Abstract
Purpose
Biochar has emerged as a promising material for limiting the bioavailability of potentially toxic elements in contaminated soils. Despite the benefits associated with the agricultural use of biochar, there is minimal information available on biochar amendment of Cd accumulation in leafy vegetable cultivars.
Materials and methods
A pot experiment was carried out to evaluate the feasibility of bamboo biochar (BB) and rice straw biochar (RSB) for decreasing cadmium (Cd) availability in artificially contaminated (Cd at 50 mg kg−1) soil, and its accumulation in Brassica chinensis. Biochar was applied at 0 (control), 0.5, 1, 2.5, and 5% (w/w).
Results and discussion
RSB at 5% significantly reduced the availability of Cd in soil (by 19.3%) compared with BB (by 8.6%). Application of BB and RSB at 1–5% markedly enhanced soil pH by 1.17–5.48 and 2.47–7.46%, respectively. Moreover, soil organic carbon was improved by 40.3–155 and 22.2–111% with 1–5% BB and RSB amendment, respectively. Soil total N was significantly increased by 24.5 and 31.1% with 5% BB and RSB application, respectively. Soil available P was increased by 10.8–24.4 and 14.6–30.7% with 2.5–5% BB or RSB amendment, respectively. Application of 2.5–5% BB and RSB significantly increased soil available K by 76.8–291 and 203–627%, respectively. Biochar at 2.5–5% ratio significantly improved nutrient (P and K) uptake in B. chinensis. Furthermore, biochar was more effective at higher rates, and RSB performed better than BB as a soil nutrient supplement. Cd accumulation in B. chinensis was significantly reduced by 12.0–48.3 and 17.0–35.4% with 0.5–5% BB and 1–5% RSB amendment, respectively. Greater biochar application further decreased bioconcentration factor (BCF) in harvested B. chinensis (all values < 1). Incorporation of 5% BB and RSB significantly reduced BCF by 51.0 and 40.6%, respectively.
Conclusions
Our results suggest that BB and RSB, especially at higher rates (> 2.5%), could be recommended as safe amendments for the immobilization of Cd in contaminated agricultural soils, thereby decreasing its threat to the food chain and human health.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Potentially toxic elements (PTEs) in soils gradually arouse worldwide concern as for their excessive concentrations and thus high risks to agricultural system and human health (Bannon et al. 2009). Cadmium (Cd) is one of the most toxic PTEs, with background level in soils less than 1 mg kg−1 (Mohamed et al. 2015). Cd contamination of soil has become an emerging environmental and health problem due to its non-biodegradability, severe toxicity, and mobility to plants and subsequent accumulation in food chain (Khan et al. 2017). The entrance of Cd into the food chain can result in many chronic health problems for humans, such as renal cell and liver function damage, lung edema, recurrent miscarriage, bone disease, and hypertension (Chen et al. 2008; Mohamed et al. 2015). Therefore, the amelioration of Cd-contaminated soils is important for reducing health risks to humans and ensuring food safety (Karami et al. 2011). Most traditional remediation methods, such as soil washing, excavation, and landfilling, are presently unsuitable due to their high costs and disruption of the environment (Mench et al. 2010). Recently, the use of soil amendments such as biochar has been considered beneficial for remediation of polluted agricultural soil through immobilization of contaminants, for example, PTEs (Karami et al. 2011; Mohamed et al. 2015; Rizwan et al. 2016; Lu et al. 2017).
Biochar typically refers to the carbon-rich solid product derived from biomass by pyrolysis in oxygen-deficient environment (Joseph et al. 2010). The increasing attention toward biochar is ascribed to its beneficial use for improving soil properties, increasing crop yield, reducing nutrient leaching, and mitigating climate change (Ding et al. 2010; Park et al. 2011; Houben et al. 2013; Hussain et al. 2017). More importantly, biochar shows exceptional adsorption capacity for PTEs due to its highly porous structure and various functional groups, e.g., carboxyls, hydroxyls, and phenols (Kei et al. 2004; Cao et al. 2009; Paz-Ferreiro et al. 2014; Li et al. 2017). Furthermore, biochar has been considered as an effective sorbent for decreasing the availability and toxicity of PTEs in soil through alteration of the stable forms (Beesley et al. 2011; Uchimiya et al. 2012; Ahmad et al. 2014; Lima et al. 2014; Khan et al. 2016). Therefore, the application of biochar has recently been suggested as a sustainable means of promoting the remediation of contaminated soils and revegetation of degraded lands (Beesley et al. 2011; Fellet et al. 2011; Qi et al. 2018).
The effect of biochar on bioavailability of PTEs is jointly controlled by soil properties, biochar properties, and environmental factors of biochar-amended soils (El-Naggar et al. 2018). On one hand, key properties of soil, such as pH, soil organic matter, clay, and cation exchange capacity (CEC), dominate the solubility, mobility, and bioavailability of PTEs (Ye et al. 2014). On the other hand, key features of biochar including pH, organic carbon content, CEC, pore structure, surface area, and surface functional groups determine the metal immobilization capacity of biochar in soils (Xie et al. 2015; Qi et al. 2017).
The characteristics of biochar depend mainly on the type of raw material and pyrolysis condition (Hossain et al. 2011; Hussain et al. 2017). Biochar pH can range from acidic to alkaline (Chan and Xu 2009). Biochar with acidic pH is generally produced under lower temperatures (Novak et al. 2009b; Hagner et al. 2016; Zhang et al. 2017), and one previous study has investigated Cd bioavailability in soils amended with acidic and neutral biochar (Qi et al. 2018). Therefore, it is desirable to investigate the effect of alkaline biochar on Cd availability in soils. Furthermore, biochar can be produced from a wide range of biomass sources, such as crop residues, woodchips, livestock manures, and sewage sludges (Paz-Ferreiro et al. 2014). Rice is a staple food for more than 3.5 billion people worldwide, and about 620 million t of rice straw was produced in Asia in 2008, with this quantity increasing every year (Liu et al. 2017). In most places, rice straw has little commercial value and is usually burned in fields or directly incorporated into soils, which results in air pollution or methane emissions, respectively. Moreover, bamboo can be selectively harvested and regenerated without replanting due to its annual increase rate of 10–30% in biomass accumulation (Ding et al. 2010), making it an attractive feedstock for biochar production. Most importantly, our previous research showed that both biochars derived from rice straw and bamboo present alkaline nature (Liu et al. 2011; Liu et al. 2016).
Leafy vegetables are frequently consumed by humans as a vital source of proteins, vitamins, carbohydrates, and minerals, but can accumulate higher concentrations of Cd than other crops (Yang et al. 2010; Ivey et al. 2012). Despite the benefits associated with the agricultural use of biochar, there is minimal information available on biochar amendment of Cd accumulation in leafy vegetable cultivars. Therefore, the objective of this research was to evaluate the extent to which biochar soil amendments derived from rice straw and bamboo chip affect Cd availability in contaminated soil, and Cd accumulation in Brassica chinensis L., a leafy vegetable cultivar. The effects of the two biochars on soil properties and nutrient (N, P, and K) uptake in B. chinensis L. were also investigated. We selected B. chinensis L. because it has the advantages of rapid growth and elevated fully harvestable biomass production, and is tolerant of higher metal concentrations than other crops (Yang et al. 2010; Wu et al. 2013). The accumulation of detrimental concentrations of Cd in aboveground organs by B. chinensis, even in soils with low concentrations of Cd, has received much attention (Chen et al. 2010; Yasmin et al. 2017). In this study, we set a relatively high concentration of Cd in the soil, with two kinds of biochar amendment. The results can provide a scientific basis for the application of biochar for remediation of Cd-polluted soil and can be of significance for safer production of agricultural products.
2 Materials and methods
2.1 Materials
Soil samples were collected from the surface layer (0–20 cm) of an experimental field in Hangzhou City, Zhejiang Province, China. The soil was air-dried and crushed to pass through a 2-mm sieve. Soil basic properties were pH (1:2.5 H2O) 6.27, soil organic matter 10.5 g kg−1, total nitrogen (N) 0.822 g kg−1, available phosphorus (P) 33.7 mg kg−1, available potassium (K) 63.3 mg kg−1, and cation exchange capacity (CEC) 13.7 cmol kg−1. Total Cd content of the tested soil was 8.70 mg kg−1. Bamboo biochar (BB) and rice straw biochar (RSB) used as soil additives were derived from pyrolysis of bamboo chips and rice straw, respectively, at 600 °C in a programmable tube furnace (Hangzhou Lantian Instrument Co., Ltd., China). Typically, the prepared biomass was batch pyrolyzed under anaerobic conditions at a heating rate of 25 °C min−1 and a residence time of 1 h. Finally, the produced biochars were allowed to cool to room temperature. Particulate biochar mass was ground to pass through a 2-mm sieve and mixed thoroughly to obtain a fine granular consistency for further use. The basic physical and chemical properties of the biochars are shown in Table 1. The Cd content of BB and RSB was 0.35 and 0.84 mg kg−1, respectively.
2.2 Experimental design
A pot experiment was conducted in a greenhouse at Zhejiang Academy of Agricultural Sciences, China. The experiment comprised ten treatments of three replicates formed from the combination of two biochar types (BB or RSB) applied at four rates (0.5, 1, 2.5, and 5%). The experimental pots (20 cm diameter × 18 cm height) were filled with 2.0 kg of soil. The source of Cd was CdCl2 with cadmium concentration of 50 mg kg−1 in soil. Both Cd and biochar rates were mixed with the surface layer soil 1 week before transplanting B. chinensis L. cv. Suzhou for equilibration under 60% water-holding capacity (WHC). Uniformly sized B. chinensis were planted one per pot and grown in the greenhouse at room temperature. Throughout the experimental period, soil moisture content was adjusted weekly to 60% WHC by the addition of tap water. Fertilizer was added as aqueous solution (0.16 g N, 0.08 g P2O5, and 0.10 g K2O per pot) in the form of urea, super-phosphate, and potassium chloride, respectively. Treatment without biochar was set as a control series (CK). Treatment without biochar or Cd was set as an experimental blank (BL).
2.3 Sampling and measurements
Biochar properties, such as pH, were measured at a solid:water ratio of 1:10. The C and N contents of biochar were determined using an elemental analyzer (Vario EL/μ cube, Elementar, Germany). P and K were measured following the protocols recommended by Lu (2000). CEC was determined by sodium acetate (1 mol L−1, pH 8.2) extraction followed by flame photometry. Specific surface area and total pore volume were measured using a BET surface area analyzer (ASAP2020, Micromeritics, USA).
The plants were harvested after 3 months, washed in tap water and distilled water to remove soil particles, and then oven-dried at 60–70 °C for 48 h and crushed to powder. Part of the plant samples was used for nutrient measurement (N, P, and K content), and the other part was digested to determine Cd concentrations.
A microwave digestion instrument (CEM-MARS 5, USA) with pressure and temperature controller was used for pretreatment of soil and plant samples. Soil samples were taken from the 0–15-cm soil layer of each plot after harvest. After air-drying, the samples were sieved < 0.15 mm. Soil pH was measured using a pH meter (Mettler-Toledo, Switzerland). Soil organic carbon (SOC) was measured by K2Cr2O7-H2SO4 oxidation (Lu 2000). Total N was measured by the semimicro-Kjeldahl method. Available P was measured by treatment with 0.5 mol L−1 NaHCO3 (pH 8.5) followed by molybdenum blue colorimetry (Lu 2000). Available K was measured by 1 mol L−1 NH4OAc extraction-flame photometry (Lu 2000).
To measure the concentrations of Cd, a 0.25 g soil sample was weighed in a polytetrafluoroethylene tube and then treated with 7 mL HNO3 (plant samples with 6 mL HNO3 and 1 mL HF) using a microwave oven for 6 min to ensure complete digestion of particles. The microwave process was following the procedure recommended by Anzano et al. (2000). Later, the sample was concentrated to about 1 mL by heating at 170 °C and then cooling to room temperature. The digested solution was diluted to 50 mL with ultrapure water and stored at 4 °C until further analysis by flame atomic absorption spectrometry (Varian AA240FS), with three replicates for each sample. The bioconcentration factor (BCF) was calculated based on the dry weight using the following formula (Li et al. 2016):
where Cdplant (mg kg−1) and Cdsoil (mg kg−1) represent Cd concentrations in the plant and soil, respectively.
2.4 Data processing
Data were analyzed by one-way analysis of variance (ANOVA) and least significant difference (LSD) multiple comparison using SPSS statistical software (version 16.0). The results are presented as mean values with standard deviation, and values of P < 0.05 are considered statistically significant. All figures were drawn using SigmaPlot 10.0 software (Systat Software Inc., USA).
3 Results and discussion
3.1 Effect of biochar on soil basic properties
The effects of biochar on soil basic properties were investigated after harvesting B. chinensis as shown in Table 2. Generally, soil properties were not significantly altered (in comparison with CK) by low input (< 1%) of either BB or RSB to soil with Cd concentration of 50 mg kg−1. Soil pH increased with increasing application ratio of RSB and BB. At BB or RSB applications > 1%, soil pH increased markedly, by 1.17–5.48 and 2.47–7.46%, respectively, compared with CK (5.57), with RSB treatment invoking significantly higher pH than BB treatment. Many previous studies have also reported that biochar application increases soil pH (Laird et al. 2010; Mukherjee et al. 2014; Butnan et al. 2015; Liu et al. 2016).
SOC is an important indicator of soil quality. Compared with CK, BB application > 0.5% significantly increased SOC by 26.8–155%, while RSB > 1% significantly increased SOC by 22.2–111%. BB treatment showed significantly higher SOC than RSB application at the same ratio, which is likely due to higher C content of BB than RSB (Table 1).
Total N in soil increased significantly by 11.7 and 15.3% with 2.5% BB or RSB application, and increased by 24.5 and 31.1% with 5% BB or RSB application, respectively, compared to CK (1.42 g kg−1). This result is in line with Borchard et al. (2014), who reported that wood biochar application at 45 t ha−1 significantly improved soil total N content.
Similarly to the finding for total N, soil available P significantly increased by 10.8–24.4 and 14.6–30.7% with 2.5–5% BB or RSB application, respectively, compared to that of CK (31.3 mg kg−1). However, there were no significant differences between the effects of BB and RSB at the same ratio. In comparison with CK, the application of > 2.5% BB significantly increased soil available K by 76.8–291%, while > 1% RSB significantly increased soil available K by 127–627%, with RSB showing significantly greater effect than BB at a given application ratio.
Previous studies have shown that soil nutrient content can be significantly improved by biochar amendment (Laird et al. 2010; Masulili et al. 2010; Liang et al. 2014). This is predominantly due to direct supplement of these nutrients, such as P and K, which were present in soluble form in biochar itself (Enders et al. 2012) and rapidly released into the soil solution (Glaser et al. 2002). In this research, soil available P and K contents were significantly increased by biochar (especially RSB) amendment. Similarly, Lai et al. (2013) found that available K content was significantly higher in the soil amended with woodchip biochar than that in unamended soil. Furthermore, from our results (Table 1), RSB contained more P and K than did BB, demonstrating that RSB appears better suited than BB as a soil nutrient supplement.
3.2 Effect of biochar on immobilizing Cd in soil
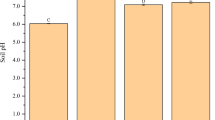
The effects of applying BB or RSB at different ratios on soil Cd availability are shown in Fig. 1. Soil Cd availability decreased with increasing biochar application. With application of 5% BB or RSB, available Cd content in soil decreased significantly by 8.6 and 19.3%, respectively, compared to that of CK (20.5 mg kg−1). Furthermore, available Cd content significantly decreased by 9.0% with 2.5% RSB application, whereas it was not significantly affected by 2.5% BB application. These results demonstrate that, at a given application rate, RSB has stronger effect than BB on immobilizing Cd in soil. This supports the results of Yi et al. (2013), who attributed this to more direct interactions between Cd and functional groups when applying RSB than when applying other biochar. Similarly, several studies have reported the positive association between biochar application and immobilization of soil Cd content (Fellet et al. 2011; Park et al. 2011; Houben et al. 2013).
Effect of biochar amendment on available Cd content in the soil. CK no soil-amendment control, BB bamboo biochar, RSB rice straw biochar. Error bars represent standard deviations of triplicate measurements. Different lowercase letters indicate the statistically significant differences at p ≤ 0.05 between the treatments
As shown by previous studies, possible mechanisms for the immobilization of Cd by biochars include cation exchange between Cd and protons or alkaline metals on biochar surface (Trakal et al. 2014; Li et al. 2017), surface complexation (ionic, covalent, or hydrogen bonding) with biochar minerals (Qian and Chen 2013; Bian et al. 2014) and oxygen-containing functional groups (Uchimiya et al. 2011; Rodríguez-Vila et al. 2015), precipitation with inorganic components (like PO43−, CO32−, SO42−, OH−) to form insoluble compounds (Cao et al. 2009; Inyang et al. 2012; Jiang et al. 2012; Wang et al. 2015), and electrostatic interactions with π electrons (C=C) (Park et al. 2011). The mechanisms for the immobilization of soil Cd content through biochar application depended on the type of biochar feedstock and the application rate. In this study, RSB was more effective than BB in immobilizing Cd when applied at 5% rate (Fig. 1). This is mainly attributed to the higher pH and CEC of RSB than that of BB (Table 1), which leads to more precipitation and cation exchange with Cd in the soil, respectively. Immobilization of Cd generally resulted from adsorption on biochars and from the increase in pH, which can affect precipitation in the soil (Uchimiya et al. 2010; Beesley and Marmiroli 2011; Karami et al. 2011). Similarly, Mohamed et al. (2015) attributed the significantly reduced availability of Cd by bamboo biochar in some soils to elevated soil pH and CEC. Biochars with high oxygen-containing functional groups are particularly effective for the stabilization of PTEs in acidic soils with low SOC (Uchimiya et al. 2011). Available Cd content usually has negative correlation with SOC in soils with biochar amendment, because the capacity for Cd adsorption can be enhanced through cation exchange and complexation by soils with higher SOC content (Yi et al. 2013). Moreover, the high capacity to adsorb Cd was also attributed to the pore structure and large surface area of biochar, which provide available sites for Cd adsorption (Lu et al. 2017).
3.3 Effect of biochar on Cd accumulation in Brassica crop
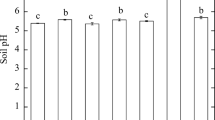
The effect of biochar on Cd accumulation in B. chinensis is shown in Fig. 2. The Cd content of the crop decreased significantly, by 12.0–48.3%, with addition of 0.5–5% BB, and by 17.0–35.4% with 1–5% RSB, compared to that of CK (47.8 mg kg−1). The results demonstrate that the application of BB or RSB can effectively immobilize Cd, thereby reducing the mobility and biotoxicity of Cd contained in soil, and consequently inhibiting the uptake of Cd by B. chinensis. Our results are in accordance with Yasmin et al. (2017), who found that Cd accumulation in plants was significantly reduced following amendment with biochar derived from cow manure, which was attributed to increased soil pH and decreased Cd availability. Similarly, Woldetsadik et al. (2016) observed that the phytoavailability of Cd to lettuce (Lactuca sativa) was reduced in heavily contaminated agricultural soil by biochar amendment. Chen et al. (2016) reported that Cd concentration was significantly reduced in Cd-accumulating rice cultivars in an acidic paddy soil by the application of wheat straw biochar, which was attributed to the formation of stable metal–organic complexes. It was found by Houben et al. (2013) that the use of 5 and 10% Miscanthus straw biochar reduced Cd concentrations in Brassica napus shoots by 47 and 75%, respectively, compared to controls, thereby confirming the vital role of biochar in limiting Cd bioavailability.
Effect of biochar amendment on Cd content in plant. CK no soil-amendment control, BB bamboo biochar, RSB rice straw biochar. Error bars represent standard deviations of triplicate measurements. Different lowercase letters indicate the statistically significant differences at p ≤ 0.05 between the treatments
3.4 Effect of biochar on BCF of Cd in Brassica chinensis L.
Data on BCF in Cd-polluted soils planted with B. chinensis with the addition of biochar are given in Table 2. Brassica chinensis tissues showed noticeably lower BCF values after biochar addition, and increasing biochar rates were associated with large decreases in BCF. The addition of 0.5–5% BB or 1–5% RSB in soils containing 50 mg Cd kg−1 significantly reduced BCF by 14.4–51.0 and 18.4–40.6%, respectively, in harvested B. chinensis.
BCF is considered an important factor for evaluating the capacity of plants to accumulate PTEs from soil (Mohamed et al. 2015). In our study, all samples showed BCF values < 1 (Table 2), indicating that phytoextraction of Cd by B. chinensis was not feasible under the tested conditions, i.e., at 0.5, 1, 2.5, and 5% biochar, and grown in soils containing 50 mg Cd kg−1 (McGrath and Zhao 2003; Houben et al. 2013; Fellet et al. 2014). The decrease of BCF values after the addition of biochar likely resulted from the high efficiency of biochar in limiting Cd uptake by B. chinensis. The absorption and translocation of metals are fundamental to assessing the potential hazards of growing plants in polluted soils.
3.5 Effect of biochar on nutrient uptake by Brassica chinensis
The effects of biochar amendment on the nutrient (N, P, and K) contents of B. chinensis are shown in Fig. 3. N content was not significantly affected by biochar amendment. In contrast, the P and K contents increased significantly with > 2.5% BB or RSB amendment, compared to CK. However, there were no significant differences between the effects of BB and RSB at a given rate, except that K content was higher at 5% RSB than 5% BB. The phytoavailability of nutrients other than N often depends on soil pH. As biochar application affects soil pH (Novak et al. 2009a), it may indirectly alter the supply of nutrients to plants (Demeyer et al. 2001; Atkinson et al. 2010). In this work, we observed relatively high soil pH following the addition of biochar (BB or RSB) at higher rates (1, 2.5, and 5%), which improved nutrient phytoavailability.
Effect of biochar amendment on nutrient (N, P, and K) contents in plant. CK no soil-amendment control, BB bamboo biochar, RSB rice straw biochar. Error bars represent standard deviations of triplicate measurements. Different lowercase letters within a group indicate the statistically significant differences at p ≤ 0.05 between the treatments
In the present study, the application of > 2.5% BB or RB increased the K content of B. chinensis by 49.7–70.5 and 62.8–97.3%, respectively, relative to CK (Fig. 3). Similarly, Lehmann et al. (2003) observed that, in addition to increasing K availability in soil, the application of biochar increased plant K uptake. Although biochar provides some nutrients when added as a fertilizer, more importantly, it can improve plant nutrient uptake efficiency when added as a soil conditioner (Peng et al. 2011). The porous structure of biochar can also result in enhanced nutrient availability through water retention and improved water balance (Oguntunde et al. 2008; Asai et al. 2009; Major et al. 2010).
4 Conclusions
The incorporation of biochar into Cd-contaminated soils limited the phytoavailability of Cd. Additionally, biochar had positive effects on soil properties (such as pH, SOC, total N, and available P and K) and on nutrient (P and K) uptake by B. chinensis, although these effects depend on the biochar feedstock materials and especially the application rates. Overall, 2.5–5.0% are recommended as suitable application rates for both biochars, and RSB is more effective than BB on soil nutrient supplement and Cd immobilization. Both BB and RSB can be used as soil amendments to alleviate health risks associated with consuming leafy vegetables grown in Cd-contaminated acidic soil.
References
Ahmad M, Rajapaksha AU, Lim JE, Zhang M, Bolan N, Mohan D, Vithanage M, Lee SS, Ok YS (2014) Biochar as a sorbent for contaminant management in soil and water: a review. Chemosphere 99:19–33. https://doi.org/10.1016/j.chemosphere.2013.10.071
Anzano JM, Asensio N, Anwar J, Martínez-Bordenave MC (2000) Zinc and manganese analysis in maize by microwave oven digestion and flame atomic absorption spectrometry. J Food Compos Anal 13(5):837–841. https://doi.org/10.1006/jfca.2000.0923
Asai H, Samson BK, Stephan HM, Songyikhangsuthor K, Homma K, Kiyono Y, Inoue Y, Shiraiwa T, Horie T (2009) Biochar amendment techniques for upland rice production in Northern Laos 1. Soil physical properties, leaf SPAD and grain yield. Field Crop Res 111(1-2):81–84. https://doi.org/10.1016/j.fcr.2008.10.008
Atkinson CJ, Fitzgerald JD, Hipps NA (2010) Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: a review. Plant Soil 337(1-2):1–18. https://doi.org/10.1007/s11104-010-0464-5
Bannon DI, Drexler JW, Fent GM, Casteel SW, Hunter PJ, Brattin WJ, Major MA (2009) Evaluation of small arms range soils for metal contamination and lead bioavailability. Environ Sci Technol 43(24):9071–9076. https://doi.org/10.1021/es901834h
Beesley L, Moreno-Jiménez E, Gomez-Eyles JL, Harris E, Robinson B, Sizmur T (2011) A review of biochars’ potential role in the remediation, revegetation and restoration of contaminated soils. Environ Pollut 159(12):3269–3282. https://doi.org/10.1016/j.envpol.2011.07.023
Beesley L, Marmiroli M (2011) The immobilization and retention of soluble arsenic, cadmium and zinc by biochar. Environ Pollut 159(2):474–480. https://doi.org/10.1016/j.envpol.2010.10.016
Bian R, Joseph S, Cui L, Pan G, Li L, Liu X, Zhang A, Rutlidge H, Wong S, Chia C, Marjo C, Gong B, Munroe P, Donne S (2014) A three-year experiment confirms continuous immobilization of cadmium and lead in contaminated paddy field with biochar amendment. J Hazard Mater 272:121–128. https://doi.org/10.1016/j.jhazmat.2014.03.017
Borchard N, Siemens J, Ladd B, Möller A, Amelung W (2014) Application of biochars to sandy and silty soil failed to increase maize yield under common agricultural practice. Soil Till Res 144:184–194
Butnan S, Deenik JL, Toomsan B, Antal MJ, Vityakon P (2015) Biochar characteristics and application rates affecting corn growth and properties of soils contrasting in texture and mineralogy. Geoderma 237–238:105–116
Cao XD, Ma LQ, Gao B, Harris W (2009) Dairy-manure derived biochar effectively sorbs lead and atrazine. Environ Sci Technol 43(9):3285–3291. https://doi.org/10.1021/es803092k
Chan KY, Xu Z (2009) Biochar: nutrient properties and their enhancement. In: Lehmann J, Joseph S (eds) Biochar for environmental management: science and technology. Earthscan, London, UK, pp 67–84
Chen D, Guo H, Li R, Li L, Pan G, Chang A, Joseph S (2016) Low uptake affinity cultivars with biochar to tackle Cd-tainted rice—a field study over four rice seasons in Hunan, China. Sci Total Environ 541:1489–1498
Chen H, Yao J, Zhou Y, Chen H, Wang F, Gai N, Zhuang R, Ceccanti B, Maskow T, Zaray G (2008) Investigation of the toxic effect of cadmium on Candida humicola and Bacillus subtilis using a microcalorimetric method. J Hazard Mater 159(2-3):465–470. https://doi.org/10.1016/j.jhazmat.2008.02.040
Chen HL, Lai HY, Wang SM, Kuo YC, Lu CJ (2010) Effect of biosolids and Cd/Pb interaction on the growth and Cd accumulation of Brassica rapa grown in Cd-contaminated soils. Water Air Soil Poll 206(1-4):385–394. https://doi.org/10.1007/s11270-009-0114-9
Demeyer A, Voundi Nkana JC, Verloo MG (2001) Characteristics of wood ash and influence on soil properties and nutrient uptake: an overview. Bioresour Technol 77(3):287–295. https://doi.org/10.1016/S0960-8524(00)00043-2
Ding Y, Liu Y, Wu W, Shi D, Yang M, Zhong Z (2010) Evaluation of biochar effects on nitrogen retention and leaching in multi-layered soil columns. Water Air Soil Poll 213(1-4):47–55. https://doi.org/10.1007/s11270-010-0366-4
El-Naggar A, Shaheen SM, Ok YS, Rinklebe J (2018) Biochar affects the dissolved and colloidal concentrations of Cd, Cu, Ni, and Zn and their phytoavailability and potential mobility in a mining soil under dynamic redox-conditions. Sci Total Environ 624:1059–1071. https://doi.org/10.1016/j.scitotenv.2017.12.190
Enders A, Hanley K, Whitman T, Joseph S, Lehmann J (2012) Characterization of biochars to evaluate recalcitrance and agronomic performance. Bioresour Technol 114:644–653
Fellet G, Marchiol L, Vedove GD, Peressotti A (2011) Application of biochar on mine tailings: effects and perspectives for land reclamation. Chemosphere 83(9):1262–1267. https://doi.org/10.1016/j.chemosphere.2011.03.053
Fellet G, Marmiroli M, Marchiol L (2014) Elements uptake by metal accumulator species grown on mine tailings amended with three types of biochar. Sci Total Environ 468–469:598–608
Glaser B, Lehmann J, Zech W (2002) Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—a review. Biol Fert. Soils 35:219–230
Hagner M, Kemppainen R, Jauhiainen L, Tiilikkala K, Setälä H (2016) The effects of birch (Betula spp.) biochar and pyrolysis temperature on soil properties and plant growth. Soil Tillage Res 163:224–234
Hossain MK, Strezov V, Chan KY, Ziolkowski A, Nelson PF (2011) Influence of pyrolysis temperature on production and nutrient properties of wastewater sludge biochar. J Environ Manag 92:223–228
Houben D, Evrard L, Sonnet P (2013) Beneficial effects of biochar application to contaminated soils on the bioavailability of Cd, Pb and Zn and the biomass production of rapeseed (Brassica napus L.) Biomass Bioenergy 57:196–204
Hussain M, Farooq M, Nawaz A, Al-Sadi AM, Solaiman ZM, Alghamdi SS, Ammara U, Ok YS, Siddique KHM (2017) Biochar for crop production: potential benefits and risks. J Soils Sediments 17(3):685–716. https://doi.org/10.1007/s11368-016-1360-2
Inyang M, Gao B, Yao Y, Xue Y, Zimmerman AR, Pullammanappallil P, Cao X (2012) Removal of heavy metals from aqueous solution by biochars derived from anaerobically digested biomass. Bioresour Technol 110:50–56
Ivey MLL, Lejeune JT, Miller SA (2012) Vegetable producers’ perceptions of food safety hazards in the Midwestern USA. Food Control 26(2):453–465. https://doi.org/10.1016/j.foodcont.2012.01.065
Jiang J, Xu RK, Jiang TY, Li Z (2012) Immobilization of Cu(II), Pb(II) and Cd(II) by the addition of rice straw derived biochar to a simulated polluted Ultisol. J Hazard Mater 229–230:145–150
Joseph SD, Camps-Arbestain M, Lin Y, Munroe P, Chia CH, Hook J, Van Zwieten L, Kimber S, Cowie A, Singh BP, Lehmann J, Foidl N, Smernik RJ, Amonette JE (2010) An investigation into the reactions of biochar in soil. Aust J Soil Res 48(7):501–515. https://doi.org/10.1071/SR10009
Karami N, Clemente R, Moreno-Jiménez E, Lepp NW, Beesley L (2011) Efficiency of green waste compost and biochar soil amendments for reducing lead and copper mobility and uptake to ryegrass. J Hazard Mater 191(1-3):41–48. https://doi.org/10.1016/j.jhazmat.2011.04.025
Kei M, Toshitatsu M, Yasuo H, Nishihara K, Nakanishi T (2004) Removal of nitrate-nitrogen from drinking water using bamboo powder charcoal. Bioresour Technol 95:255–257
Khan KY, Ali B, Cui X, Feng Y, Yang X, Stoffella PJ (2017) Impact of different feedstocks derived biochar amendment with cadmium low uptake affinity cultivar of pak choi (Brassica rapa ssb. chinensis L.) on phytoavoidation of Cd to reduce potential dietary toxicity. Ecotox Environ Safe 141:129–138. https://doi.org/10.1016/j.ecoenv.2017.03.020
Khan KY, Ali B, Cui X, Feng Y, Stoffela PJ, Pan F, Tang L, Yang X (2016) Effect of biochar amendment on bioavailability and accumulation of cadmium and trace elements in Brassica chinensis L. (Chinese cabbage). J Agric Sci 8(9):23–36. https://doi.org/10.5539/jas.v8n9p23
Lai WY, Lai CM, Ke GR, Chung RS, Chen CT, Cheng CH, Pai CW, Chen SY, Chen CC (2013) The effects of woodchip biochar application on crop yield, carbon sequestration and greenhouse gas emissions from soils planted with rice or leaf beet. J Taiwan Inst Chem Eng 44(6):1039–1044
Laird DA, Fleming P, Davis DD, Horton R, Wang B, Karlen DL (2010) Impact of biochar amendments on the quality of a typical Midwestern agricultural soil. Geoderma 158(3-4):443–449. https://doi.org/10.1016/j.geoderma.2010.05.013
Lehmann J, da Silva Jr JP, Steiner C, Nehls T, Zech W, Glaser B (2003) Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: fertilizer, manure and charcoal amendments. Plant Soil 249(2):343–357. https://doi.org/10.1023/A:1022833116184
Li H, Dong X, da Silva EB, de Oliveira LM, Chen Y, Ma LQ (2017) Mechanisms of metal sorption by biochars: biochar characteristics and modifications. Chemosphere 178:466–478. https://doi.org/10.1016/j.chemosphere.2017.03.072
Li Z, Qi X, Fan X, Du Z, Hu C, Zhao Z, Isa Y, Liu Y (2016) Amending the seedling bed of eggplant with biochar can further immobilize Cd in contaminated soils. Sci Total Environ 572:626–633. https://doi.org/10.1016/j.scitotenv.2016.05.020
Liang F, Li GT, Lin QM, Zhao XR (2014) Crop yield and soil properties in the first 3 years after biochar application to a calcareous soil. J Integr Agr 13(3):525–532. https://doi.org/10.1016/S2095-3119(13)60708-X
Lima IM, Boykin DL, Klasson KT, Uchimiya M (2014) Influence of post-treatment strategies on the properties of activated chars from broiler manure. Chemosphere 95:96–104. https://doi.org/10.1016/j.chemosphere.2013.08.027
Liu Y, Lu H, Yang S, Wang Y (2016) Impacts of biochar addition on rice yield and soil properties in a cold waterlogged paddy for two crop seasons. Field Crop Res 191:161–167. https://doi.org/10.1016/j.fcr.2016.03.003
Liu Y, Yang M, Wu Y, Wang H, Chen Y, Wu W (2011) Reducing CH4 and CO2 emissions from waterlogged paddy soil with biochar. J Soils Sediments 11(6):930–939. https://doi.org/10.1007/s11368-011-0376-x
Liu Y, Yao S, Wang Y, Lu H, Brar SK, Yang S (2017) Bio- and hydrochars from rice straw and pig manure: inter-comparison. Bioresour Technol 235:332–337. https://doi.org/10.1016/j.biortech.2017.03.103
Lu K, Yang X, Gielen G, Bolan N, Ok YS, Niazi NK, Xu S, Yuan G, Chen X, Zhang X, Liu D, Song Z, Liu X, Wang H (2017) Effect of bamboo and rice straw biochars on the mobility and redistribution of heavy metals (Cd, Cu, Pb and Zn) in contaminated soil. J Environ Manag 186(Pt 2):285–292. https://doi.org/10.1016/j.jenvman.2016.05.068
Lu R (2000) Methods of soil and agro-chemical analysis. China Agricultural Science and Technology Press, Beijing (in Chinese)
Major J, Lehmann J, Rondon M, Goodale C (2010) Fate of soil-applied black carbon: downward migration, leaching and soil respiration. Glob Chang Biol 16(4):1366–1379. https://doi.org/10.1111/j.1365-2486.2009.02044.x
Masulili A, Utomo WH, MS S (2010) Rice husk biochar for rice based cropping system in acid soil 1. The characteristics of rice husk biochar and its influence on the properties of acid sulfate soils and rice growth in West Kalimantan, Indonesia. J Agric Sci 2:39–47
McGrath SP, Zhao FJ (2003) Phytoextraction of metals and metalloids from contaminated soils. Curr Opin Biotechnol 14(3):277–282. https://doi.org/10.1016/S0958-1669(03)00060-0
Mench M, Lepp N, Bert V, Schwitzguébel JP, Gawronski SW, Schöder P, Van-gronsveld J (2010) Successes and limitations of phytotechnologies at field scale: outcomes, assessment and outlook from COST Action 859. J Soils Sediments 10(6):1039–1070. https://doi.org/10.1007/s11368-010-0190-x
Mohamed I, Zhang GS, Li ZG, Liu Y, Chen F, Dai K (2015) Ecological restoration of an acidic Cd contaminated soil using bamboo biochar application. Ecol Eng 84:67–76. https://doi.org/10.1016/j.ecoleng.2015.07.009
Mukherjee A, Lal R, Zimmerman AR (2014) Effects of biochar and other amendments on the physical properties and greenhouse gas emissions of an artificially degraded soil. Sci Total Environ 487:26–36
Novak JM, Busscher WJ, Laird DL, Ahmedna M, Watts DW, Niandou MAS (2009a) Impact of biochar amendment on fertility of a southeastern coastal plain soil. Soil Sci 174(2):105–112. https://doi.org/10.1097/SS.0b013e3181981d9a
Novak JM, Lima I, Xing B, Gaskin JW, Steiner C, Das K, Ahmedna M, Rehrah D, Watts DW, Busscher WJ (2009b) Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann Environ Sci 3:195–206
Oguntunde PG, Abiodun BJ, Ajayi AE, Van de Giesen N (2008) Effects of charcoal production on soil physical properties in Ghana. J Plant Nutr Soil Sci 171(4):591–596. https://doi.org/10.1002/jpln.200625185
Park JH, Choppala GK, Bolan NS, Chung JW, Chuasavathi T (2011) Biochar reduces the bioavailability and phytotoxicity of heavy metals. Plant Soil 348(1-2):439–451. https://doi.org/10.1007/s11104-011-0948-y
Paz-Ferreiro J, Lu H, Fu S, Méndez A, Gascó G (2014) Use of phytoremediation and biochar to remediate heavy metal polluted soils: a review. Solid Earth 5(1):65–75. https://doi.org/10.5194/se-5-65-2014
Peng X, Ye LL, Wang CH, Zhou H, Sun B (2011) Temperature- and duration-dependent rice straw-derived biochar: characteristics and its effects on soil properties of an Ultisol in southern China. Soil Till Res 112(2):159–166. https://doi.org/10.1016/j.still.2011.01.002
Qi F, Kuppusamy S, Naidu R, Bolan NS, Ok YS, Lamb D, Li Y, Yu L, Semple KT, Wang H (2017) Pyrogenic carbon and its role in contaminant immobilization in soils. Crit Rev Environ Sci Technol 47(10):795–876. https://doi.org/10.1080/10643389.2017.1328918
Qi F, Lamb D, Naidu R, Bolan NS, Yan Y, Ok YS, Rahman MM, Choppala G (2018) Cadmium solubility and bioavailability in soils amended with acidic and neutral biochar. Sci Total Environ 610–611:1457–1466
Qian L, Chen B (2013) Dual role of biochars as adsorbents for aluminum: the effects of oxygen-containing organic components and the scattering of silicate particles. Environ Sci Technol 47(15):8759–8768. https://doi.org/10.1021/es401756h
Rizwan M, Ali S, Qayyum MF, Ibrahim M, Zia-ur-Rehman M, Abbas T, Ok YS (2016) Mechanisms of biochar-mediated alleviation of toxicity of trace elements in plants: a critical review. Environ Sci Pollut R 23(3):2230–2248. https://doi.org/10.1007/s11356-015-5697-7
Rodríguez-Vila A, Asensio V, Forján R, Covelo EF (2015) Chemical fractionation of Cu, Ni, Pb and Zn in a mine soil amended with compost and biochar and vegetated with Brassica juncea L. J Geochem Explor 158:74–81. https://doi.org/10.1016/j.gexplo.2015.07.005
Trakal L, Bingöl D, Pohorělý M, Hruška M, Komárek M (2014) Geochemical and spectroscopic investigations of Cd and Pb sorption mechanisms on contrasting biochars: engineering implications. Bioresour Technol 171:442–451
Uchimiya M, Cantrell KB, Hunt PG, Novak JM, Chang SC (2012) Retention of heavy metals in a Typic Kandiudult amended with different manure-based biochars. J Environ Qual 41(4):1138–1149. https://doi.org/10.2134/jeq2011.0115
Uchimiya M, Chang SC, Klasson KT (2011) Screening biochars for heavy metal retention in soil: role of oxygen functional groups. J Hazard Mater 190(1-3):432–441. https://doi.org/10.1016/j.jhazmat.2011.03.063
Uchimiya M, Lima IM, Klasson KT, Wartelle LH (2010) Contaminant immobilization and release by biochar soil amendment: roles of natural organic matter. Chemosphere 80(8):935–940. https://doi.org/10.1016/j.chemosphere.2010.05.020
Wang Z, Liu G, Zheng H, Li F, Ngo HH, Guo W, Liu C, Chen L, Xing B (2015) Investigating the mechanisms of biochar’s removal of lead from solution. Bioresour Technol 177:308–317
Woldetsadik D, Drechsel P, Keraita B, Marschner B, Itanna F, Gebrekidan H (2016) Effects of biochar and alkaline amendments on cadmium immobilization, selected nutrient and cadmium concentrations of lettuce (Lactuca sativa) in two contrasting soils. Springer Plus 5(1):397–412. https://doi.org/10.1186/s40064-016-2019-6
Wu ZP, McGrouther K, Chen DL, Wu WD, Wang HL (2013) Subcellular distribution of metals within Brassica chinensis L. in response to elevated lead and chromium stress. J Agr Food Chem 61(20):4715–4722. https://doi.org/10.1021/jf4005725
Xie T, Reddy KR, Wang C, Yargicoglu E, Spokas K (2015) Characteristics and applications of biochar for environmental remediation: a review. Crit Rev Environ Sci Technol 45(9):939–969. https://doi.org/10.1080/10643389.2014.924180
Yang JX, Guo HT, Ma YB, Wang LQ, Wei DP, Hua L (2010) Genotypic variations in the accumulation of exhibited by different vegetables. J Environ Sci 22(8):1246–1252. https://doi.org/10.1016/S1001-0742(09)60245-X
Yasmin KK, Ali B, Cui X, Feng Y, Pan F, Tang L, Yang X (2017) Cow manure and cow manure-derived biochar application as a soil amendment for reducing cadmium availability and accumulation by Brassica chinensis L. in acidic red soil. J Integr Agr 16:725–734
Ye X, Li H, Ma Y, Wu L, Sun B (2014) The bioaccumulation of Cd in rice grains in paddy soils as affected and predicted by soil properties. J Soils Sediments 14(8):1407–1416. https://doi.org/10.1007/s11368-014-0901-9
Yi Q, Hu X, Ke Y, Liu H, Yu H (2013) Impact of different biomass black carbon amendments on retention capacity of cadmium in soil. J Agro-Environ Sci 32:88–94 (in Chinese)
Zhang RH, Li ZG, Liu XD, Wang BC, Zhou GL, Huang XX, Lin CF, Wang AH, Brooks M (2017) Immobilization and bioavailability of heavy metals in greenhouse soils amended with rice straw-derived biochar. Ecol Eng 98:183–188. https://doi.org/10.1016/j.ecoleng.2016.10.057
Acknowledgements
This research was financially supported by the Science and Technology Project of Zhejiang Province (2015C03020, 2015C03004), and Natural Science Foundation of Zhejiang Province (LY16D010004).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Yong Sik Ok
Rights and permissions
About this article
Cite this article
Liu, Y., Wang, Y., Lu, H. et al. Biochar application as a soil amendment for decreasing cadmium availability in soil and accumulation in Brassica chinensis. J Soils Sediments 18, 2511–2519 (2018). https://doi.org/10.1007/s11368-018-1927-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-018-1927-1