Abstract
Purpose
The main purpose of this study was to demonstrate the utility of the sediment fingerprinting approach to apportion surface-derived sediment, and then age date that portion using short-lived fallout radionuclides. In systems where a large mass of mobile sediment is in channel storage, age dating provides an understanding of the transfer of sediment through the watershed and the time scales over which management actions to reduce sediment loadings may be effective.
Materials and methods
In the agricultural Walnut Creek watershed, Iowa, the sediment-fingerprinting approach with elemental analysis was used to apportion the sources of fine-grained sediment (croplands, prairie, unpaved roads, and channel banks). Fallout radionuclides (7Be, 210Pbex) were used to age the portion of suspended sediment that was derived from agricultural topsoil. Age dating was performed at two different scales: 210Pbex which can date sediment to ~ 85 years and 7Be to ~ 1 year.
Results and discussion
Sediment fingerprinting results indicated that the majority of suspended sediment is derived from cropland (62%) with streambanks contributing 36%, and prairie, pasture, and unpaved roads each contributing ≤ 1%. The topsoil–derived portion of sediment (primarily agriculture) dated using 210Pbex has ages ranging from 1 to 58 years, and using 7Be, a component of much younger sediment that yields ages ranging from 44 to 205 days. The occurrence of 7Be indicates that some portion of the sediment is young, on the order of months, whereas the dating based on 210Pbex indicates that some of the surface-derived sediment has been in channel storage for decades. Published studies in Walnut Creek indicate that a large component of sediment is stored in the channel bed.
Conclusions
We conclude that the 210Pbex-based ages are a reasonable estimate for the mean age of the surface-derived fraction and that 7Be activities are evidence that there is a smaller fraction of very young sediment in the stream. We propose a geomorphic model where agricultural soil is delivered to the channel and conveyed to the watershed outlet at three time scales: a geologic-millennial time scale, decades, and a young time scale (< 1 year).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Worldwide, fine-grained sediment (<63 μm) is a major pollutant impacting aquatic habitat and infrastructure through: burial of substrate (Wood and Armitage 1997; Izagirre et al. 2009), light attenuation (Wilber and Clarke 2001; Langland and Cronin 2003), clogging of fish gills (Cavanagh et al. 2014), altering oxygen demand and effect on egg gestation (Collins et al. 2014), transporting pollutants (Warren et al. 2003; Owens et al. 2005; Yi et al. 2008), sedimentation in reservoirs (Liu et al. 2017), and clogging of municipal water filtration systems (McHale and Siemion 2014). Identifying the sources of this sediment and quantifying its contribution to the total load of streams is information useful not only to management agencies but also to our understanding of processes occurring in the critical zone, the near surface layer that covers all land on Earth. Sediment fingerprinting offers an effective tool for management agencies to identify sediment inputs. Management agencies are also interested in the volume (or mass) of sediment in channel storage and the age of this sediment, as this can inform managers of the timescales when sediment controls may show an effect. Both the sources of sediment and its residence time are important critical zone processes. Sediment fingerprinting provides information on the contributions from topsoil erosion as well as the delivery of this sediment to the fluvial system, which are critical zone processes that help us understand agricultural sustainability and aquatic ecosystem stressors.
Attempting to relate sediments to their upstream sources is challenging. However, there have been advances using geochemical and mineralogical data, including radionuclides and stable isotopes, to “fingerprint” sediment sources (Collins et al. 1997, 2010, 2017; Papanicolaou et al. 2003; Walling 2013; Evrard et al. 2016; Gellis et al. 2016; Manjoro et al. 2017). The sediment fingerprinting approach is based on characterizing each of the potential sediment sources within a watershed by a composite fingerprint, defined by a number of physical or geochemical properties of the source materials and comparing the fingerprint of sampled suspended or bed sediment (target sediment) with the fingerprints of the potential sources. By using a statistical model, it is possible to estimate the relative contributions from different source types (Collins et al. 2010; Haddadchi et al. 2013; Gellis et al. 2016). Results from sediment fingerprinting studies have been used to guide management actions to reduce erosion and sediment input worldwide (Caitcheon et al. 2006; Evans et al. 2006; Gellis and Walling 2011; Mukundan et al. 2012; Gellis et al. 2016).
Distinction between sediment transit times and residence times can be confusing (Wallbrink et al. 1998; Smith et al. 2014; Moody 2017). As sediment travels from its origin to a point of interest in the watershed, it can be deposited in storage on upland surfaces (i.e., colluvial slopes) (Smith et al. 2014), in channel storage (i.e., the active channel bed, bars) (Estrany et al. 2011; Harper et al. 2017), or on floodplains (Walling et al. 1998; Fryirs and Brierley 2001) for periods of time that range from days or weeks to millennia (Lancaster and Casebeer 2007; Pizzuto et al. 2014; Hoffmann 2015). The sediment transit time is the time it takes for sediment to travel from a starting point in the watershed to an endpoint where sediment leaves the area of interest (Matisoff et al. 2005). Residence time is the mean time sediment particles spend in a storage reservoir. Determining the transit time and residence time of sediment depends on the framework or scale of interest. An upland transit time would examine the average time it takes for a sediment particle to leave an upland source and enter the channel (Slattery et al. 2002). At the watershed scale, a sediment transit time could include the time sediment spends in all storage reservoirs, i.e., on hillslopes and on the channel bed (Bonniwell et al. 1999; Hoffmann 2015). Residence time for sediment is often computed as the mass or volume of sediment in a storage reservoir divided by the average sediment transport rate (Kelsey et al. 1987; Malmon et al. 2002). The residence time of sediment can refer to the time spent in a discrete depositional reservoir, i.e., bed or floodplain (Skalak and Pizzuto 2010; Fryirs 2013), or the entire watershed (Dominik et al. 1987; Evrard et al. 2010). At the watershed scale, sediment residence time would include time spent in both storage and transport through its journey down the fluvial system (Pizzuto et al. 2014). For this paper, the transit time is defined as the time from when sediment is delivered to the channel to when it arrives at a (downstream) sample location. The transit time includes the time sediment is in transport and channel storage but does not include the time spent in floodplain or upland storage.
The U.S. Geological Survey did an intensive study of water quality and ecology in 100 wadeable midwestern US streams in 2013 (Van Metre et al. 2018). In 99 of those streams, Gellis et al. (2017) used 7Be to age fine-grained suspended sediment and bed material to < 1 year and 210Pbex to apportion sediment sources between land surface and channel (streambanks and sediment in channel storage). Results indicated that channel sources (streambanks and sediment in channel storage) dominate; 79% of the sites had > 50% channel-derived sediment. The age of the surface-derived fraction of bed sediment ranged from 0 to 174 days with a median of 82 days and suspended-sediment sample ages ranged from 0 to 84 days with a median of 41 days. Gellis et al. (2017) noted that their approach could not determine whether the channel-derived sediment originated from streambanks or from older topsoil–derived sediment that had been in channel storage for several decades or longer. One of the 99 streams was Walnut Creek in central Iowa and is the subject of this study. Our objective was to combine the sediment fingerprinting approach, which apportions sediment into channel banks and surface-derived sediment (pasture, prairie, cropland, and unpaved roads) based on elemental analyses, with fallout radionuclides to determine ages of the surface-derived portion of this sediment.
1.1 Sediment source determination
The sediment-fingerprinting approach entails the identification of specific sediment sources through the establishment of a minimal set of physical and (or) chemical properties that uniquely define each source in the watershed. Sediment sources are typically separated into channel sources (streambanks, channel bed, and bars) and land-surface sources (classified by land use, land cover, or by geology and soil units) (Collins and Walling 2002; Collins et al. 2017). Properties that have been used to identify sediment sources include elemental analysis (Collins et al. 2012a; Gellis and Noe 2013), radionuclides (Belmont et al. 2014; Evrard et al. 2016), stable isotopes (Fox 2009; Laceby et al. 2015), compound-specific stable isotope (CSSI) analysis (Zhang et al. 2017), and soil enzyme activity (Nosrati et al. 2011).
Target sediment samples collected at a defined watershed outlet represent a composite of the source properties (fingerprints). Target sediment can be suspended sediment, channel bed, and floodplain deposits (Walling 2005; Martínez-Carreras et al. 2010; Collins et al. 2012b; Haddadchi et al. 2013; Miller et al. 2015; Gellis et al. 2016). Target samples can also include lake and reservoir cores, which enable interpretations of historical changes in sediment source from natural and anthropogenic forcings (Foster and Walling 1994; Foster et al. 2008; Belmont et al. 2011; D’Haen et al. 2013; Manjoro et al. 2017).
The apportionment of sediment sources contributing to the target sediment is determined using various statistical procedures (Collins et al. 2010; Haddadchi et al. 2013; Miller et al. 2015). Generally, two statistical approaches to apportion target sediment to its sources have appeared in the literature: a tracer reduction approach and a Bayesian approach. The tracer reduction approach uses a multi-step procedure, beginning with tests to remove potential tracers that cannot differentiate between sources using tests such as a Mann-Whitney or Kruskal-Wallis, then using stepwise discriminate function analysis (DFA) to identify an optimum composite fingerprint (Collins et al. 1997, 1998; Martínez-Carreras et al. 2010; Gellis and Noe 2013). The Bayesian approach involves probabilistic treatment of the data which allows for understanding uncertainties in the spatial and temporal variability in source and riverine sediment geochemistry (Fox and Papanicolaou 2008; Stewart et al. 2014; Cooper and Krueger 2017). Error analysis to test the sediment fingerprinting results has included a relative error comparison or goodness of fit (GOF) model (Collins et al. 1998; Manjoro et al. 2017) where the sample fingerprint properties of the target samples are compared with the corresponding values predicted by the model. Motha et al. (2003) and Collins and Walling (2007a) used a Monte Carlo design where samples of the tracer properties are randomly drawn from their corresponding normal distributions and run through the mixing model to test variability in source apportionment results.
1.2 Age determination
Short-lived fallout radionuclides have been used to examine sediment residence times in watersheds and ages of channel sediment (Dominik et al. 1987; Wallbrink et al. 2002; Evrard et al. 2010; Mabit et al. 2014). Wallbrink et al. (2002), using 210Pbex and 137Cs, estimated residence times of sediment in the channel for two river systems in Australia ranging from 0 to 21 years and 0 to 9 years. Dominik et al. (1987), using 7Be, 210Pbex, and 137Cs, proposed a two-box age model of residence times for the alpine Rhône River, with a geologic box where a component of topsoil particles travel slowly (800 to 1400 years) and a rapid box where high surface erosion rates for a small fraction of the sediment moves particles at short residence times between 1 and 220 days. Evrard et al. (2010) examined sediment residence times in a tropical watershed in central Mexico using 7Be, 210Pbex, and 137Cs and concluded that residence times were similar to the two-box model of Dominik et al. (1987); a geologic box where soil is transported to the watershed outlet at time scales of 5000 to 23,000 years and a rapid box, where once in the channel, sediment travels to the monitoring point from 50 to 200 days.
2 Materials and methods
2.1 Study area
The 52.6-km2 Walnut Creek watershed as defined here is a 4th-order system draining south-central Iowa in Jasper County (Fig. 1). The climate is humid continental with an average annual precipitation of 750 mm (Palmer et al. 2014). Soils in the watershed are primarily silty clay loams, silt loams, or clay loams formed in loess and till (Schilling et al. 2011). Upland areas in Walnut Creek are loess mantled pre-Illinoian till with Holocene alluvial fills (Schilling et al. 2011). Legacy or post-Euro-American-settlement sediment (nineteenth century) is common in the valley fills where channel banks are primarily composed of silt and clay (Schilling et al. 2011).
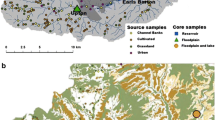
Walnut Creek, Iowa watershed study area showing location of sediment source samples and land use (Fry et al. 2011)
Walnut Creek is an agricultural basin, with 65% of the watershed in cropland, 19% in prairie, 8% in pasture, and 8% in developed land and other (Homer et al. 2015). The length of unpaved roads in the watershed obtained from 2000 and 2013 coverages (U.S. Census Bureau 2001; U.S. Department of Agriculture 2014) was 53 km or an unpaved road density of ~ 1 km−2.
Walnut Creek is one of the 99 streams selected as part of the U.S. Geological Survey’s (USGS) Midwest Stream Quality Assessment (MSQA), a study conducted by the USGS National Water-Quality Assessment (NAWQA) (Van Metre et al. 2012, 2018). The overall goals of the MSQA were to characterize water-quality stressors—contaminants, nutrients, sediment, streamflow, and habitat and ecological conditions in streams across the Midwest Region and to determine the relative effects of these stressors on aquatic organisms.
2.2 Flow, turbidity, and suspended sediment
Instantaneous discharge (10 min intervals), instantaneous suspended sediment (selected times), and turbidity (30 min intervals) were obtained from the U.S. Department of Agriculture, Walnut Creek near Vandalia, Iowa streamgage (discontinued USGS Station ID 05487550; herein called the streamgage) for the MSQA study period (23 May 2013 to 8 August 2013). Discharge, stage, and record computation followed USGS protocols and operating procedures (Kennedy 1983) which can be found in Schilling (2000). Suspended sediment was collected at least once a week during low flows using a manual dip sample, and at higher flows using isokinetic samplers with equal width increments (EWI) (Edwards and Glysson 1999). Event samples were collected using an automatic sampler to collect flow-based event composite samples comprising 100 mL subsamples of stream water taken on a discharge-volume interval of one subsample for every 18,406 m3 of flow. The 100-mL subsamples were composited in 3.8 L bottles; multiple bottles were available for large events. When the event was finished, the bottles were put in a churn splitter for mixing and a subsample was taken for suspended sediment. EWI sample results were used to determine correction coefficients to remove the bias from the grab and automatic samples. The method bias coefficient (EWI:sample) was calculated to be 0.50 for the automatic sampler suspended-sediment concentration (SSC) and 0.93 for the dip sample SSC. Suspended sediment was analyzed for concentrations at the USDA laboratory in Ames, Iowa following procedures and protocols in ASTM (2002).
Turbidity was recorded at the streamgage every 30 min using a Hydrolab DS5X sonde following standards outlined in Wagner et al. (2006). A regression model of turbidity and suspended sediment concentration (SSC) developed by USDA was used to estimate SSC during periods of missing record. A mean daily SSC was determined for low flow periods and events. On low flow days where one or more samples of SSC were obtained, samples were averaged to obtain a mean daily SSC. For periods of low flow where a SSC sample was not obtained, 10 min intervals of SSC were estimated using the time series management system (Water Information Systems by KISTERS (WISKI), Kisters North America, Citrus Heights, California, USA; www.kisters.net). For high flow days and events, SSC was estimated from the automatic sampler-collected composite sample. If the automatic sampler malfunctioned, the relation of SSC and turbidity and 30 min turbidity values were converted to SSC and averaged for the day.
Suspended-sediment daily loads were computed using the mean daily SSC and mean daily discharge, as follows:
where SSdaily is the daily suspended-sediment load (Mg day−1), Flowdaily is the mean daily flow (m3 s−1), SSCdaily is the mean daily SSC (mg L−1), and 0.0864 is a conversion coefficient based on the unit of measurement of water discharge that assumes a specific weight of 2.65 for sediment (Gray and Simões 2008).
Suspended sediment was collected for tracer analyses using passive samplers (Phillips et al. 2000) that were deployed over 14 weeks and sampled periodically (Table 1). Two pairs of PVC tubes mounted on steel struts were placed in the center of the channel with the upper tube placed on top of the lower tube. This placement of the tubes allowed flows over a range in stage to be sampled (Fig. S1, Electronic supplementary material). Sediment was retrieved from the passive samplers biweekly or after storm events and both tubes were composited into one sample; referred to herein as target samples.
The total suspended-sediment load was computed by summing the daily load for the period of each target sample and for the period of study (Table 1). Because suspended-sediment load is computed as a daily and not an hourly load, the sediment load transported for each sample period is determined from the day the sampler was deployed to the day prior to the next retrieval. The weighted sediment load is determined by dividing the sediment load for each sample by the total load for the period of study and applying this weighting to the apportionment results for each sample (Walling et al. 1999; Gellis et al. 2015). Target sample 8 retrieved on 1 August 2013 did not have enough mass (< 1 g) for radionuclide analysis; therefore, the period from 24 July 2013 to 1 August 2013 was not used to compute a total sediment load for this timeframe.
2.3 Sampling and sediment fingerprinting
The sediment-fingerprinting approach was used to determine the sources of fine-grained (< 63 μm) sediment in the Walnut Creek watershed draining to the streamgage. Nine target samples were used for source analysis between 23 May 2013 to 8 August 2013 (Table 1; sample 8 was not used). The period was chosen because it coincided with the USGS National Water Quality Assessment sampling for streams in the Midwest Region of the USA that included Walnut Creek (Van Metre et al. 2012).
Sediment-source samples were collected from pasture (n = 12), cropland (n = 13), prairie (n = 12), channel banks (n = 26), and unpaved roads (n = 6) (Fig. 1). A similar number of source samples were collected for sediment fingerprinting studies in agricultural watersheds of Chesapeake Bay (Gellis et al. 2009, 2015). Source sample locations were selected by rasterizing each of the source areas using a GIS and assigning a unique value to each cell for each source. A random number generator was used to select individual grid cells within each land use for sampling. The randomized design avoided bias in site selection and provided a random sample that is representative of land use in Walnut Creek.
Cropland, prairie, and pasture are present in Walnut Creek as large fields (10s-100s ha). In the randomly selected cropland, prairie, and pasture sites, portions of the entire field were selected for sampling that were representative of the entire field, and topsoil was collected across three to five transects each parallel to slope, ~ 30 m apart and ~ 100 m long. At each transect, topsoil was collected every 10 m from the top ~ 1.0 cm of the soil surface with a plastic hand shovel. All samples from a given site were mixed into a single sample in the field.
Each unpaved road sample was collected across three transects spaced 10 m apart that traversed the road using a hand brush and plastic dustpan and composited into one sample. Eroding streambanks were sampled by scraping the entire vertical face of the exposed streambank to a depth of ~ 1 cm with a plastic hand shovel. Three to five bank profiles spaced 10 m apart along the stream reach were sampled and composited into one sample. The channel bed is assumed to be a temporary storage reservoir of sediment originating from a variety of upstream sources (Gellis et al. 2015) and was not sampled as a source.
After collection, source samples were put on ice and transported to the Iowa State University Soil and Plant Analysis Laboratory, Ames, Iowa. Target samples were shipped on ice to the USGS MD-DC-Water Science Center in Baltimore, Maryland. Source and target samples were wet sieved with de-ionized water through a 63-μm polyester sieve to remove the sand and dried at 60 °C. The silt and clay portion (< 63 μm) of the source and target samples were split for three analyses: (1) elemental analysis using inductively coupled plasma optical emission spectrometry (ICP-OES) and inductively coupled plasma mass spectrometry (ICP-MS); (2) grain size, and (3) radionuclide analysis (7Be and 210Pbex) on target samples and for selected source samples (210Pbex).
Elemental analyses on the source and target samples were conducted at the USGS Central Mineral and Environmental Resources Science Center in Denver, Colorado, which reported 38 elemental concentrations for the source tracers and target samples used in sediment fingerprinting (Table 2; Gellis et al. 2018). Samples were dried and ground then digested at low temperature using a mixture of hydrofluoric and hydrochloric-nitric-perchloric acids, with analysis by ICP-MS (Briggs and Meier 2002). ICP-MS and ICP-OES data are acceptable if recovery of each element in reference materials analyzed with samples is within ± 15% of reference materials at five times the lower limit of determination (LOD) and if the calculated relative standard deviation (RSD) of replicates is within 15% (https://minerals.usgs.gov/science/analytical-chemistry/method17.html). Ten samples from the study were split and analyzed in replicate using the same ICP-MS and ICP-OES elemental methods as used here (Gellis et al. 2018). Samples with non-detects were not included in determining a RPD. The median relative percent difference (RPD) for the 38 elements used in sediment fingerprinting had a median RPD ranging from 0.0% (Cs) to 30.7% (Sb), and the median of the medians was 4.2%, indicating relatively good precision.
Particle size determination was performed at the USGS MD-DE-DC Water Science Center in Baltimore, Maryland using a Laser In Situ Scattering Transmissometer (LISST-100X). The LISST-100X uses laser diffraction to measure the size of sediment particles (instrument specifications can be found at http://www.sequoiasci.com/library/standards/; accessed 3 January 2017). Prior to analysis, fine sediment (< 63 μm) undergoes the following preparation procedure: an aliquot (0.0210–0.0300 g) of each sample is transferred into a 125-mL Erlenmeyer flask. A glass pipet is used to add 10 mL of a 50-g L−1 solution of hexametaphosphate (NaPO4)6 to each sample to aid in deflocculation. Each sample is placed in an ultrasonic bath for 5 min to disperse the particles and then put on a shaking table for a minimum of 16 h. The results are expressed as the median particle diameter (D50) of the sediment by the LISST software (Gellis et al. 2018).
2.4 Sediment-source apportionment using the sediment-fingerprinting approach
Several analytical and statistical steps were used to determine which tracers are most effective in defining sediment sources using the sediment-fingerprinting approach (Gellis et al. 2015, 2016). The sediment source assessment tool (Sed_SAT) was used to assist the user through all necessary statistical steps in sediment fingerprinting (Gorman Sanisaca et al. 2017). Sed_SAT is written in the statistical language R (R Core Team 2016) using Microsoft Access© (Gorman Sanisaca et al. 2017; available at https://my.usgs.gov/bitbucket/projects/SED). A full description of the decision tree is available within Gellis et al. (2016) and Gorman Sanisaca et al. (2017). The statistical defaults in Sed_SAT were used to apportion sediment in Walnut Creek. Sed_SAT follows a five-step procedure to apportion sediment: (1) option to remove outliers, (2) option to perform grain size and organic content corrections to the source data, (3) a bracket test for the conservativeness of the tracer, (4) stepwise discriminant function analysis (DFA), and (5) percent contribution from each source using an “unmixing model” (modified from Collins et al. 2010) as follows:
and
where RE is the relative minimum error term; Ci is the concentration of tracer property after size and organic correction factors are applied (i) to the suspended sediment sample; Ps is the the relative contribution from source category (s); Ssi is the mean concentration of tracer property (i) in source category (s); Wi is the tracer discriminatory weighting; n is the number of fingerprint properties comprising the optimum composite fingerprint; and m is the number of sediment source categories
where Wi is the tracer discriminatory weighting for tracer i and Pi is the percent of source-type samples classified correctly using tracer i. The percent of source-type samples classified correctly is a standard output from the DFA statistical results; Popt is the tracer that has the lowest percent of sample classified correctly. Thus, a value of 1.0 has low power of discriminating samples.
The unmixing model minimizes the relative error term in Eq. (2) through an optimization procedure using the optim() function in the programming language R.
In Walnut Creek, each target sample was apportioned to five sources: streambanks, cropland, pasture, prairie, and unpaved roads. Source percentages are presented three ways: (1) as an average for the entire study period, (2) by weighting the sediment load transported for each sample’s time period relative to the total sediment load of all samples, and (3) by the sediment concentration for each sampling period. Weighting the sediment-fingerprinting results by the sediment load is as follows:
Collectionwt(n) is the weight given to the sediment load transported for each sample’s collection period n; SSmassn is the summed daily sediment load for each sample’s collection period; n is determined using the computation method in Eq. (1); and SSmassi is the summed sediment loads transported for each sample i (from 1 to n). Note that this total does not include the period 24 July 2013 at 12:45–1 August 2013 at 09:20, where the mass of sediment sampled was too low to analyze. The final storm-weighted source percentages are determined as follows:
Sv is the storm-weighted source apportionment (in percent) for source (v) (v includes channel banks, cropland, prairie, pasture, or unpaved roads); SAvi is the sediment source apportionment from the sediment-fingerprinting results (in percent) for source (v) and sample i; and n is the number of samples (i) collected during sampling period = 8.
Apportionment of sediment sources is also presented relative to the suspended-sediment concentration (mg L−1) for each sample by taking the mean daily suspended-sediment concentration for each sampling period (Gellis et al. 2018) and multiplying it by the apportionment results for each sample.
2.5 Uncertainty in the sediment-fingerprinting approach
Uncertainty tests in the sediment-fingerprinting results determined for each target sample include: (1) the confusion matrix, (2) Monte Carlo uncertainty analysis, (3) the source verification test (SVT), and (4) a Monte Carlo leave-one-out cross validation. The confusion matrix is an output of stepwise DFA that shows the number of source samples predicted for each group versus the actual number of source samples in each group (Provost and Kohavi 1998). It is a measure of how well the tracers can discriminate the sources.
A Monte Carlo uncertainty analysis was used to quantify the uncertainties associated with the optimized sediment source contributions predicted by the unmixing model (Eq. (2) (Collins and Walling 2007b; Martínez-Carreras et al. 2010). To perform an uncertainty analysis some of the statistical properties of initial source data were used to generate random deviates. The sample median and robust-scale estimator (Qn) were proposed to generate corresponding normal distribution following Collins et al. (2012b) and Zhang et al. (2017). The present method is an alternative to the more common method of incorporating mean and standard deviation as shape and scale estimators. The notation of random deviate is:
where X is a random deviate of the sediment property of normal distribution with shape estimator—median (M) and a robust-scale estimator (Qn).
Incorporating both a random number generator and an inverse cumulative distribution function (quantile function) with obtained property statistics generated n = 1000 samples for each Monte Carlo iteration.
The SVT is designed to see how well the final set of tracers discriminates the sources. Each source sample is entered as a target sample into the unmixing model to determine how well it can correctly identify each source sample. Because soil properties in some land uses can often be similar (i.e., pasture and cropland), the SVT can be used to decide whether to combine source samples into one category (i.e., pasture and cropland into agriculture) (Gellis et al. 2015). In addition, if a sample is misclassified (e.g., < 50% of that source), the user has the option to remove this sample and start the process again.
The Monte Carlo leave-one-out cross validation was used to quantify the sensitivity of the sediment-fingerprinting results to the removal of samples (Gellis et al. 2016). In Sed_SAT, the Monte Carlo leave-one-out cross validation randomly removes one sample from each of the source groups, which affects the group means, and the unmixing model is run without these samples (Gorman Sanisaca et al. 2017). The Monte Carlo simulation is run 1000 times on each target. For each target sample, the final unmixing model results are compared with the difference from two outputs in the Monte Carlo leave-one-out cross validation output; (1) the average of the Monte Carlo results and (2) the minimum and maximum source percentage results in the Monte Carlo results.
The robustness of the source ascription results for both Monte Carlo tests were assessed using a “goodness of fit” (GOF) (Collins and Walling 2007a, b; Manjoro et al. 2017) where the sample fingerprint properties of the target samples are compared with the corresponding values predicted by the model, as follows:
where all variables are defined in Eq. (2).
The GOF test is run for each iteration of both the Monte Carlo uncertainty analysis and the Monte Carlo leave-one-out cross validation. GOF values > 0.85 are considered acceptable values (Sherriff et al. 2015).
2.6 Sediment age determinations
The age of the topsoil–derived portion (pasture, prairie, or cropland) of each target sample was determined using the radionuclides 7Be and 210Pb for two age classes: 7Be up to ~ 1 year, and 210Pb up to ~ 85 years. 7Be (half-life of 53.3 days) is a naturally occurring radionuclide produced in the upper atmosphere by cosmic ray spallation of nitrogen and oxygen. It attaches to airborne particulates and reaches the earth surface mostly through precipitation, where 7Be-bearing particles sorb to fine sediment (Baskaran et al. 1993). 7Be is typically found in the top centimeter of soil (Baskaran et al. 1993) and is negligible in streambanks.
Lead-210 (half-life 22.3 years) is a naturally occurring radionuclide of lead in the 238U decay series. 238U decays to 226Ra, which in turn decays to the noble gas 222Rn. Evasion of some of the 222Rn from continental land masses to the atmosphere occurs where it ultimately decays to 210Pb that attaches to airborne particulates and aerosols. Atmospheric fallout of this 210Pb enriches surface soils in 210Pb above the level “supported” by decay of 226Ra in geologic materials; the unsupported fraction is commonly referred to as excess 210Pb (210Pbex). 210Pbex is primarily delivered to the earth surface in the form of atmospheric aerosols during rainfall events that sorb strongly to fine sediment (Baskaran et al. 1993). The 210Pbex decays at a known rate until only the supported 210Pb remains, providing a date marker for when surface soils and sediments were last exposed to direct fallout, if the initial activity of 210Pbex is known. The activity of 210Pbex has been shown to be higher in topsoil relative to streambanks (Matisoff et al. 2005; Hancock et al. 2014) and is dependent on fallout rate and the amount of post-fallout disturbance of the soil (e.g., erosion losses, mixing by plowing). Although unpaved road material does obtain some 7Be and 210Pbex, because it is brought in from nearby quarries and applied periodically, we did not perform age dating on this surface source.
Radionuclide analysis for 7Be, and 210Pbex, were conducted at the USGS Sediment Radioisotope Laboratory in Menlo Park, California, using high-resolution germanium detector gamma spectrometers following methods described in Fuller et al. (1999) and Van Metre et al. (2004). Measured activities of 7Be were corrected for radioactive decay from the date of sample collection to the date of analysis. Excess 210Pbex is the difference between the measured total 210Pb and 226Ra, which is determined from the short-lived intermediate gamma-emitting isotopes 214Pb and 214Bi. Method detection limits (MDLs) for 7Be and 210Pbex were 6.7 mBq g−1. Because of differences in times between sample collection and analysis, the sample-specific detection limit for 7Be was determined by correcting the MDL for decay, which varies among samples (11.7 to 31.7 mBq g−1).
The reported 1-sigma uncertainty in the measured radionuclide activity (s1) was calculated from the random counting error of samples and background standard spectra at the 1 standard deviation level. Uncertainty in measured activity was typically within ± 10% of the measured activity for total 210Pb and 226Ra and ± 18% for 7Be. Uncertainty in 210Pbex was propagated from the uncertainties in total 210Pb and 226Ra activity and averaged ± 20%.
To obtain 210Pbex radionuclide activities of sediment source types in Walnut Creek (streambanks, pasture, prairie, and cropland), the samples used in sediment fingerprinting were composited into fewer samples and sent for analysis (Gellis et al. 2018). The age dating approach used here relies on the activity of 210Pbex in the target sample that originated from topsoil (pasture, prairie, and cropland). Radionuclide activities are present in lower amounts in streambanks and unpaved roads and were subtracted from activities on the target samples as shown in the following equations. Because of the time constraints in analyzing 7Be, source samples were not analyzed for 7Be. The activity of 7Be in topsoil uses the estimate found in surface soils obtained for the Midwestern USA (Gellis et al. 2017 and references therein).
7Be is first corrected to the percent of surface-derived sediment as follows:
where 7Be(corr) is the estimated surface 7Be activity (mBq g−1); 7Be(target) is the measured 7Be activity in the target sample (suspended sediment) (mBq g−1); and surface% is the percentage of surface-derived sediment from the sediment fingerprinting results (pasture + crop + prairie + unpaved roads).
The age of the target sediment up to ~ 1 year is determined as:
where 7Be (age) is the age of topsoil–derived sediment (days); 7Be(corr) is the estimated surface 7Be activity (mBq g−1) (Eqs. 8 and 9); 7Be(95) is the estimated surface material 7Be activity for Midwestern US soils (542 mBq g−1) (Gellis et al. 2017); and \( {\lambda}_{7_{\mathrm{Be}}} \)is the decay constant for 7Be = 0.01305 day−1.
The estimated topsoil activity of 210Pbex is determined as:
where 210Pbex(corr) is the estimated surface 210Pbex activity (mBq g−1); 210Pbex(target) is the measured 210Pb activity in the target sample (suspended sediment) (mBq g−1); 210Pb(roads) is the mean 210Pb activity in unpaved roads (Gellis et al. 2018); source%(roads) is the sediment-fingerprinting results in percent for unpaved roads; 210Pb(banks) is the mean 210Pb activity in streambanks (Gellis et al. 2018); source%(banks) is the sediment-fingerprinting results in percent for streambank; and surf_topsoil% is from Eqs. 8 and 9.
The age of target sediment using 210Pbex up to ~ 85 years, is as follows:
where 210Pbex(age) is the age of topsoil–derived sediment (days); 210Pbex(corr) = the estimated surface 210Pbex activity (mBq g−1) (Eq. 12); 210Pbex(surf) = the weighted surface activity of 210Pbex (75.5 mBq g−1) from samples in Walnut Creek (Gellis et al. 2018); and \( {\lambda}_{210_{\mathrm{ex}}} \) is the decay constant for 210Pbex = 8.50999 × 10−5 day−1.
For 210Pbex and 7Be, age as defined here is the transit time between when sediment enters the channel from a surface source and when the target sample is collected. Sediment that does not contain measurable quantities of 210Pbex is considered to be older than ~ 85 years. This was determined by entering the MDL for 210Pbex into Eq. (12).
3 Results
3.1 Flow and suspended sediment
The first half of the sampling period (23 May 2013 to 1 July 2013) was a period of higher flows (Fig. 2). This period of sampling accounted for 57% of the total flow and 89% of the total suspended-sediment load, where SSCs ranged from 6.4 to 3204 mg L−1 (Gellis et al. 2018). The second half of the sampling period corresponds to summertime baseflow with SSCs ranging from 13 to 149 mg L−1 (Gellis et al. 2018).
Discharge, turbidity, rainfall, and suspended sediment sample collection shown for the period (19 May 2013 to 15 August 2013) (Gellis et al. 2018)
3.2 Sediment-fingerprinting results and uncertainty
The unmixing model showed that sources varied between suspended-sediment samples and that cropland was the largest source of sediment on average (48%) and weighted by sediment loads (62%) (Fig. 3; Table 3). Based on weighting by sediment loads, channel banks accounted for most of the remainder of the sediment (36%), with prairie, pasture, and unpaved roads each contributing ≤ 1% (Table 3). For the two sampling periods with the highest mean sediment concentration (target samples 1 and 3, means of 838 and 312 mg L−1, respectively), cropland was the largest source (61 and 71%), followed by streambanks (37 and 27%) (Fig. 3b; Table 3).
The median grain size (D50) of the < 63 μm fraction of target samples was finer than most source samples and the TOC in target samples was higher than most of the source samples (Fig. 4).
Median grain size (D50) and total organic carbon (TOC) of source and target samples, Walnut Creek, Iowa (Gellis et al. 2018)
Nineteen of the thirty-eight tracers (elements) had at least one source group corrected for grain size and twenty-one tracers had at least one source group corrected for organic content (Table S1A, B, Electronic supplementary material). Only one tracer, uranium, was corrected for both size and organic corrections for the same source group (unpaved roads) (Table S1A, B, Electronic supplementary material). Smith and Blake (2014) suggest that applying both a size and organic correction to a tracer can result in over correction. Because only 1 of the 38 tracers, uranium, was corrected for both size and organics, over correcting the data was not a concern.
Results of stepwise DFA indicated that five of the tracers (Ca, Mo, Na, Sc, and TOC) were significant for all target samples with other elements being significant for one or more target samples (Fig. 5; Table S2, Electronic supplementary material). It is important to point out that a different set of tracers can be significant in discriminating the sources for any given target sample (Table S2, Electronic supplementary material). Because the size and organic content of each target sample affects the final corrected concentration of source samples, depending on the grain size and organic content of the target sample, different tracers may be significant.
Averaging all results in the confusion matrix shows that streambanks and unpaved roads have 100% of the samples classified correctly, with most samples classified correctly for pasture (87%), crop (99%), and prairie (93%) (Fig. 6). The SVT results are used to determine how well the final tracers selected in stepwise DFA can identify the source samples (Table 4; Table S3, Electronic supplementary material). Results of the SVT indicate that all sources were classified as > 73% of their source type; with prairie (73%), pasture (77%), crop (78%), and banks and unpaved roads (97%) (Table 4). Misclassified cropland samples were classified as banks (8%), pasture (4%), prairie (9%), and unpaved roads (1%) (Table 4). The majority of misclassified pasture samples were classified as prairie, and the majority of misclassified prairie samples were classified as pasture (Table 4).
Results of the Monte Carlo uncertainty analysis indicate that the cumulative distribution functions produced mean apportionment results that ranged from 0 to 8% of the original unmixing model results (Table 3). The Monte Carlo uncertainty analysis showed that confidence intervals (5 and 95 percentiles) were less than 10% except for three source apportionments which showed uncertainty of 11, 14, and 16% (Table 3; Fig. S2, Electronic supplementary material). The GOF test for all samples were > 0.90 indicating that the model provided reliable estimates of the relative contributions from the individual source types (Table 3).
The Monte Carlo leave-one-out cross validation produces a range in source group means which affects the unmixing model results. The average of the 1000 Monte Carlo iterations for each target sample were within 0 to 8% of the unmixing models results (Fig. S3, Electronic supplementary material). Six of the eight target samples showed < 16% difference from the unmixing model results to the minimum and maximum of any Monte Carlo iteration with four of the eight target samples showing a < 9% difference. Target sample 4 had 19% of the 1000 Monte Carlo iterations with a maximum difference to the unmixing model of > 10% and all occurring for pasture and prairie samples. Target sample 6 had 59% of the 1000 Monte Carlo iterations with a maximum difference to the unmixing model of >10% and all occurring for pasture and prairie samples. The GOF test showed results > 0.90 for each sample (Table 3), indicating that the model was not sensitive to the removal of samples.
3.3 Ages of fluvial sediment
Lead-210 excess and 7Be were detected in all target samples (Gellis et al. 2018). 7Be activity on the surface-derived fraction of target samples indicated that ages ranged from 44 to 205 days, averaging 131 days (Table 5). 210Pbex indicated ages from 1 to 58 years (average = 37 years) (Table 5). The age differences from the two radionuclides indicate that all of the sediment samples are comprised of sediments of a range of ages.
4 Discussion
4.1 Sediment source apportionment
The robustness of the sediment-fingerprinting results can be assessed by examining the confusion matrix (Fig. 6), Monte Carlo uncertainty analysis (Table 3; Fig. S2, Electronic supplementary material), SVT (Table 4; Table S3, Electronic supplementary material), and Monte Carlo leave-one-out cross-validation results (Fig. S3, Electronic supplementary material). The confusion matrix results indicate that the tracers used in Walnut Creek are able to correctly classify the sources (Fig. 6). Monte Carlo uncertainty analysis and the GOF results showed that most apportionment results were > 0.90, which are considered acceptable values (Sherriff et al. 2015) (Table 3). Results of the SVT indicated that banks and unpaved roads on average had ~ 99% correct classification, whereas the top-soil sources (pasture, cropland, and prairie) are often classified as other sources (Table 4). The removal of a random sample in the Monte Carlo leave-one-out cross-validation analysis, which changes the source group means, showed little difference to the unmixing model results (< 8%) except for target samples 4 and 6 which showed > 10% difference to the unmixing model results. The differences all occurred for pasture and prairie samples. Six of the eight target samples showed a maximum difference of the minimum or maximum Monte Carlo leave-one-out cross-validation result to the unmixing model of up to 16% for any iteration.
Land use has a strong effect on soil chemistry due to differences in how the soil is managed, for example, differences in erosion, compaction, biologic activity, and addition of fertilizers and nutrients (Aguilar et al. 1988; Yanai et al. 2012; Veenstra and Lee Burras 2015; Yesilonis et al. 2016). As land use is converted, soil chemistry changes are variable over time where selected elements change while others remain constant (Veenstra and Lee Burras 2015). Misclassification of the topsoil source groups in the SVT (i.e., pasture and crop) may indicate similar chemistry in soils between topsoil land use types (Miller et al. 2015) or changing land use on the same field over time (Gellis et al. 2015). Palazón et al. (2015) indicated that an overlap in DFA results between agriculture and forest was due to land use conversion of agriculture reverting to forest after land abandonment. In Linganore Creek, Maryland, an agricultural watershed in Maryland, pasture and cropland fields were often rotated over time and source samples were combined for sediment fingerprinting analysis (Gellis et al. 2015).
In Walnut Creek, the intermixing of apportionment results between crop, pasture, and prairie can also be explained by its land use history. The Neal Smith National Wildlife Refuge, located in the Walnut Creek watershed, is part of a large-scale effort by the U.S. Fish and Wildlife Service (USFWS) to restore agricultural areas in the watershed to native prairie. Beginning in 1992, an effort to convert agricultural lands to native prairie was started (Schilling et al. 2002) and by 2005, approximately 23.5% of the Walnut Creek watershed had been converted from agricultural land to native prairie (Schilling et al. 2006). Pasture and prairie show a higher percentage of interclassification (confusion matrix and SVT) (Table 4) that may be both due to prairie lands being former pasture fields and/or that both land types are dominated by grasses. Although cropland in Walnut Creek was also converted to prairie, the lower percentage of misclassified prairie samples as cropland may be due to plowing and tilling operations on cropland which continually mix the soil, whereas prairie is no longer tilled. In summary, the uncertainty results from Sed_SAT suggest that for each target sample, the final set of tracers are able to significantly discriminate between topsoil types, streambanks, and roads in Walnut Creek but that an overlap in apportionment results occurred between pasture and prairie that may be due to their land use history.
Stepwise discriminant function analysis indicated that up to 25 tracers were significant in identifying the sediment sources with 5 tracers significant for all target samples (Fig. 5). Koiter et al. (2013) proposed that an environmental basis for selection of tracers used in sediment fingerprinting, such as a relation to geology and land use, should be established. Although a rigorous evaluation of this basis for all of the tracers used here is beyond the scope of this study, logical differences are evident among the five tracers that are significant for each target sample (Ca, Mo, Na, Sc, and TOC) that may be related to pedogenesis and anthropogenic influences (Fig. S4, Electronic supplementary material). Unpaved roads have the highest Ca concentration of any land use (Fig. S4a, Electronic supplementary material). Secondary (unpaved) roads in Iowa are surfaced with either stream gravel or a crushed limestone aggregate obtained from local sources (Bergeson et al. 1990). The high Ca concentrations observed in the Walnut Creek watershed for unpaved roads is due to the high Ca content found in limestone. Because unpaved road material is obtained from mines and quarries, this material is lower in concentration of the other tracers (Mo, Na, Sc, TOC), which are related to soil properties.
Molybdenum, TOC, and Sc are highest in topsoil sources (cropland, pasture, prairie) (Fig. S4b, d, e in the Electronic supplementary material). Total organic carbon is correlated to organic matter which is commonly high in the topsoil of these land uses. The low TOC in banks reflects the decrease in organic matter commonly observed in deeper soils. Scandium is a rare earth element common in soils as a result of weathering of parent material in soil formation (Horovitz 1975; Vermeire et al. 2016). The higher concentrations of Sc observed in topsoil classes (crop, pasture, and prairie) versus streambanks and unpaved roads (Fig. S4d, Electronic supplementary material) may reflect differences due to pedogenesis. The higher concentrations of Mo in cropland, pasture, and prairie (Fig. S4b, Electronic supplementary material) might reflect the deposition of Mo from atmospheric pollution in the twentieth century. Chappaz et al. (2008, 2012) observed higher Mo concentrations over background levels in lake sediments in Canada, which were attributed to atmospheric inputs of Mo from coal combustion and ore processing plants. High levels of Mo in soil from industrial pollution in the twentieth century have also been described in Austria by (Neunhäuserer et al. 2001) and in the Midwestern USA (Elrashidi et al. 2016). Streambanks are composed of older sediment and may not be subject to atmospheric Mo deposition and thus have lower levels. The slightly higher Na concentrations in banks compared with surface soils (Fig. S4d, Electronic supplementary material) might result from the leaching of Na from surface soils, as it is a strongly hydrated monovalent cation and is relative mobile in soils (Laird et al. 2010). It also is possible that leaching of soluble constituents from surface soils is enhanced by agricultural practices. We recognize that additional research would be needed to understand the differences in Mo, Sc, and Na among these land use types.
4.2 Age of suspended sediment
Suspended sediment is a mixture of ages. The age of suspended sediment calculated from 210Pbex activities indicates that a portion of the target sediment is of decadal ages averaging 37 years (Table 5). The age of suspended-sediment samples using 7Be activity (Gellis et al. 2018) ranged from 44 to 205 days (averaging 131 days) (Table 5), indicating that a portion of fine-grained sediment is moving rapidly through Walnut Creek.
Clearly, two very different ages for the same sediment sample cannot both be accurate. The occurrence of 7Be indicates that some portion of the sediment is young, on the order of months, whereas the dating based on 210Pbex indicates that some of the surface-derived sediment has been in channel storage for decades (Table 5). Unlike dating sediment in lakes and reservoirs with 210Pbex, which assumes that each depositional layer is isolated from new sediment, the scour and fill of sediment in Walnut Creek leads to a mixture of ages. There are two processes that are reducing the activity of 7Be in surface-derived sediment below that of surface soil: radioactive decay and dilution by older sediment with no 7Be activity. The ages calculated using 7Be (mean of 131 days) assume only decay and no mixing. However, we know that much of the surface-derived sediment is much older based on 210Pbex age estimates. This implies that some smaller portion of sediment must have higher 7Be activities and be even younger than the 7Be age estimates, but there is no way at present of separating out the effects of decay and dilution on 7Be activities. Future research in age dating suspended sediment may provide some insight into this.
Based on 210Pbex, the mean age of the same surface-derived sediment fraction averaged 37 years, indicating that much of this surface-derived sediment remains in channel storage for decades. Unlike 7Be, the 210Pbex age might be a reasonable estimate of the mean age of the whole surface–derived fraction because most or all of that fraction probably entered the channel within the ~85-year time period dateable with 210Pb. We therefore conclude that the 210Pbex-based ages are a reasonable estimate for the mean age of the surface-derived fraction and that the 7Be activities are evidence that there is a smaller fraction of very young sediment being transported in the stream. Other studies have also reported that the age or transit time of sediment falls into several age groups (Table 6).
4.3 Comparison of streambank and agriculture as sediment sources in Walnut Creek
The results of this study indicate that streambank sediment is approximately one-third (36%) of the apportioned suspended sediment (Table 3), which is at the lower end of estimates from other studies in Walnut Creek (Table 7). In a program to monitor water quality, sediment, and channel changes as they relate to management activities at the Neal Smith National Wildlife Refuge in Walnut Creek, streambank contributions of sediment were estimated for three different time periods: (1) 1996 to 1998, (2) 1996 to 2005, and (3) 2004 to 2010 (Table 7). For each time period, the percent contribution from streambanks changed: period 1 banks contributed 51%, period 2 contributions ranged from 38.6 to 64.4%, and period 3 contributions ranged from 0 to 53% (Table 7).
Although the literature results in Table 7 show some periods with a higher percentage of sediment from streambanks than the current study, the literature also indicates that streambank contributions vary temporally with flow variability, where on some occasions, the contributions from streambank erosion were considered to be zero (Palmer et al. 2014). Therefore, the results shown here, where streambanks have a contribution ranging from 0 to 71% (weighted average of 36%), are still within the range of values reported in the literature for these longer-term studies.
Estimates of agricultural erosion are limited for Walnut Creek and only include modeled results using the Revised Universal Soil Loss Equation (RUSLE) (Schilling et al. 2011). For 2005, 20,490 Mg of sediment was estimated to have eroded from upland areas, a value almost three times higher than the average annual sediment load from 1996 to 2005 (7606 Mg year−1) (Table 7). Depending on the sediment delivery ratio of sediment from upland areas, upland sediment based on RUSLE could be a large source of sediment equal to or greater than streambank erosion. Our results confirm that upland sediment (cropland) is an important source of sediment in Walnut Creek.
4.4 Sediment in channel storage
The volume of sediment in channel storage can be an important component of the total sediment budget (Lambert and Walling 1988; Marttila and Kløve 2014; Piqué et al. 2014; Wilson et al. 2004), particularly in streams with accumulations of large woody debris and beaver dams (Fisher et al. 2010; Wohl and Scott 2017). In Pleasant Valley, a small 19-km2 agricultural lowland stream in Wisconsin, fine grained soft sediment stored along the channel bed is estimated to be equivalent to 8 years’ worth of annual loading exported from the watershed (Faith Fitzpatrick, USGS, written communication 9 December 2017). Stream surveys conducted in 1998 by Schilling and Wolter (2000) along the thalweg of Walnut Creek for a length of 10,123 m, showed an average thickness of 12.5 cm (dominantly silt). Much of the fine-grained sediment was stored behind logs and debris jams where in one portion of the stream, bed sediment accumulations reached > 0.3 to >0.6 m (Schilling and Wolter 2000). Based on average flow measurements for 1997, Schilling and Wolter (2000) estimated that it would take 8.8 years to remove the sediment in channel storage if no new sediment were introduced.
4.5 Sediment age model
The transit time of sediment can be viewed from two perspectives: (1) the time it takes a particle to move from its source (i.e., topsoil) to the nearest channel or from its source to a point in the channel (i.e., outlet) or (2) the time it takes a surface-derived particle to move from when it enters the channel system to the outlet (or sampling point). Here, we examine the second perspective and assume that the age of a fine-grained sediment particle begins when it enters the channel (Fig. 7), an assumption that is made for only the topsoil–derived portion of sediment. Because streambanks do not acquire substantial activities of 7Be and 210Pbex, this sediment cannot be dated. Instead, we assume that when fine-grained streambank sediment is eroded, it travels in the channel in a similar fashion to the topsoil–derived sediment.
Several studies have used radionuclides to date surface-derived fine-grained sediment at decadal scales (210Pbex, 137Cs) and days using 7Be (up to 1 year). In Old Woman Creek, Ohio, the age of suspended sediment using 7Be ranged from 46 to 79 days (Matisoff et al. 2005) (Table 6). Using 210Pbex, 137Cs, and 7Be, Evrard et al. (2010) in the Cointzio River, Mexico, and Le Cloarec et al. (2007) for the Seine River, France, both depicted a two-box model to explain sediment age times. A geologic box representing watershed storage ranging in age in Mexico from 5000 to 23,300 years and in France from 4800 to 30,000 years, and a rapid box in Mexico from 50 to 200 days and in France, to less than 1 year (Dominik et al. 1987; Evrard et al. 2010). In the agricultural Walnut Creek watershed, Iowa, we propose that sediment which is eroded from agricultural areas and streambanks can reside in three different storage age boxes: (1) a rapid box < 1 year, (2) a decadal box (10–100 years), and (3) a geologic box (100–> 1000 years (Fig. 7).
Based on 7Be results, a rapid box is depicted for Walnut Creek where a portion of the fine-grained sediment moves through the system in less than 1 year (Fig. 7). Based on 210Pbex results, a decadal storage box is depicted for Walnut Creek (Fig. 7), where some of the sediment from agricultural areas remains in storage for decadal time periods with limited transport during storm events. A decadal transit time of sediment also was observed for Australian rivers (0–21 years) (Wallbrink et al. 2002). Most of the sediment that travels in the decadal and rapid storage box in Walnut Creek originates from agricultural topsoil.
Although an analysis of the ages of floodplain sediment was not undertaken in Walnut Creek, for the geologic box, based on other studies, sediment that is deposited on floodplains is likely to remain there over mean time scales of a hundred to a thousand or more years (Lancaster and Casebeer 2007; Phillips et al. 2007; Pizzuto et al. 2014). Sediment deposited on the floodplain in Walnut Creek is depicted as the geologic box in Fig. 7, where it is assumed to remain in storage for a period of 100 years or longer.
4.6 Data limitations and uncertainty
The age model in this paper for 7Be (Eq. 10) relies on the surface activity of 7Be and, as discussed in Sect. 4.2, it is applied to the whole surface–derived sediment fraction without accounting for mixing with sediment too old to date with 7Be. It was not within the scope of this study to directly measure fallout activities. The value, 542 mBq g−1, was estimated by Gellis et al. (2017) for Midwestern US streams and is 35% less than the average value of 7Be surface soil activity in the literature (mean = 838 mBq g−1, Fig. S5, Electronic supplementary material); however, the number of studies is limited (n = 6) and the variability is large (standard deviation, ± 616) (Fig. S5, Electronic supplementary material). We recognize that the 7Be surface soil activity in Walnut Creek may be temporally and spatially variable. If the mean soil surface 7Be activity is higher or lower than our estimate, the resulting age estimates of suspended sediment will get older and younger, respectively.
Beryllium-7 and 210Pbex activity are used as indicators of the age of the surface-material component of suspended sediment. Most of the 7Be activity in surface soils is found in the top centimeter (Wallbrink et al. 1998; Huh and Su 2004). In our age model for 7Be, it is assumed that that the top most surface (~ 1 cm) is being eroded and deeper erosion of topsoil is not occurring. Deeper surface erosion would tend to lower the 7Be activity in the transported particles but not necessarily lower 210Pbex. If deeper surface erosion did occur, then our results are indicating older ages.
We recognize that some of the 7Be and 210Pbex falls directly on the wetted areas of the stream channels, which may cause an increase in activity on sediment already in suspension and channel storage. Hancock et al. (2014) examined the contribution of surface soils using fallout radionuclides and estimated that the direct contribution of 7Be to the Bowen River (9400 km2) and Logan River (3700 km2) in eastern Australia could cause a 12 and 10% increase, respectively, in the measured values. Hancock et al. (2014) further commented that this estimate is high considering that most of the rainfall fell early in the event prior to rising stage and presumably before the input of sediment to the system. Wallbrink et al. (2002) examined ratios of 210Pbex to 137Cs in source and target samples and based on the higher ratios in target samples determined that enrichment of 210Pbex on suspended sediment was occurring through direct rainfall on the channel. A similar analysis was run for Walnut Creek and results do not show enrichment of 210Pbex in target samples relative to source samples and we therefore conclude that there is not a significant contribution of 210Pbex on the target sediment from rainfall (Table S4, Electronic supplementary material).
Because 7Be and 210Pbex were not measured in rainfall in this study, correction factors cannot be applied to the direct input of 7Be to the wetted area of the channel. If rainfall inputs of 7Be directly to the stream increase suspended-sediment activities, this increase would result in a younger apparent age. For example, if 10% of the 7Be activity in the target sample is attributed to dosing from direct rainfall on the stream channel, and a correction was made to the 7Be activity, the suspended sediment would increase in the age of 8 days, a relatively small amount.
Higher radionuclide activity can be correlated to finer grain sizes (He and Walling 1996). Although a size correction factor was applied to the elemental concentrations in the sediment-fingerprinting method, because of the small amount of source samples that were analyzed for 7Be and 210Pbex (Gellis et al. 2018), we are unable to apply the same regression approach to these samples. This study did analyze the finer portion of sediment (< 63 μm) where most of the radionuclide activity is found.
Although the focus of this paper was to test the efficacy of combining the sediment fingerprinting approach with age dating, limitations related to the short duration of the study period are recognized. Sediment sources and ages may have seasonal variations that are not captured in this relatively short period of sampling. To determine how the sampling period compared with longer-term hydrologic conditions, historical monthly rainfall (NOAA) data were obtained for the Prairie City, Iowa raingage (2002–2017) (https://www.ncdc.noaa.gov/; accessed 31 March 2018) (Table S5, Electronic supplementary material), located ~ 3 km from the USGS station on Walnut Creek. Monthly rainfall summaries show that the rainfall totals for June and July 2013 (the sampling months for this study) were similar to the 2002–2017 June median and 64% less than the July median. To determine if antecedent conditions prior to sampling were different than the long-term average, monthly totals for the period January to May were summed for each year and indicated that January to May 2013 was 78% higher than the median long-term value. In summary, compared with historic records, antecedent conditions prior to sampling at Walnut Creek in 2013 were wetter and during the sampling period conditions were drier. We do not know, however, how these differences affect sediment sources and ages.
5 Conclusions
Understanding the contributions from agricultural topsoil and its residence time are important critical zone processes that can assist managers in developing actions to reduce sediment and improve the health of aquatic habitats. This study shows how the sediment fingerprinting approach combined with fallout radionuclides can apportion fine-grained sediment to its sources and date the topsoil–derived portion. Previous studies on age dating sediment using fallout radionuclides were unable to distinguish older channel sediment from sediment that was diluted by streambanks. Here, we use elemental analysis to fingerprint the sources of sediment and short-lived fallout radionuclides to date the surface-derived portion of sediment into two age groups: 7Be to < 1 year and 210Pbex to decadal ages. Results from Walnut Creek indicate that the majority (63%) of the sediment is surface-derived agricultural topsoil and has a mean age of 37 years (using 210Pbex) and includes a component of much younger sediment that results in a mean age of 131 days (using 7Be). For Walnut Creek, we propose a model of sediment ages, where sediment which is eroded from agricultural areas and streambanks can reside in three different storage age boxes: (1) a rapid box (< 1 year), (2) a decadal box (10–100 years), and (3) a geologic box (100–> 1000 years).
References
Aguilar R, Kelly EF, Heil RD (1988) Effects of cultivation on soils in Northern Great Plains rangeland. Soil Sci Soc Am 52:1081–1085
ASTM (2002) Standard test methods for determining sediment concentration in water samples. ASTM Int 3977–97:6
Baskaran M, Coleman CH, Santschi PH (1993) Atmospheric depositional fluxes of 7Be and 210Pb at Galveston and College Station, Texas. J Geophys Res 98:20555–20571
Belmont P, Willenbring JK, Schottler SP, Marquard J, Kumarasamy K, Hemmis JM (2014) Toward generalizable sediment fingerprinting with tracers that are conservative and nonconservative over sediment routing timescales. J Soils Sediments 14:1479–1492
Belmont P, Gran KB, Schottler SP, Wilcock PR, Day SS, Jennings C, Lauer JW, Viparelli E, Willenbring JK, Engstrom DR, Parker G (2011) Large shift in source of fine sediment in the Upper Mississippi River. Environ Sci Technol 45:8804–8810. https://doi.org/10.1021/es2019109
Bergeson KL, Kane MJ, Callen DO (1990) Crushed stone granular surfacing materials. Sponsored by the Iowa Limestone Producers Association and National Stone Association Research Program, report by Engineering Research Institute, Iowa State University, Ames, Iowa, USA. http://publications.iowa.gov/19899/1/IADOT_hr_2046_and_mlr_90_07_Crushed_Stone_Granular_Surfacing_Materials_1990.pdf
Bonniwell EC, Matisoff G, Whiting PJ (1999) Determining the times and distances of particle transit in a mountain stream using fallout radionuclides. Geomorphology 27:75–92
Briggs P, Meier AL (2002) The determination of forty-two elements in geological materials by inductively-coupled plasma-mass spectrometry for NAWQA. In: Taggart JEJ (ed) Analytical methods for chemical analysis of geologic and other materials. U.S. Geological Survey Open-File Report 2002-223, 16 pp
Caitcheon G, Douglas G, Palmer M (2006) Sediment source tracing in the Lake Burragorang Catchment: report to the Sydney Catchment Authority CSIRO Land and Water Science Report 47/07, Canberra, Australia. http://www.clw.csiro.au/publications/science/2007/sr47-07.pdf
Cavanagh JE, Hogsden KL, Harding JS (2014) Effects of suspended sediment on freshwater fish. Envirolink Advice Grant: 1445-WCRC129; Landcare research contract report: LC1986, 29 p, prepared for West Coast Regional Council, New Zealand, 29 p. Available at http://envirolink.govt.nz/assets/Envirolink/1445-WCRC129-Effects-of-suspended-sediment-on-freshwater-fish.pdf. Accessed 18 March 2018
Chappaz A, Gobeil C, Tessier A (2008) Geochemical and anthropogenic enrichments of Mo in sediments from perennially oxic and seasonally anoxic lakes in Eastern Canada. Geochim Cosmochim Acta 72:170–184
Chappaz A, Lyons TW, Gordon GW, Anbar AD (2012) Isotopic fingerprints of anthropogenic molybdenum in lake sediments. Sci Technol 46:10934–10940
Collins AL, Walling DE (2002) Selecting fingerprint properties for discriminating potential suspended sediment sources in river basins. J Hydrol 261:218–244
Collins AL, Walling DE (2007a) The storage and provenance of fine sediment on the channel bed of two contrasting lowland permeable catchments UK. River Res Restoration 23:429–450
Collins AL, Walling DE (2007b) Sources of fine sediment recovered from the channel bed of lowland groundwater-fed catchments in the UK. Geomorphology 88:120–138
Collins AL, Walling DE, Leeks GJL (1997) Sediment sources in the Upper Severn catchment—a fingerprinting approach. Hydrol Earth Syst Sci 1:509–521
Collins AL, Walling DE, Leeks GJL (1998) Use of composite fingerprints to determine the provenance of the contemporary suspended sediment load transported by rivers. Earth Surf Process Landf 23:31–52
Collins AL, Walling DE, Webb L, King P (2010) Apportioning catchment scale sediment sources using a modified composite fingerprinting technique incorporating property weightings and prior information. Geoderma 155:249–261
Collins AL, Zhang Y, McChesney D, Walling DE, Haley SM, Smith P (2012a) Sediment source tracing in a lowland agricultural catchment in southern England using a modified procedure combining statistical analysis and numerical modelling. Sci Total Environ 414:301–317
Collins AL, Zhang Y, Walling DE, Grenfell SE, Smith P, Grischeff J, Locke A, Sweetapple A, Brogden D (2012b) Quantifying fine-grained sediment sources in the River Axe catchment, Southwest England: application of a Monte Carlo numerical modelling framework incorporating local and genetic algorithm optimization. Hydrol Process 26:1962–1983
Collins AL, Pulley S, Foster IDL, Gellis A, Porto P, Horowitz AJ (2017) Sediment source fingerprinting as an aid to catchment management: a review of the current state of knowledge and a methodological decision-tree for end-users. J Environ Manag 194:86–108
Collins AL, Williams LJ, Zhang YS, Marius M, Dungait JAJ, Smallman DJ, Dixon ER, Stringfellow A, Sear DA, Jones JI, Naden PS (2014) Sources of sediment-bound organic matter infiltrating spawning gravels during the incubation and emergence life stages of salmonids. Agric Ecosyst Environ 196:76–93
Cooper RJ, Krueger T (2017) An extended Bayesian sediment fingerprinting mixing model for the full Bayes treatment of geochemical uncertainties. Hydrol Process 31:1900–1912
D’Haen K, Verstraeten G, Dusar B, Degryse P, Haex J, Waelkens M (2013) Unravelling changing sediment sources in a Mediterranean mountain catchment: a Bayesian fingerprinting approach. Hydrol Process 27:896–910
Dominik J, Burrus D, Vernet JP (1987) Transport of the environmental radionuclides in an alpine watershed. Earth Planet Sci Lett 84:165–180
Edwards TE, Glysson GD (1999) Field methods for measurement of fluvial sediment. U.S. Geological Survey Techniques of Water-Resources Investigations, book 3, chapter C2, 89 pp. Available at https://water.usgs.gov/osw/techniques/Edwards-TWRI.pdf
Elrashidi MA, Wysocki D, Schenenberger P (2016) Effects of land use on selected properties and heavy metal concentration for soil in the US Great Plains. Commun Soil Sci Plant Anal 47:2465–2478
Estrany J, Garcia C, Walling DE, Ferrer L (2011) Fluxes and storage of fine-grained sediment and associated contaminants in the Na Borges River (Mallorca, Spain). Catena 87:291–305
Evans DJ, Gibson CE, Rossell RS (2006) Sediment loads and sources in heavily modified Irish catchments: a move towards informed management strategies. Geomorphology 79:93–113
Evrard O, Laceby JP, Huon S, Lefèvre I, Sengtaheuanghoung O, Ribolzi O (2016) Combining multiple fallout radionuclides (137Cs, 7Be, 210Pbxs) to investigate temporal sediment source dynamics in tropical, ephemeral riverine systems. J Soils Sediments 16:1130–1144
Evrard O, Némery J, Gratiot N, Duvert C, Ayrault S, Lefèvre I, Poulenard J, Prat C, Bonté P, Esteves M (2010) Sediment dynamics during the rainy season in tropical highland catchments of Central Mexico using fallout radionuclides. Geomorphology 124:42–54
Fisher GB, Magilligan FJ, Kaste JM, Nislow KH (2010) Constraining the timescales of sediment sequestration associated with large woody debris using cosmogenic 7Be. Journal of Geophysical Research 115: F01013 https://doi.org/10.1029/2009JF001352
Foster IDL, Walling DE (1994) Using reservoir deposits to reconstruct changing sediment yields and sources in the catchment of the Old Mill Reservoir, South Devon, UK, over the past 50 years. Hydrol Sci J 39:347–368
Foster IDL, Oldfield F, Flower RJ, Keatings K (2008) Trends in mineral magnetic signatures in a long core from Lake Qarun, Middle Egypt. J Palaeolimnol 40:835–849
Fox JF (2009) Identification of sediment sources in forested watersheds with surface coal mining disturbance using carbon and nitrogen isotopes. J Am Water Resour Assoc 45:1273–1289
Fox JF, Papanicolaou AN (2008) An un-mixing model to study watershed erosion processes. Adv Water Resour 31:96–108
Fry J, Xian G, Jin S, Dewitz J, Homer C, Yang L, Barnes C, Herold N, Wickham J (2011) Completion of the 2006 National Land Cover Database for the conterminous United States. Photogramm Eng Remote Sens 77:858–864
Fryirs K (2013) Disconnectivity in catchment sediment cascades—a fresh look at the sediment delivery problem. Earth Surf Process Landf 38:30–46
Fryirs K, Brierley GJ (2001) Variability in sediment delivery and storage along river courses in Bega catchment, NSW, Australia -- implications for geomorphic river recovery. Geomorphology 38:237–265
Fuller CC, van Green A, Baskaran M, Anima R (1999) Sediment chronology in San Francisco Bay, California defined by 210Pb, 234Th, 137Cs, and 239,240Pu. Mar Chem 64:7–27
Gellis AC, Noe GB (2013) Sediment source analysis in the Linganore Creek watershed, Maryland, USA, using the sediment-fingerprinting approach: 2008 to 2010. J Soils Sediments 13:1735–1753
Gellis AC, Walling DE (2011) Sediment-source fingerprinting (tracing) and sediment budgets as tools in targeting river and watershed restoration programs. In: Simon A, Bennett S, Castro JM (eds) Stream restoration in dynamic fluvial systems. Scientific approaches, analyses, and tools, American Geophysical Union Monograph Series, vol 194. John Wiley & Sons, Chichester, pp 263–291
Gellis AC, Fuller CC, Van Metre PC (2017) Sources and ages of fine-grained sediment to streams using fallout radionuclides in the Midwestern United States. J Environ Manag 194:73–85
Gellis AC, Cole KJ, Fuller CC, Tomer MD (2018) Sediment and geomorphology data for Walnut Creek, Iowa: U.S. Geological Survey data release, https://doi.org/10.5066/F7KP8124
Gellis A, Fitzpatrick F, Schubauer-Berigan J (2016) A manual to identify sources of fluvial sediment: EPA report, EPA/600/R-16/210. 106 pp. Available at https://pubs.er.usgs.gov/publication/70182516
Gellis AC, Noe GB, Clune JW, Myers MK, Hupp CR, Schenk ER, Schwarz GE (2015) Sources of fine-grained sediment in the Linganore Creek watershed, Frederick and Carroll Counties, Maryland, 2008-10. U.S. Geological Survey Scientific Investigations Report 2014–5147, 56 p. https://doi.org/10.3133/sir20145147
Gellis AC, Hupp CR, Pavich MJ, Landwehr JM, Banks WSL, Hubbard BE, Langland MJ, Ritchie JC, Reuter JM (2009) Sources, transport, and storage of sediment at selected sites in the Chesapeake Bay watershed. U.S. Geological Survey Scientific Investigations Report 2008-5186, 95 p. http://pubs.usgs.gov/sir/2008/5186/
Gorman Sanisaca LE, Gellis AC, Lorenz DL (2017) Determining the sources of fine-grained sediment using the sediment source assessment tool (Sed_SAT). U.S. Geological Survey Open-File Report 2017-1062, 104 pp. https://doi.org/10.3133/ofr20171062
Gray JR, Simões FJM (2008) Estimating sediment discharge. In: Marcelo G (ed) Sedimentation engineering—processes, measurements, modeling, and practice. American Society of Civil Engineers Manual 110, Appendix D, pp. 1065–1086. http://water.usgs.gov/osw/techniques/Gray_Simoes.pdf
Haddadchi A, Ryder DS, Evrard O, Olley J (2013) Sediment fingerprinting in fluvial systems: review of tracers, sediment sources and mixing models. Int J Sediment Res 28:560–578
Hancock GJ, Wilkinson SN, Hawdon AA, Keen RJ (2014) Use of fallout tracers 7Be, 210Pb and 137Cs to distinguish the form of sub-surface soil erosion delivering sediment to rivers in large catchments. Hydrol Process 28:3855–3874
Harper SE, Foster IDL, Lawler DM, Mathers KL, McKenzie M, Petts GE (2017) The complexities of measuring fine sediment accumulation within gravel-bed rivers. River Res Appl 33:1575–1584
He Q, Walling DE (1996) Interpreting particle size effects in the adsorption of 137Cs and unsupported 210Pb by mineral soils and sediments. J Environ Radioact 30:117–137
Hoffmann T (2015) Sediment residence time and connectivity in non-equilibrium and transient geomorphic systems. Earth Sci Rev 150:609–627
Homer CG, Dewitz JA, Yang L, Jin S, Danielson P, Xian G, Coulston J, Herold ND, Wickham JD, Megown K (2015) Completion of the 2011 National Land Cover Database for the conterminous United States—representing a decade of land cover change information. Photogramm Eng Remote Sens 81:345–354
Horovitz CT (1975) Chapter 2: distribution in nature. In: Horovitz CT (ed) Scandium, its occurrence, chemistry, physics, metallurgy, biology, and technology. Academic Press Inc., New York, pp 18–49
Huh CA, Su CC (2004) Distribution of fallout nuclides (7Be, 137Cs, 210Pb and 239, 240Pu) in soils of Taiwan. J Environ Radioact 77:87–100
Izagirre O, Serra A, Guasch H, Elosegi A (2009) Effects of sediment deposition on periphytic biomass, photosynthetic activity and algal community structure. Sci Total Environ 407(21):5694–5700
Kelsey HM, Lamberson RL, Madej MA (1987) Stochastic model for the long-term transport of stored sediment in a river channel. Water Resour Res 23:1738–1750
Kennedy DJ (1983) Computation of continuous records of streamflow. U.S. Geological Survey Techniques of Water-Resources Investigations, Book 3, Chapter A13, 53 p
Koiter AJ, Owens PN, Petticrew EL, Lobb DA (2013) The behavioural characteristics of sediment properties and their implications for sediment fingerprinting as an approach for identifying sediment sources in river basins. Earth-Sci Rev 125:24–42
Laceby JP, Olley J, Pietsch TJ, Sheldon F, Bunn SE (2015) Identifying subsoil sediment sources with carbon and nitrogen stable isotope ratios. Hydrol Process 29:1956–1971
Laird D, Fleming P, Wang B, Horton R, Karlen D (2010) Biochar impact on nutrient leaching from a Midwestern agricultural soil. Geoderma 158:436–442
Lamba J, Karthikeyan KG, Thompson AM (2014) Using radiometric fingerprinting and phosphorus to elucidate sediment transport dynamics in an agricultural watershed. Hydrol Process 29:2681–2693
Lambert CP, Walling DE (1988) Measurement of channel storage of suspended sediment in a gravel-bed river. CATENA 15 (1):65-80
Lancaster ST, Casebeer NE (2007) Sediment storage and evacuation in headwater valleys at the transition between debris-flow and fluvial processes. Geology 35:1027–1030
Langland MJ, Cronin T (2003) A summary report of sediment processes in Chesapeake Bay and watershed. U.S. Geological Survey Water-Resources Investigations Report 03-4123, 109 pp
Le Cloarec MF, Bonté P, Lefèvre I, Mouchel JM, Colbert S (2007) Distribution of 7Be, 210Pb and 137Cs in watersheds of different scales in the Seine River basin—inventories and residence times. Sci Total Environ 375:125–139
Le Gall M, Evrard O, Foucher A, Laceby JP, Salvador-Blanes S, Maniere L, Lefèvre I, Cerdan O, Ayrault S (2017) Investigating the temporal dynamics of suspended sediment during flood events with 7Be and 210Pbxs measurements in a drained lowland catchment. Sci Rep 7:42099. https://doi.org/10.1038/srep42099
Liu C, Walling DE, Spreafico M, Ramasamy J, Thulstrup HD, Mishra A (2017) Sediment problems and strategies for their management: experience from several large river basins. United Nations Educational, Scientific and Cultural Organization, Paris, France, Publ SC-2017/WS/13, 19p. Available at: http://unesdoc.unesco.org/images/0025/002587/258795e.pdf. Accessed 21 March 2018
Mabit L, Benmansour M, Abril JM, Walling DE, Meusburger K, Iurian AR, Bernard C, Tarján S, Owens PN, Blake WH, Alewell C (2014) Fallout 210Pb as a soil and sediment tracer in catchment sediment budget investigations - a review. Earth Sci Rev 138:335–351
Malmon DV, Dunne T, Reneau ST (2002) Predicting the fate of sediment and pollutants in river floodplains. Environ Sci Technol 36:2026–2032
Manjoro M, Rowntree K, Kakembo V, Foster I, Collins AL (2017) Use of sediment source fingerprinting to assess the role of subsurface erosion in the supply of fine sediment in a degraded catchment in the eastern cape, South Africa. J Environ Manag 194:27–41
Martínez-Carreras N, Udelhoven T, Krein A, Gallart F, Iffly J, Ziebel J, Hoffmann L, Pfister L, Walling DE (2010) The use of sediment colour measured by diffuse reflectance spectrometry to determine sediment sources: application to the Attert river catchment (Luxembourg). J Hydrol 382:49–63
Marttila H, Kløve B (2014) Storage, properties and seasonal variations in fine-grained bed sediment within the main channel and headwaters of the River Sanginjoki, Finland. Hydrological Processes 28:4756–4765
Matisoff G, Wilson CG, Whiting PJ (2005) The 7Be/210Pbxs ratio as an indicator of suspended sediment age or fraction new sediment in suspension. Earth Surf Process Landf 30:1191–1201
McHale MR, Siemion J (2014) Turbidity and suspended sediment in the upper Esopus Creek watershed, Ulster County, New York: U.S. Geological Survey Scientific Investigations Report 2014–5200, 42 p. https://doi.org/10.3133/sir20145200
Miller JR, Mackin G, Orbock Miller SM (2015) Application of geochemical tracers to fluvial sediment. Springer International Publishing, Dordrecht, p 142
Moody JA (2017) Residence times and alluvial architecture of a sediment superslug in response to different flow regimes. Geomorphology 294:40–57
Motha JA, Wallbrink PJ, Hairsine PB, Grayson RB (2003) Determining the sources of suspended sediment in a forested catchment in southwestern Australia. Water Resour Res 39:1056–1070
Mukundan R, Walling DE, Gellis AC, Slattery MC, Radcliffe DR (2012) Sediment source fingerprinting: transforming from a research tool to a management tool. J Am Water Resour Assoc 48:1241–1257
Neunhäuserer C, Berreck M, Insam H (2001) Remediation of soils contaminated with molybdenum using soil amendments and phytoremediation. Water Air Soil Pollut 128:85–96
Nosrati K, Govers G, Ahmadi H, Sharifi F, Amoozegar MA, Merckx R, Vanmaercke M (2011) An exploratory study on the use of enzyme activities as sediment tracers: biochemical fingerprints? Int J Sediment Res 26:136–151
Owens PN, Batalla RJ, Collins AJ, Gomez B, Hicks DM, Horowitz AJ, Kondolf GM, Marden M, Page MJ, Peacock DH, Petticrew EL, Salomons W, Trustrum NA (2005) Fine-grained sediment in river systems -- environmental significance land management issues. River Res Appl 21:693–717
Palazón L, Gaspar L, Latorre B, Blake WH, Navas A (2015) Identifying sediment sources by applying a fingerprinting mixing model in a Pyrenean drainage catchment. J Soils Sediments 15:2067–2085
Palmer JA, Schilling KE, Isenhart TM, Schultz RC, Tomer MD (2014) Streambank erosion rates and loads within a single watershed: bridging the gap between temporal and spatial scales. Geomorphology 209:66–78
Papanicolaou, A.N., J.F. Fox, and J. Marshall. 2003. Soil fingerprinting in the Palouse Basin, USA, using stable carbon and nitrogen isotopes. International Journal of Sediment Research. 18(2):278–284.
Phillips JD, Marden M, Gomez B (2007) Residence time of alluvium in an aggrading fluvial system. Earth Surf Process Landf 32:307–316
Phillips JM, Russell MA, Walling DE (2000) Time-integrated sampling of fluvial suspended sediment: a simple methodology for small catchments. Hydrol Process 14:2589–2602
Piqué G, José A. López-Tarazón, Ramon J. Batalla, (2014) Variability of in-channel sediment storage in a river draining highly erodible areas (the Isábena, Ebro Basin). Journal of Soils and Sediments 14 (12):2031-2044
Pizzuto J, Schenk ER, Hupp CR, Gellis A, Noe G, Williamson E, Karwan DL, O'Neal M, Marquard J, Aalto R (2014) Characteristic length scales and time-averaged transport velocities of suspended sediment in the mid-Atlantic Region, USA. Water Resour Res 50:790–805
Provost R, Kohavi R (1998) On applied research in machine learning. Machine learning 30(2/3):127–132
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna URL https://www.R-project.org/
Schilling KE, Wolter CF (2000) Application of GPS and GIS to map channel features at Walnut Creek, Iowa. J Am Water Resour Assoc 36:1423–1434
Schilling, K.E. (2000) Patterns of discharge and suspended sediment transport in the Walnut and Squaw Creek Watersheds, Jasper County, Iowa. Iowa Department of Natural Resources, Iowa Geological Survey, Iowa City, Iowa. Geological Survey Bureau Technical Information Series 41
Schilling KE, Hubbard T, Luzier J, Spooner J (2006) Walnut Creek Watershed Restoration and Water Quality Monitoring Project: Iowa Geological Survey Technical Information Series No. 49, 125 pp
Schilling KE, Isenhart TM, Palmer JA, Wolter CF, Spooner J (2011) Impacts of land-cover change on suspended sediment transport in two agricultural watersheds. J Am Water Resour Assoc 47:672–686
Sherriff SC, Franks SW, Rowan JS, Fenton O, Ó’hUallacháin D (2015) Uncertainty-based assessment of tracer selection, tracer non-conservativeness and multiple solutions in sediment fingerprinting using synthetic and field data. J Soils Sediments 15:2101–2116
Skalak K, Pizzuto J (2010) The distribution and residence time of suspended sediment stored within the channel margins of a gravel-bed bedrock river. Earth Surf Process Landf 35:435–446
Slattery MC, Gares PA, Phillips JD (2002) Slope-channel linkage and sediment delivery on North Carolina coastal plain cropland. Earth Surf Process Landf 27:1377–1387
Smith HG, Blake WH (2014) Sediment fingerprinting in agricultural catchments -- a critical re-examination of source discrimination and data corrections. Geomorphology 204:177–191
Smith HG, Blake WH, Taylor A (2014) Modelling particle residence times in agricultural river basins using a sediment budget model and fallout radionuclide tracers. Earth Surf Process Landf 39:1944–1959
Stewart HA, Massoudieh A, Gellis A (2014) Sediment source apportionment in Laurel Hill Creek, PA, using Bayesian chemical mass balance and isotope fingerprinting. Hydrol Process 29(11):2545–2560
U.S. Census Bureau (2001) 2000 TIGER/line files: Washington, D.C., U.S. Department of Commerce. Available at: https://www.census.gov/geo/maps-data/data/tiger-line.html. Accessed March 2014
U.S. Department of Agriculture (2014) National Agriculture Imagery Program (NAIP). USDA Farm Service Agency, Aerial Photography Field Office (APFO) image server. Available at: http://gis.apfo.usda.gov/arcgis/rest/services/NAIP. Accessed March 2014
Van Metre PC, Frey JW, Tarquinio E (2012) The Midwest stream quality assessment. U.S. Geological Survey Fact Sheet 2012–3124, 2 pp
Van Metre PC, Mahler BJ, Carlisle D, Coles J (2018) Midwest stream quality assessment—influences of human activities on streams, U.S. Geological Survey Fact Sheet FS 2017-3087, 6 p. https://pubs.er.usgs.gov/fs20173087
Van Metre PC, Wilson JT, Fuller CC, Callender E, Mahler BJ (2004) Collection, analysis, and age-dating of sediment cores from 56 U.S. lakes and reservoirs sampled by the U.S. Geological Survey, 1992–2001. U.S. Geological Survey Scientific Investigations Report 2004-5184, 180 pp
Veenstra JJ, Lee Burras C (2015) Soil profile transformation after 50 years of agricultural land use. Soil Sci Soc Am J 79:1154–1162
Vermeire V, Cornu S, Fekiacova Z, Detienne M, Delvaux B, Cornélis JT (2016) Rare earth elements dynamics along pedogenesis in a chronosequence of podzolic soils. Chem Geol 446:163–174
Wagner RJ, Boulger RW Jr, Oblinger CJ, Smith BA (2006) Guidelines and standard procedures for continuous water-quality monitors—station operation, record computation, and data reporting. U.S. Geological Survey Techniques and Methods 1–D3, 51 p. + 8 attachments. Available at: http://pubs.water.usgs.gov/tm1d3. Accessed 16 January 2017
Wallbrink PJ, Murray AS, Olley JM, Olive L (1998) Determining sediment sources and transit times of suspended sediments in the Murrumbidgee River, NSW, Australia using fallout Cs-137 and Pb-210. Water Resour Res 34:879–887
Wallbrink P, Olley JM, Hancock G (2002) Estimating residence times of fine sediment in river channels using fallout 2l0Pb. In: Dyer FJ, Thorns MC, Olley JM (eds) The structure, function and management implications of fluvial sedimentary systems, vol 276. IAHS Press, Wallingford, pp 425–432
Walling DE (2005) Tracing suspended sediment sources in catchments and river systems. Sci Total Environ 344:159–184
Walling DE (2013) The evolution of sediment source fingerprinting investigations in fluvial systems. J Soils Sediments 13:1658–1675
Walling DE, Owens PN, Leeks GJL (1998) The role of channel and floodplain storage in the suspended sediment budget of the River Ouse, Yorkshire, UK. Geomorphology 22:225–242
Walling DE, Owens PN, Leeks GJL (1999) Fingerprinting suspended sediment sources in the catchment of the River Ouse, Yorkshire, UK. Hydrol Process 13:955–975
Warren N, Allan IJ, Cater JE, House WA, Parker A (2003) Pesticides and other micro-organic contaminants in freshwater sedimentary environments—a review. Appl Geochem 18:159–194
Wilber DH, Clarke DG (2001) Biological effects of suspended sediments: a review of suspended sediment impacts on fish and shellfish with relation to dredging activities in estuaries. North Amer J Fish Manag l21:855–875
Wilson AJ, Walling DE, Leeks GJL (2004) In-channel storage of fine sediment in rivers of southwest England. In: Golosov V, Belyaev V, and Walling DE, (Eds.) Sediment Transfer through the Fluvial System. International Association of Hydrological Sciences Publication no. 288. IAHS Press, Wallingford, 292-299
Wohl E, Scott DN (2017) Wood and sediment storage and dynamics in river corridors. Earth Surface Processes and Landforms 42:5-23
Wood PJ, Armitage PD (1997) Biological effects of fine sediment in the lotic environment. Environ Manag 21:203–217
Yanai J, Okada T, Yamada H (2012) Elemental composition of agricultural soils in Japan in relation to soil type, land use and region. Soil Sci Plant Nutr 58:1–10
Yesilonis I, Szlavecz K, Pouyat R, Whigham D, Xia L (2016) Historical land use and stand age effects on forest soil properties in the mid-Atlantic US. For Ecol Manag 370:83–92
Yi Y, Wang Z, Zhang K, Yu G, Duan X (2008) Sediment pollution and its effect on fish through food chain in the Yangtze River. Int J Sediment Res 23:338–347
Zhang Y, Collins AL, McMillan S, Dixon ER, Cancer-Berroya E, Poiret C, Stringfellow A (2017) Fingerprinting source contributions to bed sediment-associated organic matter in the headwater subcatchments of the River Itchen SAC, Hampshire, UK. River Res Appl 33:1515–1526
Acknowledgments
We would like to acknowledge the following individuals for their assistance in site selection, data collection, and handling: Jeffrey Frey and Stephen Kalkhoff, USGS and Kailyn Pederson, Justin D' Souza, and Lucas Tenborg, Iowa State University. For lab preparation and database management, we thank Lillian Gorman Sanisaca, Jeffrey Klein, and Lucas Nibert of USGS, and for GIS assistance, we thank Shannon Jackson, USGS. Staff at the Neal Smith Wildlife Refuge are also acknowledged for help with GIS coverages and site selection. Kelly McVicker for editing the manuscript and Kristin Jaeger for USGS colleague review. Data are available through U.S. Geological Survey Science Data Release https://www.sciencebase.gov/catalog/item/59889f04e4b05ba66e9ffea7. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the US Government.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Olivier Evrard
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gellis, A.C., Fuller, C.C., Van Metre, P. et al. Combining sediment fingerprinting with age-dating sediment using fallout radionuclides for an agricultural stream, Walnut Creek, Iowa, USA. J Soils Sediments 19, 3374–3396 (2019). https://doi.org/10.1007/s11368-018-2168-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-018-2168-z