Abstract
Purpose
Ultraviolet (UV) radiation plays an important role in litter decomposition, but the direction and magnitude of its effect remain inconsistent.
Materials and methods
To determine the responses of litter decomposition and C and nutrient release to UV radiation, a meta-analysis that comprised 544 observations in field experiments was performed.
Results and discussion
The change in UV radiation had significant effects on litter mass loss and litter nitrogen (N) and phosphorus (P) release in field studies, and litter types affected only the magnitude of UV radiation changes on mass loss and nutrient release. In addition, mass loss and nutrient release varied as the decomposition process continued: UV enhancement accelerated litter decomposition only after 4 months of decomposition, whereas the effect of UV attenuation on litter decomposition decreased with decomposition time. The inhabitation of UV attenuation on litter decomposition increased with a decrease in precipitation.
Conclusion
Overall, the different responses of mass loss and nutrient release in litter decomposition to changes in ultraviolet radiation may cause different C and nutrient cycling in terrestrial ecosystems in the Southern Hemisphere and Northern Hemisphere.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Litter decomposition plays a key role in carbon (C) and nutrient cycling in terrestrial ecosystems (García-Palacios et al. 2016; Prescott 2005; Xu et al. 2013; Zhang et al. 2013). An increasing number of studies have shown that UV radiation is an important driver of litter decomposition, although conclusions about the magnitude and direction of its effects remain inconsistent (Brandt et al. 2010; Brandt et al. 2009; Day et al. 2015). The amount of UV radiation reaching the Earth’s surface has changed in the past years due to the increase in human activities (Williamson et al. 2014), and increased and reduced UV radiation in the Southern Hemisphere (Herman 2010) and Northern Hemisphere (Calbo and González 2005) have been observed, respectively. Therefore, research on the effects of changing UV radiation on litter decomposition could provide important information for predicting nutrient cycling in terrestrial ecosystems under the background of UV radiation changes.
The effects of UV radiation on litter decomposition include two main processes: the breakdown of organic matter directly into C-based gases (Brandt et al. 2009; Lee et al. 2012; Rutledge et al. 2010) and the conversion of large resistant compounds to smaller compounds that are more readily degradable by soil microbes (Gallo et al. 2009; Lambie et al. 2014). UV radiation directly or indirectly affects litter decomposition through photochemical breakdown, by altering the soil microbial community or by changing the chemical and physical properties of the litter. The effect of UV on litter decomposition also depends on the plant species, litter types, and experimental conditions (Kirschbaum et al. 2011; Song et al. 2013a), and different effects have also been reported in studies due to different experimental durations (Song et al. 2013b). These studies highlight the variation in different responses of litter decomposition to UV radiation. Thus, to better understand the role of UV radiation in litter decomposition, different factors, such as litter types and experimental duration should be considered.
In arid ecosystems, litter decomposition is proportional to time and often cannot be explained by exponential models (Parton et al. 2007). UV radiation is recognized as one of the most important drivers of unexpected rapid decomposition (Austin and Ballare 2010; Brandt et al. 2009; Day et al. 2015). In addition, positive effects of UV radiation have been observed in areas with precipitation ranging from 152 to 726 mm (Day et al. 2015; Huang and Li 2017; Huang et al. 2017; Pancotto et al. 2003). Although UV radiation inhibits microbial decomposition in areas with low precipitation, sparse vegetation causes litter to receive a high level of solar radiation (Austin and Vivanco 2006), which directly increases the breakdown of organic matter and enhances the decomposition of substrates to microbes (Austin and Vivanco 2006; Brandt et al. 2010; Huang et al. 2017; Pancotto et al. 2003). However, inconsistent results from the effects of UV on litter decomposition have been observed under dry and wet conditions. For example, Brandt et al. (2007) reported stronger effects under dry conditions, but other studies found that the effects of UV were negligible under dry conditions (Uselman et al. 2011). In low-precipitation areas, litter decomposition is the balance of positive photodecomposition and negative biodecomposition due to low soil water availability. However, whether changes in UV radiation have similar effects on litter decomposition under different precipitation regimes remains unclear.
Previous studies have mainly focused on the effects of UV radiation on litter mass loss, but less attention has been paid to C and nutrient release from litter (Wang et al. 2015). There are three meta-analyses related to the effects of changes in UV radiation on litter decomposition (King et al. 2012; Song et al. 2013a; Wang et al. 2015). One of them focused only on litter mass loss and chemistry under UV enhancement (Wang et al. 2015), and the others examined only litter mass loss under changes in UV radiation (King et al. 2012; Song et al. 2013a). In general, the loss of litter mass increases with decomposition, but nutrient release may exhibit different patterns. For example, the nitrogen (N) remaining in litter increased after 15 months in semiarid Mediterranean grasslands (Almagro et al. 2017). Thus, to understand the patterns of C and nutrient release under changes in UV radiation, a clarification of the relationship between litter mass loss and nutrient release during litter decomposition is urgently needed.
To determine the impacts of UV radiation on litter decomposition as well as C and nutrient release, a meta-analysis that comprised 544 observations worldwide was performed. Specifically, we addressed the following issues: (1) how litter decomposition and C and nutrient release change under UV radiation and whether the litter types and experiment durations affect the decomposition); (2) whether litter decomposition rates change in response to UV radiation in areas with different precipitation levels; and (3) whether UV changes the relationship between C and nutrient release and litter mass loss.
2 Materials and methods
2.1 Data preparation
The following combinations of ultraviolet/UV/photodecomposition and litter decomposition/litter nutrients were employed to identify published articles in the Web of Science and articles from the Chinese Academy of Sciences and China Knowledge Resource Integrated (CNKI) databases. The following criteria were applied: (1) only field studies were included, and each study included at least one paired data set (control and treatment); (2) mass loss and the remaining nutrients measured in different durations were denoted separately; and (3) the mean, standard deviation/error, and number of replicates could be directly extracted from the text, tables, or digitized graphs. In total, 46 published papers worldwide were selected from more than 1000 published papers (Fig. S1). The list of literature sources and data are shown in the Supporting information.
For each selected study, the location and environmental variables were obtained directly from the published paper. In addition, the plant litter species, litter types (forest or grassland), leaf type (broad-leaf and needle), initial litter weight, and litter chemical properties (C, N, P, and lignin remaining or released) were recorded. All original data were extracted from the text, tables, or appendices of the publications. For those data presented graphically, we used the Get-Data Graph Digitizer (ver. 2.20, Russian Federation) to digitize and extract the numerical data.
2.2 Data analysis
The nutrients remaining during decomposition were calculated as the follows: nutrient remaining% = (Nt * Masst) / (N0 * Mass0), where Nt is the nutrient concentration at decomposition time t, Masst is the dry mass at time t, N0 is the initial nutrient concentration, and Mass0 is the initial dry mass.
The response ratios (RRs) for the mass loss and nutrient changes were evaluated using the following equation (Hedges et al. 1999):
where Xe and Xc are the responses of observations in the experimental and control treatments, respectively. The corresponding sample variance for each RR was calculated as follows:
where ne, Se, and Xe represent the sample size, standard deviation, and mean response values in the experimental group, respectively, and nc, Sc, and Xc represent the sample size, standard deviation, and mean response values in the control group, respectively. The reciprocal of the variance (w = 1 / vi) was considered to be the weight of each RR. The mean weighted response ratio (RR++) was calculated from the RR for individual pairwise comparisons between the treatment and control groups as follows:
where m is the number of groups and k is the number of comparisons in the corresponding group. In addition, the standard error of RR++ was estimated as follows:
When the decomposition time spanned more than one time category, we categorized it with the shorter decomposition period; for example, an experiment that lasted exactly 4 months was categorized as 2–4 months. A similar approach was used to categorize precipitation data. The sample size was calculated as the number of paired observations. The meta-analysis was performed using R software (version 3.1.1) (R Core Team 2014). The natural logs of the RRs for the individual and combined treatments were determined by specifying the study as a random factor in the model with the “metafor” package. The effects of changes in UV radiation on the loss of biomass and nutrient changes were considered significant if the 95% confidence interval (CI) of the RR did not overlap with zero. A regression analysis was conducted to evaluate the relationship between the nutrient release and the loss of litter weight.
3 Results
3.1 UV radiation type and litter types
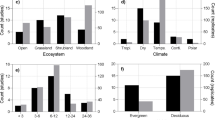
As expected, changes in UV radiation had significant effects on k decay and mass loss (Fig. 1). UV enhancement promoted N and phosphorus (P) release, with RRs of − 0.18 and − 0.09, respectively, and UV attenuation decreased the N and P release, with RRs of − 0.09 and 0.10, respectively. The effects of UV radiation on C and lignin release were not significant. Across all studies, the RR of k decay was significantly correlated with the UV radiation level (Fig. S2), which increased as the UV intensities increased. The litter types and leaf types affected litter decomposition under UV radiation changes (Fig. 2a, b); for example, UV enhancement significantly affected the mass loss and N and P release of plant litter, whereas UV attenuation significantly decreased the litter mass loss of broad-leaved and needle-leaved plants (Fig. S3).
Effects of UV treatment on litter mass loss and remaining nutrients. The black symbols indicate significant differences (p < 0.05) between the response ratios (RRs) and zero. The vertical dotted line represents a mean effect size of 0. The sample size for UV enhancement and attenuation is shown from left to right
Effects of UV enhancement (a) and attenuation (b) on litter mass loss and remaining nutrients in grassland and forest litters. The black symbols indicate significant differences (p < 0.05) between the response ratios (RRs) and zero. The sample size for each variable is shown and represents the grassland and forest litters, from left to right
3.2 Experimental duration
The RRs of mass loss and nutrient release varied with decomposition time (Fig. 3). UV enhancement had no significant effect on mass loss in the first four months of decomposition but increased mass loss from 4 to 18 months (Fig. 3a). However, UV enhancement showed no effect on litter C release (Fig. 3b) but promoted N release after 6 months of decomposition (Fig. 3c) and promoted both P and lignin release after 18 months (Fig. 3d, e). UV attenuation negatively affected mass loss, but the effect decreased with the decomposition time (Fig. 3f), and the rate as well as the direction of nutrient release differed in C, N, P, and lignin under UV attenuation (Fig. 3g–j).
Effects of UV enhancement (a–e) and attenuation (f–j) on litter mass loss and remaining nutrients during decomposition. The dashed line represents zero. The black symbols indicate significant differences (p < 0.05) between the response ratios (RRs) and zero. The sample size for each variable is shown above the symbol
3.3 Precipitation
Decomposition showed a significant correlation with precipitation under the control treatments (Fig. S4a), and k decay increased with a decrease in precipitation below 450 mm. UV attenuation significantly decreased k decay in areas with precipitation ranges from 100 to 200 mm and from 1400 to 1500 mm (Fig. 4). In addition, the RR of k decay with UV attenuation showed a significant relationship with precipitation (Fig. S4b) and decreased with a decline in precipitation less than 800 mm.
3.4 Relationships between litter mass loss and nutrient release
Similar relationships between the RRs of the remaining C, N, and P and the remaining weight were observed (Fig. 5). The slope of the RRs of the remaining C and N and the remaining weight under UV attenuation were 0.85 and 1.14, respectively, but the effects of UV radiation changes on the relationships among C, N, and P and mass loss related to the ambient environment were not significant (p > 0.05). Interestingly, UV attenuation significantly promoted lignin release compared with that in the UV enhancement (p < 0.01).
Relationships of the response ratios (RRs) of the remaining nutrients and the remaining litter weight. The p values of the relationships between the remaining nutrients and litter weight under UV enhancement (UV+) and attenuation (UV−) are shown. The grey solid and dashed lines represent the regression lines between the remaining nutrients and the remaining litter weight in UV+ or UV− conditions
4 Discussion
4.1 Differential responses of litter mass loss and nutrient release to changing UV radiation
In this study, UV enhancement significantly increased litter mass loss, whereas UV attenuation reduced mass loss (Fig. 1), which is consistent with previous studies (Almagro et al. 2017; Gehrke et al. 1995; Pancotto et al. 2003; Song et al. 2013b). This is mainly because the positive effect of UV enhancement on photodecomposition is larger than its negative effects on microbial decomposer abundance and community composition (Austin and Vivanco 2006; Smith et al. 2010; Song et al. 2014). In addition, changes in litter quality under UV radiation changes also contributed to decomposition (Figs. S5 and S6). However, UV radiation changes showed no effect on C release, which was the focus of our concern, indicating that C release has a different regulatory mechanism than litter decomposition. Due to the different changes in UV in the Southern Hemisphere and Northern Hemisphere, which would cause different responses of litter decomposition and N and P releases, UV radiation eventually had a profound effect on nutrients cycling in the Southern Hemisphere and Northern Hemisphere. However, more studies are needed to determine the effects of UV radiation changes on C release from litter. The effects of changing UV radiation on litter decomposition varied with the litter types (Fig. 2), but the litter type affected only the magnitude of litter decomposition under changing UV radiation, which might explain the difference in the litter quality that affect the decomposition rate (Day et al. 2015).
Litter decomposition is a temporally dynamic process, and the sensitivity of litter decomposition to UV varies as the decomposition process progresses (Wang et al. 2017). Different results are often observed due to the experimental duration. For example, neutral or even negative responses of litter decomposition were recorded in short-term experiments under UV enhancement (Kirschbaum et al. 2011; Lambie et al. 2014), but positive effects were frequently observed in long-term experiments (Austin and Vivanco 2006; Brandt et al. 2010). In this study, the litter decomposition rate also varied with the decomposition time and exhibited three-stage temporal dynamics (Fig. 3). In the early stage (0–4 months), UV enhancement did not affect mass loss, but it significantly promoted litter decomposition during the intermediate stage (4–18 months). This indicates that UV enhancement has a positive effect on litter decomposition given a sufficient period of UV accumulation (Wang et al. 2017) and also accelerates nutrient release. However, UV attenuation significantly reduced litter decomposition during the early stage, and the effect diminished as decomposition continued.
4.2 Response of litter decomposition to UV radiation changes under different precipitation levels
Climatic (precipitation, temperature) and litter-composition variables (C:N or lignin:N) were often used to predict mass loss due to their impacts on microorganism activity (Gallo et al. 2009). However, an increasing number of studies have indicated the key role of photodecomposition on litter decomposition in arid ecosystems (Austin and Vivanco 2006; Day et al. 2015; Gallo et al. 2009). One study reported that the attenuation of radiation could reduce decomposition by as much as 60% (Austin and Vivanco 2006). In this study, k decay was higher in areas with low precipitation (Fig. 4), indicating the important role of photodecomposition (Almagro et al. 2017; Day et al. 2015; Gallo et al. 2009), and a greater decrease in litter decomposition was observed under UV attenuation in areas with 100 to 200 mm of precipitation than in high-precipitation areas (Fig. 4). This may be mainly due to the dominant role of photodecomposition in litter decomposition in the low-precipitation areas (Brandt et al. 2010; Brandt et al. 2007). In addition, when precipitation decreased from 500 to 100 mm, k decay increased (Fig. S3a) due to the positive effect of photodecomposition (Huang et al. 2017). Moreover, the RR of k decay indicated that UV attenuation reduced litter decomposition. All of the above results demonstrated that the effects of UV on litter decomposition differed under different precipitation regimes (Ballare et al. 2011).
4.3 Relationships between mass loss and nutrient release under UV radiation changes
Across all studies, mass loss showed close relationships with nutrient release, but these relationships differed between UV enhancement and UV attenuation (Fig. 5). Under UV attenuation, the RRs of the remaining C and N were significantly correlated with the remaining weight, and the slopes were 0.85 and 1.14, which indicates less losses of C and greater losses of N than mass loss during decomposition. However, the remaining lignin was negatively correlated with the remaining weight under UV attenuation, indicating that lignin release occurred more quickly than mass loss at the beginning of decomposition but increased in the later stages of decomposition (McClaugherty and Berg 1987). The relationship between lignin and remaining mass showed a significant difference between UV enhancement treatments and the control treatment (p < 0.01).
5 Conclusions
Based on global data, the current study showed that photodecomposition played an important role in litter decomposition and became stronger as precipitation decreased below 800 mm. The effects of changes in UV radiation on litter decomposition also differed depending on the leaf types and decomposition durations. Furthermore, the relationship between mass loss and nutrient release is altered under UV radiation, suggesting that future studies should consider nutrient release as well as mass loss.
References
Almagro M, Martinez-Lopez J, Maestre FT, Rey A (2017) The contribution of photodegradation to litter decomposition in semiarid mediterranean grasslands depends on its interaction with local humidity conditions, litter quality and position. Ecosystems 20:527–542
Austin AT, Ballare CL (2010) Dual role of lignin in plant litter decomposition in terrestrial ecosystems. Proc Natl Acad Sci USA 107:4618–4622
Austin AT, Vivanco L (2006) Plant litter decomposition in a semi-arid ecosystem controlled by photodegradation. Nature 442:555–558
Ballare CL, Caldwell MM, Flint SD, Robinson A, Bornman JF (2011) Effects of solar ultraviolet radiation on terrestrial ecosystems. Patterns, mechanisms, and interactions with climate change. Photoch Photobio Sci 10:226–241
Brandt LA, King JY, Milchunas DG (2007) Effects of ultraviolet radiation on litter decomposition depend on precipitation and litter chemistry in a shortgrass steppe ecosystem. Global Change Biol 13:2193–2205
Brandt LA, Bohnet C, King JY (2009) Photochemically induced carbon dioxide production as a mechanism for carbon loss from plant litter in arid ecosystems. J Geophys Res-Biogeosciences 114:G02004
Brandt L, King J, Hobbie S, Milchunas D, Sinsabaugh R (2010) The role of photodegradation in surface litter decomposition across a grassland ecosystem precipitation gradient. Ecosystems 13:765–781
Calbo J, González JA (2005) Empirical studies of cloud effects on UV radiation: a review. Rev Geophys 43:RG2002
Day TA, Guenon R, Ruhland CT (2015) Photodegradation of plant litter in the Sonoran Desert varies by litter type and age. Soil Biol Biochem 89:109–122
Gallo ME, Porras-Alfaro A, Odenbach KJ, Sinsabaugh RL (2009) Photoacceleration of plant litter decomposition in an arid environment. Soil Biol Biochem 41:1433–1441
García-Palacios P, Shaw EA, Wall DH, Hättenschwiler S (2016) Temporal dynamics of biotic and abiotic drivers of litter decomposition. Ecol lett 19:554–563
Gehrke C, Johanson U, Callaghan TV, Chadwick D, Robinson CH (1995) The impact of enhanced ultraviolet-B radiation on litter quality and decomposition processes in Vaccinium leaves from the Sub-Arctic. Oikos 72:213–222
Hedges LV, Gurevitch J, Curtis PS (1999) The meta-analysis of response ratios in experimental ecology. Ecology 80:1150–1156
Herman JR (2010) Global increase in UV irradiance during the past 30 years (1979–2008) estimated from satellite data. J Geophys Res Atmos 115:D04203
Huang G, Li Y (2017) Photodegradation effects are related to precipitation amount, precipitation frequency and litter traits in a desert ecosystem. Soil Biol Biochem 115:383–392
Huang G, Zhao HM, Li Y (2017) Litter decomposition in hyper-arid deserts: Photodegradation is still important. Sci Total Environ 601-602:784–792
King JY, Brandt LA, Adair EC (2012) Shedding light on plant litter decomposition: advances, implications and new directions in understanding the role of photodegradation. Biogeochemistry 111:57–81
Kirschbaum MUF, Lambie SM, Zhou H (2011) No UV enhancement of litter decomposition observed on dry samples under controlled laboratory conditions. Soil Biol Biochem 43:1300–1307
Lambie SM, Kirschbaum MUF, Dando J (2014) No photodegradation of litter and humus exposed to UV-B radiation under laboratory conditions: no effect of leaf senescence or drying temperature. Soil Biol Biochem 69:46–53
Lee H, Rahn T, Throop HL (2012) An accounting of C-based trace gas release during abiotic plant litter degradation. Global Change Biol. 18:1185–1195
McClaugherty CA, Berg B (1987) Cellulose, lignin and nitrogen concentrations as rate regulating factors in late stages of forest litter decomposition. Pedobiologia 30:101–112
Pancotto VA, Sala OE, Cabello M, Lopez NI, Robson TM, Ballare CL, Caldwell MM, Scopel AL (2003) Solar UV-B decreases decomposition in herbaceous plant litter in Tierra del Fuego, Argentina: potential role of an altered decomposer community. Global Change Biol. 9:1465–1474
Parton W, Silver WL, Burke IC, Grassens L, Harmon ME, Currie WS, King JY, Adair EC, Brandt LA, Hart SC, Fasth B (2007) Global-scale similarities in nitrogen release patterns during long-term decomposition. Science 315:361–364
Prescott CE (2005) Do rates of litter decomposition tell us anything we really need to know? Forest Ecol Manag 220:66–74
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing
Rutledge S, Campbell DI, Baldocchi D, Schipper LA (2010) Photodegradation leads to increased carbon dioxide losses from terrestrial organic matter. Global Change Biol 16:3065–3074
Smith WK, Gao W, Steltzer H, Wallenstein MD, Tree R (2010) Moisture availability influences the effect of ultraviolet-B radiation on leaf litter decomposition. Global Change Biol 16:484–495
Song XZ, Peng CH, Jiang H, Zhu QA, Wang WF (2013a) Direct and indirect effects of UV-B exposure on litter decomposition: a meta-analysis. PLoS One 8:e68858
Song XZ, Zhang HL, Jiang H, Donaldson LA, Wang HL (2013b) Influence of elevated UV-B radiation on leaf litter chemistry and subsequent decomposition in humid subtropical China. J Soils Sed 13:846–853
Song XZ, Zhang HL, Jiang H, Peng CH (2014) Combination of nitrogen deposition and ultraviolet-B radiation decreased litter decomposition in subtropical China. Plant Soil 380:349–359
Uselman SM, Snyder KA, Blank RR, Jones TJ (2011) UVB exposure does not accelerate rates of litter decomposition in a semi-arid riparian ecosystem. Soil Biol Biochem 43:1254–1265
Wang J, Liu LL, Wang X, Chen YW (2015) The interaction between abiotic photodegradation and microbial decomposition under ultraviolet radiation. Global Change Biol 21:2095–2104
Wang J, Yang S, Zhang B, Liu W, Deng M, Chen S, Liu L (2017) Temporal dynamics of ultraviolet radiation impacts on litter decomposition in a semi-arid ecosystem. Plant Soil 419:71–81
Williamson CE, Zepp RG, Lucas RM, Madronich S, Austin AT, Ballaré CL, Norval M, Sulzberger B, Bais AF, McKenzie RL (2014) Solar ultraviolet radiation in a changing climate. Nat Clim Change 4:434–441
Xu S, Liu L, Sayer E (2013) Variability of above-ground litter inputs alters soil physicochemical and biological processes: a meta-analysis of litterfall-manipulation experiments. Biogeosciences 10:7423–7433
Zhang K, Cheng X, Dang H, Ye C, Zhang Y, Zhang Q (2013) Linking litter production, quality and decomposition to vegetation succession following agricultural abandonment. Soil Biol Biochem 57:803–813
Funding
The study was funded by the Natural Science Basic Research Plan in Shaanxi Province of China (2020JM-096), the National Natural Science Foundation of China (41807323), the Fundamental Research Funds for the Central Universities (2452020184), and the Association of Science and Technology Youth Talents Support Project in Shaanxi Province (2019-02-04).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Heike Knicker
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yan, W., Shangguan, Z. & Zhong, Y. Responses of mass loss and nutrient release in litter decomposition to ultraviolet radiation. J Soils Sediments 21, 698–704 (2021). https://doi.org/10.1007/s11368-020-02810-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-020-02810-0