Abstract
Background
Patient-reported outcome measures (PROMs) are an important resource for clinicians wishing to provide high-quality, patient-centered care. Finding PROMs to use in a pediatric clinical practice that are reliable, age appropriate, succinct, and not redundant is challenging.
Questions/Purposes
We sought to determine the degree of correlation between two pediatric PROMs, the Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS) and the PROMIS Pediatric Mobility (PROMIS PM) instrument, when administered at patients’ initial visit. We hypothesized that there would be a positive correlation between the two questionnaires because of their focus on physical function.
Methods
We conducted a retrospective cross-sectional review of 294 pediatric patients (mean age, 13.7 years) with localized lower-extremity joint pathologies at our high-volume urban pediatric sports practice. The patients had been asked to complete both PROMs on a computerized platform.
Results
We found a positive and statistically significant—albeit modest—correlation between the scores obtained on the HSS Pedi-FABS and the PROMIS PM. Neither instrument significantly correlated with patient age, nor were floor and ceiling effects observed.
Conclusion
The study shows that although both PROM instruments provide valuable information about pediatric physical function, they are not redundant because they measure slightly different constructs. Future studies should further investigate the correlation between these questionnaires in specific subpopulations of pediatric patients with lower-extremity pathology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patient-reported outcome measures (PROMs) have become important tools for providing patient-focused, high-quality clinical care. PROMs are validated questionnaires that can objectively measure a variety of outcomes from the patient’s perspective, including quality of life, overall health, treatment satisfaction, and functional status [25]. In addition to research applications, this information can help improve the quality of patient care delivery and allow patients to more actively participate in their recovery by learning about expected outcomes [4, 11, 25]. However, when implementing PROMs into clinical practice, it can be difficult for practitioners to find instruments that are reliable, age appropriate, succinct, and free of redundancy [4].
In an effort to facilitate the standardization of PROMs, the National Institutes of Health (NIH) funded the development of the Patient-Reported Outcome Measurement Information System (PROMIS) [21]. PROMIS is a series of validated PROMs that is used to assess the physical, mental, and social health of patients and is applicable to both acute and chronic conditions; the pediatric PROMIS domains measure the same features in children ages 8 through 17 years [21]. PROMIS has been validated in several pediatric domains, including fatigue, mobility, pain intensity, pain interference, upper-extremity function, anxiety, depressive symptoms, and peer relationships [5]. PROMIS can be administered as a computer adaptive test (CAT) that employs item response theory (IRT). IRT dynamically chooses the most appropriate next question from an item bank based on the patient’s previous responses [5, 21]. The PROMIS Pediatric Mobility instrument (PROMIS PM) works via CAT and is routinely administered to pediatric patients for both clinical care and research purposes. The Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS) is a traditional eight-item questionnaire validated as an activity rating scale in children 10 to 18 years old [7]. Both the PROMIS PM and HSS Pedi-FABS are used to assess athletic activity in children and adolescents.
The purpose of this study was to investigate for any correlation between the PROMIS PM and the HSS Pedi-FABS in pediatric patients who presented with lower-extremity complaints. We hypothesized that there would be a significant positive correlation between both scales but with moderate correlation coefficients, indicating the scales’ measurement of similar but distinct constructs. This analysis could be used to inform the use of these instruments in pediatric and adolescent patients and guide administration strategies aimed at extracting useful and novel data while avoiding questionnaire redundancy.
Materials and Methods
We conducted a retrospective cross-sectional study of children ages 10 to 18 years (but not including age 18) presenting at our tertiary care outpatient pediatric orthopedic sports medicine division; the patients had completed the relevant standard of care PROMs. The study protocol was approved by the hospital’s institutional review board. The study participants were seen by one of two pediatric orthopedic sports surgeons over a 6-month period. All pediatric patients who presented with primary lower-extremity problems and completed the PROMIS PM and HSS Pedi-FABS within a 24-h time frame were included in the study. No patients were excluded.
All pediatric PROMs were collected using a tablet device connected to a secure network. A trained technical assistant was available on-site to help as needed with software interface but not with the questionnaire items themselves. In addition to obtaining the completed questionnaires, we extracted demographic information; laterality of injury, if applicable; the affected joint; and the documented diagnosis code from the patient health records.
We administered the PROMIS PM CAT (version 1.0: Mobility) to assess physical function (sample question: “I could do sports and other exercise that kids my age could do”). IRT was employed in real time via software interface with the PROMIS Assessment Center secure data collection software. Depending on the answers provided, patients were asked between five and 11 questions, with 1 to 5 points award for each. PROMIS PM scores are calibrated with reference to US population–level statistics, each with a mean of 50 and a standard deviation of 10 [20]. Higher scores on the PROMIS PM reflect better function.
The HSS Pedi-FABS evaluates activity and activity level in a pediatric patient over the prior month [7]. This eight-item instrument focuses on mobility dynamics and the frequency with which specific activities are performed. The activities included are running, cutting, decelerating, and pivoting; activity duration and activity endurance are also assessed. Two additional items focus on the level of competition and supervision of the activities engaged in. The score is the sum of points from each question, for a total possible score ranging from 0 to 30 points. For questions on running, cutting, decelerating, pivoting, duration, and endurance, each question is scored from 0 to 4 points. The questions about competition level and supervision are scored from 0 to 3 points. A higher score on the HSS Pedi-FABS indicates greater activity level and has been shown to be predictive of performance on physical fitness metrics [7, 8]. The HSS Pedi-FABS score is normally distributed with a mean of 15.4 ± 8.5 points [9].
Statistical Analysis
We analyzed the data using SPSS version 22.0 (IBM Corp., Armonk, NY, USA). Data were expressed using descriptive statistics, including frequency and percentage (count variables), as well as mean and standard deviation (continuous variables). After confirming normality of data distribution, we performed a Pearson correlation analysis to determine the relationship between scores. We recorded bilaterality of symptoms in order not to violate the assumption of statistical independence; patients completed each test once as a measure of overall activity and function for their given condition. All analyses were two tailed, and a p value of < 0.05 was used as the threshold for statistical significance.
Results
The patient sample included 294 children and adolescents, with a mean age of 13.7 ± 2.1 years. There were 148 (50.3%) female patients and 146 (49.7%) male patients. There were 139 (47.3%) patients with left-sided symptoms, 129 (43.9%) with right-sided symptoms, and 26 (8.8%) with bilateral symptoms. Presenting symptoms were localized to specific joints: knee joint pathologies represented the most prevalent injury type, occurring in 236 (80.3%) patients, followed by the ankle (n = 43; 14.6%), hip (n = 8; 2.7%), and toe (n = 7; 24.%) joints (Table 1).
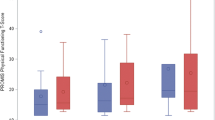
On the HSS Pedi-FABS, the mean score was 20.5 ± 8.8, with scores ranging from 0 to 30; on the PROMIS PM, it was 39.6 ± 11.1, with scores ranging from 16.5 to 61.7 (Fig. 1).
Scatter plot depicting the correlation of the PROMIS PM and the HSS Pedi-FABS. Circles represent individual samples. The correlation shows a moderate but significantly positive statistical relationship (Pearson correlation coefficient [r] = 0.189; p = 0.001). Box plots are shown for the PROMIS PM and HSS Pedi-FABS. PROMIS PM Patient-Reported Outcome Measurement Information System, HSS Pedi-FABS Hospital for Special Surgery Pediatric Functional Activity Brief Scale.
Pearson correlation analysis yielded a standard correlation coefficient of 0.189 (p = 0.001) between the HSS Pedi-FABS and the PROMIS PM, indicating a statistically significant, but modest, positive correlation. Neither the HSS Pedi-FABS nor the PROMIS PM was significantly correlated with patient age (p = 0.053 and p = 0.208, respectively).
Discussion
We found that the PROMIS PM showed a statistically significant but modest correlation to the HSS Pedi-FABS. Health status measurement instruments must possess adequate measurement properties, such as reliability, validity, and responsiveness, to be useful in either research or patient care [11]. The PROMIS was developed by the NIH in an effort to create a standardized, reliable, and validated outcome measure for use in both clinical and research arenas. The PROMIS was designed to aid in the clinical stratification of patients via streamlined data collection. Since its inception, the PROMIS PM has undergone several improvements in order to create an effective and succinct instrument [1, 13, 22]. PROMIS PM and numerous other PROMIS scales employ IRT, a computerized algorithm whereby only the most informative items targeting an individual’s functioning levels are inserted in an electronic questionnaire sequence. Similarly, the HSS Pedi-FABS was developed as a succinct traditional questionnaire for the assessment of pediatric and adolescent physical activity. During validation, the HSS Pedi-FABS was shown to have a modest and significant positive correlation with adult-focused instruments such as the Tegner activity scale, the Marx activity scale, the Noyes sports and functional activity scales, and the Physical Activities Questionnaire [7]. The HSS Pedi-FABS includes assessment of factors such as level of competitiveness and supervision of physical activity, which are important considerations in children and adolescents.
One of the limitations of our study is that analysis was limited to patients seen within a 6-month period because of the relatively recent initiation of the PROMIS PM and HSS Pedi-FABS administered via centralized software at our institution. However, our primary goal was to establish a preliminary correlation between these two instruments with a large sample before analyzing additional questionnaire characteristics. Additionally, collecting this amount of data in a short period minimizes the risk of scores being affected by changes in patient flow in the office or data collection procedures. Another limitation is that the PROMIS PM is designed for use in children and adolescents 8 through 17 years of age, whereas the HSS Pedi-FABS is designed for use in those 10 through 18 years of age [7, 12]. To allow for maximal overlap of ages for which each scale is validated, we studied only patients between 10 and 17 years of age. An additional limitation is that our patient population was generally healthy child and adolescent athletes, and these results may not apply to other, less active cohorts. Rodday et al. demonstrated when the PROMIS PM was used in subpopulations of children with neuromuscular conditions, the mean PROMIS PM form scores were as much as three standard deviations lower than the PROMIS PM–calibrated means predisposing to floor effects [23]. However, we included a variety of lower-extremity diagnoses, which increases the external validity of our findings. Future studies might evaluate questionnaire correlations in pediatric subpopulations stratified by condition and severity to better inform questionnaire qualification in these groups over a longer time period.
In an effort to perform a novel investigation and avoid scales whose use is not validated in children and adolescents, we performed a literature search and identified studies that showed correlations between pediatric PROMIS measures and other questionnaires. For example, Fedorak et al. found a strong correlation between pediatric PROMIS pain interference, mobility, and peer relationship with the SRS (Scoliosis Research Society)-22 questionnaire in patients with adolescent idiopathic scoliosis [10]. Similarly, Waljee et al. found a correlation between PROMIS Pediatric Upper Extremity with the MHQ (Michigan Hand Outcomes Questionnaire), the DASH (Disabilities of the Arm, Shoulder, and Hand) questionnaire, and the PODCI (Pediatric Outcomes Data Collection Instrument) in patients with congenital hand differences [24]. A recent study by Makhni et al. demonstrated a correlation between various PROMIS questionnaires, including physical function, pain interference, and depression, using Pearson correlation coefficients to examine overlap between the questionnaires [16]. Many other PROMIS correlation studies have been performed with patients filling out multiple questionnaires and then using a Pearson correlation coefficient to examine the relationship and overlap between PROMs [2, 3, 14, 17,18,19, 26]. We aimed in our study to use a similar methodology to determine the relationship between the HSS Pedi-FABS and the PROMIS PM.
Our results demonstrate a modest positive correlation between scores obtained on the HSS Pedi-FABS and the PROMIS PM. This indicates that although both instruments provide insight into the domain of activity and physical function, they measure slightly different constructs and may complement each other when administered concurrently. This is an important first step in trying to understand how the use of either or both of these scales can provide insight into pediatric patients’ mobility. Furthermore, these results are consistent with the individual questions, which approach similar concepts differently. For example, the HSS Pedi-FABS asks how often a patient has run in the past month, and the PROMIS PM asks patients to rate how easily they could have run a mile in the past week.
IRT, used in the administration of the PROMIS PM, is believed to have the advantage of calibrating instruments across a broad range of possible scores to overcome floor and ceiling effects that often limit PROM utility, efficiency, and sensitivity to change [6]. DeWitt et al. noted that although IRT can assist in preventing floor and ceiling effects, versions of the PROMIS PM still exhibit ceiling effects when used in moderately active populations [5]. The inclusion of factors such as level of competition, endurance, and level of supervision of sporting activity in the HSS Pedi-FABS avoids the ceiling effects when measuring physical function in an active pediatric population [7]. Important in our study, neither the PROMIS PM nor the HSS Pedi-FABS displayed a floor or ceiling effect, defined as more than 15% of our patients scoring either the minimum or maximum possible score [15].
In conclusion, although both instruments provide valuable information about pediatric physical function, they are not redundant because they measure slightly different constructs. Used together, they can inform care providers about unique elements of patient status.
References
Amtmann D, Bamer A, Cook K, Harniss M, Johnson K. Adapting PROMIS physical function items for users of assistive technology. Disabil Health J. 2010;3(2):E9.
Anthony CA, Glass NA, Hancock K, Bollier M, Wolf BR, Hettrich CM. Performance of PROMIS instruments in patients with shoulder instability. Am J Sports Med. 2017;45(2):449–453.
Bernstein DN, Mahmood B, Ketonis C, Hammert WC. A comparison of PROMIS physical function and pain interference scores in patients with carpal tunnel syndrome: research collection versus routine clinical collection. Hand (N Y). 2019. https://doi.org/10.1177/1558944719831345.
Brook EM, GK, Higgins LD, Matzkin EG. Implementing patient-reported outcome measures in your practice: pearls and pitfalls. Am J Orthop. 2017;46(6):273–278.
Dewitt EM, Stucky BD, Thissen D. Construction of the eight-item patient-reported outcomes measurement information system pediatric physical function scales: built using item response theory. J Clin Epidemiol. 2011;64(7):794–804.
Edelen MO, Reeve BB. Applying item response theory (IRT) modeling to questionnaire development, evaluation, and refinement. Qual Life Res. 2007;16 Suppl 1:5–18.
Fabricant PD, Robles A, Downey-Zayas T, et al. Development and validation of a pediatric sports activity rating scale: the Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS). Am J Sports Med. 2013;41(10):2421–2429.
Fabricant PD, Robles A, McLaren SH, Marx RG, Widmann RF, Green DW. Hospital for Special Surgery Pediatric Functional Activity Brief Scale predicts physical fitness testing performance. Clin Orthop Relat Res. 2014;472(5):1610–1616.
Fabricant PD, Suryavanshi JR, Calcei JG, Marx RG, Widmann RF, Green DW. The Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS): normative data. Am J Sports Med. 2018;46(5):1228–1234.
Fedorak GT, Larkin K, Heflin JA, Xu J, Hung M. Pediatric PROMIS is equivalent to SRS-22 in assessing health status in adolescent idiopathic scoliosis. Spine (Phila Pa 1976). 2019 Jun 12. [Epub ahead of print]. https://doi.org/10.1097/BRS.0000000000003112.
Gagnier JJ. Patient reported outcomes in orthopaedics. J Orthop Res. 2017;35(10):2098–2108.
Intro to PROMIS. HealthMeasures. Northwestern University. 2019. Available at http://www.healthmeasures.net/explore-measurement-systems/promis/intro-to-promis. Accessed 30 August 2019.
Irwin DE, Gross HE, Stucky BD. Development of six PROMIS pediatrics proxy-report item banks. Health Qual Life Outcomes. 2012;10:22.
Khechen B, Patel DV, Haws BE, et al. Evaluating the concurrent validity of PROMIS Physical Function in anterior cervical discectomy and fusion. Clin Spine Surg. 2019. https://doi.org/10.1097/BSD.0000000000000786.
Lim CR, Harris K, Dawson J, Beard DJ, Fitzpatrick R, Price AJ. Floor and ceiling effects in the OHS: an analysis of the NHS PROMs data set. BMJ Open. 2015;5(7):e007765.
Makhni EC, Meldau JE, Blanchett J, et al. Correlation of PROMIS Physical Function, Pain Interference, and Depression in Pediatric and Adolescent Patients in the ambulatory sports medicine clinic. Orthop J Sports Med. 2019;7(6):2325967119851100.
Nwachukwu BU, Beck EC, Chapman R, Chahla J, Okoroha K, Nho SJ. Preoperative performance of the PROMIS in patients undergoing hip arthroscopic surgery for femoroacetabular impingement syndrome. Orthop J Sports Med. 2019;7(7):2325967119860079. https://doi.org/10.1177/2325967119860079.
Owen RJ, Khan AZ, McAnany SJ, Peters C, Zebala LP. PROMIS correlation with NDI and VAS measurements of physical function and pain in surgical patients with cervical disc herniations and radiculopathy. J Neurosurg Spine. 2019;1–6. [Epub ahead of print]. https://doi.org/10.3171/2019.4.SPINE18422.
Owen RJ, Zebala LP, Peters C, McAnany S. PROMIS Physical Function Correlation With NDI and mJOA in the surgical cervical myelopathy patient population. Spine (Phila Pa 1976). 2018;43(8):550–555.
PROMIS. HealthMeasures. Northwestern University. 2019. Available at http://www.healthmeasures.net/score-and-interpret/interpret-scores/promis. Accessed 22 August 2019.
PROMIS Instrument Development and Psychometric Evaluation Scientific Standards. Version 2.0 (revised May 2013). HealthMeasures. Northwestern University. 2019. Available at http://www.healthmeasures.net/images/PROMIS/PROMISStandards_Vers2.0_Final.pdf. Accessed 28 Sept 2017.
Quinn H, Thissen D, Liu Y. Using item response theory to enrich and expand the PROMIS pediatric self report banks. Health Qual Life Outcomes 2014;12:160.
Rodday AM, Graham RJ, Weidner RA, Rothrock NE, Dewalt DA, Parsons SK. Leveraging pediatric PROMIS item banks to assess physical functioning in children at risk for severe functional loss. J Patient Rep Outcomes. 2017;1(1):10.
Waljee JF, Carlozzi N, Franzblau LE, Zhong L, Chung KC. Applying the Patient-Reported Outcomes Measurement Information System to assess upper extremity function among children with congenital hand differences. Plast Reconstr Surg. 2015;136(2):200e–207e.
Weldring T, Smith SM. Patient-reported outcomes (PROs) and patient-reported outcome measures (PROMs). Health Serv Insights. 2013;6:61–68.
Yee TJ, Smith BW, Joseph JR, et al. Correlation between the Oswestry Disability Index and the 4-item short forms for physical function and pain interference from PROMIS. J Neurosurg Spine. 2019;9:1–6. https://doi.org/10.3171/2019.5.SPINE19400.
Funding
The study was funded by the Hospital for Special Surgery.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Joshua Adjei, BA, Jonathan Schachne, BA, and Peter D. Fabricant, MD, MPH, declare that they have no conflicts of interest. Daniel W. Green MD, MS, FACS, FAAP, reports receiving intellectual property royalties from Arthrex, Inc., and Pega Medical; consulting fees from Arthrex, Inc.; and publishing royalties and financial or material support from Wolters Kluwer Health and Current Opinion in Pediatrics, outside the submitted work.
Human/Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2013.
Informed Consent
Informed consent was waived from all patients included in this study.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article.
Additional information
Level of Evidence: Level III: Retrospective Cross-Sectional Study
Rights and permissions
About this article
Cite this article
Adjei, J., Schachne, J., Green, D.W. et al. Correlation Between the PROMIS Pediatric Mobility Instrument and the Hospital for Special Surgery Pediatric Functional Activity Brief Scale (HSS Pedi-FABS). HSS Jrnl 16 (Suppl 2), 311–315 (2020). https://doi.org/10.1007/s11420-019-09726-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11420-019-09726-7