, Dong W, Li N, Yu X, Deng WP, Tang W. Angew Chem IntEd, 2019, 58: 16119–16123
Trost BM, Zhang T, Sieber JD. Chem Sci, 2010, 1: 427–440
Trost BM, van Vranken DL. Chem Rev, 1996, 96: 395–422
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2021YFA0804900), the Leading Innovative and Entrepreneur Team Introduction Program of Zhejiang (2022R01007), and Shanghai Frontiers Science Center of Optogenetic Techniques for Cell Metabolism.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare no conflict of interest.
Additional information
Supporting information The supporting information is available online at https://chem.scichina.com and https://springerlink.bibliotecabuap.elogim.com/journal/11426. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
Supplementary Information
11426_2023_1744_MOESM1_ESM.pdf
Palladium-catalyzed allylic alkylation enabled by ketone umpolung via Pudovik addition/[1,2]-phospha-Brook rearrangement
Rights and permissions
About this article
Cite this article
Zhang, J., Su, JY., Liu, YZ. et al. Palladium-catalyzed allylic alkylation enabled by ketone umpolung via Pudovik addition/[1,2]-phospha-Brook rearrangement. Sci. China Chem. 66, 2810–2816 (2023). https://doi.org/10.1007/s11426-023-1744-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-023-1744-2