Abstract
The ambient environmental parameters have a great bearing on the morphology of living flora and fauna. In this study, we tested this hypothesis on one of the most dominant groups of living unicellular marine microorganism, benthic foraminifera, in the dynamic region of the southeastern Arabian Sea. The living benthic foraminifera from 43 surface samples collected between 25 and 2980 m of water depth were segregated into eight morphogroups (tapered/cylindrical, flattened-ovoid, biconvex, planoconvex, flattened-tapered, spherical, rounded-trochospiral, and rounded-planispiral). We report that the high organic carbon availability is combined with deficiency of oxygen results in benthic foraminifera with low surface area to volume ratio and mostly consists of tapered/cylindrical, flattened-ovoid forms, with a preference for infaunal habitat. However, the tests of the living benthic foraminifera thriving in the oxygen-rich bottom waters have a high surface area to volume ratio, commonly reported as epifaunal, consisting of biconvex and planoconvex forms. Additionally, we also report that the abundance of other morphogroups, namely flattened-tapered, spherical, rounded-trochospiral, and rounded-planispiral, is also controlled by the distinct environmental parameters. We suggest that the living benthic foraminifera are an excellent indicator of the ambient environmental parameters and can be used to reconstruct paleoenvironments.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Everything in the cosmos has a distinct outward shape, which is referred to as morphology. All morphologies, from galaxies (Kelkar et al. 2017) to microscopic organisms (Chien et al. 2012), are shaped by its surrounding environmental parameters. The environmental parameters also control the volume and the surface area of the organisms in an ecosystem and are often interpreted as its adaptability (Holt and Miller 2010). A successful ecosystem is the result of effective adjustment of the biotic and abiotic components, where biotic component is represented by the living fauna and flora, and the abiotic components include the physico-chemical parameters. In case of marine ecosystem, benthos is one of the most successful living groups in the world ocean. The success of marine benthos in occupying diverse habitats is highly coupled with their adaptability in different kind of environmental conditions. The exoskeleton bearing unicellular microorganism benthic foraminifera are one of the most diverse and ubiquitous marine benthic organisms. Benthic foraminifera are found in a variety of marine environments, including brackish and sometimes freshwater also (Gooday 2001; Saraswat and Nigam 2013). The widespread distribution of benthic foraminifera is due to their remarkable adaptability to a variety of environmental conditions. Benthic foraminifera develop a variety of morphologies that are typically influenced by their ambient environmental parameters (Gooday 2001; Prazeres et al. 2016; O’Brien et al. 2021). The distinct morphology representing a particular habitat as well as the excellent preservation of the exoskeleton makes benthic foraminifera as one of the best-suited and most frequently used groups to reconstruct the past environmental variations (Saraswat and Nigam 2013). The abundance, distribution, and morphology of benthic foraminifera in various oceanic settings are frequently linked to seawater environmental characteristics. Temperature, salinity, pH, water depth, dissolved oxygen, and food availability have all been identified as important environmental parameters affecting benthic foraminifera abundance and diversity (Jorissen et al. 1995; Singh et al. 2021; Suokhrie et al. 2021). Furthermore, benthic foraminifera have been shown to be one of the most prominent bio-indicators of marine pollution (Frontalini et al. 2015) due to their rapid response to ambient environmental changes.
Environmental parameters considerably modulate the morphology of benthic foraminifera. The species belonging to diverse genera have previously been clubbed to represent a distinct set of environmental parameters and referred as benthic foraminifera morphogroups (Chamney 1976; Bernhard 1986). Initially, benthic foraminifera were segregated into two broad morphogroups, namely epifaunal and infaunal benthic foraminifera based on their habitat (Corliss 19851991). Epifaunal benthic foraminifera reside close to sediment water interface, whereas infaunal benthic foraminifera were spotted burrowed up to a few centimeters deep in sediments (Jorissen et al. 1995; Singh et al. 2018; Kaithwar et al. 2020). Epifaunal benthic foraminifera have high surface area to volume ratio and are usually abundant in oxygen-rich waters on the shallow continental shelf as well as deep-water regions where bottom water replenishes the oxygen (De and Gupta 2010; Khare et al. 2017). On the other hand, infaunal (occasionally also characterized as shallow infaunal, intermediate, and deep infaunal) benthic foraminifera have a low surface area to volume ratio and thrive well in low bottom water oxygen condition such as oxygen minimum zones (Nigam et al. 2007; Mazumder and Nigam 2014). A few workers refer to them as asymmetrical benthic foraminifera (AABF; sometimes also known as rectilinear forms; Nigam et al. 2007) and rounded symmetrical benthic foraminifera (RSBF) morphogroups (Manasa et al. 2016; Naik et al. 2017; Suokhrie et al. 2018). The benthic foraminifera belonging to AABF morphogroup are elongated, with the length exceeding the width of the specimen. The symmetrical forms, on the other hand, are usually rounded in shape. RSBF are abundant in the shallow waters with high energy conditions (Manasa et al. 2016) as well as in deep water where dissolved oxygen is abundant (Suokhrie et al. 2018). AABF, on the other hand, is primarily abundant in the intermediate water, where there is plenty of food but oxygen is limited (Singh et al. 2018; Suokhrie et al. 2021). Epifaunal-infaunal and AABF-RBSF, to a certain degree, represent the same morphology-based differentiation.
Later, it was realized that benthic foraminifera can be categorized further into several other morphgroups, namely, tapered/cylindrical, spherical, biconvex, planoconvex, flattened-ovoid, flattened-tapered, rounded-trochospiral, rounded-planispiral and milioline (Corliss and Chen 1988; Corliss and Fois 1990). The distribution of these morphogroups was correlated with water depth (Chamney 1976; Severin 1983; Corliss 1985; Jones and Charnock 1985; Bernhard 1986; Corliss and Fois 1990), as well as the organic carbon abundance (Corliss and Chen 1988) and microhabitat preferences (e.g., Corliss 1985; Corliss and Emerson 1990). The relative abundance of elongate-flattened, biconvex, and tapered forms increased with increasing water depth, but the planoconvex morphogroup’s abundance decreased from shallow to deep water (Severin 1983). The depth distribution of these nine benthic morphogroups from the Norwegian Sea was initially studied by Corliss and Chen (1988). The tapered-cylindrical morphogroup had a depth range of 0–2500 m but was more abundant in the upper 1000 m. The flattened tapered shapes were erratic and only found in the upper 500 m. The depth distribution of rounded-planispiral tests ranged from 20 to 3000 m, while the interval 500–1000 m had its highest abundance. The spherical morphogroup was less abundant and was reported between 200 and 1600 m. The flattened-ovoid forms were found in a wide range of water depth (50–3200 m), with the highest abundance between 700 and 1500 m. The planoconvex morphogroup, although found between 0 and 4000 m, showed two peaks of abundance, between 200 and 500 m, and in the interval of 1500–2500 m. The biconvex morphogroup was recorded at depths ranging from 0 to 4000 m, with the maximum abundance between 1500 and 4000 m. Milioline forms gradually increased from 100 to 3200 m, but as a whole, they were less abundant. The rounded-trochospiral morphogroup ranged from 0 to 4000 m, but with low abundance throughout.
Such an elaborate distinction between different benthic foraminifera morphogroups has not been applied in the Indian Ocean, with a majority of the published works either using only two major categories (AABF and RSBF) (Nigam et al. 2007; Manasa et al. 2016; Suokhrie et al. 2018), or applying it to only the agglutinated benthic foraminifera (Saalim et al. 2019). However, recently, Verma et al. (2018) categorized benthic foraminifera from a sediment core into these nine morphogroups. It was reported that planoconvex and milioline morphogroups prefer an oxygen-rich environment, whereas tapered/cylindrical forms were abundant during the period of a strengthened oxygen minimum zone.
It is also worth noting that most of the earlier efforts at categorizing benthic foraminifera into different morphogroups, based on test morphology, used dead assemblages. The dead assemblages typically comprise of a mix of benthic foraminifera resultant from a long interval and most likely various microhabitats as well (Loubere and Gary 1990). Furthermore, the selective breakdown because of dissolution and mechanical destruction significantly alters the original foraminiferal assemblage (Loubere 1997). Therefore, the environmental preferences of the morphogroups should be assessed from the living benthic foraminifera assemblages. Additionally, the living benthic foraminifera (rose Bengal stained) are now widely practiced and accepted for bio-monitoring (FOBIMO-FOraminiferal BIo-Monitoring, Schönfeld et al. 2012; Frontalini and Coccioni 2011; Saalim et al. 2017; Dubey et al. 2018; Jorissen et al. 2018). Therefore, here, we study the relationship between the different morphogroups of living benthic foraminifera and environmental parameters in a dynamic region of the southeastern Arabian Sea. The findings will be helpful in deciphering environmental preferences of the morphogroups in the present oceanic settings, which may be utilized to deduce past environmental conditions.
Physiography of the study area
The study region is in the northern Indian Ocean, where the Arabian Sea meets the Bay of Bengal. The seasonal monsoon winds in the Arabian Sea and low salinity water input from the Bay of Bengal are two oceanographic processes that have a significant impact on the biological as well as environmental parameters in the study region (Banse 1959; Smitha et al. 2008). The area encompasses a large portion of the continental shelf as well as the slope region. Additionally, a relict carbonate platform (~ 60 and 200 m water depth) is found in the shelf area (Rao et al. 2003). The sediments are very coarse sand type in the shallow depths, and finer on the slope (Sivadas et al. 2020). The sea level shift in this region is particularly apparent as the area has a broad shelf in the central portion, then a sharp increase in depth, practically a vertical drop, followed by a continental slope. Aside from that, seasonal coastal currents bring warm, high-saline water to the study area during the southwest monsoon and cold and low-saline water during the northeast monsoon (Shankar et al. 2002).
Material and methodology
The surface sediment samples (43 number) for living benthic foraminifera were collected during the post monsoon season in October 2014, onboard Sindhu Sadhana voyage SSD004 (Fig. 1). The Ocean Scientific International Limited Maxi Multi-corer was used to collect the undisturbed core-top/surface samples along with overlying water. The multi-corer tubes were 600-mm-long with 110-mm outer diameter and 100-mm internal diameter. Additionally, the overlying water collected in the multi-core top was used to measure the seawater physico-chemical parameters including temperature, salinity, and dissolved oxygen at the sediment–water interface. The sediments were sub-sampled at 1-cm interval. One half of each sub-sample in the top 10 cm was immediately stained with rose-Bengal (2 g of rose-Bengal stain dissolved in 1 L of 70% ethanol). The stained samples were stored at 4 °C for 7–8 weeks for proper staining and distinction between the living benthic foraminifera from the dead ones.
The top 2 cm of the sediment samples were processed to extract living benthic foraminifera. The samples were wet-sieved by using 63-µm sieve to segregate benthic foraminifera from the sediment matrix. The coarse fraction was oven-dried overnight at 45 °C, weighed, and stored in plastic vials. A representative aliquot of the coarse fraction was weighed and 300 benthic foraminifera were picked under Olympus SZX 12 stereo-zoom microscope. All the picked specimens were identified up to species level. Benthic foraminifera morphogroups were classified following Corliss and Fois (1990). Furthermore, canonical correspondence analysis (CCA) was computed by using MultiVariate Statistical Package software to find out the relationship between living benthic foraminifera morphogroups and the ambient environmental parameters.
For carbon and nitrogen analysis, ~ 5 g of sediment from the unstained half was freeze-dried and powdered by using a clean agate mortar pestle. The total inorganic carbon (TIC) in the sediment was analyzed by using coulometer (model CM 5015 CO2), and the total carbon and nitrogen was analyzed by using elemental analyzer (model FLASH 2000 Thermo Scientific). The organic carbon (%Corg) was calculated by subtracting TIC from the total carbon.
Results
Living benthic foraminifera
The living benthic foraminifera were found in all the samples collected from different depths. The average living depth of benthic foraminifera was ~ 2 cm in the southeastern Arabian Sea (Kaithwar et al. 2020). The absolute abundance of living benthic foraminifera in the top 2 cm varied between 3 and 448 specimen/g sediment (Fig. 2). The highest abundance was on the slope of the Gulf of Mannar (T1 transect). The foraminiferal abundance was generally high at intermediate water depths along all transects. As compared to the intermediate depths, the shallow and deeper water sediments contained < 50 specimen/g sediment. Thus, the living benthic foraminiferal abundance at shallow and deeper depths was 3–4 times less than that at intermediate water depths. A total 174 living benthic foraminifera species belonging to eight morphogroups were found. The distribution pattern of each morphogroup is discussed below in detail.
Tapered and cylindrical
The specimens belonging to a total of 52 species (Table 1) were included in the tapered/cylindrical morphogroup. This morphogroup was the most common in the southeastern Arabian Sea. Its relative abundance ranged from being absent to 100.00%, with an average of 32.30% (Table 2). The tapered and cylindrical forms were mostly abundant at the intermediate depths (1000–1500 m) of all transects. The highest relative abundance of the tapered and cylindrical forms was in the Gulf of Mannar transect (T1 transect). Furthermore, two deeper stations, MC56 and 59, have a higher proportion of living tapered and cylindrical benthic foraminifera (Fig. 3).
Spherical
The spherical morphogroup included 16 species in the southeastern Arabian Sea (Table 1). The abundance of spherical morphogroup ranged from being absent at several stations to a maximum of 15.00%, with an average of 3.26% (Table 2). Spherical morphogroup demonstrates patchy distribution pattern in the region. The maximum abundance was found along the transect T3. Interestingly, the spherical tests were relatively less abundant in the shallow water station of transects T1 and T4. Additionally, the deep-water station MC60 has higher percentage of living spherical forms (Fig. 4).
Biconvex
Biconvex morphogroup includes 15 species (Table 1) and its relative abundance ranged from being absent at several stations to 40.00%, with an average of 8.91% (Table 2). Generally, the abundance of biconvex forms increased in the intermediate water depth stations. However, in transect T4, the abundance continuously increased with water depths. Additionally, deep-water station MC58 shows higher abundance of Biconvex forms (Fig. 5).
Planoconvex
Planoconvex morphogroup comprised of specimens belonging to 15 species (Table 1). Its relative abundance ranged from being nearly absent to as high as 66.67% (Table 2), with an average of 13.79%. Planoconvex forms were abundant in transects T2, T3, and T4, whereas less abundant in T1. The higher relative abundance of planoconvex morphogroup was found at stations MC30 and MC14 at a depth of 1506 m and 1530 m, respectively. Furthermore, the relative abundance was comparatively high at intermediate water depths in all transects (Fig. 6).
Flattened-ovoid
Flattened-ovoid forms (13 species; Table 1) were comparatively rare, with abundance ranging from being absent to 39.58%, with an average of 6.33% (Table 2). It was less abundant on the upper continental shelf but has comparatively higher abundance on the upper and intermediate slope regions. The morphogroup showed lower abundance along the transects T2 and T3, and deeper slope region of transect T4. The higher abundance was at stations MC27 and MC58 at 764 m and 2750 m water depth, respectively (Fig. 7).
Flattened-tapered
Flattened-tapered morphogorup included 25 species (Table 1). It was moderately abundant in the southeastern Arabian Sea, with abundance ranging from nearly absent to 75.00% (Table 2), with an average of 19.29%. The abundance of living flattened-tapered benthic foraminifera was comparatively higher on the continental shelf and deeper slope regions, whereas the intermediate water depth stations had a very few living flattened-tapered benthic foraminifera. The highest abundance was noticed at deep-water station MC57 at the water depth of 2980 m (Fig. 8).
Rounded-trochospiral
In the southeastern Arabian Sea, rounded-trochospiral (17 species; Table 1) relative abundance ranged from being absent to 42.85% (Table 2), with an average of 8.93%. Barring a few exceptions, its relative abundance generally lower in intermediate water depths. The highest abundance was at deep-water station MC55 (2238 m) (Fig. 9).
Rounded-planispiral
The overall abundance of rounded-planispiral morphogroup (18 species; Table 1) was also moderate in the southeastern Arabian Sea. The abundance ranged from nearly absent to 33.33% (Table 2), with an average of 7.04%. The abundance shows patchy distribution in the region with increased abundance at intermediate slope, and low on the deeper slope. The higher abundance was found at MC30 and MC60 at a depth of 1506 m and 1887 m, respectively. Additionally, the relative abundance of living rounded-planispiral forms increased with depth in the transect T4 (Fig. 10).
Infaunal
The species were grouped into infaunal and epifaunal forms, following Kaithwar et al (2020). The relative abundance of living infaunal benthic foraminifera was comparatively very high with the entire assemblage comprising of only infaunal forms at several stations (Table 2). The average relative abundance of living infaunal benthic foraminifera was 68.21%. The relative abundance of infaunal forms was higher on the deep-water stations MC56 and MC57. Additionally, its abundance decreased rapidly after 1500 m in a few parts of the study area (Fig. 11).
Epifaunal
The epifaunal benthic foraminifera were comparatively less abundant in the southeastern Arabian Sea. The abundance ranged from being absent at few stations to 71.42% (Table 2), with a mean of 31.62%. The highest abundance was at MC55 (2460 m). The abundance was relatively low on the continental shelf and deeper slope, but was high at the intermediate depths (Fig. 12).
Environmental parameters
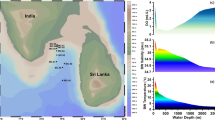
Bottom water salinity, temperature, dissolved oxygen, organic carbon concentration (%Corg), organic carbon to nitrogen ratio (%Corg/N), and water depth were the environmental parameters considered here to understand its effect on living benthic foraminifera morphogroups. The bottom water salinity varied within a narrow range (34.7–35.1 psu) and decreased with depth. Bottom water temperature also decreased with water depth and ranged between 2.5 and 27.4 °C. The depth-wise trend of both the salinity and temperature was similar in all transects, although the range was different. A large transect to transect variation was observed in %Corg, although the samples collected from the intermediate depths in all transects had a higher organic carbon content. Organic carbon concentration in the sediment varied between 0.71 and 8.32%. The degradation of organic matter causes formation of perennial oxygen deficient zone and the value ranged between 0.35 and 4.25 mL/L. Additionally, %Corg/N value ranged between 4.8 and 17.8 (Singh et al. 2021). The %Corg/N varied the maximum and showed transect to transect variation (Fig. 13; Table 2).
The temperature, salinity, and dissolved oxygen in the water at the sediment–water interface in the southeastern Arabian Sea. The organic carbon concentration (%Corg) and organic carbon to nitrogen ratio (%Corg/N) in the top 2 cm of the sediments at all the stations are also plotted with depth (modified after Singh et al. 2021). A represents whole area, T1–T4 represents transect-wise distribution of the environmental parameters
Canonical correspondence analysis
The canonical correspondence analysis helped in finding out the correlation between morphogroups and ambient environmental parameters, where the direction and length of the arrow delineate positive/negative relationship. From the CCA plot, it was evident that the flattened-tapered form was positively correlated with bottom water salinity and temperature. This form was also negatively correlated with %Corg/N and water depth. Similarly, epifaunal, biconvex, and planoconvex forms were positively correlated with bottom water–dissolved oxygen. These forms were also showing negative correlation with organic carbon percentage in the sediments. The spherical, rounded-planispiral, and rounded-trochospiral forms were positively correlated with %Corg/N and water depth and negatively correlated with bottom water salinity and temperature. The infaunal, flattened-ovoid, and tapered/cylindrical forms were positively correlated with %Corg in the sediments. These forms were also negatively correlated with bottom water–dissolved oxygen (Fig. 14).
Discussion
From the surface distribution and CCA, it was evident that several living benthic foraminifera morphogroups in the southeastern Arabian Sea were strongly modulated by the ambient environmental parameters. The major environmental parameters in the southeastern Arabian Sea included high primary productivity and concurrent perennial oxygen-deficient zone formation, as well as the bottom water salinity, temperature, water depth, and %Corg/N.
A majority of the living benthic foraminifera in the study region were tapered and cylindrical and showed a positive correlation with %Corg or, in other words, food availability. The maximum abundance of such forms was between 1000 and 1500 m water depth, in contrast to other parts of the world ocean, such as the Norwegian Sea (Corliss and Chen 1988) and the Gulf of Mexico (Corliss and Fois 1990), where maximum abundance was found in the upper 1000 m. The difference in the depth zone of increased abundance of tapered and cylindrical foraminifera could be because the lower continental slope of the southeastern Arabian Sea contains a high %Corg (Singh et al. 2021). This morphogroup is dominated by the genera Bulimina, Uvigerina, Fursenkoina, Globocassidulina, and Reophax. These genera have been well documented in the high productivity areas of the world ocean, such as the eastern Pacific and the Arabian Sea (Schumacher et al. 2007; Uchimura et al. 2017). These genera are also primarily restricted to the upper sediment column and are classified as shallow infaunal (Jorissen et al. 1995; Kaithwar et al. 2020). As a result, the abundant presence of tapered and cylindrical benthic foraminifera morphogroup in a region should be used as an indicator of high organic carbon or primary productivity. Furthermore, the high organic matter preference of these morphogroups is further confirmed by their increased abundance during the times of high productivity such as the Bølling–Allerød and early Holocene (Verma et al. 2018). Besides, this morphogroup also had a negative correlation with bottom water–dissolved oxygen, indicating that these genera thrive in regions where oxygen concentrations are depleted by the organic matter degradation and benthos respiration.
The flattened-ovoid morphogroup, like the tapered and cylindrical morphogroups, had similar environmental preference. A majority of the species belonging to the flattened-ovoid, tapered, and cylindrical morphogroups are infaunal (Corliss and Fois 1990). The flattened-ovoid forms are mostly abundant on the continental shelf and upper slope (top 1000 m) where dissolved oxygen concentration is at its lowest, as indicated by CCA (Fig. 14; Fig. 15). A similar low dissolved oxygen preference of flattened ovoid forms was also suggested in the Gulf of Mexico and the Norwegian Sea (Corliss and Chen 1988; Corliss and Fois 1990), as well as in the eastern Arabian Sea, where its abundance declined during the times of weaker OMZs, such as the Heinrich Stadial 1 (HS1; Verma et al. 2018). The infaunal benthic foraminifera have a negative relationship with dissolved oxygen and a positive relationship with %Corg. In the Arabian Sea and other parts of the world ocean, infaunal benthic foraminifera are widely used as an indicator of the past OMZ variability (Mazumder and Nigam 2014; Naik et al. 2017), and this work on living benthic foraminifera reconfirms it.
The representative species of the different morphogroups in the southeastern Arabian Sea. Tapered and cylindrical (1. Saccorhiza ramosa; 2a-b. Siphotextularia rolshauseni; 3. Bulimina aculeata; 4. Uvigerina multicostata), flattened-tapered (5a-c. Bolivina obscuranta), spherical (6. Lagena perculiaris; 7. Anturina haynesi; 8. Lagenosolenia eucerviculata), rounded-planispiral (9a-c. Melonis pompilioides), biconvex (10a-c. Cancris sagra; 11a-c. Buccella differens), planoconvex (12a-c. Cibicidoides wuellerstrofi; 13a-c Hanzawaia concentrica), flattened-ovoid (14a-b. Lenticulina lucidiformis; 15a-c. Cassidulina carinata), milioline (16. Quinqueloculina parkeri), rounded-trochospiral (17a-c. Haplophragmoides subglosum; 18a-c. Gyroidina quinqueloba). Scale = 100 µm
The epifaunal habitat of the biconvex morphogroup is well established. This group includes the genera Cancris, Eponides, Bucella, and Hoeglundina, which are usually grouped together under the epifaunal morphogroup (Nigam and Khare 1999; Manasa et al. 2016). In the southeastern Arabian Sea, epifaunal forms were abundant at very shallow depths and on the lower slope, where organic carbon content is low. This finding is also supported by CCA, indicating a negative correlation between %Corg and Biconvex morphogroup (Figs. 14 and 15). Furthermore, their increased abundance was reported from both the shallow waters in the Arabian Sea (Nigam and Khare 1999) as well as the deep water in the Norwegian Sea and Gulf of Mexico (Corliss and Chen 1988; Corliss and Fois 1990). The biconvex forms were also more abundant during the HS1 and HS2 (Verma et al. 2018), when surface primary productivity was lower. Biconvex morphogroup also showed a positive correlation with the bottom water–dissolved oxygen, indicating that it thrives in well-oxygenated bottom water, such as shallow and deep-water. Planoconvex epifaunal benthic foraminifera showed the same positive/negative relationship with ambient environmental parameters. This group primarily includes the genera Cibicidoides, Epistominella, Hanzawia, and Planulina. Their surface distribution revealed a high abundance in deep water, where dissolved oxygen is abundant. This is also supported by CCA, which suggests a positive relationship between dissolved oxygen and the planoconvex group (Figs. 14 and 15). The abundance and pore size of Cibicidoides are a well-established proxy for the bottom water oxygenation (Rathburn et al. 2018), but genera such as Epistominella are known as productivity indicators (Saraswat et al. 2005). This demonstrates that different genera/species within the same morphogroup may have distinct microhabitat preferences. Furthermore, planoconvex forms have been widely reported from the deep water in the world ocean (Corliss and Chen 1988; Corliss and Fois 1990), and their abundance was high during north Atlantic cold events such as Heinrich Stadials 1 and 2, when bottom water–dissolved oxygen concentration was high due to lower primary productivity in the eastern Arabian Sea (Verma et al. 2018).
From the CCA plot, we report a positive relationship between flattened-tapered morphogroup and bottom water salinity as well as temperature. The relative abundance of this morphogroup was negatively correlated with %Corg/N and water depth (Figs. 14 and 15). This is quite intriguing, because flattened-tapered genera, such as Bolivina, were previously thought to be OMZ indicator species. It was more abundant between 0–250 m and 1000–1500 m water depths, where %Corg concentration was comparatively low. Similarly, during HS1, when primary productivity was low, an increased percentage of flattened-tapered foraminifera was reported from the eastern Arabian Sea (Verma et al. 2018). A similar preference was reported from the Norwegian Sea, where it was found in abundance in shallow waters with low organic carbon. All these justify its negative relationship with %Corg/N, such as Heinrich Stadials when the contribution of land-derived organic matter decreased. This summarizes that this morphogroup prefers low organic carbon.
The spherical morphogroup is associated with shallow infaunal microhabitats, and its comparatively high abundance in the lower slope region suggests its preference for plenty of food availability as well as surplus dissolved oxygen. This habitat preference is confirmed by its positive correlation with %Corg/N and water depth in CCA (Figs. 14 and 15). Lagena, Baggina, Oolina, and Fissurina are the major contributors to this morphogroup and all these genera are frequently found near the high riverine influx areas such as in front of the Ganga Brahmaputra rivers (Manasa et al. 2016). The southeastern Arabian Sea is a high marine organic carbon zone, and the relative abundance of this morphogroup is lower than that of others. Other studies have confirmed its infaunal microhabitat and preference for low levels of marine organic carbon (Corliss and Chen 1988; Corliss and Fois 1990). Furthermore, Verma et al. (2018) confirmed its high abundance during the last glacial maximum, when the northeast monsoon was dominant, resulting in a high %Corg/N in the study area. This morphogroup has a negative relationship with bottom water salinity and temperature, which corresponds well with the regional hydrography and can be used to interpret the area’s past environmental conditions. The rounded-trochospiral and rounded-planispiral morphogroups exhibit similar environmental preferences (Figs. 14 and 15). This simply means that these morphogroups prefer food from the land. The rounded-trochospiral are usually epifaunal oxygen-loving species that increase in abundance with water depth, particularly after OMZ (> 1500 m). The same is true for the Gulf of Mexico, where its abundance increased abruptly after 1000 m water depth (Corliss and Fois 1990). The most common genera in this morphogroup from the southeastern Arabian Sea are Gyroidina and Haplophragmoides. The past records confirm their preference for land-derived food, as their abundance decreased during the periods of reduced rainfall, such as Heinrich Stadials 1 and 2. The rounded-planispiral forms are commonly thought of as an infaunal benthic group, with abundance increasing from southeast to northwest or from the Bay of Bengal side to the Arabian Sea side. The Bay of Bengal water contains a large amount of land-derived nutrients as well as organic matter, as evident from an increase in the %Corg/N. CCA results also revealed a positive correlation between %Corg/N and rounded-planispiral morphogroup, indicating that the land-derived food is preferred by genera such as Nonion, Nonionella, Melonis, and Pullenia. Its abundance also decreased during the periods of weaker southwest monsoon, such as HS1 and HS2.
In the southeastern Arabian Sea, two major-morphogroups, epifaunal and infaunal benthic foraminifera have a positive/negative relationship with %Corg and dissolved oxygen. This indicates its applicability in the global ocean. However, a few workers prefer to use the terms rounded symmetrical benthic foraminifera (RSBF) and angular asymmetrical benthic foraminifera (AABF) over epifaunal and infaunal, respectively. In this work, we also checked that the factors affecting the relative abundance of the RSBF and AABF (Supplementary Fig. 1). CCA of RSBF, AABF, and other morphogroups with the ambient physico-chemical parameters reveals that AABF and RSBF do not show a positive/negative relationship with %Corg and dissolved oxygen. This could be due to the fact that infuanal morphogroups such as rounded-planispiral, spherical, and flattened-ovoid are frequently considered under RSBF, not AABF (Manasa et al. 2016; Suokhrie et al. 2018). We recommend that workers should avoid using AABF and RSBF and instead focus on other morphogroups.
Conclusions
The relationship between living benthic foraminifera morphogroups and environmental parameters was tested in the southeastern Arabian Sea. We demonstrate a definite distribution pattern of a majority of the morphgroups in the surface sediment of the studied region. We conclude that infaunal morphogroups such as tapered/cylindrical and flattened-ovoid are found in an oxygen-deficient zone which is caused by the high organic carbon degradation. Additionally, epifunal morphogroups such as biconvex and planoconvex represent well-ventilated bottom water conditions. Besides this, other morphogroups, namely flattened-tapered, spherical, rounded-trochospiral, and rounded-planispiral forms were abundant at deep-water stations where land-derived organic matter was abundant (as suggested by the high %Corg/N) with low bottom water salinity and temperature. The environmental preferences of the living benthic foraminifera morphogroups delineated from the southeastern Arabian Sea can be used to reconstruct past environmental conditions.
References
Banse K (1959) On upwelling and bottom-trawling off the southwest coast of India. J Mar Biol Assoc India 1:33–49
Bernhard JM (1986) Characteristic assemblages and morphologies of benthic foraminifera from anoxic, organic-rich deposits: Jurassic through Holocene. J Foraminiferal Res 16:207–215
Chamney TP (1976) Foraminiferal morphogroup symbol for paleoenvironmental interpretation of drill cutting samples: Arctic America, Albian continental margin: maritime Sediments, Special Publication IB, pp 585–624
Chien AC, Hill N, Levin P (2012) Cell size control in bacteria. Curr Bio 22:R340–R349
Corliss BH (1985) Microhabitats of benthic foraminifera within deep-sea sediments. Nature 314:435–438
Corliss BH, Chen C (1988) Morphotype patterns of Norwegian Sea deep-sea benthic foraminifera and ecological implications. Geology 16:716–719
Corliss BH, Emerson S (1990) Distribution of Rose Bengal stained deep-sea benthic foraminifera from the Nova Scotian continental margin and Gulf of Maine. Deep-Sea Res 37:381–400
Corliss BH, Fois E (1990) Morphotype analysis of deep-sea benthic foraminifera from the northwest Gulf of Mexico. Palaios 5:589–605
Corliss BH (1991) Morphology and microhabitat preferences of benthic foraminifera from the northwest Atlantic Ocean. Mar Micropaleontol 17:195–236
De S, Gupta AK (2010) Deep-sea faunal provinces and their inferred environments in the Indian Ocean based on distribution of Recent benthic foraminifera. Palaeogeogra., Palaeoclimatol. Palaeoecol 291:429–442
Dubey R, Saraswat R, Dineshkumar PK, Nigam R (2018) Dwarf foraminifera off Kerala, India: a response to mudbank formation. J Palaeontological Soc India 63:81–90
Frontalini F, Coccioni R (2011) Benthic foraminifera as bioindicators of pollution: a review of Italian research over the last three decades. Rev Micropaléontol 54:115–127
Frontalini F, Curzi D, Giordano FM, Bernhard JM, Falcieri E, Coccioni R (2015) Effects of lead pollution on Ammonia parkinsoniana (foraminifera): ultrastructural and microanalytical approaches. Eur J Histochem 59. https://doi.org/10.4081/ejh.2015.2460
Gooday AJ (2001) Benthic Foraminifera, Editor(s): John H. Steele, Encyclopedia of Ocean Sciences, Academic Press, 274–286, ISBN 9780122274305
Holt EA, Miller SW (2010) Bioindicators: using organisms to measure environmental impacts. Nature Edu Knowl 3:8
Jones RW, Charnock MA (1985) “Morphogroups” of agglutinated foraminifera. Their life positions and feeding habits and potential applicability in (paleo)ecological studies. Rev Paleobiol 4:311–7320
Jorissen FJ, de Stigter HC, Widmark JGV (1995) A conceptual model explaining benthic foraminiferal microhabitats. Mar Micropaleontol 26:3–15
Jorissen F, Nardelli MP, Almogi-Labin A, Barras C, Bergamin L, Bicchi E, El Kateb A, Ferraro L, McGann M, Morigi C, Romano E (2018) Developing Foram-AMBI for biomonitoring in the Mediterranean: species assignments to ecological categories. Mar Micropaleontol 140:33–45
Kaithwar A, Singh DP, Saraswat R (2020) A highly diverse living benthic foraminiferal assemblage in the oxygen deficient zone of the southeastern Arabian Sea. Biodivers Conserv 29:3925–3958. https://doi.org/10.1007/s10531-020-02056-9
Kelkar K, Gray ME, Aragón-Salamanca A, Rudnick G, Milvang-Jensen B, Jablonka P, Schrabback T (2017) The effect of the environment on the structure, morphology and star formation history of intermediate-redshift galaxies. Monthly Notices Royal Astronomical Soc 469:4551–4564
Khare N, Nigam R, Mayenkar DN, Saraswat R (2017) Cluster analysis of benthic foraminiferal morpho-groups from the western margin of India reflects its depth preference. Continental Shelf Res 151:72–83
Loubere P (1997) Benthic foraminiferal assemblage formation, organic carbon flux and oxygen concentrations on the outer continental shelf and slope. J Foraminiferal Res 27:93–100
Loubere P, Gary A (1990) Taphonomic process and species microhabitats in the living to fossil assemblage transition of deeper water benthic foraminifera. Palaios 5:375–381
Manasa M, Saraswat R, Nigam R (2016) Assessing the suitability of benthic foraminiferal morpho-groups to reconstruct paleomonsoon from Bay of Bengal. J Earth Syst Sci 125:571–584
Mazumder A, Nigam R (2014) Bathymetric preference of four major genera of rectilinear benthic foraminifera within oxygen minimum zone in Arabian Sea off central west coast of India. J Earth Syst Sci 123:633–639
Naik DK, Saraswat R, Lea DW, Kurtarkar SR, Mackensen A (2017) Last glacial-interglacial productivity and associated changes in the eastern Arabian Sea. Palaeogeogra., Palaeoclimatol. Palaeoecol 483:147–156
Nigam R, Khare N (1999) Spatial and temporal distribution of foraminifera in sediments off the central west coast of India and use of their test morphologies for the reconstruction of paleomonsoonal precipitation. Micropaleontology 45:285–303
Nigam R, Mazumder A, Henriques PJ, Saraswat R (2007) Benthic foraminifera as proxy for oxygen-depleted conditions off the central west coast of India. J Geol Soc India 70:1047–1054
O’Brien PAJ, Polovodova Asteman I, Bouchet VMP (2021) Benthic foraminiferal indices and environmental quality assessment of transitional waters: a review of current challenges and future research perspectives. Water 13:1898. https://doi.org/10.3390/w13141898
Prazeres M, Uthicke S, Pandolfi J (2016) Influence of local habitat on the physiological responses of large benthic foraminifera to temperature and nutrient stress. Sci Rep 6:21936
Rao VP, Rajagopalan G, Vora KH, Almeida F (2003) Late quaternary sea level and environmental changes from relic carbonate deposits of the western margin of India. J Earth Syst Sci 112:1–25. https://doi.org/10.1007/BF02710040
Rathburn AE, Willingham J, Ziebis W, Burkett AM, Corliss BH (2018) A new biological proxy for deep-sea paleo-oxygen: pores of epifaunal benthic foraminifera. Sci Rep 8:1–8
Saalim SM, Saraswat R, Suokhrie T, Bhadra SR, Kurtarkar SR, Nigam R (2017) Benthic foraminiferal response to changes in mining pattern: a case study from the Zuari estuary, Goa, India. Environ Earth Sci 76 706 (1–10)
Saalim SM, Saraswat R, Suokhrie T, Nigam R (2019) Assessing the ecological preferences of agglutinated benthic foraminiferal morphogroups from the western Bay of Bengal. Deep Sea Res Part II: Topical Stud Oceanogra 161:38–51
Saraswat R, Nigam R, Barreto L (2005) Palaeoceanographic implications of abundance and mean proloculus diameter of benthic foraminiferal species Epistominella exigua in sub-surface sediments from distal Bay of Bengal fan. J Earth Syst Sci 114:453–458
Saraswat R, Nigam R (2013) Benthic foraminifera. Encyclopaedia Quat Sci 2:765–774
Schönfeld J, Alve E, Geslin E, Jorissen F, Korsun S, Spezzaferri S (2012) The FOBIMO (FOraminiferal BIo-MOnitoring) initiative—towards a standardised protocol for soft-bottom benthic foraminiferal monitoring studies. Mar Micropaleontol 94:1–13
Schumacher S, Jorissen FJ, Dissard D, Larkin KE, Gooday AJ (2007) Live (rose Bengal stained) and dead benthic foraminifera from the oxygen minimum zone of the Pakistan continental margin (Arabian Sea). Mar Micropaleontol 62:45–77
Severin KP (1983) Test morphology of benthic foraminifera as a discriminator of biofacies. Mar Micropaleont 8:65–76
Shankar D, Vinayachandran PN, Unnikrishnan AS (2002) The monsoon currents in the north Indian Ocean. Progr Oceanogra 52:63–120
Singh DP, Saraswat R, Kaithwar A (2018) Changes in standing stock and vertical distribution of benthic foraminifera along a depth gradient (58–2750 m) in the southeastern Arabian Sea. Mar Biodivers 48:73–88. https://doi.org/10.1007/s12526-017-0823-z
Singh DP, Saraswat R, Nigam R (2021) Untangling the effect of organic matter and dissolved oxygen on living benthic foraminifera in the southeastern Arabian Sea. Mar Poll Bull 172:112883
Sivadas SK, Singh DP, Saraswat R (2020) Functional and taxonomic (α and β) diversity patterns of macrobenthic communities along a depth gradient (19–2639 m): a case study from the southern Indian continental margin. Deep Sea Res Part I: Oceanogra Res Pap 159:103250
Smitha BR, Sanjeevan VN, Vimalkumar KG, Revichandran C (2008) On the upwelling off the southern tip and along the west coast of India. J Coast Res 24:95–102
Suokhrie T, Saalim SM, Saraswat R, Nigam R (2018) Indian monsoon variability in the last 2000 years as inferred from benthic foraminifera. Quat Int 479:128–140
Suokhrie T, Saraswat R, Nigam R (2021) Multiple ecological parameters affect living benthic foraminifera in the river-influenced west-central Bay of Bengal. Front Mar Sci 8:656757
Uchimura H, Nishi H, Takashima R, Kuroyanagi A, Yamamoto Y, Kutterolf S (2017) Distribution of recent benthic foraminifera off Western Costa Rica in the eastern equatorial Pacific Ocean. Paleontol Res 21:380–396
Verma K, Bharti SK, Singh AD (2018) Late Glacial-Holocene record of benthic foraminiferal morphogroups from the eastern Arabian Sea OMZ: paleoenvironmental implications. J Earth Syst Sci 127:1–15
Acknowledgements
Authors are thankful to the Director, CSIR-National Institute of Oceanography, Goa, India, for the permission to publish the results. DPS thanks the Indian Institute of Technology Roorkee for providing basic infrastructure for the completion of this work. RS is thankful to the Department of Science and Technology [DST/CCP/NCC&CV/130/2017(G)] and the Council of Scientific and Industrial Research, India for the financial support under the Young Scientist Research Scheme. Authors thank Dr. C.P. Babu for the help in organic carbon and nitrogen analysis. We also thank Professor Michael A. Kaminski, Department of Geosciences, College of Petroleum Engineering and Geosciences, King Fahd University of Petroleum and Minerals, and one anonymous reviewer for taking the time and effort to review our work.
Funding
The financial support is received from the Department of Science and Technology [DST/CCP/NCC&CV/130/2017(G)].
Author information
Authors and Affiliations
Contributions
DPS conceived the idea and further developed with RS and RP. DPS has done the benthic foraminifera picking and identification and wrote the original draft. RS has acquired the funding and edited and reviewed the manuscript. The manuscript has been read and approved by all named authors.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Yes.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: V.V.S.S. Sarma
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, D.P., Saraswat, R. & Pawar, R. Distinct environmental parameters influence the abundance of living benthic foraminifera morphogroups in the southeastern Arabian Sea. Environ Sci Pollut Res 29, 82541–82558 (2022). https://doi.org/10.1007/s11356-022-21492-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21492-4