Abstract
The food and feeding links and sources in an impacted tropical estuary situated along India’s western coast, the Ulhas River Estuary (URE) was analyzed employing the stable carbon and nitrogen isotopic signatures (δ13C and δ15N). Three basal carbon sources, such as mangrove leaves, particulate organic matter (phytoplankton), and detritus, were analyzed together with eight consumer groups from various trophic guilds. The δ13C varied from − 19.67 to − 24.61‰, whereas δ15N ranged from 6.31 to 15.39‰ from the primary consumer to the top predator species. The stable isotope mixing model developed for URE revealed a phytoplankton based pelagic food chain and detritus based benthic food chain in URE. The fairly larger value of SEA (Standard Ellipse Area) in the URE suggest a much broader food web structure and high trophic diversity in the ecosystem. Higher influence of detritus on the assimilated diet of majority of consumers and evidences of nitrogen enrichment in the basal sources such as detritus and particulate organic matter by anthropogenic activities in URE point towards nitrogen pollution and subsequent trophic disturbance in this tropical estuarine ecosystem.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Estuaries are considered one of the most productive ecosystems, compared to rivers and the marine ecosystems that influence them from either side (Pritchard 1967). They are important transition zones, where matter and energy from terrestrial origin intermix with that from aquatic origin. Estuarine food webs generally rely on a high level of primary production and detritus pathways in association with adjacent ecosystems like salt marshes, mangroves, and mud flats (Duarte and Cebrián 1996). They are characterized by a high degree of complexity that suffers well marked spatio-temporal variability in ecological conditions as well as biodiversity (Simenstad and Wissmar 1985; Pasquaud et al. 2008). Therefore, these ecosystems primarily support populations of aquatic species with specific ecological functions such as residence and shelter, migration channels, and feeding and breeding grounds for juvenile fish species (Cardoso et al. 2004; Elliott et al. 2007). Food webs are recognized as key components in ecology and have been structured by complex species interactions, which govern the flow of organic matter in natural systems (Summerhayes and Elton 1923). Many research works have underlined the uniqueness of the estuarine food web structure, which is more complex and dynamic than other aquatic ecosystems. Firstly, due to the high degree of intra annual fluctuations in fish diversity as a result of seasonal marine and freshwater emigrants and the subsequent community changes (Amara et al. 2000; Maes et al. 2004; Selleslagh et al. 2009; Sreekanth et al. 2019) and secondly, the monsoon mediated inflow of allochthonous inputs and the raise in primary productivity in particular seasons (Simenstad and Wissmar 1985; Pasquaud et al. 2008). These events will be more pronounced in the case of tropical estuaries, which are under the influence of monsoon (Sreekanth et al. 2019).
Characterization of food web functioning is still challenging due to the complexity in precisely measuring species interactions within a food web. Novel tracing methods that estimate species diet uptake and trophic positions are therefore required to determine food web dynamics. The application of stable isotope analysis to characterize the food webs has improved the understanding of sources and niche properties along with the quality of trophic level (TL) estimations, especially for the aquatic ecosystems, which are less amenable to the traditional tracking methods (Boecklen et al. 2011). Despite the direct methods like, visual or molecular measures of analyzing fecal or gut content analyses (Pompanon et al. 2012) that provide the snapshot of the prey ingested at a particular time, this technique provides direct estimates of the prey assimilated and a longer-term integration of the feeding habits of the consumer species (e.g. Kwak and Zedler 1997; Riera et al. 1999; Vinagre et al. 2008, 2011). The data on stable carbon isotopic ratio (δ13C) can provide important clues on the sources of primary production that forms the basis of the food web. The basal sources often differ in their δ13C values based on the photosynthetic pathway followed and also, enrichment in δ13C is much smaller between diet and consumer. Thus, δ13C can be used to trace prey–consumer connections in the food chains (Fry and Sherr 1989; Post 2002). The nitrogen isotopic ratio (δ15N) enriches more or less by 3.4‰ (±1‰) from prey to the predator (Peterson and Fry 1987; Post 2002). Therefore, δ15N has been increasingly used as a marker of trophic position and have been used for the estimation of the TL of many species and the establishment of food web structure for various ecosystems (e.g. Kwak and Zedler 1997; Le Loc’h et al. 2008; Pasquaud et al. 2010; Vinagre et al. 2011, 2012; Potapov et al. 2019). Since the last two decades, several researchers employed stable isotopic ratios for the estimation of TL and niche attributes in the estuarine and coastal ecosystems (Akin and Winemiller 2008; Lavoie et al. 2010; Le Loc’h et al. 2008; Vinagre et al. 2012; Filgueira and Castro 2011; Kwak and Zedler 1997; Pasquaud et al. 2010; Bird et al. 2018; Golikov et al. 2019; Barton et al. 2019).
The ecological or trophic structure studies from India that used stable isotopic signatures are meager and mainly focused on large oceanic predators, viz., yellowfin tuna and swordfish in the western Indian Ocean (Ménard et al. 2007), on elasmobranchs caught off Gujarat (Borrell et al. 2011), sharks from the south-western Indian Ocean (Kiszka et al. 2015), on mesopelagic communities over shallow seamounts of the south-western Indian Ocean (Annasawmy et al. 2020) etc. The stable isotopic studies in the estuarine ecosystems in India are also limited to a few, including a study on the benthic invertebrates in an estuarine mangrove ecosystem in Andhra Pradesh (Bouillon et al. 2002), and a study to characterize the sources and fate of organic matter in the Pichavaram estuarine-mangrove ecosystem, east coast of India (Prasad and Ramanatha 2009).
Stable isotope mixing models (SIMMs) have increasingly been used to determine the relative contributions of several sources to a mixture in ecological and geochemical research over the last several decades (Schwarcz 1991; Phillips and Gregg 2001). As a consumer’s tissues are ultimately derived from the dietary sources they consume, it is possible to use SIMMs to derive the assimilated diet of an individual, or a group of individuals, given the isotopic ratios of the consumers’ tissues and food sources (Phillips 2012). The earliest attempts in applying a mixing model framework were restricted to systems involving a single consumer, later Phillips and Gregg (2001) through their IsoError model, provided propagation of error calculations to establish confidence intervals around the estimates based on variances of the consumer and source isotopic measurements. Subsequently, the method was expanded in the IsoSource (Phillips and Gregg 2003) that allow for multiple sources but without explicit incorporation of source and consumer variability. The refining of the models has continued to progress through years with increasing capabilities and sophistication by various works. Recently, Bayesian mixing models have been developed that allow flexible model specification in a rigorous Bayesian statistical framework to incorporate some or all of these features such as uncertainties, concentration dependence, larger numbers of sources as well as others. These models include MixSIR (Moore and Semmens 2008; Ward et al. 2010), SIAR (Parnell et al. 2010, 2013), MixSIAR (Stock and Semmens 2013), IsotopeR (Hopkins and Ferguson 2012), FRUITS (Fernandes et al. 2014), and SIMMR (stable isotope mixing model with “R” package) (Parnell and Inger 2019) etc. SIMMR is designed as an upgrade to the SIAR package by Parnell et al. (2013). This new version contains a slightly more sophisticated mixing model, a simpler user interface, and more advanced plotting features.
Several researchers have employed stable isotope mixing models for illustrating the trophic structure of estuarine coastal ecosystems worldwide. The most important studies in this regard were undertaken by Abrantes and Sheaves, (2010) in Tropical North Queensland, Australia; Giarrizzo et al. (2011) in Curuçá estuary, northern Brazil; Vaslet et al. (2012) in mangrove ecosystems of Florida and Belize of the Tropical western Atlantic region; Shahraki et al. (2014) in Qeshm Island, Persian Gulf, Northern Indian Ocean, Sepu´lveda-Lozada et al. (2015) in Southern Gulf of Mexico; Contreras et al. (2018) in Central Coast of Colombia of the Tropical Eastern Pacific; Nelson et al. (2019) and Dillon et al.(2021) in the Northern Gulf of Mexico. However, there are no attempts so far has been reported form the Indian coastal waters and estuaries. Here we report one of the pioneer attempts in trophic structure study in a tropical estuarine ecosystem situated along the western coast of India, the Ulhas River Estuary (URE) using stable isotopic signatures. The present work aims to (1) characterize the food web structure of URE, based on the nodal links of the estuarine food web, by employing stable carbon and nitrogen isotopic signatures with the analytical tool of SIMMR; and (2) to elucidate how the isotopic trophic positions of selected fish species vary from the dietary trophic position and the reported estimates from the adjacent coastal waters.
Methods
Study area
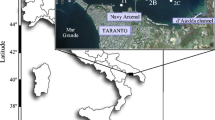
The URE (URE) is one of the transitional ecosystems situated along Mumbai, (Maharashtra) along the western coast of India. The estuary connects the Ulhas River to the Arabian Sea through Vasai creek (Fig. 1). It is a macrotidal and well-mixed estuary, which shows well marked seasonal variations in the salinity attributed by the monsoon mediated freshwater influx (Rathod et al. 2003). The estuary is also characterized by considerable land runoff that carries huge amount of sediments from its catchment area (Rathod 2005). The ecosystem is experiencing declined productivity due to heavy discharge from urban and industrial activities along the banks. Heavy pollution due to anthropogenic activities from the surrounding areas and the resultant impact on the water quality has also been reported from URE (Nikam et al. 2008; Rathod and Patil 2009). The industries situated along banks of the river acts as point sources of pollution in the Ulhas River and these pollutants are ultimately reaching the estuarine part (Mohapatra and Rengarajan 2000; Mishra 2002; Lad and Patil 2016). Sewage load from Thane city is also reported to be affecting the aquatic life and ecosystem health (Lala 2004). Moreover, the mangrove cover destruction in a greater extent along the banks due to urbanization has also adversely impacted the coastal ecosystems along the Mumbai waters in the last decades (Jagtap et al. 1994; Kantahrajan et al. 2018). Besides, Kumawat et al. (2015) reported that the fishery in the estuary is also in the wedge of damage due to the by-catch and discards from unsustainable gear operations.
Experimental fishing
Preliminary information of the seasonal patterns in the fish assemblage structure in the estuary was ascertained by experimental fishing trips conducted at three stations in the true estuarine zone (with annual average salinity range of 7.6±5.5 ppt to 33.5±0.70 ppt) between September 2017 and August 2018. Monthly sampling was carried out for an entire year in order to ensure the coverage of complete seasonal patterns in species diversity including all resident and the seasonal migrants in the estuary. Single day Dol net (stationary conical bag net) with a cod end mesh size of 10 mm was used throughout the sampling. The “dolnets” are long conical shaped bag nets with wide rectangular mouth and narrow cod-end. The length of the nets was 30 m, comprising four different segments of webbing. The mouth opening length and width were measured as 10 m and 4 m, respectively. Two wooden cylindrical spikes of 2–4 m length and 15 cm diameter driven to the bottom were used to fix the net. Two fishermen and a canoe are generally required to operate a net. For the experimental fishing, the dolnets were set against the tidal flow, moored at spikes fixed at identified stations, and operated according to the tidal pattern. The two corners of the lower end of the mouth of the dolnet were attached to the spikes, whereas the upper two corners of the mouth were tied to floats made of polystyrene to keep the mouth open. The operation was performed during the day time for a period of 3–4 h.
Un-sorted subsamples of fish catch (with catch to Subsample ratio-5:1) were brought to laboratory and care was taken to include all the fish species caught to be well represented in the sub-samples. In the laboratory, finfishes as well as shellfishes were identified up to species level based on the relevant taxonomic keys (Fischer and Bianchi 1984; Bianchi 1985). Mean trophic level of all the species were obtained from the FishBase database (Froese and Pauly 2016) as well as the information from the published literature (Vivekanandan et al. 2009). These data sources provided trophic level values for 105 species, which included 81 species of finfishes, 21 species of crustaceans, and 3 species of cephalopods. Based on the mean trophic level values, the entire species were categorized to form feeding mode trophic guilds as per the criteria followed by Vivekanandan et al. (2009), as herbivores and detritivores (HRDR—trophic level (TL): 2–2.5), omnivores (OM-TL: 2.51–3), mid-level carnivores (MLCR-TL: 3.01–3.5), high level Carnivores (HLCR–TL: 3.51–4), and top predators (TPR- >4.01) (Supplementary Table 1).
Sample collection for stable isotope analysis
The actual sampling for the stable isotope study was carried out in January 2019. The Winter season was chosen for the sampling because of two reasons. Firstly, the sediments brought by the river and terrestrial discharge remain in suspension during monsoon (July to September) and start settling down with the onset of the winter season (December to February) in the creeks and bays of the western coast of India (Rajawat et al. 2015). Secondly, along the west coast, sea level peaks during December and continue to prevail even in the subsequent months making the seasonal cycle more symmetric (Shankar 2000). Also, since the detritus pathways highly influence estuarine food webs (Mann 1998), January (winter season) is chosen as an ideal month, which is relatively stable period for the ecosystem as the impact of marine and riverine inputs in estuarine sediment sources are balanced.
Carbon sources of the estuarine food web were assumed to be the particulate organic matter (phytoplankton), detritus, and mangrove leaves and are collected during the sampling. Samples of green leaves of the mangroves of similar size and condition were collected by hand from the mangrove patches lining the estuary along the sampling stations. Detritus was taken along the mangrove system, sampling the greatest area possible. Particulate organic matter (phytoplankton) was obtained using a plankton net with a mesh size of 23 μm. Zooplankton and fish larvae samples were collected using a bolting silk plankton net of 0.02 mm mesh size from the sampling sites of the estuarine system at high tide. Additionally, sediment sample was also collected with a PVC tube (of 9 cm in diameter × 50 cm in length) at a depth of 5 cm from the fish sampling sites.
The most abundant fish species in the annual catch (pooled for 12 months) were assessed using the Relative Importance Index (RII) (De la Cruz-Aguero 1994), which represents the expression of importance of Individual species in the ecosystem estimated from the abundance values (% number of individuals, % catch weight and % frequency of occurrence of species in catch). Adult individuals of the most abundant species (species with maximum RII in the annual catches) from each trophic guild was chosen as the nodal consumer species in the food web and considered for the isotopic study and to demonstrate the trophic structure of the URE ecosystem. The representative species are chosen because of the high degree of biodiversity as well as complexity of the ecosystem with dynamic assemblage patterns with respect to seasons and considering the difficulty in analyzing the stable Isotope signatures of all the available species in all the seasons. in the study area. As per the recent estimate, about 105 species of finfishes and shell fishes has been reported from URE (Lal et al. 2021). Two species are chosen from the MLCR trophic guild due to their equally higher RII values. The species selected for the study were Nematalosa nasus (HRDR), Acetes indicus (OM), Johnius belangerii (MLCR), Escualosa thoracata (MLCR), Nemapteryx caelata (HLCR), and Lepturacanthus savala (TPR) (Table 1).
At least four samples of both basal resources and consumer species of greater size and similar dimensions were used to avoid ontogenic variability. For small species like A. indicus, more samples were used to achieve the required sample weight for the stable isotope analysis. All the samples were carefully washed in the field and preserved in ice for their transportation to the laboratory, where they were stored in a vertical freezer at −21°C until further processing.
Sample preparation
The preparation of samples for the stable isotope analysis followed the protocol given by Contreras et al. (2018). Samples were carefully washed and thawed before processing. The dorsal white muscle of the finfishes were dissected out and used for the analysis due to the relatively higher consistency of δ13C and δ15N in the part compared to other tissues (Abrantes and Sheaves 2010). For the shellfish, A. indicus, abdominal segments were only used to analyze isotopic signatures (Contreras et al. 2018). The innervenation of mangrove leaves was removed with scissors and fine blades. Water samples containing particulate organic matter were filtered using pre-combusted glass fiber filters (GC5047 mm, pore size: 0.5 μ) to extract the solid part. All samples, including detritus and sediment, were dried at 60 °C for a minimum of 48 hrs in a hot air oven and later homogenized to form fine powder using a standard mortar and pestle and sieved through a 60 µ mesh screen to ensure homogeneity. From the powdered sample, lipids are extracted using methanol-chloroform (2:1 by volume), as lipids are depleted in δ13C compared with the whole organism’s tissue lipid content (Peterson and Fry 1987; Kling et al. 1992). For the estimation of carbon isotopic signature and total organic carbon (TOC) of the sediment and detritus samples, acid pre-treatment was carried out to remove the carbonates by centrifuging the dried and powdered samples with 5% HCl in 2000–3000 rpm for 5–10 min. The procedure was repeated about 4–5 times until the acid carbonate reaction stopped, followed by distilled water wash in the centrifuge until the sample turned neutral, which was checked using a pH paper. The supernatant was discarded each time, and the sample was dried again at ~50 °C. After drying, the samples were recovered and converted it into fine powder. From the homogenized sample, one milligram (mg) of tissue (both finfish and shellfish) samples and 10 mg of sediment samples were weighed using tin capsules in a microbalance.
Stable isotope analysis
The stable carbon and nitrogen isotopic ratios (δ13C and δ15N) were analyzed using an Isotope Ratio Mass Spectrometer (IRMS): MAT 253 (Thermo Finnegan, Germany). Stable carbon and nitrogen isotopic ratios were expressed in the delta notation (δ13C and δ15N) relative to the standard reference materials (V-PDB (Pee Dee Belemnite) for δ13C and atmospheric N2 for δ15N) as:
where “X” is 13C or 15N, and “R” is the ratio of heavy to light isotopes, wherein “δX” expressed in ‰ (“Permil”).
From the stable Carbon isotopic ratios, the sources and links of the estuarine food web were predicted. From the Nitrogen isotopic ratios, the trophic positions and the food chain lengths were determined. The δ15N values alone cannot be considered to represent the trophic position, since the δ15N of primary producers (defined as organisms that convert inorganic N to organic N) are highly variable among systems (Kling et al. 1992; Cabana and Rasmussen 1996) and within systems through time (Toda and Wada 1990; Cabana and Rasmussen 1996). Hence, it is necessary to measure the isotopic signatures of each fish relative to an ecosystem-specific baseline δ15N signature (Le Loc’h et al. 2008; Post 2002; Vander Zanden and Vadeboncoeur 2002). According to Vander Zanden et al. (1997), the species to be selected as the baseline should be relatively large and long-lived primary consumer species, which integrate temporal variability in primary producer’s δ15N, thereby representing the average baseline δ15N signature. In this study, the fish trophic positions were interpreted relative to that of Gizzard shad, N. nasus, which has been chosen to be the reference organism as per the criteria explained above.
The trophic position (isotopic) for each species was calculated using the formula given by Post (2002):
Where 3.4 represents a 1.0 trophic level increment in δ15N and λ is the trophic position of the organism used to estimate δ15N base.
Comparison of dietary and isotopic trophic levels
Out of the 6 species chosen from each trophic levels, the dietary trophic positions were calculated for Johnius belangerii (MLC), Nemapteryx caelata (HLC), and Lepturacanthus savala (TPR) based on the gut content analysis. For the rest of the species, the dietary trophic level was obtained from the published literature from Indian coastal waters (Vivekanandan et al. 2009). Calculation of fish trophic position required estimation of the trophic position of prey organisms. In the present study, the simplest possible assumptions together with the reported estimates were used. Primary producers and detritus were assigned a TL of 1.00, and for zooplankton 2.00, and so on.
Dietary TL was calculated using the following equation (Christensen and Pauly 1992):
Where “TLi” is the trophic level of species ‘"i", “DCij” is the proportion of prey species “j” in the diet of species "i’, and “TLj” is the trophic level of prey species “j”. Trophic levels for prey items used in dietary calculations of trophic position are obtained from literature, as shown in Table 2. The trophic level estimated from the stable isotopic ratios was compared with the dietary estimates and the published reports. The food chain length (FCL) was also enumerated, which is defined as the trophic position of the top predator estimated as the species or taxon with the highest N value.
Stable isotope mixing model with SIMMR
Stable isotope mixing models are used to quantify the proportional contributions of various sources to a mixture (Parnel et al. 2013). Isotopic values are determined for the tissues of a consumer and its food sources, and similarity in isotopic composition between the consumer’s tissues, and its food sources (after adjustment for isotopic sorting or fractionation during digestion, metabolism, and assimilation) gives an index of the relative importance of each item in the consumer’s diet. In the present study, a Bayesian mixing model employing two isotopes (δ13C and δ15N) has been developed that allows the estimation of contributions of three sources such as detritus, particulate organic matter, and mangroves to the assimilated diet of eight consumers. Other carbon sources like sediments with similar isotopic values to detritus were excluded from the analysis. The stable isotope mixing model, depicting the food web structure of URE, was developed with SIMMR-V: 0.4.2 (stable isotope mixing model with “R” package) (Parnell and Inger 2019). For the ease of running the model, the consumer species were represented as Group 1: ZP, Group 2: FL, Group 3: NN, Group 4: AI, Group 5: ET, Group6: JB, Group 7: NC, and Group 8: LS.
This three-source mixing model is based on the fundamental linear mass balance equations (Phillips and Koch 2002):
where the subscripts “x,” “y,” “z,” and “M” represent three food sources and the mixture (i.e., the consumer), respectively; “f” represents the fractional contribution of “C” or “N” from each food source to the consumer’s diet, and ∆13C tissue–x is the trophic fractionation (change in δ13C during assimilation) between food source “x” and the consumer’s tissue. “f” represents the proportion of C from a food source and not necessarily the proportion of the biomass. This standard linear mixing model is a system of three equations in three unknowns (fx, fy, and fZ), which can be solved to determine each food source’s contribution to the diet. The present mixing model for two isotopes and three sources that do incorporate concentration variation in the sources for carbon and nitrogen, which has been enumerated separately using the Isotope Ratio Mass Spectrometer (IRMS): MAT 253 (Thermo Finnegan, Germany) before the analysis. Prior to running the mixing model, δ13C of the consumers has to be corrected (δ13Ccorr) for Trophic Enrichment Factor (TEF) and TL, using the following equation (Giarrizzo et al. 2011):
A standard TEF of 0.4±1.3‰ for δ13C and 3.4±1.0‰ for δ15N was used, as suggested by Post (2002). The present study analyses the source contributions in the assimilated diet of 8 different consumer species, considering the advantage that SIMMR can handle these data sets, provided they all share the same sources and concentration dependence values. The model has also enabled a comparison between these groups according to the relative contribution of various sources.
Estimation of isotopic niche
The ecological niche concept can provide insights into a wide variety of ecological and evolutionary problems (Martı ́-nez del Rio et al. 2009). In the stable isotopic niche analysis, convex hull area (TA) describes the niche width of the organism (Quevedo et al. 2009) or community in question (Layman and Post 2008), which is used under two distinct situations, firstly, to describe individual components of the community and secondly applied to entire communities of different species or functional groups (e.g. Layman and Post 2008; Donázar-Aramendía et al. 2019). One of the shortcomings identified herein is that application of the metrics to single community group members is sensitive to sample size, particularly across the range of sizes frequently encountered in ecological studies (n< 50). Multivariate ellipse-based metrics are more suitable than Convex Hull methods when applied to single community members, which is unbiased with respect to sample size, allows robust comparison to be made among data sets comprising different sample sizes (Jackson et al. 2011). The total convex hull area (TA) and the standard ellipse area (SEAc) were also calculated for the URE community (c indicates that the SEA was corrected for a small sample size) using the SIBER package in R.
Results
Isotopic signatures of selected species
Isotopic carbon signatures of sources varied between −26.20‰ in mangrove leaves to −20.40‰ in phytoplankton, whereas in the consumers, the values ranged between −24.38‰ in A. indicus to −19.87‰ in zooplankton (Table 3). The isotopic nitrogen signatures reflected in the consumers’ trophic level and showed substantial variation with respect to the trophic guilds considered. The ratio ranged from 6.71‰ in the primary consumer species to 15.07‰ in the top predator (Table 3). The values for sediment δ13C and δ15N were obtained as −25.3‰ and 8.70‰, respectively.
Isotopic niche
The iso-space plot is divided into four quadrants to display the species distribution in the isotopic niche space (Fig. 2). The species that had enriched δ13C and enriched δ15N were distributed on quadrant (A); depleted δ13C and enriched δ15N in quadrant (B); depleted δ13C and depleted δ15N in quadrant (C), and enriched δ13C and depleted δ15N in quadrant (D). There were notable differences among the δ13C- δ15N niches of the dominant estuarine species, represented in different trophic guilds. The species, L. savala was found at the top of the niche map, occupied in the quadrant (A) with the enriched δ13C and maximum δ15N value corresponding to the highest trophic level among the species. The groups like fish larvae, N. nasus, and E. thoracata were also included in the quadrant (A), with relatively enriched δ15N and δ13C. The species such as A. indicus, J. belangerii, and N. caelata occupied quadrant (B), indicating their utilization of depleted carbon sources compared to the other species. Concomitantly, the source detritus has also occupied in the quadrant (B) along with these species, confirming the higher utilization of more depleted carbon source, i.e. detritus. Mangrove leaves were grouped in quadrant (C) as expected, with the lowest δ15N, and the lowest trophic level with potentially depleted δ13C sources. Phytoplankton and zooplankton were observed along quadrant (D) of the niche map constructed for URE.
On the other hand, the iso-space plot of mean δ13C versus δ15N values (± SD) of basal resources and consumers from URE ecosystem indicated the dependence over the phytoplankton sources by the groups ZP, FL, NN, and ET as indicated by their closer positions occupied in the isotopic space with respect to the δ13C of POM. The sources, mangrove leaves and detritus showed close δ13C isotopic values, indicating that the detritus component has a high dependency over mangrove leaves, which might generally be sourced from the decomposition of mangrove leaves in the ecosystem. The trophic structure of URE, measured with the standard ellipse area corrected for a small sample size (SEAc) was 11.08, and the total area (TA) was 24.2. The fairly larger values of SEA in the URE suggest a much broader food web structure and high trophic diversity in the ecosystem (Fig. 3).
Stable isotope mixing model with SIMMR
The summary outputs of the isotope mixing model generated for URE based on three sources and eight consumer species (representing trophic guilds), are shown in Table 4. The SIMMR output table represents the mean values and the credible intervals (2.5 to 97.50%) containing the proportions of each sources in the consumer species (combined) analyzed in the study. In the Bayesian model, the credible interval is just the range containing a particular percentage of probable values. The results suggest that the dietary proportions for this model are quite certain. However, the credible interval for mangrove leaves was the narrowest, running from approximately 1 to 13% to the overall contribution of the estuarine fish diet. The density plot of source contributions (Fig. 4) has also indicated that all the three sources chosen in the present study are fairly separated from one another.
Source contribution to consumers
The multiple consumer-source isotopic values could be combined in SIMMR based on “multi-group data” function which yield the tracers plot representing the positioning of the consumers on the iso-space (Parnell and Inger 2019). The outputs from SIMMR depicted the contribution of various sources to consumers from different trophic guilds. Among the three sources analyzed in this study, detritus contributed maximum to the assimilated diet in the fish fauna of URE, followed by phytoplankton (Fig. 5). However, the diet sourced from mangrove leaves contributed the least to the food web. The sources analyzed in the study, such as phytoplankton, detritus, and mangrove leaves together contributed only around 30% of the assimilated diet of the consumers combined, indicating the possibility of missing food sources in the present study. When the consumers were analyzed separately for their assimilated diet contributions, the primary trophic guild such as Group 1 (zooplankton) (Fig. 6A), Group 2 (fish larvae) (Fig. 6B), and Group 3 (N. nasus) (Fig. 6C) were found to utilize the localized food sources in the estuary. The sources evaluated in this study, such as detritus, phytoplankton, and mangrove leaves together, contribute about more than 70% of their assimilated diet. For all these three primary consumer groups, source phytoplankton showed maximum contribution to the assimilated diet.
The source contribution plots of A. indicus indicated that its assimilated diet is mainly sourced from detritus and mangrove leaves (Fig. 6D). However, all three sources together contributed only about 35% of the diet components, indicate the possibilities of utilizing allochthonous dietary sources. Phytoplankton was found to be the major diet source for E. thoracata. Where the local sources considered in the study together accounted for nearly 30% of the assimilated diet (Fig. 6E). While, the isotopic source contribution plot of J. belangerii (Fig. 6F) and N. caelata (Fig. 6G) showed similar trends, dominated by detritus and mangrove leaves that contributed about 50% of their assimilated diet. In addition, the phytoplankton source also showed a minor influence on the diet of J. belangerii and N. caelata.
The source contribution plot for the top predator species, L. savala, indicated that only over 30% of its assimilated diet is accounted by the local sources, in which phytoplankton based sourced were the major contributors (Fig. 6H). The summarized results of the stable isotope mixing model is provided at Fig. 7. The entire trophic space is divided into 3 isotopic trophic zones with (a) higher phytoplankton influence, (b) mangrove-detritus influence, and (c) mixed influence. Five out of the eight consumer species chosen in the present study occupied in the mixed influence zone. Within the mixed influence zone, the species L. Savala and E thoracata showed more phytoplankton influence and A. indicus, J. belangerii, and N caelata showed more detritus influence.
Comparison of dietary and isotopic trophic levels
Among the six species chosen from each trophic guild based on the highest RII for the stable isotopic ratio analysis, the dietary trophic positions were calculated for three species such as, J. belangerii (MLCR), N. caelata (HLCR), and L. savala (TPR) for comparison. The dietary data summarized by calculating the average diet percentage (percentage of stomach volume ± 1SD) for every three species (Table 5). A. indicus, E. thoracata, and S. indicus were found to be the major diet components for all three species. A clear affinity towards the zoobenthic prey types such as polychaetes was evident for N. caelata, compared to the other two species. The dietary TL values obtained were 3.35 ± 0.77, 3.97 ± 0.49, and 4.27 ± 0.19 for J. belangerii, N. caelata, and L. savala, respectively. The trophic levels of fish species corresponding to different trophic guilds based on isotopic signatures varied between 2.00 in N. nasus to 3.75 in L. savala (Table 6). While comparing the TL estimates of these species between the diet data, stable isotope data and the available literature, the species J. belangerii showed the highest consistency between the three estimates of trophic positions (TL=3.35, 3.34, and 3.3 respectively). However, the dietary TL values for N. caelata and L. savala were more or less close to the earlier records than the isotopic TL values. The comparison of the isotopic and earlier estimates of the trophic positions revealed the relationship such that: δ15N trophic position = 0.67 × reported trophic position + 0.27; R2 = 0.67) (Fig. 8). Trophic position of fishes using isotope ratios demonstrated variations from the generally reported estimates for the majority of species; however, a few were more or less close to the estimates. The species such as A. indicus was the major outlier, where the isotopic TL showed significantly higher value than the published reports. At the same time, the predatory carnivorous species such as L. savala and N. caelata showed lower isotopic TL values compared to the literature estimates.
Discussion
Food webs are recognized as the functional components of an ecosystem and have been structured by complex species interactions (Summerhayes and Elton 1923). Characterization of food web functioning is still challenging due to the complexity in precisely measuring species interactions within a food web. Novel tracing methods that estimate species diet uptake and trophic positions are therefore required to determine food web features. The application of stable isotope analysis in the characterization of food webs has improved the understanding of sources and niche properties along with the quality of trophic level (TL) estimations, especially for the aquatic ecosystems, which are less amenable to the traditional tracing methods (Boecklen et al. 2011).
Source variations
The wide range of δ13C values indicates the diverse use of carbon sources in the estuarine habitat (Bouillon et al. 2008). In riverine ecosystems, the existence of diverse carbon sources is reported to be the result of size, the degree of seasonal fluxes, and fluvial dynamics (Caraco et al. 2010; Babler et al. 2011). Similar effects are also expected to affect the source variations in the estuarine systems. Stable isotope compositions of particulate organic matter (POM) have been used as a powerful tool to understand the sources and cycling of carbon in aquatic ecosystems (Popp et al. 1989, 1997, 1998; Bidigare et al. 1997; Rau et al. 1996). Particulate organic matter of various types, including those generated from forests, primary production in rivers, and human activity, is supplied to coastal waters via rivers. However, the nature and role of river-supplied POM in coastal waters is not universal, as the source and course characteristics shows wide range of regional variability (Komatsu et al. 2019). Since there are more than 10,000 small rivers originating from monsoon-dominated and/or mountainous regions globally, it must be appreciated that their total organic carbon contribution to the coastal system must be substantial (Shynu et al. 2015).
A study from the Mandovi-Zuari Estuaries by Shynu et al. (2015) indicated the dominance of terrestrial plant-derived organic matter and terrestrial soil-derived organic matter respectively, in the Mandovi and Zuari estuaries during wet season. It is estimated that each river contributed at least ~20% terrestrial organic carbon (TOC) to the coastal system during wet season and received similar quantity of TOC during dry season. Samanta et al. (2015) reported that δ13C values in the Ganga (Hooghly) River estuary are a result of the combined influence of seawater-river water mixing, chemical weathering, biological production, precipitation or dissolution of minerals such as calcite, and respiration and degradation of organic matter, which may be supplied via both natural as well as anthropogenic processes.
As per Kelly et al. (1998) and Coffin and Cifuentes (1999), the δ13C values are lighter (−34 to −23‰) for C3 plants and heavier (−17 to −9‰) for C4 plants (Deines 1980), whereas those derived from the marine phytoplankton range (− 22 to − 18‰) in between C3 and C4 plants sources. As the δ13C value for the detritus was about −25.4, and was found very close to the values of the mangrove leaves (−26.2), it is clear that the detritus in URE during the study period was of terrestrial-mangrove based origin. Also, the δ13C of the phytoplankton (−20.40) lies within the range of value that of the marine phytoplankton.
A comparison of the δ13C values of the carbon sources from various tropical mangrove/coastal ecosystems is given in Table 7. The carbon sources analyzed in this study (−26.20 to −20.40‰) are found to be more enriched than the carbon sources reported form the majority of the tropical coastal ecosystems (Abrantes and Sheaves 2010; Giarrizzo et al. 2011; Vaslet et al. 2012; Shahraki et al. 2014; Sepu’lveda-Lozada et al. 2015; Contreras et al. 2018; Nelson et al. 2019). The basal source, mangrove leaves showed the most depleted carbon isotopic signature among the others and found within the range of δ13C values reported from the other ecosystems. Being C3, a plant (plants that follow C3 photosynthetic pathway), mangroves are reported to have low δ13C values because of the photosynthesis with high stomatal conductance leading to depletion in δ13C values (Brugnoli and Lauteri 1991). The particulate organic matter (POM), including the phytoplankton analyzed in this study, was close to the estimates from Qeshm Island, Persian Gulf of northern Indian Ocean (Shahraki et al. 2014), and Northern Gulf of Mexico (Nelson et al. 2019). However, there exist large regional variations among the δ13C values of the POM between the study areas, which will be more of a function of the prevailing environmental conditions and composition of the plankton community and hence cannot be generalized in a broader scale (Bouillon et al. 2002).
The δ13C signal for detritus was closer to the isotopic values of mangroves in the URE, possibly indicating that these are the predominant source of detritus in the area. These results are in agreement with the values from the mangrove ecosystems of tropical Eastern Pacific (Contreras et al. 2018) as well as the southern Gulf of Mexico (Sepu’lveda-Lozada et al. 2015). However, for Qeshm Island, Persian Gulf of Northern Indian Ocean (Shahraki et al. 2014), the detrital δ13C signal was closer to that of the POM and phytoplankton. This indicating that the source of detritus carbon in the coastal ecosystems also shows region-specific variations.
Several studies reported the impact of the mangrove-seagrass sourced detritus to be the persistent source of organic matter in the soft sediments of estuaries and coastal bays (Bishop et al. 2007; Taylor et al. 2010). The sediments in the Indian estuaries are also reported to be constituted by high levels of detrital load (Chakraborty et al. 2014; Irudhayanathan et al. 2011; Rao et al. 2011). Moreover, increase of organic matter with decreasing the particle size has been reported from many areas, around the world, and is attributed to co-sedimentation of particulate organic matter with clay and mineral particles (Carter and Mitterer 1978; Sheu and Presely 1986). Nikam et al. (2008) also recorded high organic carbon load in the fine surface sediments from the URE. Hence, the closer values of the stable carbon and nitrogen isotopic signatures of the detritus and sediments observed in the present study substantiates these results. Similar pattern in the detritus-sediment isotopic composition were also reported by Contreras et al. (2018) in a Riverine Mangrove System at the Central Coast of Colombia.
Source consumer relationships
The omnivorous invertebrate representative, A. indicus, exhibited the most depleted value of δ13C among the consumer species (−24.38‰), which was highly close to the detrital δ13C values (−25.40‰). This similarity in values indicated a strong linkage of A. indicus with its benthic feeding niche. Also, the shallow coastal waters of the Northwest coast of India have been reported to have a good amount of detritus, the magnitude of which is more pronounced along the Mumbai waters (Babenerd et al. 1973). Deshmukh (2002) suggested that the phenomenal abundance of paste shrimp, A. indicus in Mumbai coastal waters compared to the other regions, is, in fact, supported by this huge detrital load in the region. Therefore, the present study clearly concurring the theories behind the detritus-paste shrimp trophic relationship. A majority of the studies that investigated the estuarine food web sources has widely used some generic factors to explain the higher productivity of estuarine consumers (Currin et al. 1995; Deegan and Garritt 1997; Ansari et al. 1995; Sreekanth et al. 2020). These factors include the marine sediments carried by tides, terrestrially derived organic matter carried by rivers, phytoplankton or benthic microalgae, and abundant biomass of marsh plants cycled through detrital pathways. Vingare et al. (2012) have also reported the high reliance of all top-level consumers towards benthic prey in the estuarine waters of Europe. Similar inferences can be made from URE ecosystem also as, A. indicus forms the primary source of prey for about 80–90% of the consumer fish species along the north western coast of India (Jaiswar and Chakraborty 2005).
Stable isotope mixing model
A Bayesian isotope mixing model has been developed using SIMMR to estimate the assimilated diet proportions of the selected consumer species representing various trophic guild in the URE. The results of the present study were in agreement with the existing hypothesis on the estuarine food webs that different trophic pathways in the system result from the diversity of resources, connectivity with other ecosystems, and trophic guild diversity of consumers. Also, the system is supported by multiple primary sources, as previously suggested by other authors (Bouillon et al. 2002, 2008; Abrantes and Sheaves 2010). Phytoplankton, detritus, and mangrove leaves together contributed only around 30% of the assimilated diet of the all consumers combined, with a slightly higher share from detritus sources. When consumers are considered separately, about 70% of the assimilated diet of lower trophic level groups, such as zooplankton, fish larvae, and N. nasus, were found to be contributed by localized sources such as phytoplankton, detritus, and mangrove leaves. However, the contribution of these sources to the assimilated diet was comparatively lower in the consumers along the higher trophic levels.
The reasons for the same can be interpreted in two ways: firstly, due to the probability that other C-enriched sources not evaluated in this study are being exploited by these consumers (Contreras et al. 2018), secondly due to the mobility of organisms in the highly dynamic estuarine environment (Costanza et al. 1993). Some of the earlier reports from mangrove ecosystems suggested that microphytobenthos as an important food source for the consumer groups (Abrantes and Sheaves 2010; Giarrizzo et al. 2011; Shahraki et al. 2014). Hence, one of the most probable missing primary carbon sources in the present study area could be the microphytobenthos. A carbon isotopic enrichment is also observed in the lower trophic level group like zooplankton (−19.87‰) compared to the sources analyzed in the present study. As per Al-Zaidan et al. (2006), the microbenthic biota are dominated by Cyanobacteria typically show the most enriched δ13C values among aquatic autotrophs by virtue of its CO2 concentrating mechanism (Price et al. 2008). This could probably explain the role of microphytobenthos in the base of the food chain, which have been missed in the present study. It is also to be noted that some of the sources considered in the earlier works such as marsh plants close to the shoreline (Sepu’lveda-Lozada et al. 2015; Nelson et al. 2019), seagrasses (Abrantes and Sheaves 2010) as well as epiphytes (Sepu’lveda-Lozada et al. 2015), marine POM or marine phytoplankton, which are not considered in the present study. The δ13C values of these higher aquatic vegetation shows high regional variability. For instance, the marsh plants with δ13C values about 32.4‰ in Southern Gulf of Mexico (Sepu’lveda-Lozada et al. 2015) and −14.5‰ along Tropical Eastern Pacific (Contreras et al. 2018) and Seagrass with −14.6 to −7.0‰ in Curuçá estuary (Giarrizzo et al. 2011) and −18.6‰ in Tropical Northern Queensland (Abrantes and Sheaves 2010). Hence, the conclusions about the missing carbon sources in the present study could not be correctly drawn out unless a detailed region specific isotopic analysis of all the available carbon sources in the ecosystem is been carried out.
The consumer species belong to lower TLs, including the larval forms with limited life cycle and mobility, may probably utilize the readily available resources in their diet and thus justifies the higher contribution values of the localized sources analyzed in the study. The studies from elsewhere on the food and feeding of fish larvae substantiate these results as planktonic preys such as mysids and copepods are often the main prey for fish larvae (Rakocinski et al. 1996; Ramos et al. 2006; Pasquaud et al. 2010). When the source contributions analyzed separately, phytoplankton was the most dominant dietary source for the groups such as zooplankton, fish larvae, and N. nasus over the other two sources. In these three consumers, phytoplankton sources alone contributed more than 50% of the assimilated diet. N. nasus is considered to be a generalized feeder on microplankton (80%), with specialization on foraminiferans and green algae in Chilika Lagoon, eastern coast of India (Mukherjee et al. 2016). The present results also strongly suggest phytoplankton based foraging habitat of the species in the URE.
Similarly, for the mid-level carnivore, E. thoracata and predatory species L. savala, the phytoplankton contribution was dominant over the other sources, although these species recorded a clear indication of the external carbon sources in their assimilated diet. Raje et al. (1994) reported that zooplankton, such as copepods, cladocerans, and crab larvae, formed the main food items of E. thoracata, whereas L. savala is principally a pelagic carnivore, feeding mainly on small pelagic fish (Stolephorus sp., Sardinella sp., Dussumieria acuta, and carangids) (Pakhmode and Mohite 2014). In the present study also, the major gut contents of L. savala were observed to be the species like E. thoracata, Sardinella gibbosa, and S. indicus, with a notable proportion of A. indicus as well. Hence a phytoplankton-based trophic pathway taken forward by zooplankton linkages, including the pelagic prey species such as E. thoracata reaching up to the top predator L. savala was evident in this study.
The contribution of mangrove leaves and detritus was dominant over the groups A. indicus, J. belangerii, and N. caelata. These results indicated their dependence on the detritus-based carbon sources, wherein more than 50% of their assimilated diet was contributed by the detritus and mangrove sources together. Also, in the niche map, J. belangerii and N. caelata exhibited more or less enriched and close values of δ13C compared to others. This observation could be a result of their dietary overlap, as suggested by Abrantes and Sheaves (2010). The dietary overlap for J. belangerii and N. caelata is reasonable, as crustaceans chiefly A. indicus was recorded to be the principal food item for both the species in the coastal and estuarine waters around Mumbai (Chakraborty et al. 2000; Raje 2006). Also, J. belangerii and N. caelata are demersal inhabitants (Chakraborty 1988) in the marine coastal waters. Therefore, a mangrove-detritus sourced pathway driven by A. indicus particularly reaching up to the demersal species such as J. belangerii and N. caelata was also could be clearly delineated from the results of the stable isotope mixing model developed in the present study. Similar results were found from the studies from a mangrove ecosystem of the Tropical Eastern Pacific, Central Coast of Colombia, where a trophic route based on mangrove-derived carbon (including detrital material) was evident (Contreras et al. 2018).
The largest negative correlation is between mangrove leaves and detritus obtained in the matrix plots (−77) has clearly indicated the least degree of segregation between these sources in the consumer diets in the present model. However, a remarkable contribution of mangrove leaves in the mid trophic level groups other than the detritus was observed for groups such as A. indicus, J. belangerii, and N. caelata from the URE. These results strongly point towards the existence of a trophic link that missed out in the present study that converts the mangrove leaves to the assimilated diet of these consumer species in the ecosystem. Shahraki et al. (2014) suggested that bacteria would play an important role in the incorporation of mangrove carbon after colonizing and increasing the nitrogen values of the organic matter. Hence, the results are indirectly identifying the role played by the bacterial component in the system as an important connecting link at the base of the aquatic food web. The present results clearly point towards a phytoplankton based pelagic food chain and detritus-based benthic food chain in the URE. Whereas, there were also clear signs that these pathways are never independent, but with interconnecting trophic linkages as evident from the occurrence of both the detrital and plankton sources in varying proportions in the assimilated diet of all the consumers analyzed for the stable isotope mixing models (Bouillon et al. 2002, 2008; Contreras et al. 2018). Also, the fish catch data of URE has indicated that the biomass of benthic groups (mainly contributed by the Acetes indicus and Sciaenids) is significant in the estuary over the pelagic sources. Therefore, the importance of a detritus-based food chain is highly evident from these results.
Food chain length
The food chain length (FCL) has long been recognized as a fundamental ecosystem attribute (Elton 1927; Pimm 1982) to characterize the food chain dynamics (Fretwell 1987), trophic cascades (Carpenter et al. 1985; Pace et al. 1999), ecosystem functions (Worm et al. 2002; Emmett Duffy et al. 2005), fish productivity (Pauly and Christensen 1995), and contaminant bioaccumulation in top predators (Kidd et al. 1995). A food chain length of 3.75 was yielded in the current study based on the isotopic signatures (δ15N) of dominant consumer fish species, indicating a food web structure with at least four trophic guilds. According to Vander Zanden and Fetzer (2007), the average trophic chain length of the marine/coastal and freshwater ecosystems is in the tune of ~4.00 and ~3.50, respectively. The results from URE defined an intermediate value compared to those reported in marine and freshwater ecosystems demanding a more detailed explanation for the interface ecosystems such as estuaries. The FCL variability between ecosystems is generally explained either by “productivity hypothesis” (Jenkins et al. 1992), productive space hypothesis (Schoener 1974), and/or dynamic stability hypothesis (Sterner et al. 1997a, b). The latter suggests that long food chains tend to be dramatically fragile towards environmental perturbation resulting in shorter food chains in ecosystems subjected to disturbances. However, Vander Zanden, and Fetzer (2007) demonstrated that although the stream and estuaries are equally subjected to disturbances, the freshwater streams will be more consistent with the dynamic stability hypothesis than estuaries. This inference is based on their observation of relatively similar and higher TCL for coastal, and marine ecosystems compared to streams in the global literature. On the other hand, Vander Zanden et al. (1999) opined that marine food chains are longer than the other aquatic systems, attributed by the inclusion of marine mammals, which results in a rise of an additional 0.6 trophic level in the overall food chain. Hence, the intermediate trophic chain length obtained in the current study may be due to the missing nodes in the higher trophic position like marine mammals in the ecosystem or their limited habitat capacity in these ecosystems (Sreekanth et al. 2019).
According to Winemiller (2005), shallow lotic and estuarine systems tends to have food webs with three or four trophic levels. Among these ecosystems also food chain length is constrained by various factors such as: energetic constraints (Odum and Smalley 1959), productivity (Pimm 1982; Briand and Cohen 1987), ecosystem size (Post et al. 2000), predator–prey population constraints (Sterner et al. 1997a, b; Pimm 1982; Holt 2002), and environmental fluctuations and disturbance (Pimm 1982). Akin and Winemiller (2006) conducted studies on Mad Island Marsh, an ecosystem situated at further south of the Mumbai megacity experiencing similar geo-climatic and disturbance situations as that of URE. They reported low trophic positions of top predators and hence the trophic chain length and inferred that it is probably due to high incidence of feeding on herbivores and detritivores (i.e., mullet, menhaden, and crabs). Also, the Mad Island Marsh tidal estuary is a relatively small and dynamic habitat subject to frequent disturbances, and hence, the ecosystem size and disturbance could be the main factors limiting food chain length. Similarly, the moderate trophic chain length observed in the study area may also be considered in line with the case of Mad Island Marsh tidal estuary.
It is also to be noted that the mean trophic level of catches can be considered to be a function of the trophic chain length in an exploited ecosystem. Pauly et al. (1998) have suggested that the mean trophic level of the fish landed can be used as a tool for ecosystem status, where the series must extend into the past to cover major changes in the relative biomass of key ecosystem components. Lal et al. (2021) have reported a mean trophic level of catch from URE to be 3.01, indicating a fishery exploiting lower to medium trophic level groups. Similar results were observed from the recent studies from the estuarine and coastal waters of India such that; 2.72 for Sundarbans mangroves (Dutta et al. 2017), 3.12 for Bay of Bengal ecosystem (Das et al. 2018), 2.34 for Hooghly Matlah estuarine System (Mukherjee et al. 2019), and 2.91 for Zuari estuary (Sreekanth et al. 2018). According to Pauly et al. (1998), globally trophic levels of fisheries landings have declined at a rate of 0.1 per decade. The present trends will lead to widespread collapse of fisheries due to a relaxation of the top-down control (Power 1992).
Trophic level variations
The δ15N values have clearly defined the trophic level of the consumer species. The herbivorous species like N. nasus confirmed the evidence of its consumption of basal sources by showing the least value of δ15N. Hence, this species would be one of the first links between the primary producers and the second-order consumers, along with the zooplankton acquaintances in the estuary. Besides N. nasus, the white sardine, E. thoracata that represented the MLCR trophic guild, also showed its isotopic trophic level, which is consistent with its dietary estimates obtained from the present study as well as that from published reports (Hajisamae et al. 2004). For other consumer species, remarkable deviations in isotopic TLs were observed from the dietary estimates and those reported from the literature. Out of these species, A. indicus was the major discrepant with a δ15N-based TL that was highly dissimilar to its feeding habits reported in the literature. The isotopic TL of A. indicus (which is considered to be an omnivore) was obtained as 3.24, made it classified under the trophic guild, MLCR as per the species segregation criteria (Vivekanandan et al. 2009). Crustaceans along the estuarine and mangrove areas show diverse grazing patterns compared to other ecosystems, comprising benthic microalgae, plant epithelial cells, detritus, and cyanobacteria (Bouillon et al. 2002). Therefore, the close association of these species with the benthic feeding habitats coupled with the incidental prey types such as; animal tissue-enriched detritus, fish larvae, or holoplanktonic organisms could explain the elevated δ15N values for A. indicus as of an MLCR or zoobenthivore.
On the other hand, the species from the higher trophic guilds (N. caelata and L. savala) showed remarkably low isotopic TL’s compared to that obtained from the reported values as well as from dietary estimates. As a result, the trophic guild of L. savala and N. caelata could be downgraded to the MLCR-HLCR trophic guild from the HLCR-TPR guild based on the isotopic TL. The hierarchical demotion of TL from the literature estimates indicated a different prey consumption pattern for these species in URE compared to the other marine coastal waters. The adults of L. savala is considered to be a piscivore with a wide range of prey preference with occasional crustacean diet in Brazilian waters (Martins et al. 2005). However, on the northwestern coast of India, the species has been reported as a predator feeding principally on crustaceans, along with fish and cephalopods (Khan 2006). Among crustaceans, sergestid, Acetes spp. is the most frequently encountered prey item in the gut contents of L. savala (Khan 2006; Jaiswar and Chakraborty 2005). Thus, the unique abundance of sergestid shrimps in the area has molded the species to be a crustacean feeder regardless of other geographical areas, where it has occupied the piscivorous niche position. This trophic positioning might be the clear justification of the significantly lower δ15N and hence, the isotopic TL of the species in the estuary. Vander Zanden et al. (1997) have attempted to verify the δ15N measure of trophic position using within-system comparisons between dietary data and δ15N. They have reported some degree of inability of dietary data to represent temporal variation in feeding and errors in trophic position estimates of the prey item. The differences in the way that the dietary and δ15N methods integrate trophic position may cause problems while attempting a direct comparison of the estimates (Vander Zanden et al. 1997). Also, use of dietary data provides a snapshot in time of an organism’s diet, which certainly does not represent the average trophic position of a species for larger temporal scales. The deviation from the dietary estimates can possibly be justified by this reason. Hence this study clearly projects the importance of δ15N-based TL calculations over dietary estimates in predicting the average trophic position of fish species over larger temporal scales in dynamic ecosystems like estuaries.
The variation in the isotopic trophic Level with the dietary and literature estimates may also be due to the constant fractionation factor used in the present study. Hussey et al. (2014) revealed that practically, the fractionation factor (δ15N) cannot be a constant, and determined an empirical linear relationship with δ15N of consumer through meta-analysis. Following this, the TL analysis used a scaled δ15N trophic framework that was a narrowing discrimination with increasing trophic levels, which provided a more accurate representation of species structuring (Reum et al. 2015). The use of the constant fractionation factor may either under estimate or overestimate the actual trophic level, based on the basal resource which may have far reaching consequences in a managerial perspective. That means, the elevated trophic levels of large apex predators indicates 1 to 2 orders of magnitude less energy available to support these fish than previously thought, assuming a transfer efficiency of approximately 10% (Trebilco et al. 2013). This suggests that for highly exploited large predators, the population numbers or biomass estimates could be much smaller than current theoretical estimates, emphasizing the need for proactive precautionary fisheries management to ensure viable populations (Hussey et al. 2014).
Stable isotopic signature as pollution indicators
It has been considered that high levels of δ15N in basal sources can be used as an alternative method to establish the level of human eutrophication since anthropogenic sources of nitrogen are generally enriched in the heavy isotope when compared to natural sources (Cole et al. 2006; Bannon and Roman 2008; Baeta et al. 2009a, b; Baeta et al. 2017). In URE the δ15N (‰) for the basal sources such as phytoplankton (5.90), and detritus (8.40) were remarkably higher compared to that for mangrove leaves (0.40). Hence, the possibility of nitrogen enrichment in the detritus and the POM/phytoplankton due to human eutrophication cannot be snubbed. Apart from that, isotopic signatures of zooplanktivorous fish and fish larvae also reflect anthropogenic pressures in the estuarine eco systems, and function as an exposure indicator of nitrogen pollution (Baeta et al. 2017). Thus, the mean δ15N (‰) of 6.71±0.35 for zooplankton observed from URE is also substantiating a possibility of nitrogen pollution in the ecosystem.
Conclusion
The present study clearly illustrated the food web structure of the URE ecosystem in terms of the major sources and links explained based on the stable carbon and nitrogen Isotopic signatures. Although the study has been conducted based on the nodal species of the food web regardless of the huge diversity in the estuarine ecosystems, it could clearly delineate the trophic pathways and the connecting links steered by the detritus and phytoplankton sources. Furthermore, the study points towards a possibility of nitrogen enrichment in the detritus and the POM/phytoplankton due to human eutrophication in URE. Hence, the present study throws light for more investigations in this line using such advanced and reliable methods for studying complex coastal ecosystems. Investigations of other spatio-temporal scales of environmental variations, including estuarine hydrography and other biotic factors in context of the heavy pollution pressure in the area, might further illustrate the dynamics of basal sources and trophic functioning of the estuary. Since the advanced tools such as stable isotopic signatures are poorly utilized for ecosystem analysis in the Indian coastal waters, future research is encouraged to further disclose the underlying facts of trophic organization in these unique tropical ecosystems.
Data availability
All the data involved in the present study are provided in the manuscript and the supplementary material.
References
Abrantes KG, Sheaves M (2010) Importance of freshwater flow in terrestrial–aquatic energetic connectivity in intermittently connected estuaries of tropical Australia. Mar Biol 157(9):2071–2086
Achyuthan H, Quade J, Roe L, Placzek C (2007) Stable isotopic composition of pedogenic carbonates from the eastern margin of the Thar Desert, Rajasthan, India. Quatern Int 162:50–60
Akin S, Winemiller KO (2006) Seasonal variation in food web composition and structure in a temperate tidal estuary. Estuaries Coasts 29(4):552–567
Akin S, Winemiller KO (2008) Body size and trophic position in a temperate estuarine food web. Acta Oecol 33:144–153
Al-Zaidan AS, Kennedy H, Jones DA, Al-Mohanna SY (2006) Role of microbial mats in Sulaibikhat Bay (Kuwait) mudflat food webs: evidence from δ13C analysis. Mar Ecol Prog Ser 308:27–36
Amara R, Lagardere F, Desaunay Y, Marchand J (2000) Metamorphosis and estuarine colonization in the common sole, Solea solea (L.): implications for recruitment regulation. Oceanologica Acta 23(4):469–484
Annasawmy P, Cherel Y, RomanovLe Loc’hMénardTernonMarsac EVFFJFF (2020) Stable isotope patterns of mesopelagic communities over two shallow seamounts of the south-western Indian Ocean. Deep Sea Res Part II 176:104804
Ansari ZA, Chatterji A, Ingole BS, Sreepada RA, Rivonkar CU, Parulekar AH (1995) Community structure and seasonal variation of an inshore demersal fish community at Goa, west coast of India. Estuar Coast Shelf Sci 41(5):593–610
Babenerd B, Boje R, Krey J, Montecino V (1973) Microbiomass and detritus in the upper 50 m of the Arabian Sea along the Coast of Africa and India during the NE monsoon 1964/65. In: Zeitzschel B, Gerlach SA (eds) The Biology of the Indian Ocean Ecological Studies, 4th edn. Springer, Berlin, pp 233–237
Babler AL, Pilati A, Vanni MJ (2011) Terrestrial support of detritivorous fish populations decreases with watershed size. Ecosphere 2(7):1–23
Baeta A, Pinto R, Valiela I, Richard P, Niquil N, Marques JC (2009a) δ15N and δ13C in the Mondego estuary food web: seasonal variation in producers and consumers. Mar Environ Res 67(3):109–116
Baeta A, Valiela I, Rossi F, Pinto R, Richard P, Niquil N, Marques JC (2009b) Eutrophication and trophic structure in response to the presence of the eelgrass. Zostera Noltii Mar Biol 156(10):2107–2120
Baeta A, Vieira LR, Lírio AV, Canhoto C, Marques JC, Guilhermino L (2017) Use of stable isotope ratios of fish larvae as indicators to assess diets and patterns of anthropogenic nitrogen pollution in estuarine ecosystems. Ecol Ind 1(83):112–121
Bannon RO, Roman CT (2008) Using stable isotopes to monitor anthropogenic nitrogen inputs to estuaries. Ecol Appl 18(1):22–30
Barton MB, Litvin SY, Vollenweider JJ, Heintz RA, Norcross BL, Boswell KM (2019) Implications of trophic discrimination factor selection for stable isotope food web models of low trophic levels in the Arctic nearshore. Mar Ecol Prog Ser 613:211–216
Bianchi G (1985) Field guide to the commercial marine and brackish-water species of Pakistan. FAO species identification sheets for fishery purposes. FAO, Rome
Bidigare RR, Fluegge A, Freeman KH, Hanson KL, Hayes JM, Hollander D, Jasper JP, King LL, Laws EA, Milder J, Millero FJ (1997) Consistent fractionation of 13C in nature and in the laboratory: growth-rate effects in some haptophyte algae. Global Biogeochem Cycles 11(2):279–292
Bird CS, Veríssimo A, Magozzi S, Abrantes KG, Aguilar A, Al-Reasi H, Barnett A, Bethea DM, Biais G, Borrell A, Bouchoucha M (2018) A global perspective on the trophic geography of sharks. Nat Ecol Evol 2(2):299–305
Bishop MJ, Kelaher BP, Alquezar R, York PH, Ralph PJ, Greg Skilbeck C (2007) Trophic cul-de-sac, Pyrazus ebeninus, limits trophic transfer through an estuarine detritus-based food web. Oikos 116(3):427–438
Boecklen WJ, Yarnes CT, Cook BA, James AC (2011) On the use of stable isotopes in trophic ecology. Annu Rev Ecol Evol Syst 42:411–440
Borrell A, Cardona L, Kumarran RP, Aguilar A (2011) Trophic ecology of elasmobranchs caught off Gujarat, India, as inferred from stable isotopes. ICES J Mar Sci 68(3):547–554
Bouillon S, Raman AV, Dauby P, Dehairs F (2002) Carbon and nitrogen stable isotope ratios of subtidal benthic invertebrates in an estuarine mangrove ecosystem (Andhra Pradesh, India). Estuar Coast Shelf Sci 54(5):901–913
Bouillon S, Borges AV, Castañeda-Moya E, Diele K, Dittmar T, Duke NC, Kristensen E, Lee SY, Marchand C, Middelburg JJ, Rivera-Monroy VH (2008) Mangrove production and carbon sinks: a revision of global budget estimates. Global Biogeochem Cycles 22(2):1–12
Briand F, Cohen JE (1987) Environmental correlates of food chain length. Science 238(4829):956–960
Brugnoli E, Lauteri M (1991) Effects of salinity on stomatal conductance, photosynthetic capacity, and carbon isotope discrimination of salt-tolerant (Gossypium hirsutum L.) and salt-sensitive (Phaseolus vulgaris L.) C3 nonhalophytes. Plant Physiol 95(2):628–35
Cabana G, Rasmussen JB (1996) Comparison of aquatic food chains using nitrogen isotopes. Proc Natl Acad Sci 93(20):10844–10847
Caraco N, Bauer JE, Cole JJ, Petsch S, Raymond P (2010) Millennial-aged organic carbon subsidies to a modern river food web. Ecology 91(8):2385–2393
Cardoso PG, Pardal MA, Lillebø AI, Ferreira SM, Raffaelli D, Marques JC (2004) Dynamic changes in seagrass assemblages under eutrophication and implications for recovery. J Exp Mar Biol Ecol 302(2):233–248
Carpenter SR, Kitchell JF, Hodgson JR (1985) Cascading trophic interactions and lake productivity. Bioscience 35(10):634–639
Carter PW, Mitterer RM (1978) Amino acid composition of organic matter associated with carbonate and non-carbonate sediments. Geochim Cosmochim Acta 42(8):1231–1238
Chakraborty SK (1988) Study on the sciaenids of Bombay waters. Ph. D. Thesis. University of Bombay, India, p 421
Chakraborty SK, Devadoss P, Manojkumar PP, Feroz Khan M, Jayasankar P, Sivakami S, Gandhi V, Appanasastry Y, Raju A, Livingston P, Ameer Hamsa KMS (2000) The fishery, biology and stock assessment of jew fish resources of India. Marine Fisheries Research and Management. CMFRI, Kochi, pp 604–616
Chakraborty P, Ramteke D, Chakraborty S, Nath BN (2014) Changes in metal contamination levels in estuarine sediments around India–an assessment. Mar Pollut Bull 78(1–2):15–25
Chidambaram S, Prasanna MV, Ramanathan AL, Vasu K, Hameed S, Warrier UK, Srinivasamoorthy K, Manivannan R, Tirumalesh K, Anandhan P, Johnsonbabu G (2009) A study on the factors affecting the stable isotopic composition in precipitation of Tamil Nadu, India. Hydrol Processes: an Int J 23(12):1792–1800
Christensen V, Pauly D (1992) ECOPATH II—a software for balancing steady-state ecosystem models and calculating network characteristics. Ecol Model 61(3–4):169–185
Coffin RB, Cifuentes LA (1999) Stable isotope analysis of carbon cycling in the Perdido Estuary, Florida. Estuaries 22(4):917–926
Cole ML, Kroeger KD, McClelland JW, Valiela I (2006) Effects of watershed land use on nitrogen concentrations and δ15 nitrogen in groundwater. Biogeochemistry 77(2):199–215
Contreras DM, Kintz JC, González AS, Mancera E (2018) Food web structure and trophic relations in a riverine mangrove system of the Tropical Eastern Pacific, Central Coast of Colombia. Estuaries Coasts 41(5):1511–1521
Costanza R, Kemp WM, Boynton WR (1993) Predictability, scale, and biodiversity in coastal and estuarine ecosystems: implications for management. Ambio 22:88–96
Currin CA, Newell SY, Paerl HW (1995) The role of standing dead Spartina alterniflora and benthic microalgae in salt marsh food webs: considerations based on multiple stable isotope analysis. Mar Ecol Prog Ser 121:99–116
Das A, Krishnaswami S, Bhattacharya SK (2005) Carbon isotope ratio of dissolved inorganic carbon (DIC) in rivers draining the Deccan Traps, India: sources of DIC and their magnitudes. Earth Planet Sci Lett 236(1–2):419–429
Das I, Hazra S, Das S, Giri S, Chanda A, Maity S, Ghosh S (2018) Trophic-level modelling of the coastal waters of the northern Bay of Bengal, West Bengal, India. Fish Sci 84(6):995–1008
Datta PS, Tyagi SK, Chandrasekharan H (1991) Factors controlling stable isotope composition of rainfall in New Delhi, India. J Hydrol 128(1–4):223–236
De la Cruz-Agüero G (194) Sistema para el análisis de comunidades ANACOM: Versión 3.0. Depto. Pesquerías y Biología Marina, CICIMAR, IPN, México, pp 99
Deegan LA, Garritt RH (1997) Evidence for spatial variability in estuarine food webs. Mar Ecol Prog Ser 147:31–47
Deines P (1980) The isotopic composition of reduced organic carbon. Handb Environ Isotope Geochem 1:329–406
Deshmukh VD (2002) Biology of Acetes indicus Milne Edwards in Bombay waters. Indian J Fish 49(4):379–388
Dillon KS, Fleming CR, Slife C, Leaf RT (2021) Stable isotopic niche variability and overlap across four fish guilds in the north-central Gulf of Mexico. Mar Coast Fish 3:213–227
Donázar-Aramendía I, Sánchez-Moyano JE, García-Asencio I, Miró JM, Megina C, García-Gómez J (2019) Human pressures on two estuaries of the Iberian Peninsula are reflected in food web structure. Sci Rep 1:1–10
Duarte CM, Cebrián J (1996) The fate of marine autotrophic production. Limnol Oceanogr 41(8):1758–1766
Dutta S, Chakraborty K, Hazra S (2017) Ecosystem structure and trophic dynamics of an exploited ecosystem of Bay of Bengal, Sunderban Estuary. India Fish Sci 83(2):145–159
Elliott M, Whitfield AK, Potter IC, Blaber SJ, Cyrus DP, Nordlie FG, Harrison TD (2007) The guild approach to categorizing estuarine fish assemblages: a global review. Fish Fish 8(3):241–268
Elton CS (1927) Animal ecology. Sidgwick and Jackson, London
Emmett Duffy J, Paul Richardson J, France KE (2005) Ecosystem consequences of diversity depend on food chain length in estuarine vegetation. Ecol Lett (3):301–309
Fernandes R, Millard AR, Brabec M, Nadeau MJ, Grootes P (2014) Food reconstruction using isotopic transferred signals (FRUITS): a Bayesian model for diet reconstruction. PLoS One 9(2):e87436
Filgueira R, Castro BG (2011) Study of the trophic web of San Simón Bay (Ría de Vigo) by using stable isotopes. Cont Shelf Res 31:476–487
Fischer W, Bianchi G (1984) FAO species identification sheets for fishery purposes: Western Indian Ocean (Fishing Area 51). v. 1: Introductory material. Bony fishes, families: Acanthuridae to Clupeidae.-v. 2: Bony fishes, families: Congiopodidae to Lophotidae.-v. 3:... families: Lutjanidae to Scaridae.-v. 4:... families: Scatophagidae to Trichiuridae.-v. 5: Bony fishes, families: Triglidae to Zeidae. Chimaeras. Sharks. Lobsters. Shrimps and prawns. Sea turtles. v. 6: Alphabetical index of scientific names and vernacular names
Fretwell SD (1987) Food chain dynamics: the central theory of ecology? Oikos 50(3):291–301
Froese R, Pauly D (2016) Fishbase World Wide Web electronic publication. http://www.fishbase.org. Accessed on 22 March 2019
Fry B, Sherr EB (1989) δ 13 C measurements as indicators of carbon flow in marine and freshwater ecosystems. Stable isotopes in ecological research. Springer, New York, pp 196–229
Giarrizzo T, Schwamborn R, Saint-Paul U (2011) Utilization of carbon sources in a northern Brazilian mangrove ecosystem. Estuar Coast Shelf Sci 95(4):447–457
Golikov AV, Ceia FR, Sabirov RM, Belyaev AN, Blicher ME, Arboe NH, Zakharov DV, Xavier JC (2019) Food spectrum and trophic position of an Arctic cephalopod, Rossia palpebrosa (Sepiolida), inferred by stomach contents and stable isotope (δ13C and δ15N) analyses. Mar Ecol Prog Ser 632:131–144
Hajisamae S, Chou LM, Ibrahim S (2004) Feeding habits and trophic relationships of fishes utilizing an impacted coastal habitat. Singap Hydrobiol 520(1–3):61–71
Holt RD (2002) Food webs in space: on the interplay of dynamic instability and spatial processes. Ecol Res 17(2):261–273
Hopkins JB, Ferguson JM (2012) Estimating the diets of animals using stable isotopes and a comprehensive Bayesian mixing model. PLoS ONE 7(1):e28478
Hussey NE, MacNeil MA, McMeans BC, Olin JA, Dudley SF, Cliff G, Wintner SP, Fennessy ST, Fisk AT (2014) Rescaling the trophic structure of marine food webs. Ecol Lett 17(2):239–250
Irudhayanathan A, Thirunavukkarasu R, Senapathi V (2011) Grain size characteristics of the Coleroon estuary sediments, Tamilnadu, East coast of India. Carpathian J Earth Environ Sci 6(2):151–157
Jackson AL, Inger R, Parnell AC, Bearhop S (2011) Comparing isotopic niche widths among and within communities: SIBER–Stable Isotope Bayesian Ellipses in R. J Anim Ecol 80(3):595–602
Jagtap TG, Untawale AG, Inamdar SN (1994) Study of mangrove environment of Maharashtra coast using remote sensing data. Indian J Mar Sci 23:90–93
Jaiswar AK, Chakraborty SK (2005) Acetes, the preferred food of fishes along the northwest coast of India. Indian J Fish 52(2):215–219
Jenkins B, Kitching RL, Pimm SL (1992) Productivity, disturbance and food-web structure at a local spatial scale in experimental container habitats. Oikos 65:249–255
Kanagaraj G, Elango L (2019) Chromium and fluoride contamination in groundwater around leather tanning industries in southern India: implications from stable isotopic ratio δ53Cr/δ52Cr, geochemical and geo-statistical modelling. Chemosphere 220:943–953
Kantharajan G, Pandey PK, Krishnan P, Ragavan P, Jeevamani JJ, Purvaja R, Ramesh R (2018) Vegetative structure and species com-position of mangroves along the Mumbai coast, Maharashtra, India. Reg Stud Mar Sci 19:1–8
Kelly EF, Blecker SW, Yonker CM, Olson CG, Wohl EE, Todd LC (1998) Stable isotope composition of soil organic matter and phytoliths as paleoenvironmental indicators. Geoderma 82(1–3):59–81
Khan MZ (2006) Fishery resource characteristics and stock assessment of ribbonfish, Trichiurus lepturus (Linnaeus). Indian J Fish 53(1):1–12
Kidd KA, Hesslein RH, Fudge RJP, Hallard KA (1995) The influence of trophic level as measured by δ15N on mercury concentrations in freshwater organisms. Mercury as a global pollutant. Springer, Dordrecht, pp 1011–1015
Kiszka JJ, Aubail A, Hussey NE, Heithaus MR, Caurant F, Bustamante P (2015) Plasticity of trophic interactions among sharks from the oceanic south-western Indian Ocean revealed by stable isotope and mercury analyses. Deep Sea Res Part I 96:49–58
Kling GW, Fry B, O’Brien WJ (1992) Stable isotopes and planktonic trophic structure in arctic lakes. Ecology 73(2):561–566
Komatsu T, Sakamoto SX, Sawayama S, Murata H, Sasa S, Montani S, Yatabe H, Yokoji N, Seko H, Nakano Y, Fujii M (2019) Toward realizing the sustainable use of and healthy marine environments in an open-type enclosed bay. In Integrated coastal management in the Japanese Satoumi, Elsevier, pp 15–57
Kumar P, Das NK, Mallik C, Bhandari RK (2011) Stable isotopes study on geothermal waters in eastern India. Curr Sci 101(9):1205–1209
Kumary KS, Azis PK, Natarajan P (2011) Sediment characteristics of Poonthura estuary (southwest coast of India) in relation to pollution. Indian J Mar Sci 30:75–80
Kumawat T, Shenoy L, Chakraborty SK, Deshmukh VD, Raje SG (2015) Compliance of bag net fishery of Maharashtra coast, India with article 7 of the FAO code of conduct for responsible fisheries. Mar Policy 56:9–15
Kwak TJ, Zedler JB (1997) Food web analysis of southern California coastal wetlands using multiple stable isotopes. Oecologia 110(2):262–277
Lad D, Patil S (2016) Diversity of intertidal meiobenthos from estuary of Bhayander and Naigaon, Thane, Maharashtra. India Int J Sci Res 5(8):245–246
Lal DM, Sreekanth GB, Shivakrishna A, Kumar R, Nayak BB, Abidi ZJ (2021) Ecosystem health status and trophic modeling of an anthropogenically impacted small tropical estuary along India’s west coast. Environ Sci Pollut Res 28(26):35073–35093
Lala KD (2004) Water supply of thane Municipal Corporation. In: Proceedings of One Day Seminar on-Pollution of Water Bodies In urban Area (Case Study), Vidya Prasarak Mandal’s Polytechnic, Thane, Sponsored by AICTE, New Delhi, pp 6–9
Lavoie RA, Hebert CE, Rail JF, Braune BM, Yumvihoze E, Hill L, Lean DRS (2010) Trophic structure and mercury distribution in a Gulf of St. Lawrence (Canada) food web using stable isotope analysis. Sci Total Environ 408:5529–5539
Layman CA, Post DM (2008) Can stable isotope ratios provide for community-wide measures of trophic structure? Reply Ecol 89(8):2358–2359
Le Loc’hHilyGrall CACJ (2008) Benthic community and food web structure on the continental shelf of the Bay of Biscay (North Eastern Atlantic) revealed by stable isotopes analysis. J Mar Syst 72(1–4):17–34
Maes J, Van Damme S, Meire P, Ollevier F (2004) Statistical modeling of seasonal and environmental influences on the population dynamics of an estuarine fish community. Mar Biol 145(5):1033–1042
Mann KH (1998) Production and use of detritus in various freshwater, estuarine, and coastal marine ecosystems. Limnol Oceanogr 33(4 2):910–930
Martínez del Rio C, Sabat P, Anderson-Sprecher R, Gonzalez SP (2009) Dietary and isotopic specialization: the isotopic niche of three Cinclodes ovenbirds. Oecologia 161(1):149–159
Martins AS, Haimovici M, Palacios R (2005) Diet and feeding of the cutlassfish Trichiurus lepturus in the Subtropical Convergence Ecosystem of southern Brazil. J Mar Biol Assoc UK 85(5):1223–1230
Mazumdar A (2004) Stable isotopic study of late Neoproterozoic-early Cambrian (?) sediments from Nagaur-Ganganagar basin, western India: possible signatures of global and regional C-isotopic events. Geochem J 38(2):163–175
Ménard F, Lorrain A, Potier M, Marsac F (2007) Isotopic evidence of distinct feeding ecologies and movement patterns in two migratory predators (yellowfin tuna and swordfish) of the western Indian Ocean. Mar Biol 153(2):141–152
Mishra V (2002) Study of macrofauna of mangrove mudflats of Ulhas River Estuary. Ph. D. Thesis, University of Mumbai, Mumbai, India
Mohapatra BC, Rengarajan K (2000) Heavy metal toxicity in the estuarine, coastal and marine ecosystems of India. CMFRI Special Publ 69:1–21
Moore JW, Semmens BX (2008) Incorporating uncertainty and prior information into stable isotope mixing models. Ecol Lett 11(5):470–480
Mukherjee A, Fryar AE, Rowe HD (2007) Regional-scale stable isotopic signatures of recharge and deep groundwater in the arsenic affected areas of West Bengal. India J Hydrol 334(1–2):151–161
Mukherjee M, Suresh VR, Manna RK, Panda D, Sharma AP, Pati MK (2016) Dietary preference and feeding ecology of Bloch’s gizzard shad, Nematalosa nasus. J Ichthyol 56(3):373–382
Mukherjee J, Karan S, Chakrabarty M, Banerjee A, Rakshit N, Ray S (2019) An approach towards quantification of ecosystem trophic status and health through ecological network analysis applied in Hooghly-Matla estuarine system, India. Ecol Ind 100:55–68
Nelson JA, Lesser J, James WR, Behringer DP, Furka V, Doerr JC (2019) Food web response to foundation species change in a coastal ecosystem. Food Webs 21:e00125
Nikam VS, Kumar A, Lalla K, Gupta K (2008) Conservation of wetlands and mangroves in Thane Creek and URE, India. In: Proceedings of Taal 2007: The 12th World Lake Conference, Jaipur, pp 1635- 1642
Odum EP, Smalley AE (1959) Comparison of population energy flow of a herbivorous and a deposit-feeding invertebrate in a salt marsh ecosystem. Proc Natl Acad Sci 45(4):617–622
Pace ML, Cole JJ, Carpenter SR, Kitchell JF (1999) Trophic cascades revealed in diverse ecosystems. Trends Ecol Evol 14(12):483–488
Pakhmode PK, Mohite SA (2014) Feeding biology of ribbonfish, Lepturacanthus savala (Cuvier, 1929) off Ratnagiri coast, Maharashtra. Int J Fish Aquat Stud 1(3):123–129
Parnell AC, Inger R, Bearhop S, Jackson AL (2010) Source partitioning using stable isotopes: coping with too much variation. PLoS ONE 5(3):e9672
Parnell AC, Phillips DL, Bearhop S, Semmens BX, Ward EJ, Moore JW, Jackson AL, Grey J, Kelly DJ, Inger R (2013) Bayesian stable isotope mixing models. Environmetrics 24(6):387–399
Parnell AC, Inger R (2019) Stable isotope mixing models in R with SIMMR: a stable isotope mixing model. Version 0.4, 1. Available from https://CRAN.R-project.org/package=simmr. Accessed 19 Sept 2020
Pasquaud S, Elie P, Jeantet C, Billy I, Martinez P, Girardin M (2008) A preliminary investigation of the fish food web in the Gironde estuary, France, using dietary and stable isotope analyses. Estuar Coast Shelf Sci 78(2):267–279
Pasquaud S, Pillet M, David V, Sautour B, Elie P (2010) Determination of fish trophic levels in an estuarine system. Estuar Coast Shelf Sci 86(2):237–246
Pauly D (1998) Tropical fishes: patterns and propensities. J Fish Biol 53:1–7
Pauly D, Christensen V (1995) Primary production required to sustain global fisheries. Nature 374(6519):255–257
Pauly D, Christensen V, Dalsgaard J, Froese R, Torres F Jr (1998) Fishing down marine food webs. Science 279(5352):860–863
Peterson BJ, Fry B (1987) Stable isotopes in ecosystem studies. Ann Rev Ecol Syst 18:293–320
Phillips DL (2012) Converting isotope values to diet composition: the use of mixing models. J Mammal 93(2):342–352
Phillips DL, Gregg JW (2001) Uncertainty in source partitioning using stable isotopes. Oecologia 127(2):171–179
Phillips DL, Gregg JW (2003) Source partitioning using stable isotopes: coping with too many sources. Oecologia 136(2):261–269
Phillips DL, Koch PL (2002) Incorporating concentration dependence in stable isotope mixing models. Oecologia 130:114–125
Pimm SL (1982) Food webs. Food webs. Springer, Dordrecht, pp 1–11
Pompanon F, Deagle E, Symondson WO, Brown DS, Jarman SN, Taberlet P (2012) Who is eating what: diet assessment using next generation sequencing. Mol Ecol 21(8):1931–1950
Popp BN, Takigiku R, Hayes JM, Louda JW, Baker EW (1989) The post-Paleozoic chronology and mechanism of 13 C depletion in primary marine organic matter. Am J Sci 289(4):436–454
Popp BN, Parekh P, Tilbrook B, Bidigare RR, Laws EA (1997) Organic carbon δ13C variations in sedimentary rocks as chemostratigraphic and paleoenvironmental tools. Palaeogeogr Palaeoclimatol Palaeoecol 132(1–4):119–132
Popp BN, Laws EA, Bidigare RR, Dore JE, Hanson KL, Wakeham SG (1998) Effect of phytoplankton cell geometry on carbon isotopic fractionation. Geochim Cosmochim Acta 62(1):69–77
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83(3):703–718
Post DM, Pace ML, Hairston NG (2000) Ecosystem size determines food-chain length in lakes. Nature 405(6790):1047–1049
Potapov AM, Scheu TAV, S, (2019) Uncovering trophic positions and food resources of soil animals using bulk natural stable isotope composition. Biol Rev 94(1):37–59
Power ME (1992) Top down and bottom-up forces in food webs: do plants have primacy. Ecology 73(3):733–746
Prasad MBK, Ramanathan AL (2009) Organic matter characterization in a tropical estuarine-mangrove ecosystem of India: preliminary assessment by using stable isotopes and lignin phenols. Estuar Coast Shelf Sci 84(4):617–624
Price GD, Badger MR, Woodger FJ, Long BM (2008) Advances in understanding the cyanobacterial CO2-concentrating-mechanism (CCM): functional components, Ci transporters, diversity, genetic regulation and prospects for engineering into plants. J Exp Bot 59(7):1441–1461
Pritchard DW (1967) What is an estuary: physical viewpoint. In: Lauff GH (ed) Estuaries, 1st edn. American Association for the Advancement of Science, Washington DC, pp 149–176
Quevedo M, Svanbäck R, Eklöv P (2009) Intrapopulation niche partitioning in a generalist predator limits food web connectivity. Ecology 90(8):2263–2274
Rajawat AS, Chauhan HB, Ratheesh R, Rode S, Bhanderi RJ, Mahapatra M, Kumar M, Yadav R, Abraham SP, Singh SS, Keshri KN (2015) Assessment of coastal erosion along the Indian coast on 1: 25,000 scale using satellite data of 1989–1991 and 2004–2006 time frames. Curr Sci 2015:347–353
Raje SG (2006) Some aspects of biology of catfishes Tachysurus caelatus (Valenciennes) and Osteogeneiosus militaris (Linnaeus) from Mumbai. Indian J Fish 53(3):330–343
Raje SG, Deshmukh VD, Das T (1994) Fishery and biology of white sardine, Escualosa thoracata (Valenciennes) at Versova, Bombay. J Indian Fish Assoc 24:51–62
Rakocinski CF, Lyczkowski-Shultz J, Richardson SL (1996) Ichthyoplankton assemblage structure in Mississippi Sound as revealed by canonical correspondence analysis. Estuar Coast Shelf Sci 43(2):237–257
Ramos S, Cowen RK, Paris C, Ré P, Bordalo AA (2006) Environmental forcing and larval fish assemblage dynamics in the Lima River estuary (northwest Portugal). J Plankton Res 28(3):275–286
Rao VP, Shynu R, Kessarkar PM, Sundar D, Michael GS, Narvekar T, Blossom V, Mehra P (2011) Suspended sediment dynamics on a seasonal scale in the Mandovi and Zuari estuaries, central west coast of India. Estuar Coast Shelf Sci 91(1):78–86
Rathod SD (2005) Effect of pollution on Mudskipper Fishery of Ulhas River Estuary with a special reference to the biology of Boleophthalmus Dussumieri (CUV. & VAL.). Minor project report, University of Mumbai, Mumbai, India
Rathod SD, Quadros G and Athalye RP (2003) Qualitative study of finfish and shell fish fauna of Thane creek and Ulhas River Estuary.Proceedings of the National Seminar on Creeks, Estuaries and Mangroves Pollution and Conservation 28th to 30th November, 2002, Thane, pp 135–141
Rathod SD, Patil NN (2009) Assessment of some hydrological parameters of Ulhas River Estuary, in the vicinity of Thane city, Maharashtra State. J Aquat Biol 24(2):103–108
Rau GH, Riebesell U, Wolf-Gladrow D (1996) A model of photosynthetic 13C fractionation by marine phytoplankton based on diffusive molecular CO2 uptake. Mar Ecol Prog Ser 133:275–285
Reum JC, Jennings S, Hunsicker ME (2015) Implications of scaled δ15N fractionation for community predator–prey body mass ratio estimates in size-structured food webs. J Anim Ecol 84(6):1618–1627
Riera P, Stal LJ, Nieuwenhuize J, Richard P, Blanchard G, Gentil F (1999) Determination of food sources for benthic invertebrates in a salt marsh (Aiguillon Bay, France) by carbon and nitrogen stable isotopes: importance of locally produced sources. Mar Ecol Prog Ser 187:301–307
Samanta S, Dalai TK, Pattanaik JK, Rai SK, Mazumdar A (2015) Dissolved inorganic carbon (DIC) and its δ13C in the Ganga (Hooghly) River estuary, India: Evidence of DIC generation via organic carbon degradation and carbonate dissolution. Geochim Cosmochim Acta 165:226–248
Schoener TW (1974) Resource partitioning in ecological communities. Science 185(4145):27–39
Schwarcz HP (1991) Some theoretical aspects of isotope paleodiet studies. J Archaeol Sci 18(3):261–275
Selleslagh J, Amara R, Laffargue P, Lesourd S, Lepage M, Girardin M (2009) Fish composition and assemblage structure in three Eastern English Channel macrotidal estuaries: a comparison with other French estuaries. Estuar Coast Shelf Sci 81(2):149–159
Sepúlveda-Lozada A, Mendoza-Carranza M, Wolff M, Saint-Paul U, Ponce-Mendoza A (2015) Differences in food web structure of mangroves and freshwater marshes: evidence from stable isotope studies in the Southern Gulf of Mexico. Wetlands Ecol Manage 23(2):293–314
Shahraki M, Fry B, Krumme U, Rixen T (2014) Microphytobenthos sustain fish food webs in intertidal arid habitats: a comparison between mangrove-lined and un-vegetated creeks in the Persian Gulf. Estuar Coast Shelf Sci 149:203–212
Shankar D (2000) Seasonal cycle of sea level and currents along the coast of India. Curr Sci 78(3):279–289
Sheu DD, Presley BJ (1986) Variations of calcium carbonate, organic carbon and iron sulfides in anoxic sediment from the Orca Basin Gulf of Mexico. Mar Geol 70(1–2):103–118
Shynu R, Rao P, Sarma VV, Kessarkar PM, Murali RM (2015) Sources and fate of organic matter in suspended and bottom sediments of the Mandovi and Zuari estuaries, western India. Curr Sci 2015:226–238
Simenstad CA, Wissmar RC (1985) d13C evidence of the origins and fates of organic carbon in estuarine and nearshore food webs. Mar Ecol Prog Ser 22:141–152
Sreekanth GB, Chakraborty SK, Jaiswar AK, Zacharia PU (2018) An inventory on the coastal finfish and shellfish species of Zuari estuary, southwest coast of India. Indian J Geo-Mar Sci 47(5):945–958
Sreekanth GB, Jaiswar AK, Zacharia PU, Pazhayamadom DG, Chakraborty SK (2019) Effect of environment on spatio-temporal structuring of fish assemblages in a monsoon-influenced tropical estuary. Environ Monit Assess 191(5):305
Sreekanth GB, Chakraborty SK, Jaiswar AK, Zacharia PU (2020) Trophic network and food web characteristics in a small tropical monsoonal estuary: a comparison with other estuarine systems. Indian J Geo-Mar Sci 47(5):945–958
Sterner RW, Bajpai A, Adams T (1997a) The enigma of food-chain length: absence of theoretical evidence for dynamic constraints. Ecology 78:2258–2262
Sterner RW, Elser JJ, Fee EJ, Guildford SJ, Chrzanowski TH (1997b) The light: nutrient ratio in lakes: the balance of energy and materials affects ecosystem structure and process. Am Nat 150(6):663–684
Stock BC, Semmens BX (2013) MixSIAR GUI user manual: version 1.0. Available from http://conserver.iugo-cafe.org/user/brice.semmens/MixSIAR. Accessed 13 Aug 2020
Summerhayes VS, Elton CS (1923) Contributions to the ecology of Spitsbergen and Bear Island. J Ecol 11:214–286
Taylor SL, Bishop MJ, Kelaher BP, Glasby TM (2010) Impacts of detritus from the invasive alga, Caulerpa taxifolia on a soft sediment community. Mar Ecol Prog Ser 420:73–81
Toda H, Wada E (1990) Use of 15 N/14 N rations to evaluate the food source of the mysid, Neomysis intermedia Czerniawsky, in a eutrophic lake in Japan. Hydrobiologia 194(1):85–90
Trebilco R, Baum JK, Salomon AK, Dulvy NK (2013) Ecosystem ecology: size-based constraints on the pyramids of life. Trends Ecol Evol 28(7):423–431
Vander Zanden M, Fetzer WW (2007) Global patterns of aquatic food chain length. Oikos 116(8):1378–1388
Vander Zanden MJ, Rasmussen JB (1996) A trophic position model of pelagic food webs: impact on contaminant bioaccumulation in lake trout. Ecol Monogr 66(4):451–477
Vander Zanden MJ, Vadeboncoeur Y (2002) Fishes as integrators of benthic and pelagic food webs in lakes. Ecology 83(8):2152–2161
Vander Zanden MJ, Cabana G, Rasmussen JB (1997) Comparing trophic position of freshwater fish calculated using stable nitrogen isotope ratios (δ15N) and literature dietary data. Can J Fish Aquat Sci 54(5):1142–1158
Vander Zanden MJ, Shuter BJ, Lester N, Rasmussen JB (1999) Patterns of food chain length in lakes: a stable isotope study. Am Nat 154(4):406–416
Vaslet A, Phillips DL, France C, Feller IC, Baldwin CC (2012) The relative importance of mangroves and seagrass beds as feeding areas for resident and transient fishes among different mangrove habitats in Florida and Belize: evidence from dietary and stable-isotope analyses. J Exp Mar Biol Ecol 434:81–93
Vinagre C, Salgado J, Costa MJ, Cabral HN (2008) Nursery fidelity, food web interactions and primary sources of nutrition of the juveniles of Solea solea and S senegalensis in the Tagus estuary (Portugal): a stable isotope approach. Estuarine Coast Shelf Sci 76(2):255–264
Vinagre C, Salgado J, Cabral HN, Costa MJ (2011) Food web structure and habitat connectivity in fish estuarine nurseries—impact of river flow. Estuaries Coasts 34(4):663–674
Vinagre C, Salgado JP, Mendonça V, Cabral H, Costa MJ (2012) Isotopes reveal fluctuation in trophic levels of estuarine organisms, in space and time. J Sea Res 72:49–54
Vivekanandan E, Gomathy S, Thirumilu P, Meiyappan MM, Balakumar SK (2009) Trophic level of fishes occurring along the Indian coast. J Mar Biol Assoc India 51(1):44–51
Ward EJ, Semmens BX, Schindler DE (2010) Including source uncertainty and prior information in the analysis of stable isotope mixing models. Environ Sci Technol 44:4645–4650
Wille M, Babechuk MG, Kleinhanns IC, Stegmaier J, Suhr N, Widdowson M, Kamber BS, Schoenberg R (2018) Silicon and chromium stable isotopic systematics during basalt weathering and lateritisation: a comparison of variably weathered basalt profiles in the Deccan Traps, India. Geoderma 314:190–204
Winemiller KO (2005) Life history strategies, population regulation, and implications for fisheries management. Can J Fish Aquat Sci 62(4):872–885
Worm B, Lotze HK, Hillebrand H, Sommer U (2002) Consumer versus resource control of species diversity and ecosystem functioning. Nature 417(6891):848–851
Acknowledgements
The authors gratefully acknowledge the Director Indian Council of Agricultural Research (ICAR)—Central Institute of Fisheries Education, Mumbai for providing necessary facilities and financial support to carry out this work. The authors express sincere gratitude to the Director, Birbal Sahni Institute of Paleosciences, Lucknow, for providing the necessary laboratory facilities and Director ICAR- Central Coastal Agricultural Research Institute, Goa for providing the technical support for accomplishing the present work. The authors also express their heartfelt thanks to the fishermen along Naigaon, Vasai, for their kind cooperation with the experimental fishing.
Funding
This work was supported by the PhD fellowship granted by ICAR-Central Institute of Fisheries Education to one of the co-author Dhanya Mohan Lal.
Author information
Authors and Affiliations
Contributions
Dhanya Mohan Lal: conceptualization; data collection, data analysis, and original draft preparation. Giri Bhavan Sreekanth: conceptualization, data analysis, and manuscript preparation. Chitra Soman: data collection and analysis. Anupam Sharma: methodology, laboratory supervision, and data interpretation. Zeba Jaffer Abidi: overall supervision and manuscript editing.
Corresponding author
Ethics declarations
Ethical approval
The authors declare that preparation of this manuscript has been duly considered the ethical aspects of manuscript publication. The authors also declare that the manuscript is prepared strictly according to the Journal format as provided in the instruction to authors.
Consent to participate
The present research does not involve any human participants.
Consent for publication
All the co-authors have agreed for submission of this manuscript to “Environmental Science and Pollution Research.”
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: V.V.S.S. Sarma
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
Matrix plots
The matrix plots obtained from the SIMMR analysis from the URE represented the source histograms on the diagonal, correlation between the sources on the lower diagonal, and contour plots of the relationship between the sources on the upper diagonal (Fig. 9).
9). Large negative correlations indicate that the model cannot discern between the two sources; they may lie close together in iso-space. In the present study, the largest negative correlation is found between mangrove leaves and detritus since they are closer in the iso-space than the other sources. These relations indicate that the model is constrained with some degree of difficulty in segregating the contribution of these sources to the overall food web.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lal, D.M., Sreekanth, G.B., Soman, C. et al. Delineating the food web structure in an Indian estuary during tropical winter employing stable isotope signatures and mixing model. Environ Sci Pollut Res 30, 49412–49434 (2023). https://doi.org/10.1007/s11356-023-25549-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-25549-w