Abstract
Raising soil contamination with cadmium (Cd2+) and salinization necessitates the development of green approaches using bio-elicitors to ensure sustainable crop production and mitigate the detrimental health impacts. Two field trials were carried out to study the individual and combined effects of foliage spraying of Moringa leaf extract (MLE) and soil application of effective microorganisms (EMs) on the physio-biochemical, osmolytes, antioxidants, and performance of sweet potato grown in Cd2+-contaminated salty soil (Cd2+ = 17.42 mg kg−1 soil and soil salinity ECe = 7.42 dS m−1). Application of MLE, EMs, or MLE plus EMs significantly reduced the accumulation of Cd2+ in roots by 55.6%, 50.0%, or 68.1% and in leaves by 31.4%, 27.6%, or 38.0%, respectively, compared to the control. Co-application of MLE and EMs reduced Na+ concentration while substantially raising N, P, K+, and Ca2+ acquisition in the leaves. MLE and EMs-treated plants exhibited higher concentrations of total soluble sugar by 69.6%, free proline by 47.7%, total free amino acids by 29.0%, and protein by 125.7% compared to the control. The enzymatic (SOD, APX, GR, and CAT) and non-enzymatic (phenolic acids, GSH, and AsA) antioxidants increased in plants treated with MLE and/or EMs application. Applying MLE and/or EMs increased the leaf photosynthetic pigment contents, membrane stability, relative water content, water productivity, growth traits, and tuber yield of Cd2+ and salt-stressed sweet potato. Consequently, the integrative application of MLE and EMs achieved the best results exceeding the single treatments recommended in future application to sweet potato in saline soil contaminated with Cd2+.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Salinity and heavy metal-induced soil contamination pose substantial challenges, leading to a major decline in soil quality and agricultural productivity (dos Santos et al. 2022). In addition, salinity stress has an inhibitory effect on plant growth (Abou-Sreea et al. 2022; Zaki and Radwan 2022). This plant growth inhibition in response to salinity may refer to (i) reduced water availability to plants due to increasing osmotic pressure in the soil solution, inducing physiological drought (Semida et al. 2021b), (ii) increased concentration of sodium and chloride ions in saline soils which has a toxic effect on plant growth (Abou-Sreea et al. 2022). Therefore, salinity induces the generation of reactive oxygen species (ROS), which cause oxidative damage, including disruption of protein synthesis, impairment to plasma membranes and cell organelles, suppression of the photosynthetic process, reduction in respiration and gas exchange (Chourasia et al. 2021; Abd El-Mageed et al. 2023). The acquisition of macro- and micro-nutrients by plants is closely linked with the accumulation of Na+ ions surrounding plant roots, which significantly alters all ionic homeostasis, causing nutritional deficiency (Abd El-Mageed et al. 2022a, b; Abd El-Mageed et al. 2022a, b).
Heavy metal-contaminated soils have seriously increased due to the rapid development of industrialization, long-term waste recycling, excessive use of chemical products, and irrigation with sewage (Li et al. 2019; Abd El-Mageed et al. 2020). Cadmium (Cd2+) is a toxic heavy metal not necessary for plants. Cadmium is characterized by high water solubility and fluidity that accelerates its absorption by the roots of plants, resulting in the suppression of cell division and root elongation (Souid et al. 2020). High Cd2+ levels in the soil evoke osmotic stress, restricting water uptake, disturbing ion homeostasis, and inducing nutrient deficiency (Rucińska-Sobkowiak 2016). Moreover, excessive Cd2+ concentration causes physiological disorders, including reduced respiration, photoinhibition, decreased water fluidity and gas exchange, the production of chlorophyll, and impairment of the plant’s antioxidant defense mechanism via motivating ROS production (Abd El-Mageed et al. 2019; Genchi et al. 2020; Rasafi et al. 2022).
Under harsh conditions like saline and/or Cd2+ contamination, grown plants enhance their machinery tolerance to combat adverse stresses by accumulating osmoregulatory active solutes, including soluble sugars and proline, and elevating their antioxidative capacity (Abd El-Mageed et al. 2020). However, in the majority of cases, the endogenous plant defense mechanisms are insufficient to sustain normal development under stress conditions, necessitating the application of exogenous support, such as the use of biostimulants, to raise their stress tolerance (Ramzan and Younis 2022). Due to increasing soil salinization and contamination with Cd2+, developing green strategies using bio-elicitors to reduce the harmful consequences caused by salinity and/or Cd2+ stress has generated massive interest worldwide (Batool et al. 2020; Safwat and Matta 2021). Therefore, the current experiment proposed effective microorganisms (EMs) and Moringa leaf extract (MLE) as natural biostimulants to mitigate the detrimental impacts on sweet potato plants grown in Cd2+-contaminated saline soils.
Effective microorganisms (EMs) are a group of collection cultures comprising favorable micro-organisms, including lactobacilli, photosynthetic bacteria, yeasts, and actinomycetes (Iriti et al. 2019; Khademian et al. 2019). EMs favor soil health by improving soil structure, decomposition of organic matter, accumulation of humus substances, and enhancing the uptake of nutrients, which in turn upgrades soil profitability and sustainability for plant production (Sagar et al. 2021; Abd El-Mageed et al. 2022a, b). The EMs also effectively contribute to improving plant growth by boosting the accessibility and uptake of nutrients, synthesizing metabolites, and producing bioactive molecules, including phytohormones, amino acids, enzymes, nucleic acids, and sugars (Talaat 2019). Therefore, due to its vital effect on plant physio-biochemical responses, using EMs significantly promotes cell division that ensures better germination, development, product quality, and yield of crops in circumstances of stress (Abu-Qaoud et al. 2021). Besides its stimulating effect on plant growth, EMs may also improve the soil’s biological and physicochemical characteristics and have a suppressive effect on pests and soil-borne diseases (De Corato 2021).
A member of the Moringaceae family, Moringa oleifera is considered one of the most widespread trees in tropical areas (Gopalakrishnan et al. 2016). The roots and leaves of these trees are regarded as sources of current plant biostimulants (Abd El-Mageed et al. 2017). The Moringa leaf extract (MLE) is a naturally rich resource of essential amino acids, carotenoids, flavonoids, proteins, vitamins (A, B2, C, and E), ascorbates, phenolics, and numerous nutrients, making it a superior plant growth bio-regulator (Abalaka et al. 2022; Irshad et al. 2022). Further, MLE was shown to be a good source of numerous antioxidants and phytohormones, such as cytokinins (zeatin), auxins, and gibberellins (Abd El-Mageed et al. 2017). The exogenous application of MLE improved plant growth, plant water relations, photosynthetic pigments, photochemical activity, and plant defenses to overcome oxidative stress by ROS scavenging via increasing the production of osmoprotectants and antioxidant enzymes (Khan et al. 2022). Foliar application of MLE on Sudan grass (Desoky et al. 2019a), quinoa (Rashid et al. 2018), wheat (Ahmed et al. 2021), bean (Howladar 2014), and potato (El-Mohamed et al. 2016) resulted in a significant yield increase under different abiotic stress.
Sweet potato (Ipomoea batatas L.) is among the Convolvulaceae family cultivated for their valuable tuberous root (Byju and George 2005). Tubers are rich in carbohydrates, especially the orange-fleshed roots, which are an excellent provider of the vitamin A precursor and β-carotene (Meng et al. 2020). Worldwide, among food crops, sweet potato occupies the seventh rank. Several reports have shown that Cd2+ and salinity stress negatively affected the productivity of sweet potato plants (Meng et al. 2020; Abd El-Mageed et al. 2022a, b).
As plant biostimulants, MLE and EMs are widely used due to their valuable applications and roles in alleviating the detrimental effects of several stresses; however, the information about their coupling application for mitigating the negative effects caused by salinity and Cd2+ toxicity on growth and physiological and biochemical responses of sweet potato plants are very limited. Thus, the current research contributes to understanding the role of applied MLE and EMs, alone or in combination, on some physiological and biochemical systems of sweet potato plants against the toxicity of Cd2+ and salts. Our research hypothesis was that treating sweet potato plants grown in Cd2+-contaminated saline soil with foliar spraying of EMs and soil application with MLE might, singly or collaboratively, alleviate the Cd2+ toxic effects through decreasing Cd2+ ion uptake and boosting morpho-physio-biochemical aspects and productivity.
Materials and methods
Location and agronomic details
Two field experiments were conducted from May to October in the summers of 2020 and 2021 in the Fayoum Governorate, Egypt (29°30′00.6″ N 30°52′32.3″ E). The methodology described by Klute and Dirksen (1986) and Page et al. (1982) was used to determine the main physicochemical properties of the tested soil (Table 1).
The soil texture analysis was 72.50% sand, 13.39% silt, and 14.11% clay, and it is considered sandy loam soil. The soil ECe (electrical conductivity) was 7.42 dS m−1, showing saline soil. The soil is slightly alkaline in pH (7.80), 17.42 mg kg−1 for Cd2+, 0.98% for organic matter content, 4.26% for CaCO3%, 0.04% for N, available K and P was 56.4, and 5.42 meq 100–1 g soil, respectively (Table 1).
Sweet potato (cv. Beauregard) vines of 30 days were relocated on May 2, 2020, and May 10, 2021, in rows of 15 m length and 0.7 m width, on which the vines were transplanted at 0.25 m apart. A drip irrigation system of a single line per row and drippers spaced 0.25 m apart with a 4 L h−1 discharge rate were used. The test plot comprises 3 planting rows and covers an area of 31.5 m2 (15 m × 2.1 m). The other recommended sweet potato farming techniques were followed exactly as advised.
Experimental design and treatments
The experimental treatments were laid out in a completely randomized block design and performed in triplicates. The four treatments were control, MLE, EMs, and MLE + EMs. After transplanting, the MLE was sprayed on the leaves thrice at 15, 30, and 45 days. To enable the best possible absorption into the sweet potato plant’s leaves, Tween-20 of 0.1% (v/v) was added to the MLE solution, and the control plants were sprayed with only water. The EMs were applied to sweet potato plants as a soil application three times (10, 20, and 30 days after transplanting).
Preparation and analysis of Moringa oleifera leaf extract (MLE)
When the Moringa trees were fully mature, they were harvested, and their fresh leaves were collected. Collected leaves were first frozen for an entire night, then crushed, and filtered twice via filter paper (Whatman No. 1). The resulting solution was refined and then centrifuged for 15 min at 8000 × g to separate the supernatant, which was then diluted to create an extraction at a 3% concentration for spraying plant foliage (Abd El-Mageed et al. 2017). The nutritional profile and chemical analysis of MLE are presented in Table S1. Tween 20 (0.1%, v/v) was used as a surfactant to enable the best possible absorption into the sweet potato plant leaves.
Application of effective microorganisms (EMs)
Five different types of beneficial microorganisms, including fermenting fungi, photosynthetic bacteria, lactic acid bacteria, actinomycetes, and yeast were present in EMs. The mixture was prepared by Egypt’s Ministry of Agriculture and Land Reclamation in Giza, as detailed by Abdelkhalik et al. (2023).
Measurements
Osmolyte accumulation assay
Sweet potato dry leaves were used for the extraction and measurement of osmolytes, i.e., total soluble sugars (TSS), total free amino acids (TFA), free proline, and soluble proteins in mg g−1 dry weight (DW). The techniques of Bates et al. (1973) were used to measure the free proline content. According to Irigoyen et al. (1992), the soluble sugar content in the leaves was determined using a Bausch & Lomb-2000 Spectrophotometer after extraction with 96% (v/v) ethanol. Using the approach of Wu et al. (2004), the amount of TFA in dried leaves was measured. Bradford (1976) recommended methods were used to quantify soluble proteins.
Assessment of enzymatic and non-enzymatic antioxidants
Bradford’s (1976) method was used to extract plant tissues as a crude enzyme extract to assess enzymatic and non-enzymatic antioxidants. For enzymatic antioxidants in U mg‒1 protein, the superoxide dismutase (SOD) enzyme activity was assessed using the approach of Giannopolitis and Ries (1977). Aebi’s (1984) approach was applied for catalase (CAT) activity quantification. According to Rao et al.’s (1996) method, the ascorbate peroxidase (APX) activity was assessed. After monitoring the GSH-dependent oxidation, the cellular glutathione reductase (GR) was measured per Rao et al.’s (1996) method. For non-enzymatic antioxidants, the phenolic acids (mg g−1 DW), reduced glutathione (GSH; µmol g−1 FW), and ascorbic acid (AsA; µmol g−1 FW) contents in fresh sweet potato leaves were quantified as outlined by Bradford (1976), Griffith (1980), and Mukherjee and Choudhuri (1983), respectively.
Photosynthetic pigments, plant water status, and membrane integrity
Following the instructions provided by Arnon (1949), chlorophyll “a” (chl a), chlorophyll “b” (chl b), total chlorophyll, and carotenoids were quantified in mg g−1 FW. Fresh leaf samples were homogenized in 50 ml of 80% (v/v) acetone before being centrifuged at 10,000 × g for 10 min. The absorbance of the extract was measured using a UV-160 A UV–vis recording spectrometer (Shimadzu, Kyoto, Japan) at 663, 645, and 470 nm for chlorophyll a, chlorophyll b, and carotenoids, respectively. According to Hayat et al. (2007), relative water content (RWC%) was assessed using the following formula:
The membrane stability index (MSI%) was estimated using the method, and the next equation was used for calculation. The following equation was used to calculate the membrane stability index (MSI%), which was determined using the Premachandra et al. (1990) approach.
where C1 is the EC of the solution after heating at 40 °C for 30 min, and C2 is the EC of the solution after boiling at 100 °C for 10 min.
Leaf mineral contents
The leaves of sweet potato (n = 10) were dried, ground into powder, and then chemically analyzed to assess the amount of N, P, K+, Ca2+, Na+, and Cd2+ in g kg−1 DW. The N content was quantified utilizing a micro-Kjeldahl equipment (Ningbo Medical Instruments Co., Ningbo, China) using the procedures of AOAC (2000). Molybdenum blue was diluted by 8% (w/v) NaHSO3-H2SO4 and H2MoO7S and used as standard reagents for the quantification of P, according to Jackson (1973). Also, a PerkinElmer Model 3300 atomic absorption spectrophotometer was used to quantify the Ca2+ level. In freeze-dried leaf powder suspension (50 mg), K+ and Na+ concentrations were measured (Lachica et al. 1973).
Roots and leaves cadmium (Cd2+) content
The Cd2+ content in sweet potato leaves and roots (tubers) was determined by spectrophotometry (PerkinElmer Model 3300) according to the Chapman and Pratt (1961) approach.
Agronomic traits
In both seasons, ten individual plants from each experimental unit were randomly collected at harvest (180 days after planting). Firstly, the leaves number of each plant was counted. A digital planimeter (Planix 7) was used to measure the leaf area (dm2 plant−1), and the leaf area index was computed by dividing the leaf area per plant by the area those plants occupied. The sweet potato shoot was oven-dried at 70 °C until it reached a constant weight, then recording the dry weight. Fifteen plants from each experimental unit were taken to determine the tuber number plant−1, weight for each tuber expressed as tuber weight (g), and tuber weight plant−1 (g). Moreover, all sweet potato plants in every experimental plot and the fifteen previously sampled were collected, cleaned, and weighed for estimated tuber yield.
Statistical analysis
For the two seasons based on RCBD, statistical analysis was performed using the GenStat tool (VSN International Ltd, Oxford, UK). The Fisher least-significant difference test was used to separate the means for each treatment at a p-value of ≤ 0.05.
Results
Growth traits
Saline soil contaminated with Cd2+ significantly diminished the growth characteristics of sweet potato (Table 2). The lowest values of leaves number plant−1 (222.1), leaf area plant−1 (119.9 dm2), leaf area index (2.99), and shoot dry weight (131.3 g) were recorded for control plots as averages in both seasons. However, the applied biostimulants, MLE, EMs, and their mixture (MLE + EMs) showed a beneficial effect on the growth traits of sweet potato (Table 2). Foliarly applied MLE or soil-treated EMs increased the leaves number plant−1 (by 88.7 or 72.6%), leaf area (by 98.2 or 46.5%), leaf area index (by 98.2 or 46.5%), and shoot dry weight (by 120.0 or 72.4%) when compared to untreated plants (Table 2). Co-application of MLE and EMs showed significant efficacy on leaf number and shoot dry plant−1, exceeding the effects of their individual application; meanwhile, no appreciable differences were found with MLE for leaf area and leaf area parameters in ameliorating stress-induced damages to sweet potato growth. Contextually, applying MLE + EMs significantly elevated the abovementioned growth traits by 98.8, 108.7, 109.0, and 184.8%, respectively, compared to the control (Table 2).
Leaves photosynthetic pigments, tissue water content, and membrane stability
The current investigation showed a noticeable deterioration in plant physiological reactions to adverse conditions created by soil salinity and contamination with Cd2+ (Table 3). In this regard, the minimum measured values (as the average of the two seasons) of relative water content (70.48%) and membrane stability index (62.81%) were given for non-ameliorated sweet potato plants (control treatment). On the other hand, plant RWC and MSI indices were substantially elevated by 8.3%, 10.6%, and 14.9% and 12.5%, 13.9%, and 17.8%, respectively, in sweet potato cultivated in Cd2+-contaminated saline soil in response to MLE, EMs, or MLE plus EMs over the control (Table 3). Similarly, Cd2+-contaminated saline soil exhibited negative impacts and significantly diminished the photosynthetic pigment contents (i.e., chl “a”, chl “b”, total chlorophyll, and carotenoids) of sweet potato plants. However, the individual or integrative application of MLE and EMs alleviated the salt and Cd2+-induced damages to the photosynthetic pigments. Sweet potato plants treated with MLE or EMs showed higher values of chl “a” by (86.6 and 35.1%), chl “b” (145.4 and 94.1%), total chlorophyll (119.0 and 67.6%), and carotenoids (118.2 and 81.8%), respectively, as compared to the untreated plants. Moreover, exogenous MLE plus soil application of EMs further increased the photosynthetic pigment contents of stressed sweet potato plants. The highest results of chl “a” (2.22), chl “b” (3.81), total chlorophyll (6.03), and carotenoids (0.26) as an average of the two seasons were measured for treated sweet potato plants with MLE + EMs. Interestingly, the MLE + EMs treatment exhibited non-significant differences with the MLE treatments for carotenoids.
Osmolyte accumulation
The data in Fig. 1 represented the accumulation of osmolytes (i.e., TSS, free proline, TFA, and soluble proteins) in sweet potato grown under Cd2+-contaminated saline soil in response to applied plant biostimulants. The accumulation of these osmoprotectants increased significantly in correspondence with the individual and co-use of MLE and EMs. The application of EMs to Cd2+-contaminated saline soil generated a significant increase in contents of TSS (by 11.0%), free proline (by 18.4%), TFA (by 53.9%), and soluble proteins (by 51.4%) in leaves of sweet potato plants in comparison to the control. In addition, applying MLE as a foliar spray on stressed sweet potato plants caused a significant increase in the contents of the osmo-regulators mentioned above by 37.1, 20.6, 60.6, and 55.9%, respectively, in contrast to the control. Combined application of MLE and EMs resulted in a substantial increase in concentrations of TSS by 69.6%, free proline by 47.7%, TFA by 29.0%, and soluble proteins by 125.7% compared with untreated plants. Meanwhile, the difference in free proline content due to the single addition of MLE or in combination with EMs was insignificant. Hence, in correspondence with used agro-management practices, these substances’ accumulation followed the subsequent descending order: MLE + EMs > MLE > EMs.
Influence of Moringa leaf extract (MLE) and effective microorganisms (EMs) on osmolytes (i.e., free proline, total free amino acids—TFA, total soluble sugars—TSS, and soluble proteins) content of sweet potato plants grown in Cd2 + -contaminated saline soil in the 2020 and 2021 seasons (as average). The various lower-case letters on the bar distinguished the treatments, corresponding to Fisher’s test at p ≤ 0.05
Antioxidative compounds
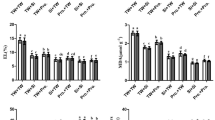
Our results indicated that the minimum values of ascorbate peroxidase (APX), glutathione reductase (GR), catalase (CAT), and superoxide dismutase (SOD) at 11.63, 10.53, 20.03, 15.43 U mg‒1 protein, respectively, were observed for non-treated sweet potato plants. Interestingly, the activity of the enzymatic antioxidants APX, GR, CAT, and SOD was upregulated in sweet potato plants subjected to Cd2+-contaminated saline soil by the application of MLE, EMs, or MLE plus EMs (Fig. 2); they elevated the APX by 35.9, 15.8, and 35.3%, the GR by 19.7, 19.9, and 29.8%, the CAT by 13.7, 13.3, and 17.3%, and the SOD by 9.1, 14.5, and 14.3%, respectively, in comparison with the control. In Cd2+-contaminated saline soil, the single or co-application of MLE and EMs accumulated higher phenolic acids, GSH, and AsA in sweet potato leaves (Fig. 3). These treatments raised phenolic acids (6.6–7.8%), GSH (5.5–7.0%), and AsA (9.9–19.6%) compared to the control plants. Single or combined application of MLE and EMs recorded similar values for phenolic acids and GSH while noting the superiority of MLE + EMs treatment in AsA.
Influence of Moringa leaf extract (MLE) and effective microorganisms (EMs) on the activity of glutathione reductase (GR), superoxide dismutase (SOD), catalase (CAT), and ascorbate peroxidase (APX) of sweet potato plants grown in Cd2 + -contaminated saline soil in the 2020 and 2021 seasons (as average). The various lower-case letters on the bar distinguished the treatments, corresponding to Fisher’s test at p ≤ 0.05
Influence of Moringa leaf extract (MLE) and effective microorganisms (EMs) on phenolic acids, ascorbic acid (AsA), and glutathione (GSH) contents of sweet potato plants grown in Cd2 + -contaminated saline soil in the 2020 and 2021 seasons (as average). The various lower-case letters on the bar distinguished the treatments, corresponding to Fisher’s test at p ≤ 0.05
Nutrient acquisition
Results listed in Fig. 4 showed significant improvements in the nutrient acquisition of sweet potato plants cultivated in Cd2+-contaminated salty soil due to the MLE and/or EMs treatments. The uptake of nutrient elements (i.e., N, P, K+, Ca2) and the K+/Na+ ratio by sweet potato plants grown in control plots was substantially reduced compared with MLE and EMs amended plants. Unlike the nutrients mentioned above, the levels of Na+ were elevated in control plots in sweet potato leaves. Nevertheless, foliar-applied MLE or EMs triggered a notable improvement in N, P, K+, Ca2+, and K+/Na+ ratio contents by 51.3 or 28.2%, 46.9 or 22.0%, 42.5 or 24.8%, 37.0 or 21.2%, and 135.0 or 126.1%, respectively, while decreasing the Na+ content by 41.0 or 44.4% in sweet potato plants relative to control. Interestingly, the integrative application of MLE and EMs magnified the macronutrient concentration, bypassing individual influences. They caused N to rise by 63.2%, P to rise by 58.8%, K+ to rise by 44.5%, Ca2+ to rise by 116.0%, and K+/Na+ ratio to rise by 159.7%, and Na+ to decrease by 43.9% when compared to control.
Influence of Moringa leaf extract (MLE) and effective microorganisms (EMs) on leaf nutrient concentrations of sweet potato grown in Cd2 + -contaminated saline soil in the 2020 and 2021 seasons (as average). The various lower-case letters on the bar distinguished the treatments, corresponding to Fisher’s test at p ≤ 0.05
Roots and leaves cadmium (Cd2+) content
Sweet potato plants grown in saline soil contaminated with Cd2+ exhibited higher accumulation of Cd2+ in the roots and leaves (Fig. 5). Nonetheless, foliar-applied MLE, soil application with EMs, or their integrative application significantly reduced Cd2+ content in the roots and leaves of sweet potato plants transplanted in Cd2+-contaminated saline soil (Fig. 5). The best results were associated with MLE + EMs treatment, which maintained the accumulation of Cd2+ ions at their lowest levels in the roots (68.1%) and leaves (38.0%) compared to the control. Also, the application of MLE or EMs induced a significant reduction of Cd2+ content in the roots by 55.6 or 50.0% and in the leaves by 31.4 or 27.6%, respectively, compared with the control.
Influence of Moringa leaf extract (MLE) and effective microorganisms (EMs) on the Cd2 + content in roots and leaves of sweet potato grown in Cd2 + -contaminated saline soil in the 2020 and 2021 seasons (as average). The various lower-case letters on the bar distinguished the treatments, corresponding to Fisher’s test at p ≤ 0.05
Yield and yield-related components
Sweet potato plants grown in Cd2+-contaminated saline soil produced the lowest tubers’ number per plant, tuber weight, tubers’ weight per plant, and tuber yield (Table 4). Nonetheless, Cd2+- and salt-stressed Ipomoea batatas plants exhibited an increment in the yield-related components when treated with MLE, EMs, or MLE plus EMs. The increments of the aforementioned yield-related characters amounted to 35.3 or 34.6%, 59.4 or 35.7%, 19.7 or 17.9%, and 59.5 or 35.7%, respectively, due to exogenous MLE or soil application of EMs against control. Meanwhile, the integrative application of MLE and EMs generated further increases in tuber number per plant by 76.4%, tuber weight by 44.7%, tubers’ weight per plant by 73.0%, and tuber yield by 73.1% (Table 4).
Relationships
The hierarchical analysis presented in a heat map graph shows the relation between the single or combined application of MLE and EMs on the studied parameters (Fig. 6), divided into two major sets (control and application of EMs, MLE, or MLE and EMs). The hierarchical analysis exhibited that the co-application MLE and EMs separated in a sub-set and performed well from the individual application of MLE or EMs. These results indicated that the foliar spraying MLE and soil application of EMs to sweet potato plants grown in Cd2+-contaminated saline soil have a significant role in enhancing the growth, yield, and defense machinery of the plant while reducing the Na+ and Cd2+ accumulation in sweet potato tissues. The impact of MLE and/or EMS on agronomic, physiological, and biochemical parameters under Cd2+ and salt stress conditions was investigated using the principal component analysis (PCA) (Fig. 7). The variability of the first and second components represents about 66.98% (PC1 = 22.31 and PC2 = 44.67%) (Fig. 7). Under Cd2+-contaminated saline soil, the PCA showed a positive correlation of the integrative application of MLE and EMs for growth, tuber yield, photosynthetic pigments, osmolyte accumulation, and antioxidant capacity. The analysis also revealed a positive correlation between Cd2+-contaminated saline soil and Na+, Cd2+ accumulation in sweet potato plants. Also, under Cd2+ and salt stress, PCA exhibited a clear positive correlation between GSH and the individual effect of MLE or EMs.
The heat map diagram displays hierarchical clustering analysis among the application of Moringa leaf extract (MLE) and/or effective microorganisms (EMs) and different studied parameters of sweet potato plants grown Cd2 + -contaminated saline soil. SOD, superoxide dismutase; RWC, relative water content; AsA, ascorbic acid; MSI, membrane stability index; CAT, catalase; APX, ascorbate peroxidase; GR, glutathione reductase; Leaf.area, leaf area plant−1; K, potassium; P, phosphorus; N, nitrogen; Ca, calcium; Na, sodium; Shoot DW, shoot dry weight; TSS, total soluble sugars; Tubers. no. plant, tubers number plant-1; GSH, glutathione; Cd. root and Cd. Leaves, roots and leaves Cd2 + content, respectively
Biplot of the principal component analysis (PCA) illustrating the relationship among the effects of Moringa leaf extract (MLE) and/or effective microorganisms (EMs) on different assessed parameters of sweet potato plants grown in Cd2 + -contaminated saline soil. Ca, calcium; Na, sodium; proline, free proline; TSS, total soluble sugars; GSH, glutathione; Shoot DW, shoot dry weight; Cd root and Cd leaves, roots and leaves Cd2 + content, respectively
Discussion
Heavy metal-contaminated salty soil seriously threatens the sustainability of vegetable crop production (Abd El-Mageed et al. 2022a, b). As a result, a new challenge arises in increasing sweet potato tolerance, reducing the unfavorable conditions brought on by the co-occurrence of Cd2+ accumulation and salinity in the soil, and promoting their long-term sustainability. Therefore, the application of biostimulants for plants has been suggested to support them to withstand Cd2+ and salt stress. Furthermore, since the co-application of MLE and EMs has never been done, it is crucial to comprehend further the mechanism of action of exogenous MLE and soil application of EMs on sweet potato under Cd2+ and salt stress.
Long-term exposure to saline soil contaminated with heavy metals perturbed several metabolic processes, including cell division, elongation, cell cycle arrest, and a high transpiration rate because of a high energy requirement (Desoky et al. 2019b). As a result, sweet potato plant growth was negatively impacted (Table 2), and tuber yield was decreased (Table 4). Plant growth suppression caused by Cd2+-contaminated saline soil-mediated stress might be attributed to ROS generation, photo-inhibition, reduced photosynthetic pigment contents, and plant water status. Also, increased Cd2+ and Na+ acquisition in sweet potato plants may cause a nutritional imbalance and ion toxicity, leading to symptoms of nutritional deficiency and growth restrictions (Isayenkov and Maathuis 2019; dos Santos et al. 2022). However, exogenous MLE combined with soil-applied EMs enhanced stressed sweet potato plants’ growth characteristics and physiological and biochemical attributes. Protein-containing MLE is necessary for protoplasm formation, vitamin C, and essential nutrients like Ca2+, Mg2+, and K+ that are involved in several natural antioxidant compounds such as flavonoids, ascorbic acid, phenolic acids, and carotenoids which in turn reflect its vital role as an excellent growth biostimulator (Talaat 2019; Abd El-Mageed et al. 2022a, b).
Furthermore, the application of auxins and cytokinins (especially zeatin) and gibberellins-containing MLE has a growth-promoting effect that improves cell division and enlargement and enhances the biosynthesis of chlorophyll molecules. Similar results showed MLE’s positive effect on wheat plants’ growth (Ahmed et al. 2021) and maize (Pervez et al. 2017). Similarly, improved growth traits and tuber yield in EMs-treated sweet potato plants could be attributed to synthesizing some useful bioactive material for plants, such as amino acids, lactic acid, vitamins, sugars, enzymes, and hormones that trigger cell division (Hu and Qi 2013). Furthermore, EMs stimulate the production of several hormones such as abscisic acid, cytokinin, and auxin; the accumulation of EPSs and enzymes (i.e., 1-aminocyclopropane-1-carboxylate deaminase (ACC deaminase); the synthesis of osmoregulatory substances (Bittencourt et al. 2023). These actions collectively induce the osmoregulation process, which may has a beneficial effect on sweet potato tolerance to saline and Cd2+ stress (Rady et al. 2023).
In response to Cd2+ or/and salt stress, plants accumulate extra osmoregulators such as soluble sugars, free proline, free amino acids, and soluble proteins to contribute to stress tolerance (Abdelkhalik et al. 2019; Semida et al. 2021a). These solutes help plants to adjust their osmotic environment, maintain turgor pressure, allow more nutrients and water uptake, inhibit free radicals’ activity, and safeguard their thylakoid membranes (Semida et al. 2020; Abdou et al. 2022). The single use of MLE or coupled addition with EMs generated further accumulation of soluble sugars, free proline, free amino acids, and soluble protein in Cd2+ and salt-stressed sweet potato plants (Fig. 1). Moreover, the greatest favorable results were assigned to the co-application of MLE and EMs. This favorable effect of MLE on stressed sweet potato plants could be associated with high content of osmolytes, phytohormones, and mineral ions (Zulfiqar et al. 2020; Khan et al. 2022). These beneficial MLE constituents were also identified in the MLE analysis conducted for this study (Table S1), which may have contributed to the accumulation of these osmoprotectants in MLE-treated plants. Exogenously applied MLE as an activator for free proline accumulation was observed in other crops, such as beans (Rady and Mohamed 2015) and potatoes (El-Mohamed et al. 2016).
In addition, when plants are exposed to unfavorable conditions, they activate the synthesis of antioxidants to mitigate the detrimental effects of oxidative stress caused by free radicals (Abd El-Mageed et al. 2022a, b; Selem et al. 2022). The present study showed significant increases in the activity of antioxidant enzymes (i.e., GR, APX, CAT, and SOD; Fig. 2) and the content of non-enzymatic antioxidants (i.e., phenolic acids, GSH, and AsA; Fig. 3). This improvement in GSH, AsA, GR, CAT, SOD, and APX levels is an ameliorative mechanism by which sweet potato plants could resist the oxidative stress generated via Cd2+ and salinity exposure. Additionally, these findings exhibited that MLE or/and EMs may combat oxidative stress by improving the AsA-GSH cycle and upregulating the enzymatic antioxidants. This significant improvement could be attributed to the effective role of MLE as a good source of amino acids, phenolic acids, nutrients, soluble sugars, and antioxidants like ascorbate and free proline (Table S1), which subsequently enhanced the accumulation of these antioxidants and enabled sweet potato plants to tolerate Cd2+ and salinity stress. Besides, its considerable levels of carotenoids (α, β carotene, lutein, and xanthin) have potent antioxidant properties (Irshad et al. 2022; Khan et al. 2022; Martínez-ispizua et al. 2022a). A similar finding was observed by Zulfiqar et al. (2020) and Khalofah et al. (2020), who reported that MLE application caused a considerable increase in GR, SOD, and APX antioxidants in common bean plants. Inculcating plants with EMs might enhance the synthesis of EPSs and beneficial enzymes, such as ACC deaminase and phytohormones (i.e., cytokinin, auxin, and abscisic acid), which in turn may lessen the harm caused by environmental stress (Bittencourt et al. 2023). This positive impact of EMs on the plant antioxidant defense system was also represented by Iriti et al. (2019) and Abd El-Mageed et al. (2022a, b). Hence, activating antioxidative enzymes, protecting the plants from oxidative damage via regulating some metabolic processes, including cell division, biosynthesis of proteins, the activity of enzymes, phytohormones, and gene expression, protecting cells from H2O2 toxicity, and maintaining membranes from oxidation and organic peroxides (Patel and Parida 2021; Martínez-ispizua et al. 2022b).
In the present study, planting sweet potatoes in Cd2+-contaminated saline soil led to a higher accumulation of Cd2+ and Na+ in plant tissue (Figs. 4 and 5), resulting in a reduction in nutrient acquisition (Fig. 4), plant water status, membrane integrity, and photosynthetic pigment contents (Table 3), as well as growth (Table 2), and tuber yield (Table 4). However, applying MLE or/and EMs ameliorated the Cd2+-contaminated saline soil-induced damage to sweet potatoes. The Cd2+ and salinity stress mediated disturbances in the ion homeostasis of sweet potato, given that they raised leaf Cd2+ and Na+ and lowered N, P, K+, Ca2+, and the K+/Na+ ratio (Fig. 4). This could be due to Na+ and Cd2+ influx that may disturb water uptake and interfere with the same N, P, K + , Ca2+, Mn, and Mg transporters, restricting their absorption (Sales and Martins 2019; Sitohy et al. 2020; Haider et al. 2021). Nonetheless, externally applied MLE or MLE plus EMs-treated soil alleviated the cumbersome influences of toxic Na+ and Cd2+ by reducing their content in the tissues and induced a considerable increase in N, P, K+, and Ca2+ acquisition, increasing the K+/Na+ ratio. As a key component of salt tolerance, maintaining high K+/Na+ ratios in tissues and, consequently, high cytosolic K+/Na+ ratios has been used to explain how Na+ uptake and transport are regulated in plants under salt stress (Assaha et al. 2017). Our study demonstrated a higher accumulation of Cd2+ in the sweet potato leaves than in the roots. In harmony with our data, Baldantoni et al. (2016) reported that the Cd2+ content in lettuce and endive leaves raised to 9 mg.kg−1, fourfold higher than in roots, and up to 30-fold higher than the Cd2+ content in the studied soil.
Foliar-applied MLE and soil-treated EMs alleviated the harmful effect of excessive Cd2+ and Na+ in root media by reducing its uptake and considerably increased the acquisition of essential macronutrients. Hence, improving the nutritional status of stressed sweet potato plants by lowering the absorption of Cd2+ and Na+ ions and raising the acquisition of N, P, K+, and Ca2+ using MLE and EMs biostimulants could be a vital strategy to counteract Cd2+ and salt stress. The nutritional profile of MLE-treated sweet potato plants was improved because MLE is a natural product abundant in vital elements, including Ca2+, Mg2+, K+, P, Fe2+, Mn2+, Cu2+, and Zn2+ (Muthuraja and Muthukumar 2022). The effective function that EMs play in preserving greater membrane integrity that consequently controls the membranes’ ability to selectively take in and transport ions (Munir et al. 2022) may be the cause of improved nutrient uptake with EMs’ application. Inoculating the soil with EMs accelerates the organic matter decomposition, which boosts nutrient release and solubility and directly impacts plant ion uptake (Soares et al. 2021; Abdelkhalik et al. 2023).
Furthermore, increased protein production in response to using MLE alone or combined with EMs on sweet potato plants might be correlated with increased content of inorganic ions such as N, P, K+, and Ca2+, and a higher K+/Na+ ratio. Plant nutritional status ameliorates the disruptive effect of osmotic stress on various enzymes, including protein biosynthesis (Talaat 2019). EMs can accumulate osmolytes, which synergistically can act with osmoprotectants produced by plants, such as free proline and total soluble sugars (Ozturk et al. 2021). Moreover, plants treated with EMs can produce exopolysaccharides (EPSs), enabling plants to have a more substantial accumulation of sugars, free proline, TFA, and soluble protein content (Khan and Bano 2019). As a result, these beneficial bacteria cause the manufacture of these osmolytes in plant cells, which protects cells from oxidative damage and increases tolerance to Cd2+ or/and salt stress (Rady et al. 2021; Ahmed et al. 2022).
High levels of Cd2+ and salt in the soil lower their osmotic potential and impose a decrease in leaf water potential, causing osmotic stress (Rucińska-Sobkowiak 2016) and a reduction of RWC as stated in the present study (Table 3). Thereafter, these salts accumulated in the plant tissues, inducing oxidative damage (Abd El-Mageed et al. 2020) and causing damage to the cellular membrane (MSI; Table 3). However, the applied bio-elicitors, MLE and/or EMs, resulted in higher RWC and MSI than Cd2+ and salt-stressed sweet potato plants, indicating that the osmotic and ionic stress was lower in MLE and/or EMs-treated plants (Talaat et al. 2015). The beneficial effect of MLE and/or EMs on tissue RWC could be attributed to their vital role in regulating the osmotic potential inside plant cells via increasing the content of TSS, TFA, free proline (Fig. 1), and inorganic ions such as K+ (Fig. 4). Also, stimulated ion acquisition (Fig. 4) in response to MLE and/or EMs application decreases the toxic effects of Na+ and Cd2+, improving the stability of membranes in stressed sweet potato plants. The enhancements in MSI generated by EMs application may be attributed to EMs’ beneficial effects on plant defense mechanisms, such as free proline (Fig. 1), enzymatic and non-enzymatic antioxidants (Figs. 2 and 3), and carotenoid accumulation (Table 3), which is crucial for maintaining cell membrane stability by preventing ROS accumulation (Talaat et al. 2015; Abd El-Mageed et al. 2022a, b). MLE is rich in Ca2+ ions required for structural roles in the cell membranes, improving Ca2+ status in plant tissues, which minimizes ion leakage and enhances membrane stability for MLE-treated plants (Farhat et al. 2018; Al-Taisan et al. 2022).
The Cd2+ and salt stress exacerbate lower physiologically active water in tissues, ROS production, and nutrient acquisition, thus activating the chlorophyllase enzyme and chloroplast deterioration, inhibiting photosynthetic pigment biosynthesis (Rasafi et al. 2022). However, the unfavorable photosynthetic pigments from salty soil contaminated with Cd2+ were alleviated by the co-application of MLE and EMs, which may be related to their positive roles in increased nutrient acquisition, osmoprotectants, and antioxidants. These results are harmonized with those demonstrated by Talaat et al. (2015) and Kalaji et al. (2016). The presence of nutrients, osmolytes, cytokinins (Zeatin), and gibberellins in MLE (Table S1) stimulates the mechanisms involved in chlorophyll and carotenoids biosynthesis, inhibits photosynthetic pigment degradation and premature leaf senescence, and maintains leaf greenish for longer periods (Desoky et al. 2019b). Applying foliar spraying with MLE significantly mitigated the observed enhancements in chlorophyll levels in wheat plants subjected to drought stress (Fozia et al. 2022). Furthermore, as a biostimulator, MLE is rich in different nutritional elements, especially Mg, a constituent of chlorophyll molecules. Also, the EMs’ application may stimulate chlorophyll biosynthesis due to its effective role in improving the absorption and translocation of essential ions such as N, P, K+, and Ca2+.
The present study found that sweet potatoes grown in saline soil contaminated with Cd2+ had a larger buildup of Cd2+ in their roots and leaves (Fig. 5). Interestingly, the integrative application of MLE and EMs substantially reduced Cd2+ accumulation in different sweet potato parts grown in Cd2+-contaminated salty soil (Fig. 5). Therefore, applying biostimulants (i.e., MLE and EMs) is considered an effective strategy for Cd2+-phytoremediation when applied as foliar and soil applications. EMs may be able to adsorb Cd2+ ions (Treesubsuntorn et al. 2018; Matos et al. 2019), thus reducing their transportation into sweet potato plants. Furthermore, increased availability of Cd2+-divalent cations such as Ca2+ and Mg2+ may create competition with Cd2+, decreasing Cd2+ uptake in rice plants cultivated in Cd2+-contaminated soil (Treesubsuntorn et al. 2018). Cd2+ reduction in sweet potato by exogenous MLE might be a result of the production of enzymatic (Fig. 2) and non-enzymatic antioxidants (Fig. 3) and increased nutrient acquisition to counteract Cd2+ sequestration in plants (Khalofah et al. 2020, Abalaka et al. 2022).
The positive effects of used MLE and/or EMs on sweet potato plants’ growth and physiological and biochemical responses are reasonable for improved water use efficiency, yield, and related parameters (Table 4). In another study by Abd El-Mageed et al. (2022a, b), saline soil-applied EMs increased sweet potato plants’ average tuber weight, number, and yield.
Our study demonstrated the possibility of foliar application with MLE and/or soil application of EMs to improve sweet potato growth and productivity in Cd2+-contaminated saline soil. Greater effectiveness was demonstrated in decreasing stress brought on by Cd2+ and salinity when MLE and EMs were applied together rather than separately. Moreover, the present results may have practical importance as they highlight the potential of both EM and MLE as successful plant biostimulants for future use in reducing oxidative damage and promoting sustainable crop production in arid and semiarid areas.
Conclusion
The results showed that the integrative application of Moringa leaf extract and effective microorganisms can effectively enhance sweet potato tolerance to salinity and cadmium stress. Stress tolerance in sweet potato is a result of increased nutrient acquisition, osmolytes accumulation, and antioxidants in sweet potato plants compared with control. Moreover, Moringa leaf extract and effective microorganisms beneficially affected stressed plants by reducing the cadmium and sodium uptake in sweet potato tissues. Therefore, using these biostimulants may help plants thrive in challenging environments brought on by salt and cadmium stressors. The photosynthetic pigments, tissue water status, and membrane stability were improved in plants treated with Moringa leaf extract and effective microorganisms; subsequently, the growth and tuber yield of sweet potato plants were promoted. Therefore, using Moringa leaf extract and effective microorganisms to increase the productivity of sweet potato plants cultivated in cadmium-contaminated saline soils may be a promising sustainable strategy for the future.
Data availability
The data behind this article will be shared with the appropriate authors upon reasonable request.
References
Abalaka SE, Oyelowo FO, Akande MG et al (2022) Correction to: effects of Moringa oleifera leaves extract, vitamin C, and taurine co-exposures on calcium and metallothionein levels, oxidative stress, and gill histopathological changes in Clarias gariepinus exposed to sub-lethal cadmium. Environ Sci Pollut Res 29:40279. https://doi.org/10.1007/s11356-022-20149-6
Abd El-Mageed TA, El-Sherif AMA, Abd El-Mageed SA, Abdou NM (2019) A novel compost alleviate drought stress for sugar beet production grown in Cd-contaminated saline soil. Agric Water Manag 226:105831. https://doi.org/10.1016/j.agwat.2019.105831
Abd El-Mageed TA, Abdurrahman HA, El-Mageed SAA (2020) Residual acidified biochar modulates growth, physiological responses, and water relations of maize (Zea mays) under heavy metal – contaminated irrigation water. Environ Sci Pollut Res 27:22956–22966
Abd El-Mageed TA, Gyushi MAH, Hemida KA et al (2022b) Coapplication of effective microorganisms and nanomagnesium boosts the defenses against salt stress in Ipomoea batatas. Front Plant Sci 13(883274):1–14. https://doi.org/10.3389/fpls.2022.883274
Abd El-Mageed TA, Semida WM, Rady MM (2017) Moringa leaf extract as biostimulant improves water use efficiency, physio-biochemical attributes of squash plants under deficit irrigation. Agric Water Manag 193:. https://doi.org/10.1016/j.agwat.2017.08.004
Abd El-Mageed, Shaaban A, Abd El-Mageed SA, et al (2020) Defensive role in maize (Zea mays L.) against drought stress and metals-contaminated irrigation water. Silicon. https://doi.org/10.1007/s12633-020-00690-0
Abd El-Mageed TA, Abd El-Mageed SA, El-Saadony MT, et al (2022a) Plant growth-promoting rhizobacteria improve growth, morph-physiological responses, water productivity, and yield of rice plants under full and deficit drip irrigation. Rice 15:. https://doi.org/10.1186/s12284-022-00564-6
Abd El-Mageed TA, Semida WM, Abdou NM, El-Mageed SAA (2023) Coupling effects of potassium fertilization rate and application time on growth and grain yield of wheat (Triticum aestivum L.) plants grown under Cd-contaminated saline soil. J Soil Sci Plant Nutr. https://doi.org/10.1007/s42729-022-01104-3
Abdelkhalik A, Pascual B, Nájera I et al (2019) Deficit irrigation as a sustainable practice in improving irrigation water use efficiency in cauliflower under Mediterranean conditions. Agronomy 9(732):732. https://doi.org/10.3390/agronomy9110732
Abdelkhalik A, Abd El-mageed TA, Mohamed IAA et al (2023) Soil application of effective microorganisms and nitrogen alleviates salt stress in hot pepper (Capsicum annum L.) plants. Front Plant Sci 13:1079260. https://doi.org/10.3389/fpls.2022.1079260
Abdou NM, El-Saadony FMA, Roby MHH, et al (2022) Foliar spray of potassium silicate, aloe extract composite and their effect on growth and yielding capacity of roselle (Hibiscus sabdariffa L.) under water deficit stress conditions. Saudi J Biol Sci. https://doi.org/10.1016/j.sjbs.2022.02.033
Abou-Sreea AIB, Roby MHH, Mahdy HAA et al (2022) Improvement of selected morphological, physiological, and biochemical parameters of roselle (Hibiscus sabdariffa L.) grown under different salinity levels using potassium silicate and Aloe saponaria extract. Plants 11:1–20. https://doi.org/10.3390/plants11040497
Abu-Qaoud H, Al-Fares H, Shtaya MJY, Shawarb N (2021) Effect of effective microorganisms on wheat growth under salt stress condition. Chil J Agric Res 81:351–356. https://doi.org/10.4067/S0718-58392021000300351
Aebi H (1984) Catalase in vitro. Methods Enzymol Methods Enzym 6(105):121
Ahmed EA, Shoala T, Abdelkhalik A et al (2022) Nanoinhibitory impacts of salicylic acid, glycyrrhizic acid ammonium salt, and boric acid nanoparticles against phytoplasma associated with faba bean. Molecules 27:1467. https://doi.org/10.3390/molecules27051467
Ahmed T, Elezz AA, Khalid MF (2021) Hydropriming with moringa leaf extract mitigates salt stress in wheat seedlings. Agriculture 11:. https://doi.org/10.3390/agriculture11121254
Al-Taisan WA, Alabdallah NM, Almuqadam L (2022) Moringa leaf extract and green algae improve the growth and physiological attributes of Mentha species under salt stress. Sci Rep 12:1–9. https://doi.org/10.1038/s41598-022-18481-5
AOAC (2000) Official methods of analysis, 17th edn. The Association of Official Analytical Chemists. Gaithersburg, Gaithersburg, MD. USA
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenol-oxidase in Beta vulgaris L. Plant Physiol 24:1–5
Assaha DVM, Ueda A, Saneoka H, Al-yahyai R (2017) The role of Na+ and K+ transporters in salt stress adaptation in glycophytes. Front Physiol 8:. https://doi.org/10.3389/fphys.2017.00509
Baldantoni D, Morra L, Zaccardelli M, Alfani A (2016) Cadmium accumulation in leaves of leafy vegetables. Ecotoxicol Environ Saf 123:89–94. https://doi.org/10.1016/j.ecoenv.2015.05.019
Bates L, Waldren R, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil 207:205–207
Batool S, Khan S, Basra SMA (2020) Foliar application of moringa leaf extract improves the growth of moringa seedlings in winter. South African J Bot 129:347–353. https://doi.org/10.1016/j.sajb.2019.08.040
Bittencourt PP, Alves AF, Ferreira MB, et al (2023) Mechanisms and applications of bacterial inoculants in plant drought stress tolerance. Microorganisms 11:. https://doi.org/10.3390/microorganisms11020502
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Byju G, George J (2005) Potassium nutrition of sweet potato. Adv Hortic Sci 19:221–239. https://doi.org/10.1400/14433
Chapman HD, Pratt PF (1961) Methods of analysis for soils, plants and waters. University of California, Los Angeles
Chourasia KN, Lal MK, Tiwari RK, et al (2021) Salinity stress in potato: understanding physiological, biochemical and molecular responses. Life 11:. https://doi.org/10.3390/life11060545
De Corato U (2021) Effect of value-added organic co-products from four industrial chains on functioning of plant disease suppressive soil and their potentiality to enhance soil quality: a review from the perspective of a circular economy. Appl Soil Ecol 168:104221. https://doi.org/10.1016/j.apsoil.2021.104221
Desoky ESM, Elrys AS, Rady MM (2019b) Integrative moringa and licorice extracts application improves Capsicum annuum fruit yield and declines its contaminant contents on a heavy metals-contaminated saline soil. Ecotoxicol Environ Saf 169:50–60. https://doi.org/10.1016/j.ecoenv.2018.10.117
Desoky E, Merwad A-R, Ibrahim S (2019a) Humus materials and Moringa ( Moringa oleifera Lam.) leaf extract modulate the harmful effect of soil salinity stress in Sudan grass (Sorghum vulgare L.). Egypt J Agron:. https://doi.org/10.21608/agro.2019.6844.1141
dos Santos TB, Ribas AF, de Souza SGH et al (2022) Physiological responses to drought, salinity, and heat stress in plants: a review. Stresses 2:113–135. https://doi.org/10.3390/stresses2010009
El Rasafi T, Oukarroum A, Haddioui A et al (2022) Cadmium stress in plants: a critical review of the effects, mechanisms, and tolerance strategies. Crit Rev Environ Sci Technol 52:675–726. https://doi.org/10.1080/10643389.2020.1835435
El-Mohamed RSR, Abdalla AM, Ghoname AA (2016) Field application of chitosan and Moringa oleifera extracts as fungicides alternatives to control early blight and improvement growth and yield quality of potato. Plant Pathol J 15:135–143. https://doi.org/10.3923/ppj.2016.135.143
Farhat F, Arfan M, Wahid A, Sadia B (2018) Modulation of ionic and water status by Moringa oleifera extract against cadmium toxicity in wheat (Triticum aestivum). Int J Agric Biol 20:2692–2700
Fozia F, Muhammad A, Abdul W, Bushra S (2022) The impact of bio-stimulants on Cd-stressed wheat (Triticum aestivum L.): insights into growth, chlorophyll fluorescence, Cd accumulation, and osmolyte regulation. Front Plant Sci 13:1–15. https://doi.org/10.3389/fpls.2022.850567
Genchi G, Sinicropi MS, Lauria G et al (2020) The effects of cadmium toxicity. Int J Environ Res Public Health 17:1–24. https://doi.org/10.3390/ijerph17113782
Giannopolitis C, Ries S (1977) Superoxide dismutases. I. Occurrence in Higher Plants Plant Physiol 59:309–314
Gopalakrishnan L, Doriya K, Kumar DS (2016) Moringa oleifera: a review on nutritive importance and its medicinal application. Food Sci Hum Wellness 5:49–56. https://doi.org/10.1016/j.fshw.2016.04.001
Griffith W (1980) Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal Biochem 106:207–212
Haider FU, Liqun C, Coulter JA et al (2021) Cadmium toxicity in plants : impacts and remediation strategies. Ecotoxicol Environ Saf 211:111887. https://doi.org/10.1016/j.ecoenv.2020.111887
Hayat S, Ali B, Hasan SA, Ahmad A (2007) Brassinosteroid enhanced the level of antioxidants under cadmium stress in Brassica juncea. Environ Exp Bot 60:33–41. https://doi.org/10.1016/j.envexpbot.2006.06.002
Howladar SM (2014) A novel Moringa oleifera leaf extract can mitigate the stress effects of salinity and cadmium in bean (Phaseolus vulgaris L.) plants. Ecotoxicol Environ Saf 100:69–75. https://doi.org/10.1016/j.ecoenv.2013.11.022
Hu C, Qi Y (2013) Long-term effective microorganisms application promote growth and increase yields and nutrition of wheat in China. Eur J Agron 46:63–67. https://doi.org/10.1016/j.eja.2012.12.003
Irigoyen JJ, Einerich DW, Sánchez-Díaz M (1992) Water stress induced changes in concentrations of proline and total soluble sugars in nodulated alfalfa (Medicago sativa) plants. Physiol Plant 84:55–60. https://doi.org/10.1111/j.1399-3054.1992.tb08764.x
Iriti M, Scarafoni A, Pierce S, et al (2019) Soil application of effective microorganisms (EM) maintains leaf photosynthetic efficiency, increases seed yield and quality traits of bean (Phaseolus vulgaris L.) plants grown on different substrates. Int J Mol Sci 20:. https://doi.org/10.3390/ijms20092327
Irshad S, Matloob A, Iqbal S et al (2022) Foliar application of potassium and moringa leaf extract improves growth, physiology and productivity of kabuli chickpea grown under varying sowing regimes. PLoS ONE 17:1–19. https://doi.org/10.1371/journal.pone.0263323
Isayenkov SV, Maathuis FJM (2019) Plant salinity stress: many unanswered questions remain. Front Plant Sci 10:80. https://doi.org/10.3389/fpls.2019.00080
Jackson ML (1973) Soil chemical analysis. New Delhi, India. Prentice Hall India Pvt Ltd
Kalaji HM, Cetner MD, Samborska IA et al (2016) Effective microorganisms impact on photosynthetic activity of Arabidopsis plant grown under salinity stress conditions. Ann Warsaw Univ Life Sci-SGGW L Reclam 48:153–163. https://doi.org/10.1515/sggw-2016-0012
Khademian R, Asghari B, Sedaghati B, Yaghoubian Y (2019) Plant beneficial rhizospheric microorganisms (PBRMs)mitigate deleterious effects of salinity in sesame (Sesamum indicum L.): Physio-biochemical properties, fatty acids composition and secondary metabolites content. Ind Crops Prod 136:129–139. https://doi.org/10.1016/j.indcrop.2019.05.002
Khalofah A, Bokhari NA, Migdadi HM, Alwahibi MS (2020) Antioxidant responses and the role of Moringa oleifera leaf extract for mitigation of cadmium stressed Lepidium sativum L. South African J Bot 129:341–346. https://doi.org/10.1016/j.sajb.2019.08.041
Khan N, Bano A (2019) Exopolysaccharide producing rhizobacteria and their impact on growth and drought tolerance of wheat grown under rainfed conditions. PLoS ONE 14:1–19. https://doi.org/10.1371/journal.pone.0222302
Khan S, Ibrar D, Bashir S et al (2022) Application of Moringa leaf extract as a seed priming agent enhances growth and physiological attributes of rice seedlings cultivated under water deficit regime. Plants 11:1–17. https://doi.org/10.3390/plants11030261
Klute A, Dirksen C (1986) Hydraulic conductivity and diffusivity: laboratory methods. Methods of soil analysis: part 1—physical and mineralogical methods. Soil Science Society of America, American Society of Agronomy, pp 687–734
Lachica M, Aguilar A, Yanez J (1973) Analisis foliar: Métodos Utilizados enla estaciln experimental del zaidin. An Edafol Agrobiol 32:1033–1047
Li T, Liu Y, Lin S et al (2019) Soil pollution management in China: a brief introduction. Sustain 11:1–15. https://doi.org/10.3390/su11030556
Martínez-ispizua E, Calatayud Á, Marsal JI et al (2022) Postharvest changes in the nutritional properties of commercial and traditional lettuce varieties in relation with overall visual quality. Agronomy 12:403. https://doi.org/10.3390/agronomy12020403
Martínez-ispizua E, Calatayud Á, Marsal JI et al (2022) The nutritional quality potential of microgreens, baby leaves, and adult lettuce: an underexploited nutraceutical source. Foods 11:423. https://doi.org/10.3390/foods11030423
Matos D, Sá C, Cardoso P et al (2019) The role of volatiles in Rhizobium tolerance to cadmium: effects of aldehydes and alcohols on growth and biochemical endpoints. Ecotoxicol Environ Saf 186:109759. https://doi.org/10.1016/j.ecoenv.2019.109759
Meng X, Liu S, Dong T et al (2020) Comparative transcriptome and proteome analysis of salt-tolerant and salt-sensitive sweet potato and overexpression of IbNAC7 confers salt tolerance in Arabidopsis. Front Plant Sci 11:1–15. https://doi.org/10.3389/fpls.2020.572540
Mukherjee S, Choudhuri MA (1983) Implications of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol Plant 58:166–170. https://doi.org/10.1111/j.1399-3054.1983.tb04162.x
Munir N, Hanif M, Abideen Z et al (2022) Mechanisms and strategies of plant microbiome interactions to mitigate abiotic stresses. Agronomy 12:1–33. https://doi.org/10.3390/agronomy12092069
Muthuraja R, Muthukumar T (2022) Co-inoculation of halotolerant potassium solubilizing Bacillus licheniformis and Aspergillus violaceofuscus improves tomato growth and potassium uptake in different soil types under salinity. Chemosphere 294:133718. https://doi.org/10.1016/j.chemosphere.2022.133718
Ozturk M, Turkyilmaz Unal B, García-Caparrós P et al (2021) Osmoregulation and its actions during the drought stress in plants. Physiol Plant 172:1321–1335. https://doi.org/10.1111/ppl.13297
Page AL, Miller RH, Keeney DR (1982) Methods of soil analysis Part 2. American Society of Agronomy Inc, Madison (WI), Chemical and microbiological properties
Patel M, Parida AK (2021) Chapter 3 - Role of hydrogen sulfide in alleviating oxidative stress in plants through induction of antioxidative defense mechanism, and modulations of physiological and biochemical components. In: Singh S, Singh VP, Tripathi DK, Prasad SM, Chauhan DK, Dubey NK (eds) Hydrogen sulfide in plant biology. Academic Press, pp 55–85. https://doi.org/10.1016/B978-0-323-85862-5.00006-3
Pervez K, Ullah F, Mehmood S, Khattak A (2017) Effect of Moringa oleifera Lam. leaf aqueous extract on growth attributes and cell wall bound phenolics accumulation in maize (Zea mays L.) under drought stress. Kuwait J Sci 44:110–118
Premachandra GS, Saneoka H, Ogata S (1990) Cell membrane stability, an indicator of drought tolerance, as affected by applied nitrogen in soyabean. J Agric Sci 115:. https://doi.org/10.1017/S0021859600073925
Rady MM, Mohamed GF (2015) Modulation of salt stress effects on the growth, physio-chemical attributes and yields of Phaseolus vulgaris L. plants by the combined application of salicylic acid and Moringa oleifera leaf extract. Sci Hortic (amsterdam) 193:105–113. https://doi.org/10.1016/j.scienta.2015.07.003
Rady MM, Boriek SHK, El-mageed TAA et al (2021) Exogenous gibberellic acid or dilute bee honey boosts drought stress tolerance in Vicia faba by rebalancing osmoprotectants, antioxidants, nutrients, and phytohormones. Plants 10(748):1–23. https://doi.org/10.3390/plants10040748
Rady MM, Alharby HF, Desoky EM et al (2023) Citrate-containing lemon juice, as an organic substitute for chemical citric acid, proactively improves photosynthesis, antioxidant capacity, and enzyme gene expression in cadmium-exposed Phaseolus vulgaris. South African J Bot 160:88–101. https://doi.org/10.1016/j.sajb.2023.07.004
Ramzan F, Younis A (2022) Chapter 20 - Use of biostimulants in tolerance of drought stress in agricultural crops, Editor(s): Tariq Aftab, M. Naeem, Emerging Plant Growth Regulators in Agriculture, Academic Press. 429–446
Rao MV, Paliyath G, Ormrod DP (1996) Ultraviolet-B- and ozone-induced biochemical changes in antioxidant enzymes of Arabidopsis thaliana. Plant Physiol 110:125–136. https://doi.org/10.1104/pp.110.1.125
Rashid N, Basra SMA, Shahbaz M et al (2018) Foliar applied moringa leaf extract induces terminal heat tolerance in quinoa. Int J Agric Biol 20:157–164. https://doi.org/10.17957/IJAB/15.0469
Rucińska-Sobkowiak R (2016) Water relations in plants subjected to heavy metal stresses. Acta Physiol Plant 38:. https://doi.org/10.1007/s11738-016-2277-5
Safwat SM, Matta ME (2021) Environmental applications of effective microorganisms: a review of current knowledge and recommendations for future directions. J Eng Appl Sci 68:1–12. https://doi.org/10.1186/s44147-021-00049-1
Sagar A, Rathore P, Ramteke PW, et al (2021) Plant growth promoting rhizobacteria, arbuscular mycorrhizal fungi and their synergistic interactions to counteract the negative effects of saline soil on agriculture: key macromolecules and mechanisms. Microorganisms 9:. https://doi.org/10.3390/microorganisms9071491
Sales J, Martins LL (2019) The effect of Cd stress in mineral nutrient uptake in plants. In: Hasanuzzaman M, Prasad MNV, Fujita M (eds) Cadmium toxicity and tolerance in plants. Elsevier Inc., pp 327–348
Selem E, Hassan AASA, Awad MF, et al (2022) Impact of exogenously sprayed antioxidants on physio-biochemical, agronomic, and quality parameters of potato in salt-affected soil. Plants 11:. https://doi.org/10.3390/plants11020210
Semida WM, Abdelkhalik A, Rady MOA et al (2020) Exogenously applied proline enhances growth and productivity of drought stressed onion by improving photosynthetic efficiency, water use efficiency and up-regulating osmoprotectants. Sci Hortic (amsterdam) 272:109580. https://doi.org/10.1016/j.scienta.2020.109580
Semida WM, Abd El-mageed TA, Abdelkhalik A et al (2021) Selenium modulates antioxidant activity, osmoprotectants, and photosynthetic efficiency of onion under saline soil conditions. Agronomy 11:855. https://doi.org/10.3390/agronomy11050855
Semida WM, Abdelkhalik A, Mohamed GF et al (2021) Foliar application of zinc oxide nanoparticles promotes drought stress tolerance in eggplant (Solanum melongena L.). Plants 10:421. https://doi.org/10.3390/plants10020421
Sitohy MZ, Desoky ESM, Osman A, Rady MM (2020) Pumpkin seed protein hydrolysate treatment alleviates salt stress effects on Phaseolus vulgaris by elevating antioxidant capacity and recovering ion homeostasis. Sci Hortic (amsterdam) 271:109495. https://doi.org/10.1016/j.scienta.2020.109495
Soares TFSN, da Silva AVC, Muniz EN (2021) Moringa leaf extract: a cost-effective and sustainable product to improve plant growth. South African J Bot 141:171–176. https://doi.org/10.1016/j.sajb.2021.04.007
Souid G, Sfar M, Timoumi R et al (2020) Protective effect assessment of Moringa oleifera against cadmium-induced toxicity in HCT116 and HEK293 cell lines. Environ Sci Pollut Res 27:23783–23792. https://doi.org/10.1007/s11356-020-08730-3
Talaat NB (2019) Effective microorganisms: an innovative tool for inducing common bean (Phaseolus vulgaris L.) salt-tolerance by regulating photosynthetic rate and endogenous phytohormones production. Sci Hortic (amsterdam) 250:254–265. https://doi.org/10.1016/j.scienta.2019.02.052
Talaat NB, Ghoniem AE, Abdelhamid MT, Shawky BT (2015) Effective microorganisms improve growth performance, alter nutrients acquisition and induce compatible solutes accumulation in common bean (Phaseolus vulgaris L.) plants subjected to salinity stress. Plant Growth Regul 75:281–295. https://doi.org/10.1007/s10725-014-9952-6
Treesubsuntorn C, Dhurakit P, Khaksar G, Thiravetyan P (2018) Effect of microorganisms on reducing cadmium uptake and toxicity in rice (Oryza sativa L.). Environ Sci Pollut Res 25:25690–25701. https://doi.org/10.1007/s11356-017-9058-6
Wu FB, Chen F, Wei K, Zhang GP (2004) Effect of cadmium on free amino acid, glutathione and ascorbic acid concentrations in two barley genotypes (Hordeum vulgare L.) differing in cadmium tolerance. Chemosphere 57:447–454. https://doi.org/10.1016/j.chemosphere.2004.06.042
Zaki HEM, Radwan KSA (2022) The use of osmoregulators and antioxidants to mitigate the adverse impacts of salinity stress in diploid and tetraploid potato genotypes (Solanum spp.). Chem Biol Technol Agric 9:1–21. https://doi.org/10.1186/s40538-022-00286-3
Zulfiqar F, Casadesús A, Brockman H, Munné-Bosch S (2020) An overview of plant-based natural biostimulants for sustainable horticulture with a particular focus on moringa leaf extracts. Plant Sci 295:110194. https://doi.org/10.1016/j.plantsci.2019.110194
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
All authors had the formulation of this research goal and designed the methodology. A. A., M. A. H. G., S. A. A., K. A. H., and T. A. A were responsible for conducting the research, data analysis, and writing the initial manuscript. N. M. A and A. S contributed to reviewing and revising the manuscript, and T. A. A was responsible for communication with the editorial office.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All authors were involved in the experiments and writing process.
Consent for publication
All authors of this article agreed to be published.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Gangrong Shi
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdelkhalik, A., Abdou, N.M., Gyushi, M.A.H. et al. Enhancing sweet potato (Ipomoea batatas) resilience grown in cadmium-contaminated saline soil: a synergistic approach using Moringa leaf extract and effective microorganisms application. Environ Sci Pollut Res 31, 32464–32479 (2024). https://doi.org/10.1007/s11356-024-33295-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-33295-w