Abstract
The interest of biofuel producers in Neotropical species that have high growth rates, slight wood density variability, and elemental composition that does not compromise the environment has increased in recent decades. We investigated the density and chemical characteristics of wood of Schizolobium parahyba var. amazonicum (Huber × Ducke) Barneby as a source for the generation of bioenergy. Apparent radial wood density profiles (X-ray densitometry (XRD)) and the elemental distribution (X-ray fluorescence (XRF)) of Cl, S, K, and Ca in the wood of nine S. parahyba var. amazonicum trees, divided into three diameter classes (I = 15.5, II = 19.5, and III = 23.5 cm) were analyzed. The high heating value (HHV) of the wood samples was determined, and the energy density was estimated by the product of the HHV and the apparent density. Trees that grew better (classes II and III) produced wood with higher density. These trees showed higher concentrations of K and S, and lower concentrations of Ca and Cl. The highest Cl concentrations were observed in classes with smaller diameters. The chlorine levels met the standards for use of this wood as fuel, but the sulfur levels were higher than the threshold recommended by the ISO 17225–3:2021 guidelines, which can limit the use of the species for certain energy uses. The wood of S. parahyba var. amazonicum had interesting characteristics for the production of bioenergy due to its low density, so it can be used in the production of solid biofuels such as pellets and briquettes. Monitoring chlorine and sulfur is important, since during the combustion of biomass they are released into the atmosphere and can negatively contribute to the effects of climate change.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forest biomass is a cheap and abundant source of energy (Yu et al. 2021), mainly in tropical regions. However, its use requires the classification of species according to their potential to produce biofuels through the evaluation of tree growth, physical properties, and chemical characteristics of wood (Gonçalves et al. 2018). The wood for bioenergy production must have characteristics such as high calorific value, high fixed carbon content, low ash content, and high density (Protásio et al. 2021).
The assessment of wood density is relevant for knowledge of the variability of energy density, which is important in the biofuels industry (Protásio et al. 2019). Basic density is usually one of the simplest parameters to ascertain the quality of wood (Zobel and van Buijtenen 2012; Protásio et al. 2021). However, the methods applied for its evaluation limit the detailed analysis of the heterogeneity of trunk wood density (Moreno-Fernández et al. 2018; Ortega Rodriguez and Tomazello-Filho 2019). In this sense, studies based on tree rings have the advantage of offering annual resolution and high sensitivity to analyze wood density (Roque and Tomazelo Filho 2015; Andrade et al. 2017; Quintilhan et al. 2021). One of the most traditional methods to analyze these wood density patterns is X-ray densitometry (Jacquin et al. 2017), whose main advantages are the simple preparation of samples and the fast data acquisition (Hervé et al. 2014), facilitating the analysis of wood density variability of tropical species (Roque and Tomazelo Filho 2015; Gaitan-Alvarez et al. 2019). In addition to density, the chemical composition of biomass is important to validate fuel use as a fuel, especially a low concentration of harmful elements.
Plant biomass with low concentrations of hazardous elements is therefore increasingly required by the biofuel industry (Stumm and Morgan 1970; ENplus 2015). In tropical regions, the diversity of species with varied levels of chemical elements (Oliveira et al. 1997; Amais et al. 2021; Ortega Rodriguez et al. 2022) can be a detrimental aspect for energy producers (Nurek et al. 2019). Some elements, such as Cl and S, released into the air during combustion result in toxic compounds such as organochlorines, sulfur oxides, furans, and PAHs, which are dangerous to both human health and the environment (Zhao et al. 2017; Dias Júnior et al. 2018). International standards regulate the use of both woody and non-woody materials as biomass for biofuel production (ENplus 2015; ISO 2021), especially the contents of hazardous compounds, such as chlorine and sulfur, which must be below 300 and 400 mg kg−1 BW, respectively (Mello 2001; Agraniotis et al. 2010; ENplus 2015). Moreover, the analysis of the main elements of physiological processes, such as K, which is involved in cambium activity (Fromm 2010), and Ca, involved in cell wall formation (Lautner and Fromm 2009), has aroused the interest of researchers investigating nutritional indicators of forestry treatments that favor tree growth (Hevia et al. 2018; Ortega Rodriguez et al. 2018). In tropical regions, such as Brazil, we can highlight species of the genera Eucalyptus and Pinus, more commonly employed to produce charcoal and pellets, respectively, which are widely used for bioenergy production in different sectors. Even so, other species are being studied to increase the variability of the renewable energy mix.
Fast-growing species with a cutting cycle of around 5 to 7 years, although structurally heterogeneous, are preferentially used as raw material due to their low density and easy densification for the production of clean and economical fuels that are calorically efficient and compact (e.g., pellets or briquettes) (Sette Jr et al. 2020). A tropical species that has outstanding qualities to supply this demand is Schizolobium parahyba var. amazonicum (Huber × Ducke) Barneby, commonly called “Paricá” (Ferreira et al. 2020). This species occurs naturally throughout the Neotropical Amazon region (Tourne et al. 2016; Schwartz et al. 2017). Furthermore, estimates point to the existence of 90,811 ha of plantations of the species in Brazil in 2018 (IBÁ 2020). S. parahyba var. amazonicum has stood out in forestry projects and agroforestry systems in the North, Midwest, and part of the Northeast regions of Brazil (Schwartz et al. 2017; Silva et al. 2020). Despite being found naturally in other countries in South and Central America, such as Bolivia, Colombia, Ecuador, Costa Rica, and Honduras (Barneby 1996; Turchetto-Zolet et al. 2012), S. parahyba var. amazonicum is only commercially planted in Brazil, due to its fast growth (between 20 and 30 m3 ha year−1), with trees that can reach between 15 and 40 m in height and 50 to 100 cm in diameter at breast height (Almeida et al. 2013; Vidaurre et al. 2018), with a straight and cylindrical shaft and good natural pruning (Barneby 1996; Almeida et al. 2013). Despite the information already available, studies that evaluate the energy potential of this species are essential, especially those that evaluate the chemical characteristics of the species that can influence its energy use.
Studying the distribution of elements such as Ca and K is important to verify growth patterns, wood quality for bioenergy and the effect of these elements on ash content and other important aspects of the fuel. On the other hand, studying the distribution of elements such as Cl and S is important to establish the use of these biomasses as fuels, since international regulations limit the content of these elements in solid biomass fuels (mainly pellets). High concentrations of Cl and S can result in greater amounts of hazardous elements released into the atmosphere, which can considerably reduce the air quality (Cui et al. 2018; Aas et al. 2019). The need to study these elements for the bioenergy area poses some challenges, especially involving the techniques used to analyze these materials.
Regarding the methods of elemental analysis of the plant biomass used as biofuel, most studies have used destructive techniques such as atomic absorption spectroscopy (AAS), flame emission spectrometry (FES), and/or inductively coupled plasma mass spectroscopy (ICP-MS) (Mendoza Martinez et al. 2019; Jagodzińska et al. 2021). These methods have low detection limits (Prohaska et al. 1998; Barrelet et al. 2006), thus being restricted to identifying inter- and intra-annual patterns of elemental variability in the radial profiles of trees, besides demanding laborious, costly, and time-consuming sample preparation (Balouet and Chalot 2015; Ortega Rodriguez et al. 2020). As an analytical alternative, XRF requires simple sample preparation (no digestion or combustion processes) and provides high spatial resolution (micrometers to millimeters). It also enables simultaneous evaluation of elements with atomic number > 11 (sodium), thus being fast, cheap, and environmentally friendly (Hevia et al. 2018; Ortega Rodriguez et al. 2018, 2020). This technique has shown promising results for assessing environmental and climate forest assets worldwide (Balouet et al. 2009; Hevia et al. 2018; Ortega Rodriguez et al. 2022). We hypothesized that nondestructive techniques (XRD and XRF) can be used as efficient tools to evaluate the quality of biomass for energy. To test this hypothesis, we evaluated the energetic potential of S. parahyba var. amazonicum individuals using these two techniques.
The aim of this study was thus to investigate the radial variability of density and elemental distribution, important aspects for the verification of new sustainable sources of clean energy, using X-ray densitometry and X-ray fluorescence microanalysis (µ-XRF), little explored for wood quality assessment for bioenergy use. In particular, we investigated the apparent density, energy density and distribution of the elements Cl, S, K and Ca in the wood of S. parahyba var. amazonicum as a potential source for the production of bioenergy.
Materials and methods
Study area and forest plantation
Wood samples were extracted from Schizolobium parahyba var. amazonicum (Huber × Ducke) Barneby trees in a commercial plantation covering 278 ha located in the municipalities of Paragominas and Ulianópolis (3° 21′ 40″ S, 47° 36′ 57″ W, 90 m a.s.l.) in the state of Pará, on the border with the state of Maranhão, in the Amazon region, Northern Brazil. According to the Köppen climate classification, the region has an Awi climate, tropical rainy, with an extended dry period between July and September. The predominant soil is yellow latosol. Average air temperatures range from 25.6 to 27.8 °C and annual precipitation is between 858 and 2788 mm, with an average of 1802 mm (Alvares et al. 2014).
The planting spacing was 4 × 4 m. Nine trees of S. parahyba var. amazonicum at 7 years of age were selected for cutting and separated according to the diameter at breast height (DBH, 1.30 m from the ground). The trees were classified into three diameter classes, representative of the population and free of phytosanitary problems. The variety of diameters was defined for each class according to the proportion of individuals of different diameters in the area (Table 1).
Sample collection and preparation
Cross Sects. (40 mm thick) were removed at breast height of the selected trees (Fig. 1), dried in the open air for 30 days and sanded (120–600 grains) to better visualize their transverse surface. Ten diametric sections of each tree, with four subsamples per radius, whose size varied according to the diameter of each individual, were cut from the discs for densitometry and XRF analyses, avoiding defects and reaction wood. Then, thin cross Sects. (1.7 mm thick) were obtained with the aid of a double circular saw. The material was conditioned in a climatic chamber at 20 °C and 60% relative humidity until reaching a stable moisture content of 12% (Quintilhan et al. 2021). The pith-bark radius of the cross section with the best surface finish was selected for density scanning using X-ray densitometry, and for determination of the chemical elements based on the µ-XRF technique.
For X-ray densitometry, the samples were scanned for wood density profile (pith-bark direction) with a collimated X-ray beam (80 μm step) in an X-ray densitometer (QTRS-01X, Quintek Measurement Systems), which allowed obtaining the mean apparent density (Ortega Rodriguez and Tomazello-Filho 2019).
The high heating value (HHV) of the wood samples was determined in an IKA C200® calorimeter, according to DIN 51,900–1 (DIN 2000). Based on these data, we calculated the net heating value (NHV) (Dias Júnior et al. 2021b). The energy density was estimated by the product of the NHV and the apparent density of the wood (Dias Júnior et al. 2021b), as determined by X-ray densitometry.
Elemental analysis
Linear scanning in the pith-bark transverse direction was carried out to analyze the temporal absorption and quantification of the inorganic elements Cl, S, K, and Ca, in the transect of the growth rings of the species. Analyses were performed with a µ-XRF spectrometer (Orbis PC EDAX, USA), with an Rh anode and a 30 mm2 drift silicon fluorescence detector (25 kV, 500 µA). The data were recorded to optimize detection of Cl, S, K, and Ca (100 s per point, with 32 points per line). An X-ray beam with 30 µm width and an Al filter with 25 µm vacuum condition (< 0.5 Torr) and dead time lower than 10% were used.
The intensity counts of Cl, S, K, and Ca were analyzed to distinguish the detected signal from the background with reasonable certainty for the analytical quantification process. A quantification threshold (Eq. 1) was used to assign an intensity threshold below which the elemental peak from quantification estimates were removed (EDAX Insight 2017). Also, the statistical intensity threshold value for each element was considered as its limit of quantification.
where NThreshold = statistical intensity threshold (cps); σ = sigma factor; set to 6 as default from the Orbis software; BG = background (cps); and s = time of each measurement point.
Thus, the elements with peak intensity above the applicable thresholds (S > 2.99, K > 4.47, Ca > 4.84, and Cl > 10.96) were quantified in “mg kg−1 DW” using the fundamental parameters method with a Shimadzu spectrometer (EDX-720) operating with a Rh (50W) and Si (Li) tube detector. The Cl and S values were compared to the maximum values allowed by ISO 17225–3:2021 (ISO 2021).
Data analysis
All data were normally distributed (Shapiro–Wilk test) with homogeneous variances (Levene test), thus satisfying statistical assumptions. After these assumptions were confirmed, the Pearson test was applied (t; p < 0.05) to identify possible correlations between the distribution patterns of the investigated elements. Analysis of variance was performed using the F-test, and when the interaction was significant, regression analysis was applied to verify trends as a function of the variables. Subsequently, the t-test was applied to individually analyze the equation coefficients. In the graphs, we present the average of the values and how the distribution of the values occurred by repetition, for each analyzed element. For the analysis of the apparent density of the wood, the Tukey test was applied (p < 0.05) to verify the differences between the means. In addition to the tests mentioned above, descriptive statistics of the data were calculated. All tests were conducted at 95% probability with the aid of the R Studio and SigmaPlot 13® software.
Results and discussion
Wood density and energy density
Wood density (WD) and energy density (ED) profiles for all diameter classes increased in the pith-bark direction (Fig. 2; Fig. S1).
Annual tree rings of S. amazonicum defined by differential X-ray attenuations (A) and radial energy density (ED) variability for each diametric classes (B). ED estimated based on wood density (WD) values obtained by X-ray densitometry. Average values of WD and ED for each diametric classes are also presented at the top left. Pairwise comparison (Tukey’s test): treatments with different letters (a and b) indicate statistically significant differences (p values < 0.05)
The S. parahyba var. amazonicum density profile was similar to that of other shade-tolerant Leguminosae tree species of the Amazon Forest (Woodcock and Shier 2002; Lehnebach et al. 2019). Although S. parahyba var. amazonicum is considered a fast-growing pioneer species due to its morphological and physiological characteristics (Lopes et al. 2015), its wood has a radial density trend similar to that of early-successional species (Woodcock and Shier 2002). The values of WD and ED increased in the first 2 cm near the pith, then remained stable, and increased again in the last 2 cm near the bark (Fig. 2; Fig. S1).
S. parahyba var. amazonicum showed distinct growth rings delimited by thickening of the fiber wall, flattening of radial wall and marginal parenchyma band (Fig. 2) (Marcati et al. 2008). Growth rings were characterized by stable-flatted values of density in the earlywood, followed by a progressive increment between the early- to latewood transition, related to thick-walled fiber formation. Then, the density values fell sharply at the end of the latewood, which was related to the formation of marginal parenchyma delimiting the growth rings, as in other tropical species with similar ring boundaries (Quintilhan et al. 2021; Ortega Rodriguez et al. 2022). Both anatomical and wood density characteristics of growth rings also make S. parahyba var. amazonicum a potential species for dendrochronological studies (Latorraca et al. 2015), and allow the annual monitoring of tree growth and quality of wood.
Table 2 shows significant differences of wood density among the diameter classes. Class II presented the highest average density values (apparent wood density, WD = 0.36 g cm−3; basic density, BD = 0.30 g cm−3). The pith region presented the lowest wood density (WD = 0.23 g cm−3; BD = 0.19 g cm−3), regardless of the diameter class. The xylem region between the pith and bark presented the most stable wood density values and the lowest standard deviation (< 0.08 g cm−3) regardless of the diameter classes. The region towards the bark had the highest wood density values (WD = 0.41, 0.47, 0.47 g cm−3 and BD = 0.34, 0.39, 0.39 g cm−3 for classes I, II, and III, respectively) and the highest standard deviation (> 0.11 g cm−3, regardless of the diameter class). The wood basic density of 7-year-old S. parahyba var. amazonicum trees, ~ 0.27 g cm−3 (estimated according to Silva et al. 2019) was similar to the values reported in plantations with trees of 5 to 11 years old in the same region, between 0.25 and 0.36 g cm−3 (Baptista Vidaurre et al. 2018; Melo et al. 2018). These are significantly lower values compared to other fast-growing species (BD = ~ 0.51 g cm−3) from upland (terra firme) forest stands in central Amazonia (Nogueira et al. 2005).
We also observed an increase in the standard deviation with increasing diameter classes, that is, the apparent WD was more heterogeneous in samples with larger diameters (Table 2). A more heterogeneous WD is expected in young fast-growing species, since competitive ecological processes result in an increase of intra-annual density fluctuations (IADFs) (Micco et al. 2016). IADFs are anatomical anomalies (such as alterations in the lumen size and cell-wall thickness of both vessels and fibers), caused by an abnormal xylogenesis process during the growing season, which results in gradual, abrupt, or indistinguishable ring boundaries (Venegas-González et al. 2015; Micco et al. 2016). WD can be considered one of the main characteristics for the selection of biomass for energy use, directly influencing other properties, such as ED.
ED represents the amount of energy present in a given volume of fuel. It is very relevant in the selection of compacted solid fuels or for the comparison of firewood (Dias Júnior et al. 2021b). Since the higher calorific value was practically constant in the samples, the energy density did not vary greatly. Based on this data, we calculated the NHV, which was equal to 15 (± 1) MJ kg−1 and used this data to calculate the ED. Figure 2 shows that the energy density followed the same behavior as the apparent density along the radial direction. This analysis is important to support decision-making on the appropriate time for felling the tree based on these parameters. It is desirable to obtain higher density values, since factors such as transport costs and power efficiency in energy systems are affected by this property (Protásio et al. 2020).
The data and methodology of the present study should be used to support decisions in sectors that need energy from biomass, especially rural producers who work in regions with low availability of other fast-growing species, such as Eucalyptus spp. For practical purposes, producers should choose materials that have higher energy density, so the analysis of this property, when carried out with adequate frequency, can help to improve energy production from biomass. In agroforestry systems that use S. parahyba var. amazonicum together with another species (Cordeiro et al. 2015), the evaluation of this property is even more important, enabling detailed monitoring during the growth of the trees, making it possible to better adjust the silvicultural treatments and the interaction between species to favor energy production. In the case of the wood evaluated in this work, we observed a positive correlation between diameter class and wood density. Therefore, in order to favor the energy density of wood, two factors stand out: (i) effective silvicultural treatments and growing conditions, to increase wood density (Moreno-Fernández et al. 2018; Rocha et al. 2020), and (ii) tree age, since the greater the age of the tree, the greater will be the natural tendency for higher basic wood density (Santos et al. 2021).
Distribution and quantification of chemical elements
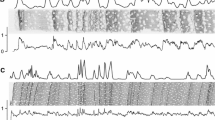
Ca showed a decreasing trend in the radial direction in all diameter classes, whereas K presented an opposite trend (Fig. 3). Similar radial distribution of Ca and K have been reported for different angiosperm and gymnosperm tree species worldwide (Hevia et al. 2018; Ortega Rodriguez et al. 2018, 2022; Alves et al. 2021). Cl and S showed unclear trends; both elements increased in the pith, between the second to third annual tree ring, and close to the bark (Fig. 3). Less mobile elements such as Ca decreased from the pith to bark, possibly related to the diminishing binding capacities of the youngest xylem formed by the cambium (Momoshima and Bondietti 1990; Prohaska et al. 1998). Increased mobile elements such as K, S, and Cl close to the bark, on the other hand, are attributed to the strong transport through the symplast in outer tree rings (Smith et al. 2014). The high-mobility elements occur from older to younger tissues, creating an age trend (Colin-Belgrand et al. 1996; Fairchild et al. 2009). Besides this, xylem sap’s contents of K, Ca, and Cl, which form other substances in the tree development phase, increase from trunk base to crown (Glavac et al. 1990).
Radial distribution of S, Cl, K, and Ca (mg kg−1 DW) in wood samples of S. amazonicum recorded by µ-XRF (net intensity), with the respective diameter classes: 15.5 (a), 19.5 (b), and 23.5 (c) cm. The dashed white line represents the investigated region (line scan). * The upper black numbers indicate the limits of the annual tree rings
Calcium, involved in cell wall expansion, is considered a less mobile element, fixed in the xylem cell walls, whereas K, involved in the cell differentiation, is a mobile element, easily translocated among different xylem cells (Smith et al. 2014; Scharnweber et al. 2016). Both elements are essential to cambial activity and wood formation (Fromm 2010). Our results showed that suppressed trees (with shorter diameters and less dense wood) contained higher amounts of Ca and lower amounts of K, possibly aiming to generate larger cells in compensation for lower xylem cell differentiation and formation (Figs. 2 and 4).
On the other hand, dominant trees with larger diameters and denser wood stored higher K contents and lower Ca contents, which probably contributed to greater differentiation and cell formation in compensation for lesser expansion of their cell walls. In a Pinus taeda L. plantation fertilized with pulp mill sludge, wider rings stored more Ca than K and denser rings, in turn, stored more K than Ca (Ortega Rodriguez et al. 2018). This may mean different strategies should be applied for the use of K and Ca in the formation of xylem tissues, depending on the type of species (gymnosperm or angiosperm) (Scharnweber et al. 2016). Cl, considered an essential micronutrient for plants (Robson and Pitman 1983), and S, stored in the primary cell wall, in which proteins (P-and S-bearing) define the cell architecture (Fairchild et al. 2009), are mobile elements for uptake and translocation within the tree (Baligar et al. 2001; Alves et al. 2021).
We also observed that dominant trees with larger diameters and denser wood stored higher S contents and lower Cl contents compared to suppressed trees (Figs. 2 and 4). For each element, the values of each repetition are presented as points on the graph, while above, on the bar graph, the average values are presented divided by diameter class.
Low concentrations of Cl are usually stable in woody tissues after fixation due to ionic binding (Balouet et al. 2012), whereas high concentrations of Cl (especially in high-saline environments) can precipitate with cations such as Na, Ca, and K (Alves et al. 2021). S forms stable structural entities within the wood, even with high supply of S (Fairchild et al. 2009), so its storage in xylem tissues functions as a source for foliage production (Barrelet et al. 2006).
The ionic binding of Cl and S with Ca and K can be seen in the significant positive relationship between these elements (Fig. 5). The fitted models of each equation are presented in Table 3. Fertilization based on K and Ca can play an important role in the kinetics of Cl and S absorption and in the precipitation of these elements in the growth rings of trees (Fahad et al. 2017). In soils with low natural availability of Cl and S, another possible source for trees is exposure to different pollutants such as SO2 and Cl2, among others released into the air, soil, and water in industrialized areas (Xiao et al. 2009; Zhao et al. 2017). The stability of woody S and Cl fixation implies that both elements should be reliable as indicators of environmental pollution (Fairchild et al. 2009; Balouet et al. 2012; Alves et al. 2021).
From the perspective of energy generation, K and Ca remain after combustion in the form of ash (Neina et al. 2020). In general, when analyzing the energy potential of biomass the ash content from mineral elements such as K and Ca negatively impacts the potential, since it reduces the HHV of the biomass (Dias Junior et al. 2020). Moreover, high ash contents can allow the ash to melt and crystallize, compromising the combustion equipment used (Dias Júnior et al. 2021b). The lowest Cl content was found in the class with the largest diameter, 23.5 cm (Fig. 4), whereas the smallest classes had the highest concentration. This high variability in the values of classes I and II is related to the distribution of the elements along the woody tissue of the studied individual, as seen in Fig. 4. The other elements investigated showed no significant differences among the diameter classes evaluated (Fig. S2).
For bioenergy production purposes, whether traditional (firewood, direct combustion) or modern techniques (pellets, briquettes), Cl concentrations were below the standard required by the European Union (EU) (ISO 2021) for the investigated diameter classes. A sulfur concentration above the permitted level for export to the EU (ISO 2021) was found in most of the samples of the studied diameter classes. When analyzing the diameter class relationship of S. parahyba var. amazonicum and levels of Cl and S, the main question is to determine when it is possible to obtain higher energy productivity (energy density) and lower concentrations of these elements that form of toxic compounds, such as organochlorines. Observing these aspects is relevant for food safety actions, since fuels can be used for cooking food. Burning biomass can also negatively impact occupational safety and environmental quality due to the release various substances, however without exposing all biotic organisms to dangerous levels of gaseous emissions.
Attention to the chlorine level has intensified in recent years since it forms organochlorine compounds, usually found in small amounts in nature. These originate from the contact of chlorine-based substances with compounds based on carbon chains (Varise et al. 2014). They are neurotoxic, highly lipophilic, chemically stable, and persistent in nature. They are toxic to some plants and insects and can be synthetically produced by the action of elemental chlorine on aromatic hydrocarbons (Baird 1998; Williams et al. 2000). Sulfur, when oxidized during biomass combustion, can generate sulfur dioxide (SO2) and other compounds that are classified as pollutants, in addition to causing damage to human health (Ren et al. 2021). Depending on the type of equipment used and the ventilation of the place, people who use these fuels for cooking can develop a number of health problems related to exposure to these compounds generated from incomplete combustion (Simoneit 2002). On a larger scale, the release of these compounds into the atmosphere can contribute to the greenhouse effect, resulting in aggravation of climate change (Ren et al. 2021).
The chemical characterization and monitoring of gaseous effluents make it possible to assess the feasibility of applying different plant biomasses for energy generation. In particular, this study sheds light on the use of S. parahyba var. amazonicum as a raw material for bioenergy generation, based on its high density and low concentrations of chlorine and sulfur. These results indicate the species has potential for use both in direct combustion and production of compact fuels, such as pellets and briquettes, which are widely used worldwide.
Practical applications and future perspectives
S. parahyba var. amazonicum is a species with high potential for the establishment of tropical forest restoration strategies that can generate financial returns in addition to environmental benefits (Schwartz et al. 2017). Based on our results, S. parahyba var. amazonicum is a fast-growing species with potential for the production of wood and energy byproducts in several systems, such as mixed ones that integrate restoration of forest systems with wood production (Dias Júnior et al. 2021a). Previous studies have already demonstrated the possibility of using juvenile wood (5 years old) from S. parahyba var. amazonicum for energy production (Vidaurre et al. 2012). This possibility of using wood from a short rotation cycle is interesting, indicating that S. parahyba var. amazonicum is an alternative to the species traditionally used for energy. Furthermore, planting this species for energy uses can reduce pressure on native tropical forest species, increase job generation and sustainable development in the region.
With the methodology used, we observed the density parameters and dynamics of the chemical elements of interest in trees with different diameter classes. These methods can be improved to become tools for producers of biomass for bioenergy, especially in industrial systems that demand increasingly faster, more economical, and environmentally friendly processes. Knowledge about the characteristics of biomass will allow choosing wood with a low concentration of pollutants, and consequently will generate less pollutants during burning, hence helping to achieve the Sustainable Development Goals (SDGs) (United Nations 2015), especially goals 7 (Affordable and clean energy), 12 (Responsible consumption and production), 13 (Climate action), and 15 (Life on land). This characterization is even more important in countries like Brazil, whose National Energy Plan (Empresa de Pesquisa Energética 2020) aims to increase the participation of biomass in the national energy mix. For this to be carried out in a responsible and sustainable manner, the characterization and control of gaseous emissions from biomass burning are extremely important.
Although our study presents some innovative advances for the characterization of S. parahyba var. amazonicum biomass for energy, other gaps still need to be addressed. We suggest future studies to further investigate the correlation between the elements present in the wood and elements present in the gaseous effluents during the burning of these fuels. Understanding this correlation would allow better adjustments during the biomass production stage, which might result in the reduction of gaseous pollutants released during combustion. In addition, studies that evaluate the species’ genetic characteristics and possible strategies to improve energy production are also suggested, together with studies that verify the yield of biomass per area, taking into account the density and volume of each individual in the plantation.
Conclusion
In the wood of S. parahyba var. amazonicum, the chlorine levels meet the standards for use of this material as fuel. Due to the low density and good NHV (15 MJ kg−1), this wood can also be used for the production of solid biofuels, such as pellets and briquettes. However, the sulfur levels found are higher than recommended, so this is a limiting factor for energy uses. The combustion of materials rich in sulfur can intensify atmospheric pollution. Nondestructive techniques based on X-ray spectrometry proved to have potential to characterize physical and chemical lignocellulosic materials for energy purposes. In addition, they helped to identify the chemical components present in these materials that, when released during burning, can cause environmental and health problems.
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Aas W, Mortier A, Bowersox V, Cherian R, Faluvegi G, Fagerli H, Hand J, Klimont Z, Galy-Lacaux C, Lehmann CMB et al (2019) Global and regional trends of atmospheric sulfur. Sci Rep 9:953. https://doi.org/10.1038/s41598-018-37304-0
Agraniotis M, Nikolopoulos N, Nikolopoulos A, Grammelis P, Kakaras E (2010) Numerical investigation of solid recovered fuels’ co-firing with brown coal in large scale boilers – evaluation of different co-combustion modes. Fuel 89:3693–3709. https://doi.org/10.1016/j.fuel.2010.07.030
Alvares CA, Stape JL, Sentelhas PC, de MoraesGonçalves JL, Sparovek G (2014) Köppen’s climate classification map for Brazil. Meteorol Zeitschrift 22:711–728. https://doi.org/10.1127/0941-2948/2013/0507
Alves EEN, Ortega Rodriguez DR, de Rocha PA, Vergütz L, Santini Junior L, Hesterberg D, Pessenda LCR, Tomazello-Filho M, da Costa LM (2021) Synchrotron-based X-ray microscopy for assessing elements distribution and speciation in mangrove tree-rings. Results Chem 3:100121. https://doi.org/10.1016/j.rechem.2021.100121
Amais RS, Moreau PS, Francischini DS, Magnusson R, Locosselli GM, Godoy-veiga M, Ortega Rodriguez DR, Tomazello-filho M, Arruda MAZ (2021) Trace elements distribution in tropical tree rings through high-resolution imaging using LA-ICP-MS analysis. J Trace Elem Med Biol 68:126872. https://doi.org/10.1016/j.jtemb.2021.126872
Baird C (1998) Environmental Chemistry. New York
Baligar VC, Fageria NK, He ZL, Baligar VC, Fageria NK, He ZL (2001) Nutrient use efficiency in plants. Commun Soil Sci Plant Anal 32:921–950. https://doi.org/10.1081/CSS-100104098
Balouet C, Chalot M (2015) Sample preparation for trace element analysis
Balouet JC, Smith KT, Vroblesky D, Oudijk G (2009) Use of dendrochronology and dendrochemistry in environmental forensics: does it meet the daubert criteria? Environ Forensics 10:268–276. https://doi.org/10.1080/15275920903347545
Balouet JC, Burken JG, Karg F, Vroblesky D, Smith KT, Grudd H, Rindby A, Beaujard F, Chalot M (2012) Dendrochemistry of multiple releases of chlorinated solvents at a former industrial site. Environ Sci Technol 46:9541–9547. https://doi.org/10.1021/es300318v
Baptista Vidaurre G, Vital BR, De A, Oliveira C, Tarcísio J, Oliveira DS, Moulin JC, Missia JG, Silva D, Soranso DR (2018) Physical and mechanical properties of juvenile of Schizolobium amazonicum wood. Rev Árvore 42
Barneby RC (1996) Neotropical Fabales at NY: asides and oversights. Brittonia 48:174. https://doi.org/10.2307/2807811
Barrelet T, Ulrich A, Rennenberg H, Krähenbühl U (2006) Seasonal profiles of sulphur, phosphorus, and potassium in Norway spruce wood. Plant Biol 8:462–469. https://doi.org/10.1055/s-2006-924044
Colin-Belgrand M, Ranger J, Bouchon J (1996) Internal nutrient translocation in chestnut tree stemwood: III. Dynamics across an age series of Castanea sativa (Miller). Ann Bot 78:729–740. https://doi.org/10.1006/anbo.1996.0183
Cordeiro IMCC, de Barros PLC, Lameira OA, GazelFilho AB (2015) Assessment of paricá (Schizolobium parahyba var.amazonicum (Huber ex Ducke) Barneby) plantations at different ages and cultivation systems in aurora do pará (Pará state-Brazil). Ciência Florest 25:679–687. https://doi.org/10.5902/1980509819618
Cui J, Duan L, Zhou L, Zhao C (2018) Effects of air pollution control devices on the chlorine emission from 410 t/h circulating fluidized bed boilers co-firing petroleum coke and coal. Energy Fuels 32:4410–4416. https://doi.org/10.1021/acs.energyfuels.7b03106
de Almeida DH, de Scaliante RM, de Macedo LB, Macêdo AN, Dias AA, Christoforo AL, Calil Junior C (2013) Caracterização completa da madeira da espécie amazônica Paricá (Schizolobium amazonicum Herb) em peças de dimensões estruturais. Rev Árvore 37:1175–1181. https://doi.org/10.1590/S0100-67622013000600019
de Andrade ES, Garcia S dos SC, Albernaz ALKM, Tomazello Fillho M, Moutinho VHP (2017) Growth ring analysis of Euxylophora paraensis through x-ray microdensitometry. Ciência Rural 47. https://doi.org/10.1590/0103-8478cr20150895
da Ferreira VRS, Cademartori PHG de, Lima EA De, Ferraz FA, Aguiar OJR de, Silva DA Da (2020) Produção e avaliação de briquetes de Schizolobium parahyba var. amazonicum (Huber ex Ducke) Barneby. Sci For 48. https://doi.org/10.18671/scifor.v48n128.12
de Latorraca JVF, Souza MT, Silva LDSAB, Ramos LMA (2015) Dendrocronologia de árvores de Schizolobium Parahyba (Vell.) S. F. Blake de ocorrência na Rebio de Tinguá-RJ. Rev Árvore 39:385–394. https://doi.org/10.1590/0100-67622015000200018
de Protásio TP, Scatolino MV, de Araújo ACC, de Oliveira AFCF, de Figueiredo ICR, de Assis MR, Trugilho PF (2019) Assessing proximate composition, extractive concentration, and lignin quality to determine appropriate parameters for selection of superior eucalyptus firewood. BioEnergy Res 12:626–641. https://doi.org/10.1007/s12155-019-10004-x
de Protásio TP, Scatolino MV, Lima MDR, de Araújo ACC, de Figueiredo ICR, Bufalino L, Hein PRG, Trugilho PF (2020) Insights in quantitative indexes for better grouping and classification of Eucalyptus clones used in combustion and energy cogeneration processes in Brazil. Biomass Bioenergy 143:105835. https://doi.org/10.1016/j.biombioe.2020.105835
de Protásio TP, Roque Lima MD, Scatolino MV, Silva AB, Rodrigues de Figueiredo IC, Gherardi Hein PR, Trugilho PF (2021) Charcoal productivity and quality parameters for reliable classification of Eucalyptus clones from Brazilian energy forests. Renew Energy 164:34–45. https://doi.org/10.1016/j.renene.2020.09.057
Dias Júnior AF, Andrade CR, Brito JO, Lira SP, Andrade AM de, Souza ND de (2018) Polycyclic aromatic hydrocarbons in the organic phase extracted from charcoal for barbecue. Rev Árvore 41. https://doi.org/10.1590/1806-9088201700050001
Dias Junior AF, Esteves RP, da Silva ÁM, Sousa Júnior AD, Oliveira MP, Brito JO, Napoli A, Braga BM (2020) Investigating the pyrolysis temperature to define the use of charcoal. Eur J Wood Wood Prod 78:193–204. https://doi.org/10.1007/s00107-019-01489-6
Dias Júnior AF, Pincelli ALMS, Câmara da Silva AP, da Silva ÁM, de Souza ND, TommasielloFilho M, Brito JO (2021a) Integrating species and successional classes for wood production in a mixed forest restoration system in a neotropical region. J for Res. https://doi.org/10.1007/s11676-021-01297-w
Dias Júnior AF, Suuchi MA, Sant’AnnaNeto A, da Silva JGM, da Silva ÁM, de Souza ND, Protásio T de P, Brito JO, (2021b) Blends of charcoal fines and wood improve the combustibility and quality of the solid biofuels. BioEnergy Res 14:344–354. https://doi.org/10.1007/s12155-020-10179-8
DIN DI für N (2000) DIN 51900–1 - Determining the gross calorific value of solid and liquid fuels using the bomb calorimeter, and calculation of net calorific value. 1–16
EDAX Insight (2017) Spectral background fitting and peak noise statistics
Empresa de Pesquisa Energética (2020) National Energy Plan (PNE) 2050. Brasília, Brazil
ENplus (2015) Quality certification scheme for wood pellets. European Pellet Council (EPC)
Fahad S, Bajwa AA, Nazir U, Anjum SA, Farooq A, Zohaib A, Sadia S, Nasim W, Adkins S, Saud S et al (2017) Crop production under drought and heat stress: plant responses and management options. Front Plant Sci 8:1147. https://doi.org/10.3389/fpls.2017.01147
Fairchild IJ, Loader NJ, Wynn PM, Frisia S, Thomas PA, Lageard JGA, De Momi A, Hartland A, Borsato A, La Porta N et al (2009) Sulfur fixation in wood mapped by synchrotron X-ray studies: Implications for environmental archives. Environ Sci Technol 43:1310–1315. https://doi.org/10.1021/es8029297
Fromm J (2010) Wood formation of trees in relation to potassium and calcium nutrition. Tree Physiol 30:1140–1147. https://doi.org/10.1093/treephys/tpq024
Gaitan-Alvarez J, Moya R, Berrocal A (2019) The use of X-ray densitometry to evaluate the wood density profile of Tectona grandis trees growing in fast-growth plantations. Dendrochronologia 55:71–79. https://doi.org/10.1016/j.dendro.2019.04.004
Glavac V, Koenies H, Ebben U (1990) Seasonal variation of calcium, magnesium, potassium, and manganese contents in xylem sap of beech (Fagus sylvatica L.) in a 35-year-old limestone beech forest stand. Trees-Structure Funct 4:75–80
Gonçalves AC, Malico I, Sousa AMO (2018) Solid biomass from forest trees to energy: a review. Renew Resour Biorefineries. https://doi.org/10.5772/INTECHOPEN.79303
Hervé V, Mothe F, Freyburger C, Gelhaye E, Frey-Klett P (2014) Density mapping of decaying wood using X-ray computed tomography. Int Biodeterior Biodegradation 86:358–363. https://doi.org/10.1016/j.ibiod.2013.10.009
Hevia A, Sánchez-Salguero R, Camarero JJ, Buras A, Sangüesa-Barreda G, Galván JD, Gutiérrez E (2018) Towards a better understanding of long-term wood-chemistry variations in old-growth forests: a case study on ancient Pinus uncinata trees from the Pyrenees. Sci Total Environ 625:220–232. https://doi.org/10.1016/j.scitotenv.2017.12.229
IBÁ IB de Á (2020) Relatório Anual IBÁ 2019
ISO (2021) ISO 17225–3:2021 - Solid biofuels - fuel specifications and classes - part 3: graded wood briquettes. 7
Jacquin P, Longuetaud F, Leban J-M, Mothe F (2017) X-ray microdensitometry of wood: a review of existing principles and devices. Dendrochronologia 42:42–50. https://doi.org/10.1016/j.dendro.2017.01.004
Jagodzińska K, Garcia Lopez C, Yang W, Jönsson PG, Pretz T, Raulf K (2021) Characterisation of excavated landfill waste fractions to evaluate the energy recovery potential using Py-GC/MS and ICP techniques. Resour Conserv Recycl 168:105446. https://doi.org/10.1016/j.resconrec.2021.105446
Lautner S, Fromm J (2009) Calcium-dependent physiological processes in trees. Plant Biol 12:268–274. https://doi.org/10.1111/j.1438-8677.2009.00281.x
Lehnebach R, Bossu J, Va S, Morel H, Amusant N, Nicolini E, Beauchêne J (2019) Wood density variations of legume trees in French Guiana along the shade tolerance continuum: Heartwood effects on radial patterns and gradients. Forests 10:80. https://doi.org/10.3390/f10020080
Lopes MJ, Dias-Filho MB, Menezes Neto MA, Moreira Dos Santos JU, Cruz ED, Dias DSDS, H, (2015) Morphological and physiological responses to shade in seedlings of Parkia gigantocarpa Ducke and Schizolobium parahyba var. amazonicum (Huber ex Ducke) Barneby (Leguminosae). Sci for Sci 43:573–580
Marcati CR, Milanez CRD, Machado SR (2008) Seasonal development of secondary xylem and phloem in Schizolobium parahyba (Vell.) Blake (Leguminosae: Caesalpinioideae). Trees - Struct Funct 22:3–12. https://doi.org/10.1007/s00468-007-0173-8
Mello W (2001) Precipitation chemistry in the coast of the Metropolitan Region of Rio de Janeiro, Brazil. Environ Pollut 114:235–242. https://doi.org/10.1016/S0269-7491(00)00209-8
Melo LEDL, de Silva CJ, de Protásio TP, da Mota GS, Santos IS, Urbinati CV, Trugilho PF, Mori FA (2018) Planting density effect on some properties of Schizolobium parahyba wood. Maderas Cienc y Tecnol 20:381–394. https://doi.org/10.4067/S0718-221X2018005003901
Mendoza Martinez CL, Sermyaginade Cassia Oliveira Carneiro A, Vakkilainen E, Cardoso M E (2019) Production and characterization of coffee-pine wood residue briquettes as an alternative fuel for local firing systems in Brazil. Biomass and Bioenergy 123:70–77. https://doi.org/10.1016/j.biombioe.2019.02.013
Micco V, Campelo F, De Luis M, Bräuning A, Grabner M, Battipaglia G, Cherubini P (2016) Intra-annual density fluctuations in tree rings: how, when, where, and why? IAWA J 37:232–259. https://doi.org/10.1163/22941932-20160132
Momoshima N, Bondietti EA (1990) Cation binding in wood: applications to under- standing historical changes in divalent cation availability to red spruce. Can J for Res 20:1840–1849
Moreno-Fernández D, Hevia A, Majada J, Cañellas I (2018) Do common silvicultural treatments affect wood density of Mediterranean montane pines ? Forests 9:80. https://doi.org/10.3390/f9020080
Neina D, Faust S, Joergensen RG (2020) Characterization of charcoal and firewood ash for use in African peri-urban agriculture. Chem Biol Technol Agric 7:5. https://doi.org/10.1186/s40538-019-0171-2
Nogueira EM, Nelson BW, Fearnside PM (2005) Wood density in dense forest in central Amazonia, Brazil. For Ecol Manage 208:261–286. https://doi.org/10.1016/j.foreco.2004.12.007
Nurek T, Gendek A, Roman K, Dąbrowska M (2019) The effect of temperature and moisture on the chosen parameters of briquettes made of shredded logging residues. Biomass Bioenergy 130:105368. https://doi.org/10.1016/j.biombioe.2019.105368
Oliveira H, Fernandes EAN, Haselberger N, Markowicz A, Ferraz ESB (1997) A brazilian tree collection analyzed by X-ray fluorescence. In: International workshop on biomonitoring of atmospheric pollution (with emphasis on trace elements) - BioMAP. Lisbon, pp 136–142
Ortega Rodriguez DR, Tomazello-Filho M (2019) Clues to wood quality and production from analyzing ring width and density variabilities of fertilized Pinus taeda trees. New for 50:821–843. https://doi.org/10.1007/s11056-018-09702-9
Ortega Rodriguez DR, de Carvalho HWP, Tomazello-Filho M (2018) Nutrient concentrations of 17- year-old Pinus taeda annual tree-rings analyzed by X-ray fluorescence microanalysis. Dendrochronologia 52:67–79. https://doi.org/10.1016/j.dendro.2018.09.009
Ortega Rodriguez DR, de Almeida E, Tomazello-Filho M, Pereira de Carvalho HW (2020) Space-resolved determination of the mineral nutrient content in tree-rings by X-ray fluorescence. Sci Total Environ 708:134537. https://doi.org/10.1016/j.scitotenv.2019.134537
Ortega Rodriguez DR, Hevia A, Sánchez-Salguero R, Santini L, de Carvalho HWP, Roig FA, Tomazello-Filho M (2022) Exploring wood anatomy, density and chemistry profiles to understand the tree-ring formation in Amazonian tree species. Dendrochronologia 71:125915. https://doi.org/10.1016/J.DENDRO.2021.125915
Prohaska T, Stadlbauer C, Wimmer R, Stingeder G, Latkoczy C, Hoffmann E, Stephanowitz H (1998) Investigation of element variability in tree rings of young Norway spruce by laser-ablation-ICPMS. Sci Total Environ 219:29–39. https://doi.org/10.1016/S0048-9697(98)00224-1
Quintilhan MT, Santini L, Ortega Rodriguez DR, Guillemot J, Cesilio GHM, Chambi-Legoas R, Nouvellon Y, Tomazello-Filho M (2021) Growth-ring boundaries of tropical tree species: aiding delimitation by long histological sections and wood density profiles. Dendrochronologia 69:125878. https://doi.org/10.1016/J.DENDRO.2021.125878
Ren Y, Shen G, Shen H, Zhong Q, Xu H, Meng W, Zhang W, Yu X, Yun X, Luo Z et al (2021) Contributions of biomass burning to global and regional SO2 emissions. Atmos Res 260:105709. https://doi.org/10.1016/j.atmosres.2021.105709
Robson AD, Pitman MG (1983) Interactions between nutrients in higher plants. In: Läuchli A, Bieleski RL (eds) Inorganic plant nutrition. Encyclopedia of Plant Physiology (New Series), vol 15., 1st edn. Springer, Berlin, Heidelberg, pp 147–180
Rocha SMG, Vidaurre GB, Pezzopane JEM, Almeida MNF, Carneiro RL, Campoe OC, Scolforo HF, Alvares CA, Neves JCL, Xavier AC et al (2020) Influence of climatic variations on production, biomass and density of wood in eucalyptus clones of different species. For Ecol Manage 473:118290. https://doi.org/10.1016/j.foreco.2020.118290
Roque RM, TomazeloFilho M (2015) Relationships between anatomical features and intra-ring wood density profiles in Gmelina arborea applying x-ray densitometry. Cerne 13:384–392
Santos LMH, de Almeida MNF, da Silva JGM, Vidaurre GB, Hein PRG, da Silva GF, Zanuncio AJV, FragaFilho CV, Campinhos EN, Mafia RG et al (2021) Variations in heartwood formation and wood density as a function of age and plant spacing in a fast-growing eucalyptus plantation. Holzforschung 75:979–988. https://doi.org/10.1515/hf-2020-0215
Scharnweber T, Hevia A, Buras A, van der Maaten E, Wilmking M (2016) Common trends in elements? Within- and between-tree variations of wood-chemistry measured by X-ray fluorescence — a dendrochemical study. Sci Total Environ 566–567:1245–1253. https://doi.org/10.1016/j.scitotenv.2016.05.182
Schwartz G, Pereira PCG, Siviero MA, Pereira JF, Ruschel AR, Yared JAG (2017) Enrichment planting in logging gaps with Schizolobium parahyba var. amazonicum (Huber ex Ducke) Barneby: A financially profitable alternative for degraded tropical forests in the Amazon. For Ecol Manage 390:166–172. https://doi.org/10.1016/j.foreco.2017.01.031
Sette CR Jr, da Cunha TQG, Coneglian A, Hansted ALS, da Silva DA, Lima PAF, da Silva MF, Yamaji FM (2020) Does the presence of bark in the wood of fast-growing forest species significantly change the energy potential? BioEnergy Res 13:222–228. https://doi.org/10.1007/s12155-020-10115-w
Silva CBR, dos Santos Junior JA, Araújo AJC, Sales A, Siviero MA, Andrade FWC, Castro JP, de FigueiredoLatorraca JV, de Lima Melo LE (2020) Properties of juvenile wood of Schizolobium parahyba var. amazonicum (paricá) under different cropping systems. Agrofor Syst 94:583–595. https://doi.org/10.1007/s10457-019-00422-3
Silva RT, Sette Junior CR, Ferreira A, Chagas MP, Filho MT (2019) Wood and briquette density under the effect of fertilizers and water regimes. Floresta e Ambient 26. https://doi.org/10.1590/2179-8087.047116
Simoneit BR (2002) Biomass burning — a review of organic tracers for smoke from incomplete combustion. Appl Geochemistry 17:129–162. https://doi.org/10.1016/S0883-2927(01)00061-0
Smith KT, Balouet JC, Shortle WC, Chalot M, Beaujard F, Grudd H, Vroblesky DA, Burken JG (2014) Dendrochemical patterns of calcium, zinc, and potassium related to internal factors detected by energy dispersive X-ray fluorescence (EDXRF). Chemosphere 95:58–62. https://doi.org/10.1016/j.chemosphere.2013.08.017
Stumm W, Morgan JJ (1970) Aquatic chemistry an introduction emphasizing chemical equilibria in natural waters. Wiley-Interscience, New York
Tourne DCM, Martorano LG, Brienza Junior S, dos Dias CTS, Lisboa LS, Sartorio SD, Vettorazzi CA (2016) Potential topoclimatic zones as support for forest plantation in the Amazon: Advances and challenges to growing paricá (Schizolobium amazonicum). Environ Dev 18:26–35. https://doi.org/10.1016/j.envdev.2015.11.002
Turchetto-Zolet AC, Cruz F, Vendramin GG, Simon MF, Salgueiro F, Margis-Pinheiro M, Margis R (2012) Large-scale phylogeography of the disjunct Neotropical tree species Schizolobium parahyba (Fabaceae-Caesalpinioideae). Mol Phylogenet Evol 65:174–182. https://doi.org/10.1016/j.ympev.2012.06.012
United Nations (2015) Agenda of Sustainable Development Goals 2030. https://sdgs.un.org/goals. Accessed 17 Sep 2021
Varise TG, Estrela C, Guedes DFC, Sousa-Neto MD, Pécora JD (2014) Detection of organochlorine compounds formed during the contact of sodium hypochlorite with dentin and dental pulp. Braz Dent J 25:109–116. https://doi.org/10.1590/0103-6440201302404
Venegas-González A, von Arx G, Chagas MP, Filho MT (2015) Plasticity in xylem anatomical traits of two tropical species in response to intra-seasonal climate variability. Trees - Struct Funct 29:423–435. https://doi.org/10.1007/s00468-014-1121-z
Vidaurre GB, Carneiro ACO, Vital BR, dos Santos RC, Valle MLA (2012) Energy properties of the wood and coal of paricá (Schizolobium amazonicum) tree. Rev Arvore 36:365–371. https://doi.org/10.1590/S0100-67622012000200018
Vidaurre GB, Vital BR, Colodette JL, Oliveira A de C, Silva JGM da, Moulin JC, Soranso DR (2018) Anatomical and chemical properties of juvenile Schizolobium amazonicum wood. Rev Árvore 42. https://doi.org/10.1590/1806-90882018000300001
Vieilledent G, Fischer FJ, Chave J, Guibal D, Langbour P, Gérard J (2018) New formula and conversion factor to compute basic wood density of tree species using a global wood technology database. Am J Bot 105:1653–1661. https://doi.org/10.1002/ajb2.1175
Williams PL, James RC, Roberts S (2000) Principles of toxicology: environmental and industrial applications, 2nd edn. John Wiley & Sons, New York
Woodcock D, Shier A (2002) Wood specific gravity and its radial variations: the many ways to make a tree. Trees - Struct Funct 16:437–443. https://doi.org/10.1007/s00468-002-0173-7
Xiao HY, Tang CG, Xiao HW, Liu XY, Liu CQ (2009) Identifying the change in atmospheric sulfur sources in China using isotopic ratios in mosses. J Geophys Res Atmos 114. https://doi.org/10.1029/2009JD012034
Yu Q, Wang Y, Van Le Q, Yang H, Hosseinzadeh-Bandbafha H, Yang Y, Sonne C, Tabatabaei M, Lam SS, Peng W (2021) An overview on the conversion of forest biomass into bioenergy. Front Energy Res 9:348. https://doi.org/10.3389/FENRG.2021.684234/BIBTEX
Zhao H, Yang W, Wu F, Tan B (2017) Mixed forest plantations can efficiently filter rainfall deposits of sulfur and chlorine in Western China. Sci Rep 7:1–12. https://doi.org/10.1038/srep41680
Zobel BJ, van Buijtenen JP (2012) Wood variation: its causes and control, 2nd edn. Springer Science & Business Media, New York
Acknowledgements
We thank the research group for Bioenergy and Forest-Based Bioproducts and the Biomass Energy Laboratory of Federal University of Espírito Santo (LEB/UFES) in Brazil for support in carrying out the various analyses.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES) – Finance Code 001, and Fundação de Amparo à Pesquisa e Inovação do Espírito Santo (FAPES) (CAPES/FAPES collaboration). This study is also supported by the Wood Anatomy and Tree-Ring Laboratory (LAIM) (FAPESP; grant umber 2009/53951–7) of the Department of Forest Sciences of Luiz de Queiroz College of Agriculture (ESALQ) and the Laboratory for Nuclear Instrumentation (LIN) (FAPESP projects: 2015/05942–0 and 2015/19121–8), Center for Nuclear Energy in Agriculture (CENA), University of Sao Paulo (USP). Daigard Ricardo Ortega Rodriguez is supported by FAPESP through research fellowship, grant number 2018/22914–8.
Author information
Authors and Affiliations
Contributions
Ananias Francisco Dias Júnior, Mario Tomazello-Filho, and José Otávio Brito contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by Daigard Ricardo Ortega Rodriguez and Analder Sant’anna Neto. The first draft of the manuscript was written by Ananias Francisco Dias Júnior and João Gilberto Meza Ucella-Filho, and all authors commented on previous versions of the manuscript. The final review of the manuscript was conducted by Elias Costa de Souza, João Gilberto Meza Ucella-Filho, João Gabriel Missia da Silva, Analder Sant’anna Neto, Ana Paula Câmara da Silva, and Lourdes Maria Hilgert Santos. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ta Yeong Wu
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ucella-Filho, J.G.M., Dias Júnior, A.F., de Souza, E.C. et al. Clues about wood density and trace-element variability of Schizolobium parahyba var. amazonicum (Huber ex Ducke) Barneby for bioenergy use. Environ Sci Pollut Res 30, 56161–56173 (2023). https://doi.org/10.1007/s11356-023-26343-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-26343-4