Abstract
Purpose
The production of cellulase enzymes (CE) has been identified as one major contributor towards the life cycle environmental and economic impacts of second-generation lignocellulosic bioethanol (LCB) production. Despite this knowledge, the literature lacks consistent and transparent life cycle assessments (LCA) which compare CE production based on the three more commonly proposed carbon sources: cornstarch glucose, sugar cane molasses and pre-treated softwood. Furthermore, numerous LCAs of LCB omit CE production from their system boundaries, with several authors citing the lack of available production data.
Methods
In this article, we perform a comparative attributional LCA for the on-site production of 1 kg CE in full broth via submerged aerobic fermentation (SmF) based on the three alternative carbon sources, cases A, B and C, respectively. We determine life cycle inventory (LCI) material consumption using stoichiometric equations and volume flow, supplemented with information from the literature. All LCIs are provided in a consistent and transparent manner, filling the existing data gaps towards performing representative LCAs of LCB production with on-site CE production. Life cycle impact assessment (LCIA) results are determined with SimaPro 8 software using CML 1A baseline and non-baseline methods along with cumulative energy demand and are compared to results of similar studies. Sensitivity analysis is performed both for all major assumptions and for market changes with the application of advanced attributional LCA (AALCA).
Results and discussion
We find that CE production from pre-treated softwood (case C) provides the lowest environmental impacts, followed by sugar cane molasses (case B) and then cornstarch glucose (case A), with global warming potentials of 7.9, 9.1 and 10.6 kg CO2 eq./kg enzyme, respectively. These findings compare well with those of similar studies, though great variation exists in the literature. Through sensitivity analysis, we determine that results are sensitive to assumptions made concerning carbon source origin, applied allocation, market changes, process efficiency and electricity supply.
Conclusions
Furthermore, we find that the contribution of CE production towards the overall life cycle impacts of LCB is significant and that the omission of this sub-process in LCAs of LCB production can compromise their representativeness.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
With the implementation of the Renewable Energy Directive (EC 2009) and its predecessors, the European Union (EU) has set a 10 % target for use of renewable energy in the transport sector by 2020. Furthermore, the European Commission reported in a recently released scenario that the total energy demand of all transport in the EU 27 + 2 was 381 million tonnes oil equivalent (Mtoe) in 2010, of which road transport was 307 Mtoe, with biofuels contributing 13 Mtoe (Hamje et al. 2014). The same report predicts that by 2020, energy demand in road transport will decrease by 8.5 % though biofuel’s contribution will increase to 21.5 Mtoe, with lignocellulosic bioethanol (LCB) contributing 0.7 Mtoe, or approximately 1.38 × 109 l. The promotion of LCB is partially based on the environmental, social and economic concerns surrounding first-generation biofuels (Tilman et al. 2009), many of which resulted from so-called transfer effects (Holden and Gilpin 2013).
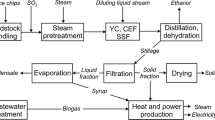
LCB is one of several second-generation biofuels, which are defined as biofuels produced from cellulose, hemicellulose and lignin (Sims et al. 2008), with biomass sources from silviculture, agriculture waste streams and fast-growing high-yield energy crops. The most commonly proposed method for converting lignocellulosic biomass into bioethanol is via the biochemical process of enzymatic hydrolysis and fermentation. Central to this process is the enzymatic hydrolysis of cellulose and hemicellulose into fermentable sugars by cellulase enzymes (CE), see Fig. 1.
Simplified process overview of LCB production via enzymatic hydrolysis and fermentation (Humbird et al. 2011)
Large quantities of CE are required for the enzymatic hydrolysis of lignocellulosic biomass, with CE consumption ranges of approximately 0.3–2.1 g of CE per MJ LCB proposed in the literature (Hong et al. 2013; Humbird et al. 2011; MacLean and Spatari 2009), see Electronic Supplementary Material.
The method most commonly proposed in life cycle assessment (LCA) literature to produce CE results from their secretion by the fungus Trichoderma reesei cultivated by submerged aerobic fermentation (SmF) and fed on a carbon source, see Table 1, though numerous CE-secreting organisms and cultivation methods exist. CE production is a resource-intensive process and represents one of the greatest uncertainties and current high costs related to LCB production (Foust et al. 2009; Luo et al. 2009). Specifically, the high uncertainty and costs are related to the provision of the carbon source, for which glucose, molasses and pre-treated softwood are alternatives, see Table 1.
1.1 Goal definition
The goal of this LCA study is to compare the environmental impacts associated with European production of CE that uses the three alternative carbon sources: cornstarch glucose, sugar cane molasses and pre-treated softwood. In addition, we aim to fill the existing data gaps in existing LCAs of LCB by presenting consistent, detailed and transparent life cycle inventory (LCI) data for CE production. In doing so, we answer the following research questions:
-
1.
Which cellulase enzyme production path, with respect to carbon source, provides the lowest environmental impacts?
-
2.
How do the environmental impacts determined in this study compare with those determined in similar studies?
-
3.
If current enzyme production methods are over- or underestimated, or omitted, what inferences can be made with respect to the representativeness of existing LCAs of LCB?
The LCA is performed under the guidance of both the International Organization for Standardization standards 14040 series for LCA (ISO 2006a, b) and the European Commission’s “International Reference Life Cycle Data System (ILCD) Handbook” (EC 2010). We apply attributional LCI modelling methods, and prioritize representative, publically available, consistent and transparent data sources over proprietary commercial data. The effect of this choice on results, along with other assumptions described in more detail later, is tested by sensitivity analyses. Classifying this study as situation A (EC 2010) and applying attributional LCA methodology present limitations for interpreting and applying the results, which are not intended to be used for meso- or macro-level decision-making. Instead, the results are intended to provide guidance to researchers and business developers, and to other public and private stakeholders working in the field of CE and LCB development.
1.2 Scope definition
“Cellulase enzyme” is an umbrella term for a mixture of endo-p-glucanases, exo-P-glucanases and β-glucosidase proteins. These three proteins work in sequence, with endo-p-glucanases randomly breaking the cellulose molecular chain into glucose and cello-oligo saccharides, followed by exo-P-glucanases, which then work on the exposed chain ends reducing these to glucose and cellobiose. Finally, the β-glucosidase protein hydrolyse the cellobiose to glucose. The result is slurry of fermentable, low-molecular-weight hexose (e.g. glucose) and pentose (e.g. xylose) monosaccharides (Miyamoto 1997).
Figure 2 presents the general system boundary for on-site CE production via the SmF method, from which the specific cases are derived. The product of this process is a full broth containing CE, a full broth which is then utilized in on-site LCB production, specifically the enzymatic hydrolysis of cellulose and hemicellulose to fermentable sugars, step 2 in Fig. 1. The functional unit (FU) of on-site CE production is 1 kg of CE (protein) in full broth, which is the same FU chosen by all authors of on-site CE-specific assessments in Table 1.
Process overview of CE production via the SmF method (Davis et al. 2015)
LCA presents the potential environmental impacts of a system or product over its life cycle by using measurable data of existing processes and products with known uncertainty (EC 2010). In this study, foreground process LCIs are formulated according to the literature review and are presented in the succeeding case descriptions. Background processes are derived from commercial databases available in the SimaPro 8 LCA software package (Goedkoop et al. 2013).
Neither CE production nor any of the foreground processes are multi-functional processes. Of the significant background processes, both corn wet-milling and sugar cane processing are multi-functional processes. Corn wet-milling has the co-products corn steep liquor, corn oil and cornstarch (input to glucose production) among others, and sugar cane processing has the co-product molasses among others. As recommended by ISO (2006a, b), the methods of subdivision and allocation were considered, in that order, for dealing with multi-functionality. Using the same logic as Würdinger et al. (2003) (i.e. because of the complexity of corn wet-milling and sugar cane processing, and because of a lack of data with suitable resolution), the preferable solution of subdivision was not feasible. Therefore, and with the aim of methodological consistency, allocation has been applied to all multi-functional processes. Specifically, a preferred causal physical relation does not exist for the co-products of corn wet-milling, and considering that the primary motivation of corn wet-milling operators is profit, background LCI data using economic allocation were selected (Würdinger et al. 2003). Sugar and molasses, the co-products of sugar cane processing, share the causal physical relation of energy; therefore, background LCI data using energetic allocation were chosen (van Zeist et al. 2012). Remaining allocation decisions for significant background processes are selected according to the most suitable causal relation, and clearly identified in the LCI tables in this article and in its Electronic Supplementary Material.
Life cycle impact assessment (LCIA) was performed using SimaPro 8 LCA software (Goedkoop et al. 2013). CML 1A baseline and CML 1A non-baseline methods (Pré 2015) were used for determining global warming potential 100-year time horizon (GWP; measured in kg CO2 eq.), eutrophication potential (EP; measured in g PO4 eq.), acidification potential (AP; measured in g SO2 eq.), ozone layer depletion (ODP; measured in mg CFC-11 eq.), photochemical oxidation potential (POP; measured in g C2H4 eq.) and land use (measured in m2a). Cumulative energy demand, measured in MJ, was determined using methods developed by Frischknecht et al. (2007).
Sensitivity analysis involves two approaches: first, through traditional testing of sensitivity towards changes in CE yield, source of electricity and various aspects concerning the carbon source. The second approach involves testing the sensitivity of results towards market effects by multiplying global change mix factors (GCMF) by hexose and pentose flows, a method known as advanced attributional LCA (AALCA) (Andrae 2015). We apply AALCA because of the common criticism of ALCA methodology, that is, its limitation to account for future market effects. Andrae (2015) has shown that AALCA can be performed as a sensitivity check of comparative ALCA results. By applying GCMFs based on real or future market changes, the interdependence of rising and declining global markets and micro-level LCA shifts can be taken into account in comparative micro-level ALCA studies. Therefore, AALCA represents an improvement over ALCA but cannot fully claim, as consequential LCA can, to fully reflect consequences of decisions, and as such, is not a replacement for consequential LCA.
2 Methods and case descriptions
In this LCA study, we compare three cases of CE production that use the three more relevant carbon sources: cornstarch glucose in case A, sugar cane molasses in case B and pre-treated softwood in case C. All percentages (%) are given as weight (wt.) percentages unless otherwise stated.
The National Renewable Energy Laboratory (NREL) process descriptions for CE production (Humbird et al. 2011; Wooley et al. 1999) form the general system from which all cases are developed. These designs along with succeeding/preceding LCB assessments produced by NREL, see Table 1, are based on Schell et al. (1991). We have chosen the NREL designs for two reasons: first, NREL provides transparent foreground LCIs of CE production, and second, these designs have been adopted in numerous other studies (Dunn et al. 2012; MacLean and Spatari 2009; Sheehan et al. 2003; Zhuang et al. 2007). This choice facilitates both the comparison with and the transfer of results to these and other studies.
The on-site SmF production of CE in full broth involves three steps:
-
1.
Media preparation—A carbon source, water and other nutrients are mixed in fixed quantities, along with small amounts of slip-stream-produced sophorose, which induces T. reesei to produce CE.
-
2.
Seed train—T. reesei fungus is stimulated to multiply in optimal conditions and fed by fraction of the media prepared in step 1, producing a T. reesei inoculum for step 3.
-
3.
Aerobic cultivation—In this step, the T. reesei from step 2 is introduced into a fermenter under submerged aerobic conditions (SmF), where it feeds on the carbon source and nutrients prepared in step 1 and secretes CE, see Fig. 2.
Common to all three cases is the assumption that the production of cell mass (T. reesei) and CE, steps 2 and 3, respectively, requires the stoichiometrically balanced dosing of the reactants: carbon source, oxygen (O2), ammonia (NH3) and sulphur dioxide (SO2) with the resulting products: carbon dioxide (CO2), water (H2O), CE and cell mass, see Table 2. We assume that the entire reactive carbon source, see Table 3, is consumed during steps 2 and 3. Humbird et al. (2011), citing Atkinson and Mavituna (1991) and proprietary information provided by novozymes, provide the elemental compositions for CE and cell mass, respectively, for cases A and B and Wooley et al. (1999), citing Wooley and Putsche (1996), provide the elemental composition for CE and cell mass for case C, see Table 2.
The efficiency of CE production can be expressed by the molar selectivity of the reactive carbon source’s carbon atoms towards the formation of CO2, CE and cell mass. The higher the carbon selectivity towards CE, the higher the yield of CE per kilogramme of reactive carbon source. Davis et al. (2015, 2013) and Humbird et al. (2011) all apply the same molar selectivity with glucose as the carbon source, and these authors provide the molar selectivity for cases A and B, see Table 2. Wooley et al. (1999) assume one molar selectivity for soluble sugars and a different molar selectivity for sugar polymers; these combine to form the molar selectivity presented in Table 2 for case C. By assessing the elemental compositions of the reactants and products and molar selectivity, we were able to balance the aforementioned stoichiometric equations, see Table 2.
CE production requirements for nutrients and antifoaming agents are based on Schell et al. (1991), see Electronic Supplementary Material. LCI data for polysorbate 80 and potassium phosphate were not found in the literature or in commercial databases; accordingly, we have determined GWP and cumulative energy demand for polysorbate 80 by using the Swiss Method (Wernet et al. 2008, 2009) and have constructed an LCI for the production of potassium phosphate based on Freilich and Petersen (2005), see Electronic Supplementary Material.
Energy requirements (electricity, heating and cooling) for cases A and B are adapted from Humbird et al. (2011) and Wooley et al. (1999) for case C. Furthermore, Humbird et al. (2011) and Wooley et al. (1999) describe an on-site combined heat and power (CHP) generation plant using LCB production waste streams as feedstock. In this study, CHP generation lies outside the evaluated system’s boundary and representative LCIs for CHP generation using organic waste feedstock have been selected; the validity of this selection is tested by sensitivity analysis. Cooling is produced from the same electricity source and scaled with respect to CE production. Aeration in the form of compressed air is scaled according to the oxygen uptake rate determined stoichiometrically, see Table 2. In doing so, we assume that the oxygen transfer rate is proportional to the oxygen uptake rate (Humbird et al. 2011). In addition, we assume the same reactor geometries, equipment and media characteristics as presented in Humbird et al. (2011) for cases A and B and Wooley et al. (1999) for case C.
Table 4 presents the final LCIs for the base cases A, B and C. The specific descriptions of carbon source production are provided subsequent sections.
2.1 Cornstarch glucose
Hobbs (2009) and EIA (2015) provide the LCI data for cornstarch glucose production; this data has been conditioned with respect to glucose output based on Tsiropoulos et al. (2013) and lower dry matter (DM) (85 %), see Table 5. In the modelled process, cornstarch containing slurry is treated with hydrochloric acid to lower the pH under high pressure and high temperature for a short residence time, resulting in the longer starch molecules being cleaved into lower-molecular-weight glucose. After conversion to glucose has been achieved, the pH is raised using soda ash (sodium carbonate). The resulting slurry is then centrifuged to remove impurities, then treated with active carbon to remove undesirable flavour and colour. The final step adjusts the pH further and condenses the liquor through evaporation (Hobbs 2009). The composition of reactive carbon sources available for CE production per 1000 kg glucose syrup is presented in Table 3.
2.2 Sugar cane molasses
Agri-footprint (2014) citing van Zeist et al. (2012) provide the consumption mix and LCI data for sugar cane molasses production and transport to Europe (Netherlands). Furthermore, we have adopted the energetic allocation applied by van Zeist et al. (2012), with 41 % of inputs allocated to molasses and the remainder to sugar. We assume that the consumption mix of molasses for the Netherlands is representative of the mix for Europe. Molasses is one product derived from the multi-product process of refining sugar cane; other products are cane sugar, bagasse and filter cake. Specifically, molasses is the co-product of the multi-stage process of forming and separating crystallized cane sugar (sucrose) from cane juice. The precursor to molasses, cane juice, is dried to form various grades of molasses depending on consumption (i.e. edible molasses or feed-grade molasses (blackstrap)) (EPA 1995). The DM content of the raw molasses is assumed to be 73 % (van Zeist et al. 2012). DM composition is based on Olbrich (2006), see Electronic Supplementary Material, and has a higher ratio of sucrose to glucose and fructose than the ratio used by He et al. (2014). The composition of reactive carbon sources available for CE production per 1000 kg of molasses is presented in Table 3.
2.3 Pre-treated softwood biomass
The production of pre-treated softwood, including woodchip handling and pre-treatment processes, has been adapted for softwood chips from Wooley et al. (1999), see Table 6. The softwood chips entering handling and pre-treatment have a DM content of 47.9 %, and a DM composition adopted from Ferraro et al. (1999), see Electronic Supplementary Material, which is similar to that used by Barta et al. (2010) and Olofsson et al. (2015). In this process, the softwood chips are initially screened and cleaned of impurities and contaminants. Then, a dilute-acid treatment is applied at high temperatures, and during the treatment, the hemicellulose polysaccharides are hydrolysed into soluble monosaccharides. This choice of pre-treatment is in agreement with the majority of assessments of LCB (Wiloso et al. 2012). In addition, a portion of the cellulose is converted to glucose, and otherwise exposed for conversion by CE. Lime is then added to raise the pH, and then precipitated as gypsum and removed. The resulting product is a hydrolysate containing cellulose and soluble sugars, among others (Wooley et al. 1999). The hydrolysis reactions for cellulose and hemicellulose are adopted from Davis et al. (2015), see Electronic Supplementary Material. The composition of reactive carbon sources available for CE production per 1000 kg of pre-treated softwood is presented in Table 3.
3 Results
Table 7 summarizes the LCIA results of the present study and compares these with the results of other relevant studies. From Table 7, we observe two things: first, the present results for GWP, EP, AP, POP and cumulative energy demand are within acceptable ranges. All of these impacts (excl. EP for case A) fall within the standard deviation (2σ or 95 % confidence) of the weighted mean of the relevant studies, when considering that non-formulated enzymes can have impacts (GWP) 36 % lower than formulated enzymes do (Hong et al. 2013). It should be noted that it was not possible to determine the weighted mean and standard deviation for several impact categories, because of limited sample size. The second observation is that considering all impact categories, case C provides the lowest environmental impacts for the production of CE.
Figures 3, 4, 5, 6, 7, 8 and 9 present the LCIA results for the base cases A, B and C. The results are disaggregated for all sub-processes which contribute significantly towards each impact (≥5 %). Contribution analysis results are provided in the Electronic Supplementary Material.
Based on the contribution analyses presented in Figs. 3, 4, 5, 6, 7, 8 and 9 and Electronic Supplementary Material, it can be observed that the carbon source and electricity are the most significant contributors to all impact categories with the exception of GWP, where CE production-process emissions are significant (36–55 %) as well. CE production-process emissions (CO2) have been determined stoichiometrically based on the assumed molar selectivity of the carbon source, see Table 2; therefore, CE production-process emissions are directly correlated to the assumed molar selectivity and carbon source.
Furthermore, for the impact category land use (Fig. 8), the provision of carbon sources is the largest contributor. Differences in land use impacts are the result of differences in carbon source yield (per m2a). Considering corn and sugar cane are high-yield annual crops, and that softwood is a lower-yielding and slower-growing crop, softwood will occupy the same area for several years to produce the same mass of carbon source, resulting in higher land use values per kilogramme reactive carbon source.
A sensitivity analysis is performed for the two significant sub-processes, carbon source and electricity, observing changes in the arbitrarily selected impact categories: GWP and cumulative energy demand. Hereafter, we refer to the previously determined results for cases A, B and C as base cases.
In the base-case scenarios, we have assumed a carbon source molar selectivity of 31 % for CE production for cases A and B (Davis et al. 2015) and 29 % for CE for case C (Wooley et al. 1999). Both Davis et al. (2015) and Wooley et al. (1999) have assumed these selectivities for future nth plant performance based on Meerman et al. (2004) and Hamilton (1998), respectively. Similar to Hong et al. (2013), in the sensitivity analysis, we reduce selectivity for CE production arbitrarily by 5 and 10 %, with resulting increases in selectivity for cell mass production of 5 and 10 %, respectively. These changes in molar selectivity for CE production can be interpreted as a reduction from Davis et al.’s (2015) and Wooley et al.’s (1999) nth plant assumptions. The results of this first sensitivity analysis are presented in Figs. 10 and 11. From these figures, two observations can be made: first, GWP and cumulative energy demand are sensitive to changes in process efficiency, and second, that case C is least sensitive to changes in carbon source molar selectivity. It should be noted that whereas Humbird et al. (2011) (case A) and Wooley et al. (1999) (case C) based their molar selectivity and yields on laboratory results, this study has assumed the same molar selectivity of case A for case B, using results from studies by Olofsson et al. (2015), He et al. (2014) and Barta et al. (2010). From the laboratory results of He et al. (2014), one can determine a molar selectivity of approximately 6 % for CE. Therefore, a cautious interpretation of results would choose the 10 % reduction for case B as more appropriate.
Additional assumptions were made with regard to carbon sources in the base-case LCI models. For glucose, we have constructed a LCI for glucose production. This LCI results in a GWP value of 1.34 kg CO2 eq./kg glucose DM, which is slightly higher than the 0.95 kg CO2 eq./kg glucose DM determined by An et al. (2012) and within the range of 0.7 ± 0.2–1.1 ± 0.2 kg CO2 eq./kg glucose DM determined by Tsiropoulos et al. (2013). Disregarding differences in LCIA, these differences can be attributed to impacts associated with cornstarch production and variations in LCI modelling approaches as explained by both Tsiropoulos et al. (2013) and An et al. (2012). Würdinger et al. (2003) and van Zeist et al. (2012) provide cornstarch LCIs which result in GWP values ranging from 1.41 kg CO2 eq./kg cornstarch DM (global, economic allocation) to 0.53 kg CO2 eq./kg cornstarch DM (US, energy allocation), respectively. In this study we have applied a cornstarch LCI resulting in GWP of 1.29 kg CO2 eq./kg cornstarch DM (German, economic allocation). The application of Würdinger et al.’s (2003) and van Zeist et al.’s (2012) cornstarch LCIs results in GWP values of 1.43 kg CO2 eq./kg glucose DM and 0.92 kg CO2 eq./kg glucose DM, respectively. Furthermore, Fig. 12 depicts the sensitivity analysis results from substituting into the complete LCI for case A these two alternative cornstarch LCIs. Figure 12 indicates that GWP results for case A are sensitive to changes in cornstarch’s feedstock, geography of origin and applied allocation.
For molasses, we have based our LCI model on van Zeist et al. (2012) for sugar cane molasses, assuming this LCI is suitably representative. This LCI, when applying energetic allocation, results in a GWP of 0.76 kg CO2 eq./kg molasses DM. LCAs of molasses production are scarce because of the predominance of integrated sugar and molasses-ethanol production (Gopal and Kammen 2009), and the resulting lack of sufficiently disaggregated LCIs. Van Zeist et al. (2012) provides values ranging from 0.16 kg CO2 eq./kg DM for sugar beet molasses (Netherlands, economic allocation) to 1.43 kg CO2 eq./kg DM for sugar cane molasses (Brazil, mass allocation). These values equate to a 79 % decrease and an 87 % increase in GWP, respectively, relative to the base-case scenario. As a sensitivity analysis, we consider both an 80 % decrease and an 80 % increase in molasses GWP, or 0.15 and 1.37 kg CO2 eq./kg molasses DM, respectively. Figure 13 presents the results of this sensitivity analysis and indicates that GWP results for CE production based on molasses are sensitive to molasses’ feedstock, geography of origin and applied allocation. In comparison, Olofsson et al. (2015) cite a GWP of 0.14 kg CO2 eq./kg molasses, which upon review is assumed to be for sugar beet molasses. Thus, the 80 % decrease column in Fig. 13 is more representative of CE production that uses sugar beet molasses, whereas this study’s results are representative for CE production that uses sugar cane molasses.
For pre-treated softwood, we have assumed softwood cellulose and hemicellulose pre-treatment hydrolysis reactions (conversion) based on Davis et al. (2015), see Electronic Supplementary Material. Wooley et al. (1999) provide another set of pre-treatment reactions, see Electronic Supplementary Material. As a sensitivity analysis, we consider the effect of applying Wooley et al.’s (1999) set of hydrolysis reactions to our pre-treatment process; Fig. 14 presents these results. The reduction in GWP when using Wooley et al.’s (1999) hydrolysis reactions is directly related to their assumption that less cellulose is converted to soluble sugars in pre-treatment, and as a result, more cellulose is available for CE production in succeeding stages. However, the differences in conversion efficiency are only slight, and these differences effect on GWP results for case C are less significant.
Considering electricity, in the base-case scenarios, electricity was modelled using CHP generation from organic waste feedstock (Humbird et al. 2011; Wooley et al. 1999), see Electronic Supplementary Material. In the sensitivity analysis, we have substituted this method for softwood chip boiler CHP generation and the EU 27 consumption mix, see Figs. 15 and 16. These figures indicate that both GWP and cumulative energy demand results are sensitive to direct electricity supply. Particularly, the substitution of organic waste CHP for softwood chip CHP has little effect on GWP and cumulative energy demand results for all cases. The substitution of organic waste CHP for the EU 27 consumption mix significantly increases impacts for case C, though less so for cases A and B. The sensitivity to changes in electricity supply is explained by the variation in direct electricity consumption between cases A, B and C in increasing order, see Table 4, making case C more sensitive to changes in direct electricity supply.
As a final sensitivity analysis, as an estimate of the effect of market changes on carbon source GWP results, historical GCMF for cornstarch glucose (case A), sugar cane molasses (case B) and pre-treated softwood (case C) are estimated, and these GCMF are then multiplied by the reactive carbon source flows, see Table 3, for each case A, B and C.
The cornstarch and sugar cane markets are large and diverse, and in 2013, more than 2 billion metric tonnes of each were produced (Geohive 2015a). Cornstarch glucose and sugar cane molasses can be derived from starches and sugar cane, respectively, and pre-treated softwood can be derived from woodchips.
For cornstarch glucose, the GCMF are obtained from the global corn production (Geohive 2015b) and from the share of corn sweetener of US corn production (USDA 2015). Globally in 2000 and 2015, 592 million metric tonnes (MMT) (Geohive 2015b) and 972 MMT (WOC 2015) of corn were produced, respectively, and the shares used to make corn sweeteners were 7.69 and 5.5 %, respectively (i.e. 46 MMT and 53 MMT, respectively).
For sugar cane molasses, the GCMF are obtained from the global sugar cane production (Geohive 2015a) and from the yield of molasses per tonne of sugar cane, which is approximately 3 % (MM 2015). In 2000 and 2015, 1256 and 2005 MMT of sugar canes were produced, respectively (Geohive 2015a, b) (i.e. 38 and 60 MMT of molasses cane syrup, respectively).
For pre-treated softwood, the GCMF are obtained from the annual woodchip production, which is used as a starting point for estimating softwood-based hydrolysate. The global annual woodchip production increased from around 30 to 60 MMT between 2000 and 2015 (Ekstrom 2011), and the theoretical amount of hydrolysate that can be extracted from woodchips is 1–12 % DM content (Gladyshko 2011). Extraction of hemicelluloses by acid-catalysed hydrolysis is not equal to the actual production of wood hydrolysate for glucose applications, because a large share of hydrolysate is used for biofuels and renewable products (Dahlman et al. 2014). Still, if an average 6 % hydrolysate from woodchips is used, the pre-treated softwood production thereby increased from 1.8 to 3.6 MMT between 2000 and 2015, respectively.
Alternatively, global corn sweetener production (Geohive 2015b; USDA 2015; WOC 2015) and the market share of hydrolysates related to the sweeteners glucose syrup, modified starch, maltodextrin and cyclodextrin (MM 2015) can be used. In 2000 and 2015, the market shares of hydrolysates are estimated to be 7.2 and 11.4 %, respectively (MM 2015) (i.e. 4 and 7 MMT of hydrolysates, respectively).
The values used for determining GCMF are summarized in Tables 8 and 9, and the results are presented in Fig. 17. The results indicate that case A, or more specifically, GWP associated with glucose production, is highly sensitive to market changes. The reasons are the slow market growth of cornstarch glucose leading to a relatively low GCMF, implying a relatively low carbon source allocation in Table 4 in combination with the relatively high share of the carbon source of the total GWP score (Fig. 3). Pre-treated softwood also obtains a low carbon source allocation because of a low share of the applied GCMF mix. However, the share of the carbon source of the total GWP score for pre-treated softwood is relatively small (Fig. 3), and therefore, the effect is less significant, as shown in Fig. 17. The sugar cane molasses market grows quicker than the markets of cornstarch glucose and pre-treated softwood, and therefore, is allocated a relatively high GCMF (Table 9). The share of the carbon source of the total GWP score for sugar cane molasses is relatively high (Fig. 3), leading to a similar GWP score for ALCA and AALCA (Fig. 17).
The results of the sensitivity analysis indicate that case A is most sensitive to assumptions made concerning glucose’s feedstock, geography of origin and applied allocation and market changes, whereas case C is most sensitive to assumptions made concerning molar selectivity and direct electricity supply. Case B indicates the least sensitivity to those assumptions tested in the sensitivity analysis.
4 Discussion
A semi-quantitative comparison of the results of this study with the results of similar non-formulated enzyme assessments, see Table 7, reveals that this study’s results are within the determined range (2σ) of Hong et al. (2013) and MacLean and Spatari (2009). Furthermore, when one considers that Hong et al. (2013) found that formulated enzymes can have impacts (GWP) 57 % higher than non-formulated enzymes do, the results of this study, when compared with those for formulated enzymes, seem acceptable. However, the results of this study do not share similarities with Agostinho et al.’s (2014) cumulative energy demand or with Harding’s (2008) GWP results, see Table 7. Agostinho et al.’s (2014) high value of 1664 MJ/kg CE can be attributed to the use of paper pulp as a carbon source, which accounts for 77 % of cumulative energy demand. Harding’s (2008) low (negative) GWP impacts are the result of the LCI modelling approach he has taken, where CO2 uptake from agricultural inputs during cultivation exceeds CO2 releases during cellulase production. Remaining minor differences can be attributed to variations in the background LCIs chosen and variations in LCI modelling.
Considering the CE consumption values presented in the introduction, and the results of this study, one can estimate the environmental impact associated with CE production per MJ LCB based on this study, see Electronic Supplementary Material. The GWP range for all cases is 2–22 g CO2 eq. CE/MJ LCB for low CE consumption (case C) to high CE consumption (case A), respectively. These results are similar to the results of Hong et al. (2013) (12 g CO2 eq. CE/MJ LCB) and of MacLean and Spatari (2009) (3.3–3.6 g CO2 eq. CE/MJ LCB) for non-formulated enzymes, and to the higher results of Olofsson et al. (2015) (18–30 g CO2 eq. CE/MJ LCB) and of Dunn et al. (2012) (4.6 g CO2 eq. CE/MJ LCB) for formulated enzymes.
When one compares these values with the total GWP of forest-to-gate LCB production determined by other authors (9.3–50.3 g CO2 eq./MJ LCB: Bright and Strømman (2009), Dunn et al. (2012) and Olofsson et al. (2015), to cite several), and with a recent review of 53 studies by Morales et al. (2015) revealing values ranging from 1.6 to 123.4 g CO2 eq./MJ LCB, one can conclude that CE production is significant with respect to the total environmental impacts of LCB production.
Alarmingly, Borrion et al. (2012), who reviewed 53 LCAs of LCB, found that in many instances, it is not clear whether CE production is within the evaluated system boundary, and Wiloso et al. (2012), who reviewed 22 LCAs of LCB, revealed that only 15 studies incorporated enzyme production in their inventory analyses, several of which have been cited in this article. Morales et al. (2015), Borrion et al. (2012), Wiloso et al. (2012), Singh et al. (2010) and Luo et al. (2009) all point to the existing data gaps concerning CE production and the necessity to include this process in LCAs of LCB in order to represent the true environmental impacts associated with LCB production.
Finally, future research, development and implementation will lead to a better understanding of the best-suited pre-treatments (Kumar et al. 2009), enzyme strains (Seiboth et al. 2011) and location of CE production (Olofsson et al. 2015), in addition to compiling LCIs suitable for assessing additional LCIA categories, for example, water consumption (An et al. 2012).
5 Conclusions
The results of this study show that CE production using pre-treated softwood as a carbon source provides lower environmental impacts than does CE production using cornstarch glucose and sugar cane molasses carbon sources. However, to varying degrees, results are sensitive to assumptions.
From the results, we conclude that other studies evaluating the environmental impacts of CE production neither overestimate nor underestimate CE production’s environmental impact. Instead, from using sensitivity analysis and from reviewing these sources, we conclude that particular attention should be paid to correct selection of background LCIs, particularly carbon source and electricity, and even nutrient requirements. Additionally, consistent LCI modelling methodology should be applied and transparently described.
Furthermore, we observe that according to estimates for CE’s contribution towards the GWP impacts of LCB production, CE production’s exclusion from LCB assessments can lead to significant deviations from the true impacts associated with LCB production.
As CE production increases because of the foreseen increase in LCB production, experience will lead to advances in understanding and technology, advances which could prove either favourable or detrimental to assessing the environmental impacts associated with CE production. It is, however, of the utmost importance to find (the most) sustainable processes and to avoid competition for finite resources, in line with the philosophy of industrial ecology. This study has filled some of the data gaps associated with the production of LCB in general and has specifically provided a clear and transparent indication of the resource use and environmental impacts of on-site SmF CE in full broth production. Future research could include defining a pure consequential LCA looking at the consequences of changing cornstarch glucose, sugar cane molasses and pre-treated softwood demands in CE production to substantiate or disregard the findings of AALCA sensitivity analysis.
References
Agostinho F, Ortega E (2013) Energetic-environmental assessment of a scenario for Brazilian cellulosic ethanol. J Clean Prod 47:474–489
Agostinho F, Bertaglia AB, Almeida CM, Giannetti BF (2014) Influence of cellulase enzyme production on the energetic–environmental performance of lignocellulosic ethanol. Ecol Model 315:45–56
Agri-footprint (2014) Agri-footprint description of data V1.0. Blonk Agri-footprint BV, Gouda, the Netherlands
An V, Evelien D, Katrien B (2012) Life cycle assessment study of starch products for the European starch industry association (AAF): sector study. Flemish Institute for Technological Research NV, Boeretang.
Andrae AS (2015) Method based on market changes for improvement of comparative attributional life cycle assessments. Int J Life Cycle Assess 20:263–275
Atkinson B, Mavituna F (1991) Biochemical engineering and biotechnology handbook. Stockton Press, New York
Barta Z, Kovacs K, Reczey K, Zacchi G (2010) Process design and economics of on-site cellulase production on various carbon sources in a softwood-based ethanol plant. Enzyme Research 2010:1–8
Borrion AL, McManus MC, Hammond GP (2012) Environmental life cycle assessment of lignocellulosic conversion to ethanol: a review. Renew Sust Energ Rev 16:4638–4650
Bright RM, Strømman AH (2009) Life cycle assessment of second generation bioethanols produced from Scandinavian boreal forest resources. J Ind Ecol 13:514–531
Dahlman O, Soederqvist LM, Parkaas J, Albertsson AC, Edlund U (2014) Utilization of a wood hydrolysate. Google Patents. http://www.google.com/patents/WO2009068525A1?cl=en. Accessed 11 June 2015
Davis R, Tao L, Tan ECD, Biddy MJ, Beckham GT, Scarlata C, Jacobson J, Cafferty K, Ross J, Lukas J, Knorr D, Schoen P (2013) Process design and economics for the conversion of lignocellulosic biomass to hydrocarbons: dilute-acid and enzymatic deconstruction of biomass to sugars and biological conversion of sugars to hydrocarbons. National Renewable Energy Laboratory, Golden
Davis R, Tao L, Scarlata C, Tan ECD, Ross J, Lukas J, Sexton D (2015) Process design and economics for the conversion of lignocellulosic biomass to hydrocarbons: dilute-acid and enzymatic deconstruction of biomass to sugars and catalytic conversion of sugars to hydrocarbons. National Renewable Energy Laboratory, Golden
Dunn JB, Mueller S, Wang M, Han J (2012) Energy consumption and greenhouse gas emissions from enzyme and yeast manufacture for corn and cellulosic ethanol production. Biotechnol Lett 34:2259–2263
Ekstrom H (2011) Global trade of wood chips was up in 2010 After sharp decline in 2009. Business Insider. http://www.businessinsider.com/global-trade-of-wood-chips-was-up-in-2010-after-sharp-decline-in-2009-2011-3?IR=T. Accessed 11 June 2015
Embassy of the Federal Democratic Republic of Ethiopia in London (EIA) (2015) 50. Profile on the production of glucose. Ethiopian Investment Agency. http://www.ethioembassy.org.uk/trade_and_investment/Investment%20Profiles%20EIA/Chemicals%20and%20Pharmaceuticals/Glucose.pdf. Accessed 1 Oct 2015
Environmental Protection Agency (EPA) (1995) Sugarcane processing. In: Compilation of air pollutant emission factors Volume 1: Stationary point and area sources. United States Environmental Protection Agency, Research Triangle Park, North Carolina
European Commission (EC) (2010) International Reference Life Cycle Data System (ILCD) Handbook: General guide for life cycle assessment–detailed guidance. European Commission, Joint Research Centre, Institute for Environmental and Sustainability. Publications office of the European Union, Luxembourg
European Council (EC) (2009) Directive 2009/28/EC of the European parliament and of the Council of 23 April 2009 on the promotion of the use of energy from renewable sources and amending and susbsequently repealing Directives 2001/77/EC and 2003/30/EC. European Parliament, Council of the European Union. EUR-Lex. http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32009L0028. Accessed 1 Oct 2015
Ferraro F, Kraft M, Penka K, Sharpe J, Voiles D, Kadam K, Keller F, Nguyen Q, Posey-Eddy F, Tucker M, Aden A, Wooley B, Yancey M, Selya E (1999) Softwood biomass to ethanol feasibility study. National Renewable Energy Laboratory, Battelle
Foust TD, Aden A, Dutta A, Phillips S (2009) An economic and environmental comparison of a biochemical and thermochemical lignocellulosic ethanol conversion process. Cellulose 16:547–565
Freilich MB, Petersen RL (2005) Potassium compounds. In: Kirk-Othmer encyclopedia of chemical technology. Wiley, New Jersey
Frischknecht R, Jungbluth N, Althaus H, Bauer C, Doka, G, Dones R, Hischier R, Hellweg S, Humbert S, Köllner T, Loerincik Y, Margni M, Nemecek T (2007) Implementation of life cycle impact assessment methods. Swiss Centre for Life Cycle Inventories, Dübendorf, Switzerland
Geohive (2015a) World: production of crops in metric tonnes. Geohive. http://www.geohive.com/charts/ag_crops.aspx. Accessed 11 June 2015
Geohive (2015b) World: maize (corn) production in metric tonnes. Geohive http://www.geohive.com/charts/ag_maize.aspx. Accessed 11 June 2015
Gladyshko Y (2011) Extraction of hemicelluloses by acid catalyzed hydrolysis. Bachelor thesis, Saimia University of Applied Sciences
Goedkoop M, Oele M, Leijting J, Ponsioen T, Meijer E (2013) Introduction to LCA with SimaPro. Pré Consultants, Amersfoort, the Netherlands
Gopal AR, Kammen DM (2009) Molasses for ethanol: the economic and environmental impacts of a new pathway for the lifecycle greenhouse gas analysis of sugarcane ethanol. Environ Res Lett 4:1–5
Hamilton J (1998) Cellulase production experiment no. 36: air/oxygen 22 factoral design. National Renewable Energy Laboratory, Golden
Hamje HDC, Hass H, Lonza L, Maas H, Reid A, Rose KD, Venderbosch T (2014) EU renewable energy targets in 2020: revised analysis of scenarios for transport fuels. European Commission–Joint Research Center, Luxembourg
Harding K (2008) A generic approach to environmental assessment of microbial bioprocesses through life cycle assessment (LCA). Dissertation, University of Cape Town
Harding K, Harrison S (2011) Life cycle assessment (LCA) and comparison of various cellulase production methods. Life Cycle Management Conference (LCM 2011), Berlin
He J, Wu A, Chen D, Yu B, Mao X, Zheng P, Yu J, Tian G (2014) Cost-effective lignocellulolytic enzyme production by Trichoderma Reesei on a cane molasses medium. Biotechnology for Biofuels 7:1–9
Hobbs L (2009) Sweeteners from starch: production, properties and uses. In: Starch: chemistry and technology. Academic Press, Elsevier, London, pp. 797–832
Holden E, Gilpin G (2013) Biofuels and sustainable transport: a conceptual discussion. Sustainability 5:3129–3149
Hong Y, Nizami A, Bafrani MP, Saville BA, MacLean HL (2013) Impact of cellulase production on environmental and financial metrics for lignocellulosic ethanol. Biofuel Bioprod Bior 7:303–313
Humbird D, Davis R, Tao L, Kinchin C, Hsu D, Aden A, Schoen P, Lukas J, Olthof B, Worley M, Sexton D, Dudgeon D (2011) Process design and economics for biochemical conversion of lignocellulosic biomass to ethanol: dilute-acid pretreatment and enzymatic hydrolysis of corn stover. National Renewable Energy Laboratory, Golden
ISO (2006a) 14040- Environmental management—Life cycle assessment—Principles and framework
ISO (2006b) 14044- Environmental management—Life cycle assessment—Requirements and guidelines
Kim S, Jiménez-González C, Dale BE (2009) Enzymes for pharmaceutical applications—a cradle-to-gate life cycle assessment. Int J Life Cycle Assess 14:392–400
Klein-Marcuschamer D, Oleskowicz-Popiel P, Simmons BA, Blanch HW (2012) The challenge of enzyme cost in the production of lignocellulosic biofuels. Biotechnol Bioeng 109:1083–1087
Kumar P, Barrett DM, Delwiche MJ, Stroeve P (2009) Methods for pretreatment of lignocellulosic biomass for efficient hydrolysis and biofuel production. Ind Eng Chem Res 48:3713–3729
Luo L, van der Voet E, Huppes G (2009) An energy analysis of ethanol from cellulosic feedstock–corn Stover. Renew Sust Energ Rev 13:2003–2011
MacLean HL, Spatari S (2009) The contribution of enzymes and process chemicals to the life cycle of ethanol. Environ Res Lett 4:1–10
Meerman HJ, Kelley AS, Ward M (2004) Advances in protein expression in filamentous fungi. Horizon Bioscience, Norfolk, p. 345
Micromarket Monitor (MM) (2015) Global hydrolysates market research report. Micromarket Monitor. http://www.micromarketmonitor.com/market-report/hydrolysates-reports-9767589706.html. Accessed 11 June 2015
Miyamoto K (1997) Renewable biological systems for alternative sustainable energy production Vol. 128. Food and Agriculture Organization of the United Nations, Rome
Morales M, Quintero J, Conejeros R, Aroca G (2015) Life cycle assessment of lignocellulosic bioethanol: environmental impacts and energy balance. Renew Sust Energ Rev 42:1349–1361
Nielsen PH, Oxenbøll KM, Wenzel H (2007) Cradle-to-gate environmental assessment of enzyme products produced industrially in Denmark by Novozymes A/S. Int J Life Cycle Assess 12:432–438
Olbrich H (2006) The molasses. In: Principles of sugar technology. Biotechnologie Kempe GmbH, Kleinmachnow, pp. 511–697
Olofsson J, Barta Z, Börjesson P, Wallberg O (2015) Life cycle assessment and techno-economical analysis of on-site enzyme production in 2nd generation bioethanol. The Swedish Knowledge Center For Renewable Transportation Fuels, Göteborg
Pré (2015) SimaPro database manual–methods library. Pré Consultants, Amersfoort
Schell D, Riley C, Bergeron P, Walter P (1991) Technical and economic analysis of an enzymatic hydrolysis based ethanol plant. Solar Energy Research Institute, Golden
Seiboth B, Ivanova C, Seidl-Seiboth V (2011) Trichoderma reesei: a fungal enzyme producer for cellulosic biofuels. In: Bernardes MADS (ed) Biofuel Production-Recent Developments and Prospects. Intech. http://www.intechopen.com/books/biofuel-production-recent-developments-and-prospects/trichoderma-reesei-a-fungal-enzyme-producer-for-cellulosic-biofuels. Accessed 1 Oct 2015
Sheehan J, Aden A, Paustian K, Killian K, Brenner J, Walsh M, Nelson R (2003) Energy and environmental aspects of using corn stover for fuel ethanol. J Ind Ecol 7:117–146
Sims R, Taylor M, Saddler J, Mabee W (2008) From 1st-to 2nd- generation biofuel technologies An overview of current industry and RD&D activities. International Energy Agency, Paris
Singh A, Pant D, Korres NE, Nizami AS, Prasad S, Murphy JD (2010) Key issues in life cycle assessment of ethanol production from lignocellulosic biomass: challenges and perspectives. Bioresource Technol 101:5003–5012
Tilman D, Socolow R, Foley JA, Hill J, Larson E, Lynd L, Pacala S, Reilly J, Searchinger T, Somerville C, Williams R (2009) Beneficial biofuels—the food, energy, and environment trilemma. Science 325:270–271
Tsiropoulos I, Cok B, Patel MK (2013) Energy and greenhouse gas assessment of European glucose production from corn—a multiple allocation approach for a key ingredient of the bio-based economy. J Clean Prod 43:182–190
United States Department of Agriculture (USDA) (2015) Table 27–U.S. use of field corn, by crop year. United States Department of Agriculture–Economic Research Service. http://www.ers.usda.gov/datafiles/Sugar_and_Sweeteners_Yearbook_Tables/Corn_Sweetener_Supply_Use_and_Trade/Table27.xls. Accessed 11 June 2015
van Zeist WJ, Marinussen M, Broekema R, Groen E, Kool A, Dolman M, Blonk H (2012) LCI data for the calculation tool Feedprint for greenhouse gas emissions of feed production and utilization. Blonk Consultants, Wageningen
Wernet G, Hellweg S, Fischer U, Papadokonstantakis S, Hungerbühler K (2008) Molecular-structure-based models of chemical inventories using neural networks. Environ Sci Technol 42:6717–6722
Wernet G, Papadokonstantakis S, Hellweg S, Hungerbühler K (2009) Bridging data gaps in environmental assessments: modeling impacts of fine and basic chemical production. Green Chem 11:1826–1831
Wiloso EI, Heijungs R, de Snoo GR (2012) LCA of second generation bioethanol: a review and some issues to be resolved for good LCA practice. Renew Sust Energ Rev 16:5295–5308
Wooley RJ, Putsche V (1996) Development of an ASPEN PLUS physical property database for biofuels components. National Renewable Energy Laboratory, Golden
Wooley R, Ruth M, Sheehan J, Ibsen K, Majdeski H, Galvez A (1999) Lignocellulosic biomass to ethanol process design and economics utilizing co-current dilute acid prehydrolysis and enzymatic hydrolysis current and futuristic scenarios. National Renewable Energy Laboratory, Golden
World of Corn (WOC) (2015) World corn production. World of Corn. http://www.worldofcorn.com/#world-corn-production. Accessed 11 June 2015
Würdinger E, Roth U, Wegener A, Peche R, Rommel W, Kreibe S, Nikolakis A, Rüdenauer I, Pürschel C, Ballarin P, Knebel T, Borken J, Detzel A, Fehrenbach H, Giegrich J, Möhler S, Patyk A, Reinhardt GA, Vogt R, Mühlberger D, Wante J (2003) Kunststoffe aus nachwachsenden rohstoffen: vergleichende ökobilanz für loose-fill-packmittel aus stärke bzw. polystyrol. Deutsche Bundesstiftung Umwelt, Augsburg, Germany
Zhuang J, Marchant M, Nokes S, Strobel H (2007) Economic analysis of cellulase production methods for bio-ethanol. Appl Eng Agric 23:679–687
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Niels Jungbluth
Electronic supplementary material
ESM 1
(DOCX 34 kb)
Rights and permissions
About this article
Cite this article
Gilpin, G.S., Andrae, A.S.G. Comparative attributional life cycle assessment of European cellulase enzyme production for use in second-generation lignocellulosic bioethanol production. Int J Life Cycle Assess 22, 1034–1053 (2017). https://doi.org/10.1007/s11367-016-1208-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11367-016-1208-4