Abstract
Purpose
Soil organic carbon (SOC) dynamics are central to understanding the effects of environmental change on the carbon cycle of ecosystems. Vegetation and soil stable carbon isotope composition (δ13C), especially the difference of δ13C between surface soils and source vegetation (Δδ13C), can provide useful information about the SOC dynamics. The variations and patterns of vegetation and soil δ13C and Δδ13C along climatic and edaphic gradients were analyzed to improve the understanding of SOC dynamics in temperate grassland ecosystems.
Materials and methods
Soil and plant samples were collected along climatic and edaphic gradients. Meteorological data were extracted from a regional climate database, which was spatially interpolated based on the records at 107 climatic stations located in Inner Mongolia. δ13C, carbon and nitrogen contents of soil and plant, soil pH, soil clay, silt, and sand contents were determined. Δδ13C and soil C:N were calculated. The integrative effects of these factors were further estimated using stepwise regression, redundancy analysis (RDA) and T value biplots.
Results and discussion
As expected, soil δ13C was positively related to vegetation δ13C, and higher than the vegetation δ13C. Vegetation and soil δ13C and Δδ13C were all related to growing season precipitation (GSP) and growing season temperature (GST). However, when climatic and edaphic factors were considered together, vegetation δ13C was positively related to GST and soil C:N, soil δ13C was positively related only to GST, and Δδ13C was negatively related only to soil C:N. Unlike previous research conducted at the species level which only included C3 species, GST, instead of precipitation, was the dominant controlling factor of vegetation δ13C.
Conclusions
GST influences vegetation δ13C mainly through its effect on relative abundance of C4 plants. Soil C:N rather than climatic factors is the dominant controlling factor of SOC decomposition, and the effects of climatic factors on SOC decomposition are indirect and induced by their effects on soil C:N through influencing species composition and plant C:N. Measuring vegetation δ13C, soil δ13C, and △δ13C simultaneously is necessary to comprehensively understand how environmental changes influence ecosystem carbon cycles.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Soil organic carbon (SOC) stocks greatly exceed the amount of carbon stored in both the atmosphere and terrestrial vegetation (Scharlemann et al. 2014). It has been suggested that CO2 released from the decomposition of SOC makes an important contribution to climate change. Furthermore, global warming is likely going to increase carbon release from soil to the atmosphere by accelerating decomposition of SOC (Lin et al. 2011; Sanderman et al. 2003). Understanding of SOC dynamics has attracted much attention as it is central to the evaluation of ecosystem carbon balance, and the feedbacks of this to climate change (Kirschbaum 2000; Campbell et al. 2009; Wang et al. 2013; Averill 2014).
It is difficult to detect statistically significant changes of the SOC stock over short-time periods, due to its large pool size, slow turnover rate, and huge spatial heterogeneity (Van Groenigen et al. 2014). Moreover, studies of the microbial processes controlling SOC turnover are necessary to understand the mechanisms governing SOC dynamics (Schimel and Schaeffer 2012); however, these process-based studies can be expensive and time consuming in the field (Campbell et al. 2009). Consequently, carbon stable isotope composition (δ13C) measurements of more easily collected plant biomass and soil samples offer an alternative and relatively cost-effective method to comprehend SOC dynamics (Garten et al. 2000; Acton et al. 2013; Wang et al. 2013; Yang et al. 2015).
Some studies have estimated SOC turnover and degree of SOC decomposition and explored the mechanisms governing SOC dynamics by investigating the pattern and variation of soil δ13C along environmental gradients (Garten et al. 2000; Bai et al. 2012; Peri et al. 2012; Wang et al. 2013; Acton et al. 2013; Angelo and Pau 2015). In general, soil δ13C decreases with mean annual precipitation (MAP) (Lee et al. 2005; Feng et al. 2008; Peri et al. 2012), and increases with mean annual temperature (MAT) (Cheng et al. 2011). The effects of climatic factors on soil δ13C are mainly attributed to their effects on plant δ13C, relative abundance of C4 plants (PC4) or both (Lee et al. 2005; Feng et al. 2008; Cheng et al. 2011; Peri et al. 2012). Mostly, soil δ13C decreased with soil C:N, SOC, and soil slit content (Wang et al. 2015; Yang et al. 2015), and increased with soil sand content (Wang et al. 2013). Edaphic factors may affect soil δ13C by changing microbial activities, substrate quality and availability and stabilizing effect (Wynn et al. 2006; Xu et al. 2016).
Previous studies have demonstrated that climatic and edaphic variables have important roles in shaping patterns of soil δ13C and consequently soil carbon dynamics (Peri et al. 2012; Wang et al. 2013; Yang et al. 2015). However, the controlling factors of soil δ13C differ for various ecosystems and regions. For instance, Peri et al. (2012) investigated relationships of soil δ13C in Southern Patagonia’s native forests with 28 climatic and soil variables, and found that MAP, but not soil properties, was the controlling factor of soil δ13C. At a continental-scale in Australia, Wynn and Bird (2008) found that climate and soil texture (percent of mineral particles < 63 μm) were the dominant controllers of soil δ13C. Wang et al. (2013) found that MAT and SOC were the controlling factors of soil δ13C in alpine meadow of the Tibetan Plateau.
Compared with soil δ13C, the difference of δ13C between surface soils and source plants (Δδ13C) may be a more direct and profound indicator of soil carbon dynamics along environmental gradients (Yang et al. 2015). This is because soil δ13C is not only determined by the rate of SOC decomposition but also by the δ13C value of source vegetation (Ehleringer et al. 2000; Peri et al. 2012). Thus, Δδ13C can eliminate the effect of δ13C value of source vegetation on soil δ13C, and so provide a more accurate indicator of soil carbon dynamics, i.e., large ∆δ13C translates to faster SOC decomposition rates (Yang et al. 2015). However, previous studies have mostly concentrated on soil δ13C and rarely on Δδ13C (Yang et al. 2015; Wang et al. 2018). Although several studies have investigated the relationships of Δδ13C with climatic and edaphic factors in alpine grassland and temperate forest ecosystems (Garten et al. 2000; Yang et al. 2015), the results indicated that controlling factors of Δδ13C differed among ecosystems. MAT was the dominant controlling factor of Δδ13C in temperate forest ecosystems (Garten et al. 2000). Soil silt content and C:N were the dominant controlling factors of Δδ13C in alpine meadow, while in alpine steppe it was SOC (Yang et al. 2015). Moreover, the soil properties, climatic conditions, and vegetation types of these studies are very different from those of temperate grassland ecosystems. Thus, it is important to conduct related research on a temperate grassland ecosystem to determine if its relationships of Δδ13C with climate and soil factors differ from those of other ecosystems.
Xilingele League, Inner Mongolia, China, is a region with typical temperate grassland ecosystems. Thus, we conducted research in this region with the following primary objectives: (1) to verify the hypothesis that in temperate grasslands soil δ13C is positively correlated to vegetation δ13C and is higher than vegetation δ13C, as soil organic matter is mainly derived from plant inputs and SOC decomposition discriminates 13C; and (2) to quantify the individual and combined effects of climatic and edaphic variables on vegetation, soil δ13C and Δδ13C, in order to determine the controlling factors of soil carbon dynamics in temperate grasslands.
2 Materials and methods
2.1 Study area
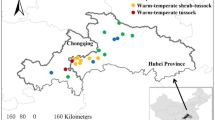
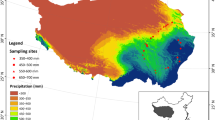
Plant and soil samples were obtained from 29 grassland sites along northeast–southwest transects located at longitude 111° 25′~ 119° 58′, latitude 41° 3′~46° 45′ in the Xilingele League, Inner Mongolia, China (Fig. 1).
The Xilingele League has an average elevation of about 1200 m. It has an arid and semi-arid continental climate with water availability being the major abiotic factor limiting plant growth. MAP ranges from 100 to 450 mm, MAT ranges from − 3 to 6 °C. There is a significant negative correlation between MAP and MAT (r2 = 0.74, P < 0.001). The natural vegetation cover is temperate grassland. The main grassland types, in transition from east to west, are temperate meadow steppe, temperate typical steppe, and temperate desert steppe (DAHV and CISNR 1996). Dominant plant species are as follows: Stipa grandis, Stipa baicalensis, Stipa krylovii, Stipa tianschanica, Leymus chinensis, Agropyron cristatum, Cleistogenes squarrosa, Cleistogenes songorica, Artemisia frigida, Salsola passerine, Salsola arbuscula, Chenopodium glaucum, Allium bidentatum, and Allium mongolicum (DAHV and CISNR 1996). For more detailed information, see Table A1 - ESM. The soils are mostly chernozems, kastanozems, calcisols, and cambisols (Fu et al. 2011).
2.2 Sampling
Plant and soil samples were obtained in August 2015. To minimize influences of short-term human disturbance, such as grazing, sampling sites were at least 500 m away from human habitation, fenced, and not grazed in the sampling year. Sites were separated from each other by about 50 km and their geographic positions were recorded using GPS (Garmin, Montana 650, USA). At each site, an approximately 10 m × 10 m plot was established, and within that plot, three 1 m × 1 m quadrats were randomly located. All aboveground standing plant biomass in each quadrat was cut to ground level and put in a large envelope. Three replicate soil samples from the top 10 cm of each quadrat were collected using a 70-mm inner diameter soil auger.
2.3 Sample analysis
All aboveground plant biomass samples were washed with deionized water to remove dust particles, de-enzymed at 105 °C for 15 min, oven-dried at 80 °C to constant weight, and then were ground and thoroughly mixed using a plant grinding machine (FW100, China). Finally, in preparation for analysis, subsamples of about 20 g were finely ground using a ball mill (Retsch MM400, Germany). After air-drying, soil samples were sieved to 2 mm, and plant residues were removed. Before isotope measurements, soil samples were ground to fine powder as for plant samples, and treated with 0.1 N HCl solution for 24 h at room temperature to remove any carbonates, then washed with distilled water and finally oven-dried at 65 °C to constant weight.
The carbon and nitrogen concentrations and carbon isotope compositions (δ13C, ‰) of plant and soil samples were determined by a coupled elemental analyzer (FlashEA1112; Thermo Electron) and isotope ratio mass spectrometer (Finnigan MAT-253; Thermo Electron). δ13C values of samples were calculated as:
where Rsample and Rstandard, respectively, are the 13C/12C ratios in the sample and the standard. The universally accepted standard of Vienna Pee Dee Belemnite (VPDB) was used. The precision for isotope composition measurement was 0.1‰.
Percentages of clay, silt, and sand contents in the soil samples were measured using a particle size analyzer (Malvern Mastersizer 2000; Malvern Instruments Ltd., Worcestershire, UK) after removal of organic matter and calcium carbonates by adding 30% H2O2 and heating at 100 °C for 45 min. Soil pH was determined using a pH electrode in a mixture of soil and water, with a water:soil ratio of 2.5:1 (Bao 2005).
2.4 Meteorological data
Mean monthly temperature (MMT), MAT, mean monthly precipitation (MMP), and MAP for each sampling site were extracted from a regional climate database with 1-km spatial resolution. This database was spatially interpolated based on the records of 107 climatic stations located in Inner Mongolia using the software package Anuspline (ver. 4.2). Using MMT and MMP, we then calculated the growing season temperature (GST) and growing season precipitation (GSP) of each sample site.
2.5 Data analysis
Paired-sample T tests were conducted to compare the mean values of vegetation and soil δ13C. Simple regression analysis was applied to investigate the relationship between vegetation and soil δ13C and also the relationships of vegetation δ13C, soil δ13C, and Δδ13C with climatic and edaphic variables (that is, GSP, MAP, GST, MAT, soil pH, clay content, silt content, sand content, soil organic carbon, and soil C:N). Then, stepwise regression was used to evaluate integrative effects of climatic and edaphic variables on vegetation δ13C, soil δ13C, and Δδ13C. Further, redundancy analysis (RDA) was conducted and T value biplots were drawn to explore primary controlling factors for vegetation δ13C, soil δ13C, and Δδ13C. Data analyses were conducted using SPSS (ver.18.0; SPSS Inc., USA) and Canoco (ver. 5.0; Microcomputer Power, Ithaca, NY, USA).
3 Results
3.1 Variation of vegetation and soil δ13C among sampling sites
Soil δ13C values ranged from − 24.7 to − 19.5‰, with a mean of − 22.4 ± 0.3‰, and with 55% of these values within the range of − 24 to − 22‰ (Fig. A1a, Electronic Supplementary Material - ESM). Vegetation δ13C values ranged from − 26.4 to − 21.6 ‰, with a mean of − 24.5 ± 0.2‰, and with 69% of these values within the range of − 26 to − 24‰ (Fig. A1b - ESM).
3.2 Relationship between vegetation and soil δ13C
Soil δ13C increased linearly with vegetation δ13C (Fig. 2). There was a clear pattern of soil being 13C enriched relative to the source vegetation, and this was more pronounced at sites with lower vegetation δ13C (Fig. 2). On average, △δ13C was 2.1‰ and higher than 1.3‰ for 59% of the sample sites.
3.3 Variation of vegetation δ13C, soil δ13C, and Δδ13C along climatic gradients
All soil δ13C, vegetation δ13C, and △δ13C showed linear increasing trends with GST and MAT, and decreasing trends with GSP and MAP (Fig. 3 and Fig. A2 - ESM). Moreover, the relationships of soil δ13C with climate factors were stronger than those of vegetation δ13C (Fig. 3).
3.4 Variation of vegetation δ13C, soil δ13C, and Δδ13C along edaphic gradients
Soil δ13C was positively related to soil pH (Fig. 4a), and negatively related to SOC content and soil C:N (Fig. 4b, c). Vegetation δ13C was not related to soil pH, SOC content, and soil C:N (Fig. 4d~f). △δ13C was negatively related to SOC content and soil C:N (Fig. 4h, i), but was not related to soil pH (Fig. 4g). None of vegetation δ13C, soil δ13C, and △δ13C were related to soil texture characteristics (clay, slit, and sand content; Fig. A3 - ESM).
3.5 Integrative effects of climatic and edaphic variables on vegetation δ13C, soil δ13C, and Δδ13C
When the integrative effects of climatic and edaphic variables were evaluated in stepwise regression analyses, only GST entered the model for soil δ13C and explained 74% of the variation in soil δ13C (soil δ13C = − 38.38 + 0.96 × GST, r2 = 0.74). GST and soil C:N entered the model for vegetation δ13C and together explained 45% of the variation in vegetation δ13C (vegetation δ13C = − 41.59 + 0.74 × GST + 0.61 × C:N, r2 = 0.45). Only soil C:N entered the model for △δ13C and explained 51% of the variation in △δ13C (△δ13C = 9.71–0.97 × C:N, r2 = 0.51).
The ordination diagram of RDA showed that most variation of δ13C was explained by climatic factors, soil C:N and SOC, but not by soil texture (Fig. 5a). Further, T value biplots of RDA showed that GST was related positively to soil and vegetation δ13C, but was not related significantly to △δ13C (Fig. 5b), soil C:N was related negatively to △δ13C and positively to vegetation δ13C, but was not related significantly to soil δ13C (Fig. 5e). None of GSP, SOC, clay content, silt content, and soil pH were related significantly to soil δ13C, vegetation δ13C or △δ13C (Fig. 5 c, d, and f~h). There was no T value biplot for sand content as the relationships between sand content and δ13C was too weak. Thus, the results of RDA confirm the results of stepwise regression analyses.
Ordination diagram and T value biplots of redundancy analysis (RDA) plotted with the first axis shown in horizontal (X) direction and second axis shown in vertical (Y) direction. Response variables (soilδ13C, vegetation δ13C, and △δ13C) were displayed as solid arrows, explanatory variables (GST, growing season temperature; GSP, growing season precipitation; SOC, soil organic carbon content; C:N, soil carbon:nitrogen ratio; clay, soil clay content; silt, soil slit content; sand, soil sand content; soil pH) were displayed as hollow arrows. Ordination diagram (a); T value biplots with Van Dobben Circle drawn for GST (b), GSP (c), SOC (d), C:N (e), clay (f), silt (g), and soil pH (h). Response variables with arrows that end in Van Dobben Circles had significant relationships with particular explanatory variables
4 Discussion
4.1 Potential mechanisms for soil 13C enrichment relative to vegetation
As expected, soil δ13C was higher than vegetation δ13C and this is consistent with other findings (Peri et al. 2012; Yang et al. 2015). An explanation for soil 13C enrichment relative to vegetation is the Suess effect (Revelle and Suess 1957). This arises from a shift to lighter C in the atmosphere due to the burning of fossil fuels (Garten et al. 2000). The maximum of the Suess effect on 13C-CO2 is about 1.3% (Graven et al. 2017). But, the mean △δ13C of our study was 2.1‰, suggesting that the Suess effect cannot be the only mechanism for soil 13C enrichment in our study. Thus, other mechanisms, such as isotopic fractionation during SOC decomposition, could also play an important role (Yang et al. 2015).
Soil δ13C increased linearly with vegetation δ13C (Fig. 2), a result in accord with previous observations in alpine grasslands and temperate forests (Peri et al. 2012; Yang et al. 2015). This indicates that the positive relationship between soil δ13C and vegetation δ13C is common to various ecosystems and that soil δ13C reflects the δ13C of source plants due to soil organic matter being derived mainly from plant inputs.
4.2 Effects of climate and edaphic factors on vegetation δ13C
Although GST, MAT, GSP, and MAP were all related significantly to vegetation δ13C (Fig. 3 and Fig. A2 - ESM), when they were considered together, only GST was related to vegetation δ13C. Firstly, MAT and GST are both expressions of temperature, but GST can more accurately reflect the effect of temperature on vegetation δ13C than MAT, because MAT largely depends on winter temperatures which always remain below biological zero in the region of the study and have no effect on growing vegetation. Secondly, this result indicated that temperature is the dominant controlling factor for vegetation δ13C, a result inconsistent with those showing that precipitation is the dominant controlling factor for δ13C of C3 species in Inner Mongolia (Wittmer et al. 2008; Zhou et al. 2016). The reason for this inconsistency is that the previous research was conducted at the species-level and only included C3 species, whereas the present study was conducted at the community-level and included both C3 and C4 species. There is a significant difference of δ13C between C3 and C4 species, with mean δ13C values of − 12‰ for C4 species, and − 26‰ for C3 species (O'Leary 1981). Thus, when there are only C3 species present, precipitation is the major determinant of plant δ13C in the Inner Mongolia region (Zhou et al. 2016). However, when both C3 and C4 species are present, the relative abundance of C4 plants (PC4) is the major determinant of vegetation δ13C in the region (Chen et al. 2007). Moreover, there is abundant evidence that PC4 is primarily controlled by temperature and it has been found that PC4 increased with GST in Inner Mongolia grassland ecosystems (Wittmer et al. 2010; Auerswald et al. 2012). Thus, at the community-level, GST is the dominant climatic factor determining vegetation δ13C in the temperate grasslands of Inner Mongolia.
Although there was no correlation between vegetation δ13C and soil C:N in simple regression (Fig. 4f), when the climate and edaphic factors were considered together, vegetation δ13C was positively related to soil C:N (Fig. 5e). The mechanisms underlying the positive correlation between vegetation δ13C and soil C:N are not clear. Possibly it is because soil C:N is positively related to PC4. To test this hypothesis, data on PC4 across soil C:N gradients in our study area is required. However, there was significantly positive correlation between plant and soil C:N (P = 0.007) in our study, and leaf C:N of C4 species was significantly higher than that of C3 species (Zheng and Shangguan 2007), which supports our hypothesis above.
4.3 Effects of climate and edaphic factors on △δ13C
Although there were significant positive relationships of △δ13C with GST, MAT, GSP. and MAP in simple regression (Fig. 3 and Fig. A2 - ESM), when the climate and edaphic factors were considered together, the climate factors were not significantly related to △δ13C (Fig. 5b, c). This indicates that compared with edaphic factors, climate factors are not the predominant controlling factors of SOC decomposition. This result is inconsistent with the finding of Peri et al. (2012), but is in accord with the finding of Yang et al. (2015) that edaphic rather than climatic factors controlled over △δ13C in alpine grasslands on the Tibetan Plateau, i.e., △δ13C regulated by soil C:N and silt content. We found that △δ13C was regulated only by soil C:N as shown by a negative association between C:N and △δ13C (Fig. 5). This negative association could be largely driven by the higher rate of SOC decomposition in soils with lower C:N ratios (Yang et al. 2015).
It is known that in many ecosystems, soil texture can play an important role in regulating SOC dynamics. Generally, fine textured soils can increase physical and hydrological protection of SOC by inhibiting decomposition, and stabilizing SOC (Schimel et al. 1994; Wynn et al. 2006; Xu et al. 2016). This would reduce gaseous carbon losses to the atmosphere and the leaching of dissolved organic carbon leading to lower △δ13C. Indeed, previous studies have found that △δ13C was negatively related to soil silt content (Powers and Schlesinger 2002; Yang et al. 2015). However, △δ13C was not related to soil silt content or soil clay or sand content in our study (Fig. A3 - ESM). Wynn et al. (2006) found that SOC decomposition was regulated by soil texture in some climatic regions, but not in others. Furthermore, previous studies have demonstrated that the importance of clay content on SOC decomposition could vary across soils with different clay mineralogy (Spain 1990; Percival et al. 2000). Thus, the absence of association between △δ13C and soil texture might be attributed to the climate and the physical and chemical properties of clay and silt minerals in our study area. Clearly, these hypotheses need to be further considered and tested in future studies.
Based on the results and analysis above, we suggest that climate factors might indirectly affect SOC decomposition through their effects on species composition and plant C:N, and subsequently soil C:N. The significant positive relationships between plant and soil C:N (P = 0.007), and significant relationships between climate factors and plant and soil C:N (GST vs. plant C:N, P < 0.001; GSP vs. plant C:N, P < 0.001; GST vs. soil C:N, P < 0.001; GSP vs. soil C:N, P < 0.001) support this suggestion.
It is relevant to note that nitrogen deposition may affect △δ13C by changing plant species composition and microbial activity (Berendse et al. 2001; Waldrop et al. 2004). Thus, it is necessary to investigate the relationship between nitrogen deposition and △δ13C in future. Moreover, another factor requiring attention is that: as it is difficult to obtain source vegetation δ13C information, present vegetation δ13C is mostly used as proxy for source vegetation δ13C. In this context, using Δδ13C to accurately assess soil carbon dynamic is based on the assumption that the present vegetation δ13C reflects the source vegetation δ13C of soil. Thus, in certain situations, i.e., where there has recently been large plant composition (especially C4/C3 abundance) transformation, this approach is not entirely satisfactory for understanding SOC dynamics. In regard to consideration of interannual variation of vegetation δ13C, the average value of vegetation δ13C for several years should more accurately reflect the source vegetation δ13C. Unfortunately, the present study only considered vegetation δ13C for 1 year.
4.4 Effects of climate and edaphic factors on soil δ13C
We found that GST had a positive effect on soil δ13C. An explanation for this is that as SOC is derived mainly from plant inputs, SOC largely preserves the isotopic signal of the plants it derives carbon from, and that GST has a positive effect on vegetation δ13C by its effect on PC4 (Figs. 3c and 5b). Thus, GST can indirectly affect soil δ13C through its effect on vegetation δ13C. However, unexpectedly the relationship between soil δ13C and GST was stronger than that between vegetation δ13C and GST. This indicates that GST controls soil δ13C not only through its effects on vegetation δ13C but also through other mechanisms, e.g., SOC decomposition which can, to some extent, lead to 13C enrichment, resulting in higher soil δ13C (Garten et al. 2000). Based on the relationships of △δ13C with climatic and edaphic factors, we suggest that climate factors might indirectly affect SOC decomposition through their effects on plant species composition and plant C:N, and then soil C:N. This suggestion is supported by the negative relationship between GST and soil C:N (P < 0.001).
As there was a significant relationship between soil C:N and △δ13C (Figs. 4c and 5e), soil C:N should have an effect on SOC decomposition, and then to have an effect on soil δ13C. However, when the climatic and edaphic factors were considered together, there was no relationship between soil δ13C and soil C:N (Fig. 5e). This may be because soil C:N had a positive effect on vegetation δ13C and a negative effect on SOC decomposition (Fig. 5e), i.e., the effect of soil C:N on soil δ13C was eliminated, due to its offsetting effects on δ13C value of source vegetation and SOC decomposition.
5 Conclusions
Firstly, this study finds a positive association between soil and plant δ13C and soil 13C enrichment relative to vegetation in temperate grassland in the Inner Mongolia, that is similar to previously observed isotopic patterns in alpine grasslands. Secondly, the study demonstrates that GST is the dominant controlling factor of vegetation δ13C, most likely through its effect on PC4. More importantly, by exhibiting that △δ13C was regulated by soil C:N, our results indicates that edaphic rather than climatic factors are the dominant controlling factor of SOC decomposition. Further, the effects of climatic factors on △δ13C are indirect and induced by their effects on soil C:N through influencing species composition and plant C:N. Finally, we suggest that compared with soil δ13C, △δ13C may provide more accurate information about soil carbon dynamics, i.e., larger △δ13C means faster SOC decomposition rate, as soil δ13C may either underestimate or overestimate the effects of some factors on SOC decomposition. This emphasizes that measuring plant δ13C, soil δ13C, and △δ13C simultaneously are necessary to comprehensively understand how environmental change influences the carbon cycles of ecosystems.
References
Acton P, Fox J, Campbell E, Rowe H, Wilkinson M (2013) Carbon isotopes for estimating soil decomposition and physical mixing in well-drained forest soils. J Geophys Res Biogeosci 118:1532–1545
Angelo CL, Pau S (2015) Root biomass and soil δ13C in C3 and C4 grasslands along a precipitation gradient. Plant Ecol 216:615–627
Auerswald K, Wittmer MHOM, Bai Y, Yang H, Taube F, Susenbeth A, Schnyder H (2012) C4 abundance in an Inner Mongolia grassland system is driven by temperature–moisture interaction, not grazing pressure. Basic Appl Ecol 13:67–75
Averill C (2014) Mycorrhiza-mediated competition between plants and decomposers drives soil carbon storage. Nature 505:543–545
Bai E, Boutton TW, Liu F, Ben Wu X, Hallmark CT, Archer SR (2012) Spatial variation of soil δ13C and its relation to carbon input and soil texture in a subtropical lowland woodland. Soil Biol Biochem 44:102–112
Bao S (2005) Agricultural and chemistry analysis of soil. Agriculture Press, Beijing
Berendse F, Van Breemen N, Rydin H, Buttler A, Heijmans M, Hoosbeek MR, Lee JA, Mitchell E, Saarinen T, Vasander H (2001) Raised atmospheric CO2 levels and increased N deposition cause shifts in plant species composition and production in Sphagnum bogs. Glob Chang Biol 7:591–598
Campbell JE, Fox JF, Davis CM, Rowe HD, Thompson N (2009) Carbon and nitrogen isotopic measurements from southern Appalachian soils: assessing soil carbon sequestration under climate and land-use variation. J Environ Eng 135:439–448
Chen SP, Bai YF, Lin GH, Huang JH, Han XG (2007) Variations in δ13C values among major plant community types in the Xilin River Basin, Inner Mongolia, China. Aust J Bot 55:48–54
Cheng X, Luo Y, Xu X, Zhang Q (2011) Soil organic matter dynamics in a North America tallgrass prairie after 9 yr of experimental warming. Biogeosciences 8:1487–1498
Department of Animal Husbandry and Veterinary, Institute of Grasslands, Chinese Academy of Agricultural Sciences (DAHV), Commission for Integrated Survey of Natural Resources, Chinese Academy of Sciences (CISNR) (1996) Rangeland resources of China. China Agricultural Science and Technology Press, Beijing
Ehleringer JR, Buchmann N, Flanagan LB (2000) Carbon isotope ratios in belowground carbon cycle processes. Ecol Appl 10:412–422
Feng ZD, Wang LX, Ji YH, Guo LL, Lee XQ, Dworkin SI (2008) Climatic dependency of soil organic carbon isotopic composition along the S–N transect from 34°N to 52°N in central-East Asia. Palaeogeogr Palaeoclimatol Palaeoecol 257:335–343
Fu Y, Yu Y, Yao H (2011) Changes of soil organic carbon of grassland in the Xilinguole, Inner Mongolia from 2000 to 2007. Pratacult Sci 28:1589–1597
Garten CT, Cooper LW, Post WM, Hanson PJ (2000) Climate controls on forest soil C isotope ratios in the southern Appalachian Mountains. Ecology 81:1108–1119
Graven H, Allison CE, Etheridge DM, Hammer S, Keeling RF, Levin I, Meijer HA, Rubino M, Tans PP, Trudinger CM (2017) Compiled records of carbon isotopes in atmospheric CO2 for historical simulations in CMIP6. Geosci Model Dev 10:4405–4417
Kirschbaum MUF (2000) Will changes in soil organic carbon act as a positive or negative feedback on global warming? Biogeochemistry 48:21–51
Lee X, Feng Z, Guo L, Wang L, Jin L, Huang Y, Chopping M, Huang D, Jiang W, Jiang Q (2005) Carbon isotope of bulk organic matter: a proxy for precipitation in the arid and semiarid Central East Asia. Glob Biogeochem Cycles 19:2833–2845
Lin X, Zhang Z, Wang S, Hu Y, Xu G, Luo C, Chang X, Duan J, Lin Q, Xu B (2011) Response of ecosystem respiration to warming and grazing during the growing seasons in the alpine meadow on the Tibetan Plateau. Agric For Meteorol 151:792–802
O'Leary M (1981) Carbon isotope fractionation in plants. Phytochemistry 20:553–567
Percival HJ, Parfitt RL, Scott NA (2000) Factors controlling soil carbon levels in New Zealand grasslands: is clay content important? Soil Sci Soc Am J 64:1623–1630
Peri PL, Ladd B, Pepper DA, Bonser SP, Laffan SW, Amelung W (2012) Carbon (δ13C) and nitrogen (δ15N) stable isotope composition in plant and soil in southern Patagonia’s native forests. Glob Chang Biol 18:311–321
Powers JS, Schlesinger WH (2002) Geographic and vertical patterns of stable carbon isotopes in tropical rain forest soils of Costa Rica. Geoderma 109:141–160
Revelle R, Suess HE (1957) Carbon dioxide exchange between atmosphere and ocean and the question of an increase of atmospheric CO2 during the past decades. Tellus 9:18–27
Sanderman J, Amundson RG, Baldocchi DD (2003) Application of eddy covariance measurements to the temperature dependence of soil organic matter mean residence time. Glob Biogeochem Cycles 17:193–228
Scharlemann JP, Tanner EV, Hiederer R, Kapos V (2014) Global soil carbon: understanding and managing the largest terrestrial carbon pool. Carbon Manag 5:81–91
Schimel J, Schaeffer SM (2012) Microbial control over carbon cycling in soil. Front Microbiol 3:348
Schimel DS, Braswell BH, Holland EA, Mckeown R, Ojima DS, Painter TH, Parton WJ, Townsend AR (1994) Climatic, edaphic, and biotic controls over storage and turnover of carbon in soils. Glob Biogeochem Cycles 8:279–294
Spain AV (1990) Influence of environmental conditions and some soil chemical properties on the carbon and nitrogen contents of some tropical Australian rainforest soils. Aust J Soil Res 28:825–839
Van Groenigen KJ, Qi X, Osenberg CW, Luo Y, Hungate BA (2014) Faster decomposition under increased atmospheric CO2 limits soil carbon storage. Science 344:508–509
Waldrop MP, Zak DR, Sinsabaugh RL, Gallo M, Lauber C (2004) Nitrogen deposition modifies soil carbon storage through changes in microbial enzymatic activity. Ecol Appl 14:1172–1177
Wang S, Fan J, Song M, Yu G, Zhou L, Liu J, Zhong H, Gao L, Hu Z, Wu W (2013) Patterns of SOC and soil 13C and their relations to climatic factors and soil characteristics on the Qinghai–Tibetan Plateau. Plant Soil 363:243–255
Wang G, Jia Y, Wei L (2015) Effects of environmental and biotic factors on carbon isotopic fractionation during decomposition of soil organic matter. Sci Rep 5:11043
Wang C, Houlton BZ, Liu D, Hou J, Cheng W, Bai E (2018) Stable isotopic constraints on global soil organic carbon turnover. Biogeosciences 15:987–995
Wittmer M, Auerswald K, Tungalag R, Bai YF, Schaufele R, Schnyder H (2008) Carbon isotope discrimination of C3 vegetation in Central Asian grassland as related to long-term and short-term precipitation patterns. Biogeosciences 5:913–924
Wittmer M, Auerswald K, Bai YF, Schaufele R, Schnyder H (2010) Changes in the abundance of C3/C4 species of Inner Mongolia grassland: evidence from isotopic composition of soil and vegetation. Glob Chang Biol 16:605–616
Wynn JG, Bird MI (2008) Environmental controls on the stable carbon isotopic composition of soil organic carbon: implications for modelling the distribution of C3 and C4 plants, Australia. Tellus Ser B-chem Phys Meteorol 60:604–621
Wynn JG, Bird MI, Lins V, Emilie GC, John C, Berry SL (2006) Continental-scale measurement of the soil organic carbon pool with climatic, edaphic, and biotic controls. Glob Biogeochem Cycles 20:630–637
Xu X, Shi Z, Li D, Rey A, Ruan H, Craine JM, Liang J, Zhou J, Luo Y (2016) Soil properties control decomposition of soil organic carbon: results from data-assimilation analysis. Geoderma 262:235–242
Yang Y, Ji C, Chen L, Ding J, Cheng X, Robinson D (2015) Edaphic rather than climatic controls over 13C enrichment between soil and vegetation in alpine grasslands on the Tibetan Plateau. Funct Ecol 29:839–848
Zheng S, Shangguan Z (2007) Spatial patterns of leaf nutrient traits of the plants in the loess plateau of China. Trees 21:357–370
Zhou YC, Cheng XL, Fan JW, XU XY (2016) Spatial pattern of foliar δ13C in C3-dominated grasslands and its responses to climatic factors in Inner Mongolia, China. J Northeastern Univ (Nat Sci) 37:273–279
Acknowledgements
We thank Hongli Zhang for her assistance with field sampling and laboratory analyses.
Funding
The study was supported by the National Natural Science Foundation of China (No. 31400413); the Fundamental Research Funds for the Central Universities of China (No. N130301001, N160102001); the Doctoral Startup Foundation of Liaoning Province (No. 20141017); and the Visiting Scholar Program of China Scholarship Council (No. 201706085003).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Zhihong Xu
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Fig. A1
Frequency distribution of soil and vegetation δ13C values from 29 temperate grassland sites on the Xilingele League, Inner Mongolia, China (DOCX 63 kb)
Fig. A2
Relationships of soil δ13C, vegetation δ13C and △δ13C with MAT and MAP in temperate grasslands on the Xilingele League, Inner Mongolia, China. MAT, mean annual temperature; MAP, mean annual precipitation (DOCX 107 kb)
Fig. A3
Relationships of soil δ13C, vegetation δ13C and △δ13C with soil texture in temperate grasslands on the Xilingele League, Inner Mongolia, China (DOCX 74 kb)
ESM 1
(DOCX 18 kb)
Rights and permissions
About this article
Cite this article
Zhou, Y., Zhang, W., Cheng, X. et al. Factors affecting 13C enrichment of vegetation and soil in temperate grasslands in Inner Mongolia, China. J Soils Sediments 19, 2190–2199 (2019). https://doi.org/10.1007/s11368-019-02248-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-019-02248-z