Abstract
Methotrexate (MTX) and azathioprine (AZA) are chemotherapeutic, immunosuppressive, cytotoxic drugs with reported adverse effects, including oxidative damage to testis. This study aims to evaluate the potential effect of grape seed extract (GSE; gervital) to prevent testicular damage caused by MTX and AZA. Male albino rats were separated into six groups: group I, normal control group; group II, GSE (150 mg/kg/day); group III, MTX (8 mg/kg/week); group IV, AZA (15 mg/kg/day); group V, GSE (150 mg/kg/day) + MTX (8 mg/kg/week); group VI, GSE (150 mg/kg/day) + AZA (15 mg/kg/day). All rats were sacrificed, blood samples were obtained for testosterone analysis, and testis was removed for histological and ultrastructural studies and oxidation measurements. A reduction in relative body and testis weight, along with a significant decrease in testosterone levels, was observed. Histopathological and ultrastructural alterations induced by MTX or AZA included reduced spermatozoa, sloughing, marked reduction of spermatogenic cells, and pyknosis of some nuclei. Significant oxidative stress manifested as reduced glutathione (GSH) levels and catalase (CAT) and superoxide dismutase (SOD) activities, as well as increased malondialdehyde (MDA) levels. GSE administration showed an ameliorative effect on testosterone levels and histopathological and ultrastructural changes. GSE treatment also suppressed the increases in MDA levels and the decreases in GSH levels and CAT and SOD activities. In conclusion, these findings confirm that GSE is an effective antioxidant that protects testis from histopathological and ultrastructural damage induced by MTX and AZA. Therefore, GSE is a promising candidate for future use to minimize and alleviate MTX and AZA risks.
Graphical abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Chemotherapy is the utilization of chemical agents to suppress or kill proliferating cancer cells through several mechanisms, including cell membrane damage, intercalation into DNA, inhibition of DNA replication, or free radical generation. The target of anticancer drugs is not only the tumor but also other cells, thus causing the same deterioration among both defective and normal cells (Granados-Principal et al. 2010).
Methotrexate (MTX) and azathioprine (AZA) are classified as cytotoxic drugs and are utilized in the treatment of cancer. They have devastating reported side effects, such as damage to DNA/RNA and body organs (Elelaimy et al. 2012). AZA and MTX increase oxidative stress (OS) in testicular tissues and decrease testosterone hormones, and both have a destructive effect on the functions and morphology of the testes in adult Wistar rats, including the disruption of seminiferous tubules (Akinlolu et al. 2014).
As mentioned, MTX is commonly utilized as a cytotoxic chemotherapeutic agent for the treatment of certain cancers (Zhang et al. 2022) and causes several side effects if used for long or short periods (Alahmadi and Abduljawad 2021). Numerous studies have documented the role of apoptotic cell death driven by MTX-associated testicular impairment (Maremanda and Jena 2017). Previous research has found that MTX causes a decrease in the diameter of the lumen of the seminiferous tubule, and the degeneration and shedding of germ cells (Liu et al. 2015).
AZA is a cytotoxic immunomodulatory drug that is commonly used to treat inflammatory bowel disease, autoimmune disorders, organ transplant rejection, and cancer (Reggio et al. 2019; Bergasa 2022). AZA leads to interstitial spaces being poorly defined, inadequate spermatogenic cells and spermatozoa, and disrupted and shrunken seminiferous tubules (Shaikh et al. 2020b).
Antioxidants are considered an important line of defense against free-radical destruction and help maintain ideal health and well-being. They play a key role against reactive oxygen species (ROS) in the body’s defense system (Hasanuzzaman et al. 2020). Grapes (Vitis vinifera) are one of the most frequently consumed fruits in the world and are among the world’s major fruit crops (Raja et al. 2020).
Grape seed extract (GSE), also known as gervital, is a potent antioxidant compound because it can surmount superoxide radicals in living cells through its primary proanthocyanidin components. It protects cells from harmful diseases and is considered to be anti-diabetic, anti-tumor, anti-microbial, anti-aging, and anti-inflammatory (Kwatra 2020). This study aimed to evaluate the potential role of GSE in preventing testicular damage resulting from MTX or AZA, and clarify the ultrastructural changes induced by AZA since little is known about it from previous studies.
Materials and methods
Drug and treatment
MTX tablets containing 2.5 mg were obtained from Orion Corporation (Espoo, Finland). AZA tablets containing 50 mg were purchased from RPG Life Sciences Limited (Mumbai, India). GSE proanthocyanidin capsules containing 150 mg were purchased from Enshas-Sharkeya (Egypt) Pharmaceutical and Medicinal Plants Arab Company (MEPACO–MEDIFOOD).
Animals
A total of 36 male albino rats, 140–200 g each and aged 6–8 weeks, were collected from the Nahda University Animal House Facility Center. All experimental rats were housed and maintained in a clean and well-ventilated rodent room under standard temperature (25 ± 5 °C) and humidity conditions, with 12-h dark/light cycles, and were provided free access to a standard diet of pellets and tap water. They remained under observation for 15 days before the experiment began to rule out any intercurrent infections. The animals received care in compliance with the recommendations of the Committee for the Control Purpose. Animal care was in accordance with the European Community Directive (86/609/EEC Edition 8). This study was previously approved by the Committee of Zoology, Beni-Suef University, Egypt. IACUC (permit number BSU/FS/2018/1).
Experimental design
Animals were randomly allocated into six groups, as demonstrated in Table 1. The dosage regimen of MTX (8 mg/kg/week) and AZA (15 mg/kg/day) is in accordance to previous research (Akinlolu et al. 2014), and the dose of GSE (150 mg/kg/day) was selected according to previous research (Bagchi et al. 2001).
Sample collection and biochemical estimation
The rats were sacrificed after day 35 of the experiment under light diethyl ether anesthesia. Each rat had been examined weekly for bodyweight assessment. Blood samples were taken from the retro-orbital venous plexus into serum tubes with the aid of a capillary tube (Kumar et al. 2011). Sera were separated by centrifugation for effect on serum testosterone according to the method of Maruyama et al. (1987).
Tissue homogenate preparation and oxidative stress–related marker assay
The testes were rapidly excised from surrounding tissues and weighed. Testis homogenate (25%) was maintained in isotonic ice-cooled normal saline. The resulting homogenate was centrifuged for 15 min at 10,000 × g. The supernatant was preserved at − 20 °C before analysis. The testicular homogenate was used for malondialdehyde (MDA) estimation according to Altintas et al. (2014). Testicular superoxide dismutase (SOD) and catalase (CAT) activities were measured (Marklund & Marklund 1974; Claiborne 1985), and glutathione (GSH) was calculated using the method of Sedlak and Lindsay (1968).
Histological examination
Left testis tissues were cut into 0.5-cm3 pieces and then fixed for 24 h in 10% neutral formalin buffer. Testis specimens were cleaned to remove the residual fixative and then dehydrated for 45 min each in ascending grades of ethanol, then for 30 min each in two levels of absolute ethyl alcohol. This was followed by clearance of 30 min each in two xylene changes. The tissues were then soaked for 3 h at 60 °C with paraplast plus (three changes) and then embedded in paraplast plus. For histopathological tests, 4–5-μm-thick sections were stained with hematoxylin and eosin (Bancroft and Gamble 2008).
Ultrastructural examination
Testis specimens were cut into pieces measuring approximately 1 mm3 and immediately fixed for 18–24 h in fresh 3% glutaraldehyde-formaldehyde at 4 °C. The specimens were then washed in a phosphate buffer (pH 7.4) and then set for 1 h at 4 °C in isotonic 1% osmium tetroxide (Bain and Mercer 1966). Alcohol series dehydration was performed. The specimens were then transmitted through a propylene oxide solution two times for 10 min each. Embedding of the specimens in Epon epoxy resin began by infiltrating the specimens in a propylene oxide–resin mixture overnight. The specimens were transferred to fresh-resin capsules for polymerization to obtain hard blocks. Semithin sections were cut using an ultracut Reichert-Jung ultramicrotome from these blocks with the aid of glass knives and were then stained with toluidine blue stain. According to prior research (Bozzola and Russell 1999), ultrathin sections were then prepared and stained with uranyl acetate and lead citrate, and then examined with a JEOL CX 100 transmission electron microscope operating at 60 kV.
Statistical analysis
The Statistical Package for the Social Sciences (version 20.0 for Windows; IBM, Armonk, NY, USA) was used. The results were elucidated as the mean ± standard error, and all statistical comparisons were calculated using one-way ANOVA testing (Rao et al. 1985). This was followed by Duncan’s multiple range test analysis. P < 0.05 was considered statistically significant.
Results
Body weight
MTX and AZA therapy affected the percentage of change between the two body weights, where the male albino rats suffered from a marked decrease (P < 0.05) as compared with the control and GSE groups, while GSE showed an insignificant increase in body weights in the MTX plus GSE, and AZA plus GSE groups, in comparison with MTX and AZA, respectively (Fig. 1).
Percentage changes between initial and final body weights in control, gervital (GSE), methotrexate (MTX), azathioprine (AZA), gervital plus methotrexate (GSE + MTX), and gervital plus azathioprine (GSE + AZA) groups. Values with different superscript letters are considered significantly different (P < 0.05). Groups with the same superscript letter do not differ significantly
Testis relative weight
MTX and AZA therapy affected relative testis weight, where the therapy rats exhibited a marked decrease (P < 0.05) in relative testis weight when compared with the control, GSE, MTX plus GSE, and AZA plus GSE groups. In addition, co-administration of MTX with GSE, or AZA with GSE, induced significant increases in relative testis weight compared with the two groups which were treated with MTX or AZA only (Fig. 2).
Changes in testis relative weights in control, gervital (GSE), methotrexate (MTX), azathioprine, gervital plus methotrexate (GSE + MTX), and gervital plus azathioprine (GSE + AZA) groups. Values with different superscript letters are considered significantly different (P < 0.05). Groups with the same superscript letter do not differ significantly
Effect on serum testosterone
A significant decrease in testosterone (P < 0.05) in the MTX and AZA groups was observed in comparison with the control, GSE, MTX plus GSE, and AZA plus GSE groups. GSE treatment in the MTX plus GSE, and AZA plus GSE groups produced a significant augmentation of testosterone in both MTX and AZA-treated groups, compared with the two groups which were treated with MTX or AZA only (Fig. 3).
Changes in testosterone in control, gervital (GSE), methotrexate (MTX), azathioprine (AZA), gervital plus methotrexate (GSE + MTX), and gervital plus azathioprine (GSE + AZA) groups. Values with different superscript letters are considered significantly different (P < 0.05). Groups with the same superscript letter do not differ significantly
Testis antioxidant parameters
Lipid peroxidation (LP), articulated as MDA concentration, was measured as a biomarker of testis OS state. It demonstrated a significant increase in MTX and AZA-treated groups when compared to control, GSE, MTX plus GSE, and AZA plus GSE-treated groups. GSE treatment in MTX plus GSE, and AZA plus GSE groups, produced a marked LP level decrease in comparison with MTX and AZA-treated groups, respectively. Concerning the enzymatic and non-enzymatic antioxidant defense system, CAT, SOD, and GSH all revealed noticeable reductions in the MTX and AZA groups (P < 0.05). GSE in MTX plus GSE, and AZA plus GSE groups significantly increased the activities of these antioxidant enzymes and increased GSH levels, respectively, in the treated groups (Tables 1 and 2).
Histopathological changes
Examination of testicular tissue of both control and GSE-treated animals revealed normal seminiferous tubules with active spermatogenesis, spermatogonia, and triangular Sertoli cells resting upon the basement membrane. In addition, Leydig cells and clusters of spermatozoa were seen within the lumen. Primary spermatocytes were recognized by their large nuclei containing coarse clumps of chromatin. Furthermore, spermatids appeared with rounded nuclei in the control (Fig. 4a, b) and GSE (Fig. 4c, d).
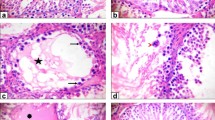
A photomicrograph of testis sections of control rats revealing normal seminiferous tubules (arrow) with active spermatogenesis and spermatogonia (SG) resting upon the basement membrane (a, c). Interstitial tissue containing Leydig cell (LC) and a cluster of spermatozoa (SP) in the lumen (Lu) is seen. High magnification showing primary spermatocytes (PS), recognized by their large nuclei containing coarse clumps of chromatin (b, d). Spermatids (SD) with rounded nuclei and spermatozoa (SP) were observed. Triangular Sertoli cell (SC) and spermatogonia (SG) at the basement membrane and the interstitial tissue containing Leydig cell (LC) are seen. e Photomicrograph of testis sections of rats treated with methotrexate showing severe degenerated and variable-sized seminiferous tubules (thin arrow), where many tubules appeared with a marked decrease in the spermatogenic cells and few or no sperms. Detachment of spermatogenic cells from the basal lamina (arrowhead) and vacuolated cytoplasm (V) and noticeable sloughing of spermatogenic cells into the lumen of seminiferous tubules (thick arrow) are seen. f Photomicrograph of testis sections of rats treated with azathioprine revealing degenerated seminiferous tubules with ruptured basement membrane (*). Notice the detachment of spermatogenic cells from the basal lamina (arrowhead) and vacuolated cytoplasm (V). Reduction of spermatogenic cells and pyknosis of some nuclei (arrow) are also observed. Interstitial tissue (IT) containing degenerated Leydig cell (LC). g Photomicrograph of testis sections of rats treated with methotrexate and gervital showing approximate recovery of seminiferous tubules. Amelioration of spermatogenic cells and spermatozoa (SP) except a few vacuoles (V) and normal Leydig cells are seen. h Photomicrograph of testis sections of rats treated with azathioprine plus gervital revealing improvement of spermatogenesis and nearly normal structure in most seminiferous tubules. Compact spermatogenic layers with few degenerative germ cells, except a few vacuoles (V) and normal Leydig cells (LC), are seen
Animals treated with MTX showed severely degenerated and variably sized seminiferous tubules, and many tubules appeared with a marked decrease in spermatogenic cells and few or no sperm. Detachment of spermatogenic cells from the basal lamina and vacuolated cytoplasm was also observed, and there was sloughing of spermatogenic cells into the lumen of the seminiferous tubules (Fig. 4e).
Animals treated with AZA revealed degenerated seminiferous tubules with ruptured basement membranes. The detachment of spermatogenic cells from the basal lamina and vacuolated cytoplasm was noticed, and the reduction of spermatogenic cells and pyknosis of some nuclei were also observed (Fig. 4f).
Examination of the testes of animals treated with GSE plus MTX revealed some recovery of seminiferous tubules, amelioration of spermatogenic cells, and spermatozoa, although few vacuoles were observed. In addition, normal Leydig cells were seen in the interstitial tissue (Fig. 4g). Examination of testes of animals treated with GSE and AZA showed improved spermatogenesis, and nearly normal structure in most seminiferous tubules. Compact spermatogenic layers with few degenerative germ cells and normal Leydig cells were also observed (Fig. 4h).
Ultrastructural changes
The electron microscopic examination of the control testis revealed spermatogonia resting on the basement membrane, myoid cells, Sertoli cells with triangular nuclei, primary spermatocytes with mitochondria, and large spherical nuclei (Fig. 5a). In addition, there were rounded spermatids with spherical nuclei, acrosomal cap, and peripherally located mitochondria observed (Fig. 5b). Also, lumen containing normal spermatozoan at the midpiece and tail region were observed (Fig. 5c). Normal interstitial tissue containing Leydig cells with a large nucleus, thin rims of chromatin, prominent nucleolus, lipid droplets, and capillaries were also seen (Fig. 5d).
a–d Electron micrograph of a portion of a seminiferous tubule of control testis showing a the spermatogonia (SG) resting on the basement membrane (BM), myoid cell (MC), the Sertoli cell (SC) having a triangular nucleus, and primary spermatocyte (PS) with mitochondria (M) and large spherical nucleus. b Normal-looking early rounded spermatid (SD) with spherical nuclei, acrosomal cap (AC), and peripherally located mitochondria (M). c Lumen (Lu) contains normal spermatozoan (SP) at the mid-piece and tail region. d Normal interstitial tissue containing Leydig cell (LC) with large nucleus (N), thin rim of chromatin, prominent nucleolus, lipid droplet (Li), and capillaries were observed
The ultrastructure mitochondria in spermatogonia, Sertoli cells, and primary spermatocytes. The observation of the testes from animals which had been administered MTX revealed many vacuoles and vacuolated lysosomes in the Sertoli cells, and spermatogonia was seen (Fig. 6a). Distorted spermatid with marked cytoplasmic vacuolation and rarified cytoplasm was surrounded by a large lytic area, and presence of some lysosome and some degenerated mitochondria was observed (Fig. 6b). A marked decrease in the number of sperm in the lumen of the seminiferous tubule was seen (Fig. 6c), and abnormal interstitial tissue with degenerated Leydig cells with an irregular nucleus having a thick rim of heterochromatin, few lipids drops, and dilated smooth endoplasmic reticulum were also observed (Fig. 6d).
a–d Electron micrograph of a portion of a seminiferous tubule of testis treated with methotrexate showing a many vacuoles (V), vacuolated mitochondria (M) in addition to the presence of some lysosome (LY) in spermatogonia (SG), Sertoli cell (SC), and primary spermatocyte (PS). b Distorted spermatid (SD) with marked cytoplasmic vacuolation (V) and rarified cytoplasm surrounded with the large lytic area (*). Notice the presence of some lysosome (LY) and some degenerated mitochondria (M). c Marked decrease in the number of sperms (SP) in the lumen (Lu) of the seminiferous tubule. d Degenerated Leydig (LC) cell with irregular nucleus (N), a thick rim of heterochromatin, few lipid droplets (Li), and dilated smooth endoplasmic reticulum (arrow) was also observed
The ultrastructure observation of the animal testes of AZA-treated group showed many vacuoles in both Sertoli cells and spermatogonia cells, and an overall decrease in cytoplasmic ground substance (Fig. 7a). Thick ruptured irregular basement membranes, vacuolated primary spermatocytes, and severe degenerated Sertoli cells with many vacuoles and some lysosomes were detected (Fig. 7b). There was distorted and vacuolated early spermatid. The dissolution of some cells was noticed (Fig. 7c). Degenerated vacuolated spermatid and dissolution of some cells were also observed (Fig. 7d), in addition to degenerated Leydig cells. Noteworthy were the irregular nuclear envelopes with dark clumps of heterochromatin adjacent to the nuclear membranes, as well as the presence of few lysosomes (Fig. 7e).
a–e Electron micrograph of a portion of a seminiferous tubule of testis treated with azathioprine showing a degenerated Sertoli (SC) cells and spermatogonia cells (SG) with many vacuoles (V) and an overall decrease in cytoplasmic ground substance. b Thick irregular basement membrane (BM). Vacuolated (V) primary spermatocyte (PS) and severe degenerated Sertoli cells (SC) with many vacuoles (V) and many lysosomes (LY) were seen. c Degenerated and vacuolated (V) spermatid (SD). Notice the dissolution of some cells (D). d Abnormal spermatid (SD) with vacuolated cytoplasm (V), degenerated nucleus, and some lysosomes (LY) in addition to an obvious decrease in the number of sperms (SP) in the lumen (Lu). e Abnormal interstitial tissue with degenerated Leydig cell (LC), its nucleus (N) with dark clumps of heterochromatin, and presence of few lysosomes (LY)
The ultrastructure observation of the testes from animals which had been administered GSE and MTX showed amelioration in the structure of the spermatogonium, Sertoli cells, primary spermatocytes, and few degenerated mitochondria (Fig. 8a). Normal spermatid appeared with a rounded nucleus and peripherally located mitochondria except for a few cytoplasmic vacuolations and a few lysosomes (Fig. 8b). Lumen were seen with marked recovery in transverse sections, and normal sperm at midpiece and tail regions (Fig. 8c). Leydig cells with large nuclei, smooth endoplasmic reticulum, lipid droplets, and blood capillaries were also seen (Fig. 8d).
a–d Electron micrograph of a portion of a seminiferous tubule of testis treated with methotrexate plus gervital showing a amelioration in the structure of spermatogonium (SG), Sertoli cell (SC), and primary spermatocyte (PS) except few degenerated mitochondria (M). b Normally rounded spermatid (SD) with a rounded nucleus and peripherally located mitochondria (M) except few cytoplasmic vacuolations (V) and few lysosomes. c Lumen (Lu) with marked recovery in transverse sections of normal sperms (SP) at the mid-piece and tail region. d Normal interstitial tissue that contained Leydig cells with a large nucleus (N), prominent nucleolus, smooth endoplasmic reticulum, lipid droplets (Li), and blood capillaries (C)
The ultrastructure observation of testes from animals which had been administered GSE and AZA revealed a relatively normal basement membrane, improvement in the structure of cells except vacuolation in Sertoli cells, spermatogonia, primary spermatocyte, and spermatid with rounded nuclei and normal acrosomal cap (Fig. 9a). In addition, a round spermatid appeared with an acrosomal cap, peripherally located mitochondria, and few cytoplasmic vacuolations (Fig. 9b). Round spermatids and a moderate number of cross-sections at the midpiece and tail region of sperm were also observed in the lumen (Fig. 9c). Interstitial tissue and Leydig cells with normal nuclei and a moderate number of lipid droplets were also observed (Fig. 9d).
a–d Electron micrograph of a portion of a seminiferous tubule of testis treated with azathioprine plus gervital showing a slightly normal basement membrane (BM), improvement in the structure of cells except vacuolation in Sertoli cell (SC), spermatogonia (SG), primary spermatocyte, and spermatid (SD) with a rounded nucleus and normal acrosomal cap (AC). b Normal-looking early rounded spermatid (SD) with a rounded nucleus, acrosomal cap (AC), and peripherally located mitochondria (M) with few cytoplasmic vacuolation (V). c Spermatid and a moderate number of cross-sections at mid-piece and tail region of sperms (SP) in the lumen (LU). d Interstitial tissue and Leydig cell with normal nucleus (N) and a moderate number of lipid droplets (Li)
Discussion
Testis is affected by MTX and AZA as chemotherapeutic drugs, where they induce OS, apoptosis, and histopathological changes. This research primarily focused on analyzing the antioxidant and anti-apoptotic properties of GSE which improve OS and protect against testicular injury.
MTX belongs to the anti-metabolic class of chemotherapeutic agents. As an analog of folic acid, MTX inhibits dihydrofolate reductase that is required for DNA synthesis (Rajagopalan et al. 2002). AZA is a prodrug, which is converted into the active metabolite of 6-mercaptopurine and 6-thieosinic acid in the body that impairs the immune response and inhibits purine synthesis (Abd-Elfatah et al. 2021). Grape seed extract contains a high value of polyphenols which are powerful antioxidants. GSE is a remarkable biological agent with promising chemotherapeutic and chemopreventive effects. GSE is a clinical alternative for its antioxidant, anti-inflammatory, nerve-protecting, heart-protecting, kidney-protecting, and anti-ovulatory properties (Khan et al. 2021).
Chemotherapy drugs can cause testicular apoptosis, which is an obviously harmful side effect (Abu-Risha et al. 2022). GSE is a powerful antioxidant as demonstrated by previous studies (Abdul-Hamid et al. 2022; Priyadarshi et al. 2022) which compared the efficacy of GSE and Ginkgo biloba to reduce hepatotoxicity induced by antidysrhythmic drugs. The present study revealed a significant reduction in both body weight and relative testes weight in MTX-treated rats when compared with the control and GSE groups. Similar results were observed by Arisha (2017) who demonstrated that the reduction may reflect direct toxicity, low feed intake, and water intake. Animals exhibited diarrhea, the probable cause of which could be the corrosive and irritating impact of MTX on gastrointestinal mucosa, as reported by Patel et al. (2014). This study manifested decreases in both body weight and relative testis weight in association with AZA, and this was also reported recently by Shaikh et al. (2020a). Testis relative weight returned to near normality after treatment with GSE, and this concurs with a study conducted by Hajizadeh et al. (2016) who found that male albino rats treated with GSE after treatment with fluoxetine significantly increased testis relative weight. Testes are the most seriously affected goal organs by OS as a result of their high polyunsaturated membrane lipid content (Belhan et al. 2017). An imbalance between the formation and elimination of free radicals can produce a pathological condition (Bhattacharya 2015). OS can harm biological molecules, such as lipids, proteins, polysaccharides, and DNA (Yüncü et al. 2019). The present study observed disruptions in testis oxidation associated with MTX or AZA which were manifested by increased MDA levels, decreased GSH production, and reduced CAT and SOD activity. This concurs with the findings of Akinlolu et al. (2014). In testicular development and spermatogenesis, SOD plays a critical function. Changes in this enzyme can cause sperm development to cease and testicular function to be affected (Prahalathan et al. 2004). In the present study, male albino rats treated with GSE plus MTX, or rats treated with GSE plus AZA, revealed a decrease in MDA levels and an increase in GSH, CAT, and SOD values in accordance with Hui et al. (2020) who confirmed the radical scavenger activity of GSE. The present study revealed decreases in testosterone levels in MTX or AZA-treated group. The production and secretion of testosterone drops due to the loss of sexual cells, Sertoli cell damage, atrophy of interstitial cells, and OS-caused decreases in the anabolic effect of testosterone (Ramadan et al. 2018). Treatment with GSE plus MTX restored serum testosterone levels to normal, and these results concurred with Arisha (2017) who found that treatment with GSE following MTX administration restored serum testosterone levels to normal. The present study also found that treatment with GSE following AZA exposure restored serum testosterone levels to normal. Similarly, Mohamadpour et al. (2020) reported that GSE enabled the resumption of normal levels of serum testosterone. The present light microscopic examination revealed that the testis of MTX or AZA-exposed rats demonstrated histopathological changes in the seminiferous tubules when compared with the control and GSE groups. This agreed with the prior results of Akinlolu et al. (2014). In the present study, the MTX-exposed group showed degenerated and variable-sized seminiferous tubules, with a marked decrease in spermatogenic cells. A similar result was seen by Shrestha et al. (2007), who indicated that the size of seminiferous tubule cellular contents changed significantly and that this may be because primary spermatocytes and spermatids were unsuccessful in DNA replication because of the inhibition of dihydrofolate reductase, an important enzyme essential for normal DNA synthesis. This study observed many tubules with damage to the structure of germ cells. Moreover, Ateşşahin et al. (2006) found that damage to the structure of germ cells was caused by OS. The current AZA-treated group revealed degenerated seminiferous tubules, marked decrease in spermatogenic cells, pyknotic nuclei, and vacuolated cytoplasm. The cytoplasmic vacuolation may be due to Sertoli cell damage since Sertoli cells control the spermatogenic process as explained in a recent research by Abdelbaky et al. (2020). The present ultrastructural study of the MTX group revealed several extreme changes in spermatogonia and Sertoli cells. Richburg (2000) observed that when Sertoli cell numbers diminish, the number of germinal cells declines dramatically. The present study also revealed cytoplasmic vacuolations and marked decreasing of sperm number in the lumen of seminiferous tubules. Reduced relative testicular weight, damage to sperm DNA, as well as sperm count (Padmanabhan et al. 2009) and spermatozoa have all been highly susceptible to LP resulting from ROS due to high polyunsaturated fatty acid content, as seen by Alvarez et al. (1987). The current ultrastructural study of the AZA group revealed a germ cell with pyknotic nuclei and vacuolated cytoplasm. Degenerated Sertoli cells and spermatogonia cells with many vacuoles were also observed. This result is in agreement with Karawya and El-Nahas (2006) who demonstrated that the decrease in testis weight is indirectly indicative of the effect on spermatogenesis. The current study also revealed a marked decrease in the number of sperm in the lumen. Similar results were observed by Onanuga et al. (2014). The present light microscopic examination and ultrastructural study of testis treated with GSE plus MTX, and testis treated with GSE plus AZA, showed nearly normal structures in most seminiferous tubules, improvement of spermatogenic cells, Sertoli cell, and primary spermatocytes, except for a few degenerated mitochondria. These results are similar to several studies conducted on the use of antioxidant substances to reduce the side effects of MTX (Yucel et al. 2017; Yüncü et al. 2019). Furthermore, Arisha (2017) concluded that GSE improved the histology and ultrastructure of testes resulting from toxicity induced by MTX. Another study found that GSE prevents AZA toxicity in male albino rats (El-Ashmawy et al. 2010). The current study agreed with another study that showed that GSE possesses cytoprotective ultrastructure and antitoxic effects in hepatocytes during CCL4 intoxication (Hovnanyan et al. 2014).
The current study has some limitations including the investigation of the exact mechanism of MTX or AZA-induced testicular damage and also the inflammatory and apoptotic biomarkers which could potentially reveal the anti-inflammatory and anti-apoptotic role of GSE.
Conclusion
The present study determined that GSE had preventive effects against MTX- or AZA-prompted testicular damage. This was confirmed through histological, ultrastructural, and biochemical studies. The preventive effects of GSE may be attributed to its antioxidant potential against free radicals. This study revealed marked amelioration after treatment with GSE on toxicity induced by MTX and AZA, but fewer improvements were observed in the MTX-treated group compared with the AZA-treated group.
Data availability
The authors confirmed that all data generated or analyzed during this study are included in this published article.
Change history
12 December 2022
Article is ineligible, the Funding text should be removed.
13 June 2024
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1007/s11356-024-33995-3
References
Abdelbaky NW, Abdelazem AZ, Hashem KS (2020) Thymoquinone attenuates 6-mercaptopurine induced testicular toxicity in albino rats: possible mechanisms are involved. Adv Anim Vet Sci 8(6):653–660. https://doi.org/10.17582/journal.aavs/2020/8.6.653.660
Abd-Elfatah, MH, Abd-Elhady, EE, El-Moslemany, AM (2021) Effect of dried kiwi and kumquat fruits against azathioprine induced liver toxicity in male albino rats. Egypt J Nutr 36:1–36. https://ejn.journals.ekb.eg/article_177743.html
Abdul-Hamid M, Abdel-Reheim, ES, Hegazy, W et al (2022) Effect of gervital in attenuating hepatotoxicity caused by methotrexate or azathioprine in adult albino rats. Environ. Sci Pollut Res 1–14. https://doi.org/10.1007/s11356-022-18903-x
Abu-Risha SE, Mousa MA, Elsisi AE (2022) Protective role of irbesartan against cyclophosphamide-induced testicular damage in rats via up-regulating PPAR-γ signaling and ameliorating NF-κB/NLRP3/IL-18 inflammatory axis. Life Sci 289:120218. https://www.sciencedirect.com/science/article/pii/S0024320521012054
Akinlolu A, Akinola O, Khobe P et al (2014) Azathioprine and methotrexate impaired the morphology and functions of the testes in adult wistar rats. J Morphol Sci 31(02):075–081. https://doi.org/10.4322/jms.057513.pdf
Alahmadi, Ahlam A, and Eman A Abduljawad (2021) Equisetum arvense L. extract ameliorates oxidative stress, inflammation and testicular injury induced by methotrexate in male rats. J Pharm Res Int 101–114. https://doi.org/10.4322/jms.057513
Altintas R, Polat A, Parlakpinar H et al (2014) The effect of melatonin on acetylsalicylic acid-induced kidney and testis damage. Hum Exp Toxicol 33(4):383–395. https://doi.org/10.1177/0960327113506240
Alvarez JG, Touchstone JC, Blasco L et al (1987) Spontaneous lipid peroxidation and production of hydrogen peroxide and superoxide in human spermatozoa superoxide dismutase as major enzyme protectant against oxygen toxicity. J Androl 8(5):338–348. https://doi.org/10.1002/j.1939-4640.1987.tb00973.x
Arisha SM (2017) Effect of grape seed extract, gervital, against methotrexate induced histological and ultrastructural alterations in testes of albino rats. World J Pharm Res 6 (7):98–126. https://wjpr.s3.ap-south-1.amazonaws.com/article_issue/1498804823.pdf
Ateşşahin A, Şahna E, Türk G et al (2006) Chemoprotective effect of melatonin against cisplatin-induced testicular toxicity in rats. J Pineal Res 41(1):21–27. https://doi.org/10.1111/j.1600-079X.2006.00327.x
Bagchi D, Ray S, Patel D et al (2001) Protection against drug- and chemical-induced multiorgan toxicity by a novel IH636 grape seed proanthocyanidin extract. Drugs Exp Clin Res 27(1):3–15. https://europepmc.org/article/med/11276828
Bain JM, Mercer F (1966) Subcellular organization of the developing cotyledons of Pisum sativum L. Aust J Biol Sci 19(1),49–68. https://www.publish.csiro.au/BI/BI9660049
Bancroft JD, Gamble M (2008) Theory and practice of histological techniques. Elsevier health sciences.https://books.google.com/books?hl=ar&lr=&id=CERPDwAAQBAJ&oi=fnd&pg=PP1&dq=Bancroft,+John+D,+and+Marilyn+Gamble+2008%09Theory+and+practice+of+histological+techniques:+Elsevier+health+sciences.&ots=ytXMqltoNQ&sig=XxTxALWNwukuvupdjCVIBnwOuQw
Belhan S, Özkaraca M, Kandemir FM et al (2017) Effectiveness of hesperidin on methotrexate-induced testicular toxicity in rats. Membr Biol 23 (5), 779–786. https://www.researchgate.net/profile/Fatih-Kandemir/publication/318311623_Effectiveness_of_Hesperidin_on_Methotrexate-Induced_Testicular_toxicity_in_rats/links/59628325a6fdccc9b1488f33/Effectiveness-of-Hesperidin-on-Methotrexate-Induced-Testicular-toxicity-in-rats.pdf
Bergasa NV (2022) Autoimmune hepatitis. Clinical cases in hepatology. Springer, 85–122. https://doi.org/10.1007/978-1-4471-4715-2_4
Bhattacharya S (2015) Reactive oxygen species and cellular defense system. Free radicals in human health and disease. Pp. 17–29: Springer. https://doi.org/10.1007/978-81-322-2035-0_2
Bozzola JJ, Russell LD (1999) Electron microscopy: principles and techniques for biologists. Jones & Bartlett Learning. https://books.google.com/books?hl=ar&lr=&id=zMkBAPACbEkC&oi=fnd&pg=PR21&dq=Bozzola,+John+J,+and+Lonnie+Dee+Russell+1999+Electron+microscopy:+principles+and+techniques+for+biologists:+Jones+%26+Bartlett+Learning.&ots=AdNVWmmFK4&sig=7ff0R-_uh_q6EKwJyZoKEp7rm5E
Claiborne A (1985) Handbook of methods for oxygen radical research. Florida: CRC Press, Boca Raton:283–284.
El-Ashmawy IM, Gad SB, Salama OM (2010) Grape seed extract prevents azathioprine toxicity in rats. Phytother Res 24(11):1710–1715. https://doi.org/10.1002/ptr.3200
Elelaimy I, Elfiky S, Hassan A et al (2012) Genotoxicity of anticancer drug azathioprine (Imuran): role of omega-3 (ω-3) oil as protective agent. J App Pharm Sci 2(4),14–23. http://www.japsonline.com/admin/php/uploads/421_pdf.pdf
Granados-Principal S, Quiles JL, Ramirez-Tortosa CL et al (2010) New advances in molecular mechanisms and the prevention of adriamycin toxicity by antioxidant nutrients. Food Chem Toxicol 48(6),1425–1438. https://www.sciencedirect.com/science/article/pii/S0278691510002218
Hajizadeh Z, Soleimani Mehranjani M, Najafi G et al (2016) Black grape seed extract modulates fluoxetine-induced oxidative stress and cytotoxicity in the mouse testis. Jundishapur J Nat. Pharm Prod 11(2). https://sites.kowsarpub.com/jjnpp/articles/18430.html
Hasanuzzaman M, Bhuyan M, Zulfiqar F et al (2020) Reactive oxygen species and antioxidant defense in plants under abiotic stress: revisiting the crucial role of a universal defense regulator. Antioxidants 9(8), 681. https://www.mdpi.com/783108
Hovnanyan K, Mamikonyan V, Margaryan A et al (2014) Antioxidants as effective remedies at hepatotoxic action of carbon tetrachloride. J Biochem. Biophys 2014. https://www.scirp.org/html/1-7100195_41999.htm
Hui WE, Li YZ, Fang PG et al (2020) Grape seed procyanidin extract attenuate sodium fluoride-induced oxidative damage and apoptosis in rat kidneys. Biomed Environ Sci. 33(6):454–457. http://www.besjournal.com/fileSWYXYHJKX/journal/article/swyxyhjkx/2020/6/PDF/19278.pdf
Karawya FS, El-Nahas AF (2006) The protective effect of vitamin C on azathioprine induced seminiferous tubular structural changes and cytogenetic toxicity in albino rats. Cancer Ther 4:125–34. https://www.academia.edu/download/51078224/The_protective_effect_of_vitamin_C_on_Az20161227-1274-18vkpy7.pdf
Khan R, Ali S, Mumtaz, S et al (2021) Ameliorating and pharmacological intervention potential of grape seed extract against lead- and cadmium-induced toxicity. International Journal of Environmental Science and Technology, 1–16. https://springerlink.bibliotecabuap.elogim.com/article/https://doi.org/10.1007/s13762-021-03541-6
Kumar AS, Deepthi KB, Prasad MDV et al (2011) Evaluation of the protective effects of omega-3 fatty acids against methotrexate induced testicular toxicity in male albino mice. Int J Phytopharm 2(2),48–52. https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.1065.5949&rep=rep1&type=pdf
Kwatra B. (2020) A review on potential properties and therapeutic applications of grape seed extract. World J Pharm. Res 9,2519–2540. https://www.researchgate.net/profile/Bharat-Kwatra/publication/341134392_A_REVIEW_ON_POTENTIAL_PROPERTIES_AND_THERAPEUTIC_APPLICATIONS_OF_GRAPE_SEED_EXTRACT/links/5eb06afa45851592d6b8c451/A-REVIEW-ON-POTENTIAL-PROPERTIES-AND-THERAPEUTIC-APPLICATIONS-OF-GRAPE-SEED-EXTRACT.pdf
Liu Z, Sun Y, Su L (2015) Effects of cisplatin on testicular enzymes and Sertoli cell function in rats. Fundamental Toxicological Sciences 2(4),137–145. https://www.jstage.jst.go.jp/article/fts/2/4/2_137/_article/-char/ja/
Maremanda KP, Jena G (2017) Methotrexate-induced germ cell toxicity and the important role of zinc and SOD1: investigation of molecular mechanisms. Biochem Biophys Res Commun. 483(1),596–601. https://www.sciencedirect.com/science/article/pii/S0006291X16321416
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47(3):469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Maruyama Y, Aoki N, Suzuki Y et al (1987) Sex-steroid-binding plasma protein (SBP), testosterone, oestradiol and dehydroepiandrosterone (DHEA) in prepuberty and puberty. Eur J Endocrinol 114(1),60–67. https://eje.bioscientifica.com/view/journals/eje/114/1/acta_114_1_009.xml
Mohamadpour M, Mollajani R, Sarabandi F et al. (2020) Protective effect of grape seed extract on dexamethasone-induced testicular toxicity in mice. Crescent J Med Biol Sci 7(1),59–65. http://eprints.bmsu.ac.ir/9068/
Onanuga IO, Ibrahim RB, Amin A et al (2014) Evaluation of the effects of ascorbic acid on azathioprine-induced alteration in the testes of adult Wistar rats. Int j biol chem sci. 8(2),426–433. https://www.ajol.info/index.php/ijbcs/article/view/107045
Padmanabhan S, Tripathi D, Vikram A et al (2009) Methotrexate-induced cytotoxicity and genotoxicity in germ cells of mice: intervention of folic and folinic acid. Mutat Res Genet Toxicol Environ Mutagen . 673(1),43–52. https://www.sciencedirect.com/science/article/pii/S1383571808003616
Patel N, Ghodasara D, Pandey S et al (2014) Subacute toxicopathological studies of methotrexate in Wistar rats. Vet World 7(7). http://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.587.1840&rep=rep1&type=pdf
Prahalathan C, Selvakumar E, Varalakshmi P (2004) Remedial effect of DL-α-lipoic acid against adriamycin induced testicular lipid peroxidation. Molecular and cellular biochemistry 267:209–214. https://doi.org/10.1023/B:MCBI.0000049385.13773.23
Priyadarshi R, Riahi Z, Rhim J.-W (2022) Antioxidant pectin/pullulan edible coating incorporated with Vitis vinifera grape seed extract for extending the shelf life of peanuts. Postharvest Biol Technol 183, 111740. https://www.sciencedirect.com/science/article/pii/S0925521421002799
Raja W, Dubey A, Verma P (2020) Evaluation of phytochemicals screening and antioxidant activity of vitis vinifera (grapes) fruit extract using fenton reaction. Eur J Biomed 7(7),582–587. https://www.researchgate.net/profile/Wasim-Raja-2/publication/344298218_Evaluation_of_Phytochemicals_Screening_and_Antioxidant_Activity_of_Vitis_Vinifera_Grapes_Fruit_Extract_using_Fenton_Reaction/links/5f648b8492851c14bc841130/Evaluation-of-Phytochemicals-Screening-and-Antioxidant-Activity-of-Vitis-Vinifera-Grapes-Fruit-Extract-using-Fenton-Reaction.pdf
Rajagopalan, PR, Zhang, Z, McCourt, L et al (2002) Interaction of dihydrofolate reductase with methotrexate: ensemble and single-molecule kinetics. Proc Natl Acad Sci 99, 13481–13486. https://www.pnas.org/doi/abs/10.1073/pnas.172501499
Ramadan, BK, Schaalan MF, Mahmoud ES( 2018) Protective effect of taurine on thiopurine-induced testicular atrophy in male albino rats. J Steroids Horm Sci 9(192). https://m.23michael.com/open-access/protective-effect-of-taurine-on-thiopurineinduced-testicular-atrophy-inmale-albino-rats-2157-7536-1000192.pdf
Rao, M, Blane K, Zonneberg M (1985) PC-STAT, one and two way analysis of variance. UGA Version 1A (C) copyright.
Reggio A, Spada F, Rosina M et al (2019) The immunosuppressant drug azathioprine restrains adipogenesis of muscle fibro/adipogenic progenitors from dystrophic mice by affecting AKT signaling. Sci Rep. 9(1):1–13. https://www.nature.com/articles/s41598-019-39538-y
Richburg JH (2000) The relevance of spontaneous- and chemically-induced alterations in testicular germ cell apoptosis to toxicology. Toxicol Lett. 112,79–86. https://www.sciencedirect.com/science/article/pii/S0378427499002532
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman's reagent. Anal Biochem 25, 192–20 https://www.researchgate.net/file.PostFileLoader.html?id=56cba9af6307d91d708b4567&assetKey=AS%3A332089951047681%401456187822637
Shaikh SP, Kumar H, Ali A et al (2020b) Ameliorative effect of zinc chloride in azathioprine induced reduction in body and testes weight in albino rats. JSMC. http://jsmc.pk/index.php/jsmc/article/view/322
Shaikh SP, Mohiuddin M, Kumar H et al (2020a) Protective effects of zinc chloride on azathioprine-induced toxicity on germinal epithelium of albino rats: a morphometric study. Pak J Surg 36: 83–89.https://www.pjs.com.pk/journal_pdfs/jan_mar20/83.pdf
Shrestha S, Dhungel S, Saxena A et al (2007) Effect of methotrexate (MTX) administration on spermatogenesis: an experimental on animal model. Nepal Med Coll J 9: 230–233. https://www.nmcth.edu/images/gallery/Editorial/Ry8oPs_shrestha.pdf
Zhang S, Ye C, Zhao W et al (2022) Product identification and toxicity change during oxidation of methotrexate by ferrate and permanganate in water. Front Environ Sci Eng 16:1–13. https://doi.org/10.1007/s11783-021-1501-8
Yucel Y, Oguz E, Kocarslan S et al (2017) The effects of lycopene on methotrexate-induced liver injury in rats. Bratisl Lek Listy 118(4), 212–216. https://avesis.medeniyet.edu.tr/yayin/ac33f305-3a8f-4280-b487-f4e07de7bc7d/the-effects-of-lycopene-on-methotrexate-induced-liver-injury-in-rats/document.pdf
Yüncü M, Bükücü N, Bayat N et al (2019) Protective effect of proanthocyanidin against methotrexate-induced testicle damage in rats. Eur J Ther 25(1), 51–57. https://www.researchsquare.com/article/rs-10900/latest.pdf
Acknowledgements
The authors acknowledge Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2022R5), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Funding
Princess Nourah bint Abdulrahman University Researchers Supporting Project number PNURSP2022R5, Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia
Author information
Authors and Affiliations
Contributions
M.A., E.S.A., and S.H.A. have contributed to suggesting the design of the work, preparation, and analysis of the results. A.A., S.I.O., and H.A. contributed to interpretation of data and discussion. W.H. has performed the experiments, analyzed the data and drafted the manuscript. All authors are in agreement with the contents of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experimental procedures were performed in accordance with recommendations, instructions, and guidelines of the regulatory committee. Animal care followed the Direction of the European Community (86/609/EEC Edition 8). This has been accepted by the Committee of Zoology, Beni-Suef University, Egypt. The IACUC Permit Number is BSU/FS/2018/1.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1007/s11356-024-33995-3
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdul-Hamid, M., Abdel-Reheim, E.S., Hegazy, W. et al. RETRACTED ARTICLE: Impact of gervital against histopathological, ultrastructural, and biochemical alterations caused by methotrexate or azathioprine in albino rat testis. Environ Sci Pollut Res 30, 21914–21926 (2023). https://doi.org/10.1007/s11356-022-23588-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23588-3