Abstract
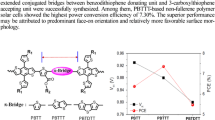
Two new conjugated copolymers, PBDT-T6-TTF and PBDT-T12-TTF, were derived from a novel 4-fluorobenzoyl thienothiophene (TTF). In addition, two types of benzodithiophene (BDT) units with 2,3-dihexylthienyl (T6) and 2,3-didodecylthienyl (T12) substituents, respectively, were successfully synthesized. The effect of the dual two-dimensional (2D) substitutions of the building blocks upon the optoelectronic properties of the polymers was investigated. Generally, the two polymers exhibited good solubility and broad absorption, showing similar optical band gaps of ∼1.53 eV. However, PBDT-T6-TTF with its shorter alkyl chain length possessed a larger extinction coefficient in thin solid film. The highest occupied molecular orbital (HOMO) level of PBDT-T6-TTF was located at −5.38 eV while that of PBDT-T12-TTF was at −5.51 eV. In space charge-limited-current (SCLC) measurement, PBDT-T6-TTF and PBDT-T12-TTF displayed respective hole mobilities of 3.0×10−4 and 1.6×10−5 cm2 V−1s−1. In polymer solar cells, PBDT-T6-TTF and PBDT-T12-TTF showed respective power conversion efficiencies (PCEs) of 2.86% and 1.67%. When 1,8-diiodooctane (DIO) was used as the solvent additive, the PCE of PBDT-T6-TTF was remarkably elevated to 4.85%, but the use of DIO for the PBDT-T12-TTF-blend film resulted in a lower PCE of 0.91%. Atomic force microscopy (AFM) indicated that the superior efficiency of PBDT-T6-TTF with 3% DIO (v/v) should be related to the better continuous phase separation of the blend film. Nevertheless, the morphology of the PBDT-T12-TTF deteriorated when the 3% DIO (v/v) was added. Our results suggest that the alkyl-chain length on the 2D BDT units play an important role in determining the optoelectronic properties of dual 2D BDT-TT-based polymers.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Günes S, Neugebauer H, Sariciftci NS, Conjugated polymer-based organic solar cells. Chem Rev, 2007, 107: 1324–1338
Li YF, Zou YP. Conjugated polymer photovoltaic materials with broad absorption band and high charge carrier mobility. Adv Mater, 2008, 20: 2952–2958
Chen JW, Cao Y. Development of novel conjugated donor polymers for high-efficiency bulk-heterojunction photovoltaic devices. Acc Chem Res, 2009, 42: 1709–1718
Liu Z, Xu F, Yan DD. New progress in the device physics of polymer-fullerene solar cells. Acta Chim Sinica, 2014, 72: 171–184
Liang YY, Wu Y, Feng DQ, Tsai ST, Son HJ, Li G, Yu P. Development of new semiconducting polymers for high performance solar cells. J Am Chem Soc, 2008, 131: 56–57
Hou JH, Chen HY, Zhang SQ, Chen RI, Yang Y, Wu Y, Li G. Synthesis of a low band gap polymer and its application in highly efficient polymer solar cells. J Am Chem Soc, 2009, 131: 15586–15587
Chen HY, Hou JH, Zhang SQ, Liang YY, Yang GW, Yang Y, Yu LP, Wu Y, Li G. Polymer solar cells with enhanced open-circuit voltage and efficiency. Nat Photon, 2009, 3: 649–653
Liang YY, Xu Z, Xia JB, Tsai S-T, Wu Y, Li G, Ray C, Yu LP. For the bright future—Bulk heterojunction polymer solar cells with power conversion efficiency of 7.4%. Adv Mater, 2010, 22: E135–E138
He ZC, Zhong CM, Huang X, Wong WY, Wu HB, Chen LW, Su SJ, Cao Y. Simultaneous enhancement of open-circuit voltage, short-circuit current density, and fill factor in polymer solar cells. Adv Mater, 2011, 23: 4636–4643
He ZC, Zhong CM, Su SJ, Xu M, Wu HB, Cao Y. Enhanced power-conversion efficiency in polymer solar cells using an inverted device structure. Nat Photon, 2012, 6: 591–595
Kim HJ, Lee BH, Lee KC, Kim GJ, Yu JY, Kim N, Lee SH, Lee K. Role of the side chain in the phase segregation of polymer:fullerene bulk heterojunction composites. Adv Energy Mater, 2013, 3: 1575–1580
Fan J, Yuen JD, Wang MF, Seifter J, Seo JH, Mohebbi AR, Zakhidov D, Heeger A, Wudl F. High-performance ambipolar transistors and inverters from an ultralow bandgap polymer. Adv Mater, 2012, 24: 2186–2190
Chen ZH, Cai P, Chen JW, Liu XC, Zhang LJ, Lan LF, Peng JB, Ma YG, Cao Y. Low band-gap conjugated polymers with strong interchain aggregation and very high hole mobility towards highly efficient thick-film polymer solar cells. Adv Mater, 2014, 26: 2586–2591
Lei T, Wang JY, Pei J. Roles of flexible chains in organic semiconducting materials.Chem Mater, 2013, 26: 594–603
Akkerman HB, Mannsfeld SCB, Kaushik AP, Verploegen E, Burnier L, Zoombelt AP, Saathoff JD, Hong S, Atahan-Evrenk S, Liu XL, Aspuru-Guzik A, Toney MF, Clancy P, Bao ZN. Effects of odd-even side chain length of alkyl-substituted diphenylbithiophenes on first monolayer thin film packing structure. J Am Chem Soc, 2013, 135: 11006–11014
Liang YY, Feng DQ, Wu Y, Tsai S-T, Li G, Ray C, Yu LP. Highly efficient solar cell polymers developed via fine-tuning of structural and electronic properties. J Am Chem Soc, 2009, 131: 7792–7799
Huo LJ, Hou JH, Zhang SQ, Chen HY, Yang Y. A polybenzo[1,2-b:4,5-b′]dithiophene derivative with deep HOMO level and its application in high-performance polymer solar cells. Angew Chem Int Ed, 2010, 49: 1500–1503
Huo LJ, Guo X, Zhang SQ, Li YF, Hou JH. PBDTTTZ: a broad band gap conjugated polymer with high photovoltaic performance in polymer solar cells. Macromolecules, 2011, 44: 4035–4037
Wu Y, Li ZJ, Guo X, Fan HL, Huo LJ, Hou JH. Synthesis and application of dithieno[2,3-d:2[prime or minute],3[prime or minute]-d[prime or minute]]benzo[1,2-b:4,5-b[prime or minute]]dithiophene in conjugated polymer. J Mater Chem, 2012, 22: 21362–21365
Huo LJ, Ye L, Wu Y, Li ZJ, Guo X, Zhang MJ, Zhang SQ, Hou JH. Conjugated and nonconjugated substitution effect on photovoltaic properties of benzodifuran-based photovoltaic polymers. Macromolecules, 2012, 45: 6923–6929
Ye L, Zhang SQ, Huo LJ, Zhang MJ, Hou JH. Molecular design toward highly efficient photovoltaic polymers based on two-dimensional conjugated benzodithiophene. Acc Chem Res, 2014, 47: 1595–1603
Huo LJ, Zhang SQ, Guo X, Xu F, Li YF, Hou JH. Replacing alkoxy groups with alkylthienyl groups: A feasible approach to improve the properties of photovoltaic polymers. Angew Chem Int Ed, 2011, 50: 9697–9702
Kim JH, Song CE, Kim BS, Kang I-N, Shin WS, Hwang DH. Thieno[3,2-b]thiophene-substituted benzo[1,2-b:4,5-b′]dithiophene as a promising building block for low bandgap semiconducting polymers for high-performance single and tandem organic photovoltaic cells. Chem Mater, 2013, 26: 1234–1242
Cui CH, Wong WY, Li YF. Improvement of open-circuit voltage and photovoltaic properties of 2D-conjugated polymers by alkylthio substitution. Energy Environ Sci, 2014, 7: 2276–2284
Huo LJ, Li ZJ, Guo X, Wu Y, Zhang MJ, Ye L, Zhang SQ, Hou JH. Benzodifuran-alt-thienothiophene based low band gap copolymers: substituent effects on their molecular energy levels and photovoltaic properties. Polym Chem, 2013, 4: 3047–3056
Huang Y, Huo LJ, Zhang SQ, Guo X, Han CC, Li YF, Hou JH. Sulfonyl: a new application of electron-withdrawing substituent in highly efficient photovoltaic polymer. Chem Commun, 2011, 47: 8904–8906
Wu Y, Li ZJ, Ma W, Huang Y, Huo LJ, Guo X, Zhang MJ, Ade H, Hou JH. PDT-S-T: a new polymer with optimized molecular conformation for controlled aggregation and π-π stacking and its application in efficient photovoltaic devices. Adv Mater, 2013, 25: 3449–3455
Homyak PD, Tinkham J, Lahti PM, Coughlin EB. Thieno[3,4-b]-thiophene acceptors with alkyl, aryl, perfluoroalkyl, and perfluorophenyl pendants for donor-acceptor low bandgap polymers. Macromolecules, 2013, 46: 8873–8881
Yamamoto T, Ikai T, Kuzuba M, Kuwabara T, Maeda K, Takahashi K, Kanoh S. Synthesis and characterization of thieno[3,4-b]thiophene-based copolymers bearing 4-substituted phenyl ester pendants: facile fine-tuning of HOMO energy levels. Macromolecules, 2011, 44: 6659–6662
Meager I, Ashraf RS, Mollinger S, Schroeder BC, Bronstein H, Beatrup D, Vezie MS, Kirchartz T, Salleo A, Nelson J, McCulloch I. Photocurrent enhancement from diketopyrrolopyrrole polymer solar cells through alkyl-chain branching point manipulation. J Am Chem Soc, 2013, 135: 11537–11540
Sun JM, Zhu YX, Xu XF, Lan LF, Zhang LJ, Cai P, Chen JW, Peng JB, Cao Y. High efficiency and high V oc inverted polymer solar cells based on a low-lying HOMO polycarbazole donor and a hydrophilic polycarbazole interlayer on ITO cathode. J Phys Chem C, 2012, 116: 14188–14198
Yang TB, Qin DH, Lan LF, Huang WB, Gong X, Peng JB, Cao Y. Inverted polymer solar cells with a solution-processed zinc oxide thin film as an electron collection layer. Sci China Chem, 2012, 55: 755–759
Jiang ZL, Yang D, Wang N, Zhang FJ, Zhao B, Tan ST, Zhang J. Inverted polymer solar cells with TiO2 electron extraction layers prepared by magnetron sputtering. Sci China Chem, 2013, 56: 1573–1577
Zhu YX, Xu XF, Zhang LJ, Chen JW, Cao Y. High efficiency inverted polymeric bulk-heterojunction solar cells with hydrophilic conjugated polymers as cathode interlayer on ITO. Sol Energy Mater Sol Cells, 2012, 97: 83–88
Gao L, Zhang J, He C, Zhang Y, Sun QJ, Li YF. Effect of additives on the photovoltaic properties of organic solar cells based on triphenylamine-containing amorphous molecules. Sci China Chem, 2014, 57: 966–972
Chen ZH, Cai P, Zhang LJ, Zhu YX, Xu XF, Sun JM, Huang J, Liu XC, Chen JW, Chen HZ, Cao Y. Donor-acceptor copolymers based on phenanthrene as electron-donating unit: synthesis and photovoltaic performances. J Polym Sci Part A, Polym Chem, 2013, 51: 4966–4974
Huang J, Zhu Y, Zhang LJ, Cai P, Xu XF, Chen JW, Cao Y. Low band-gap D-A conjugated copolymers based on anthradithiophene and diketopyrrolopyrrole for polymer solar cells and field-effect transistors. J Polym Sci Part A, Polym Chem, 2014, 52: 1652–1661
Liao SH, Jhuo HJ, Cheng YS, Chen SA. Fullerene derivative-doped zinc oxide nanofilm as the cathode of inverted polymer solar cells with low-bandgap polymer (PTB7-Th) for high performance. Adv Mater, 2013, 25: 4766–4771
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liu, Z., Sun, J., Zhu, Y. et al. Low band-gap benzodithiophene-thienothiophenecopolymers: the effect of dual two-dimensional substitutions on optoelectronic properties. Sci. China Chem. 58, 267–275 (2015). https://doi.org/10.1007/s11426-014-5223-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-014-5223-7