Abstract
Himalaya, the highest mountain system in the world and house of important biodiversity hotspot, is sensitive to projected warming by climate change. Rhizocarpon geographicum (map lichen), a crustose lichen, grows in high mountain ranges, is a potential indicator species of climate change. In the present study, MaxEnt species distribution modeling algorithm was used to predict the suitable habitat for R. geographicum in current and future climate scenarios. Nineteen bioclimatic variables from WorldClim database, along with elevation, were used to predict the current distribution and three representative concentration pathway (RCP) scenarios by integrating three general circulation models (GCMs) for future distribution of species covering years 2050 and 2070. Furthermore, we performed change analysis to identify the precise difference between the current and future distribution of suitable areas of the species for delineating habitat range expansion (gain), habitat contraction (loss), and stable habitats. The final ensemble model obtained had average test value 0.968, and its predicted ~ 27.5% of the geographical area in the Indian Himalayan Region is presently climatically suitable for the species. The predicted highly suitable area for R. geographicum is observed to be declining in Northwestern Himalaya, and it is shifting towards the higher elevation areas of the Eastern Himalaya. The projected distribution in future under the RCP scenarios (RCP 4.5, 6.0, and 8.5) showed the range expansion towards higher elevations, and it is more pronounced for the extreme future scenarios (RCP 8.5) than for the moderate and intermediate climate scenarios (RCP 4.5 and RCP 6.0). However, assuming that species can migrate to previously unoccupied areas, the model forecasts a habitat loss of 10.86–16.51% for R. geographicum, which is expected due to increase in mean annual temperature by 1.5–3.7 °C. The predictive MaxEnt modeling approach for mapping lichen will contribute significantly to the understanding of the impact of climate change in Himalayan ecosystems with wide implications for drawing suitable conservation plans and to take adaptation and mitigation measures.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Climate change not only alters the natural ecosystems but also affects each and every species on the earth to a lesser or greater extent (Walther et al. 2002). Habitat reduction and shifting, physiological and behavioral changes in biota, and extinction of species have been observed as impacts of global climate change (Forrest et al. 2012; Bajpai et al. 2016a). Species are shifting their elevation ranges, latitudinal distribution, and phenologies in response to changing climate (Lavorel and Ganier 2002; Wilson et al. 2005; Hamid et al. 2019; Kumar et al. 2019). The Fifth Assessment Report (AR5) of the Intergovernmental Panel on Climate Change (IPCC) predicted that global climate warming will be continued and the average temperature on the earth will be increased by 0.3–4.5 °C through the end of the twenty-first century (relative to the 1986–2005 baselines) (Stocker et al. 2014). To mitigate the effects of climate change on forest ecosystems and their functioning, we can effectively target conservation strategies by modeling species distribution to identify the suitable habitat areas where key species exist or probability of existence. For effective modeling of species, detailed information about the ecology and spatial distribution pattern of species is a prerequisite. Predicting critical suitable habitat availability will also influence better understanding of ecological systems and the distribution patterns of genetic variation within species (Wang et al. 2012).
Predictive models are widely used powerful tools to obtain an initial understanding of species distribution in response to climate change (Thuiller et al. 2006; Pereira et al. 2010). The ecological niche modeling (ENM) tool is commonly used in predicting the geographic range of species, using presence records and environmental predictors which are assumed to influence its distribution (Raxworthy et al. 2003; Anderson and Martinez-Meyer 2004; Elith and Leathwick 2009; Elith et al. 2011; Peterson et al. 2011; Kong et al. 2021). In recent years, ENM using maximum entropy model (MaxEnt) has emerged as a useful tool in modeling of rare and economically important plant species, including even those with a narrow range and only few presence records (Phillips et al. 2006; Pearson et al. 2007; Elith et al. 2011; Garcia et al. 2013; Hamid et al. 2019; Kumar et al. 2019).
Lichens are among the most vulnerable organisms to changes in environment and climate, induced by both natural and anthropogenic factors, while this sensitivity is mostly due to their low physiological tolerance (Kukwa and Kolanowska 2016; Żołkoś et al. 2013; Wolseley et al. 2006). Lichens (lichenized fungi) are an artificial group of various, distantly related fungal lineages that comprise a mutualistic dependence on a photoautotrophic partner (Miądlikowska et al. 2014). They are poikilohydric in nature and lack vascular system thus cannot regulate their water content actively and absorb water and nutrients passively from their surrounding environment (Green et al. 2011). Because of this, lichens are particularly sensitive to climate changes and air pollution (Pinho et al. 2011; Branquinho et al. 2015). Lichens occur in a variety of land ecosystems from Antarctica to desert and up to the highest mountain (Nash III et al. 1990). Their mechanisms of growth and physiology are significantly regulated by moisture and temperature. These biological and eco-physical barriers make them ideal organisms for examining the effects of climate change.
The Himalaya is experiencing warming at a higher rate than the global average (three times higher, 0.06 °C/year; Shrestha et al. 2012). The Hindu Kush Himalaya Assessment report indicated that if the average global warming will be limited to 1.5 °C above the pre-industrial period, this region will warm by 1.80 ± 0.40 °C (Krishnan et al. 2019). The region represents a rich repository of biodiversity and thus designated as one of the 36 global biodiversity hostspots (Mittermeier et al. 2011). Considering the rich biodiversity, endemism, and higher sensitivity towards climate change, studies are sparse on the species responses to changing climate (Saran et al. 2010; Kumar 2012; Forrest et al. 2012; Telwala et al. 2013; Shrestha and Bawa 2014; Manish et al. 2016; Hamid et al. 2019; Kumar et al. 2019). Because of its distinct evolutionary history and rich species diversity, the Himalayan region warrants more understanding on species responses and ecosystem functioning with regard to climate change (Pandit and Babu 1998; Pandit et al. 2007).
In earlier studies, particularly in Europe and North America, the ENM tools have been used to test the distribution patterns (Szczepańska et al. 2015), taxonomic research on Fuscopannaria confusa (Carlsen et al. 2012), local conservation planning (Cameron et al. 2011; Binder and Ellis 2008), and influence of climate change on the future distribution of several lichen species (Ellis et al. 2007a, 2007b; Ellis et al. 2014; Allen and Lendemer 2016; Kukwa and Kolanowska 2016; Rubio-Salcedo et al. 2017; Ellis 2019). Using ensemble modeling algorithm, the current and the future distribution of Rhizocarpon geographicum (Map Lichen) were mapped across the Indian Himalayan Region (IHR) in view of climate change. R. geographicum is a crustose lichen, which grows on the exposed rocks surface in higher Himalayan mountains and is well-suited as climate change indicator species in lichenometry (Armstrong 2004). To date, no attempt has been made to understand the distribution modeling of R. geographicum at regional or global scale. The objective of the present study aimed (i) to determine the important climatic variables which enhance habitat prediction accuracy of R. geographicum in current climatic condition, (ii) to assess the potential distribution of R. geographicum under the current and future climatic conditions, (iii) to determine the susceptibility of R. geographicum to the possible future range contraction/expansion, and (iv) to propose conservation strategies and management options under different scenarios.
Material and methods
Study area
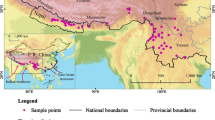
The study was carried out in the IHR which comprises four states (viz., Himachal Pradesh, Uttarakhand, Sikkim, and Arunachal Pradesh) and two Union Territories (viz., Jammu and Kashmir and Ladakh), covering approximately 12% of total geographical area of India (Fig. 1). Physiographically, the region has four zones from south to north, viz., the shivaliks, the lesser Himalaya, the greater Himalaya, and Trans-Himalaya zones. The mountain ranges in IHR are distributed up to an altitude of 8586 m and extending 8° (26° to 34° N) in latitude and 28° (69° to 97° E) in longitude. This huge mountain ranges of IHR have been historically divided into two eco-sensitive regions: the Eastern Himalaya, which covers Arunachal Pradesh and Sikkim states of north-eastern region of India, and the Western Himalaya, covering the Uttarakhand, Himachal Pradesh, Jammu and Kashmir, and Ladakh.
According to the State of Forest Report-2019, forest cover in the IHR is ~ 41% of the total geographical area out of which 16.9% area is under very dense forest cover, 45.4% under moderate forest cover, and the remaining 37.7% under open forest category (ISFR 2019). The Himalayan forests are extensive and diverse and they differ significantly from both tropical and temperate forests with respect to structure, growth cycle and function as well as in terms of ecosystem processes (Zobel and Singh 1997). Forest vegetation in the region ranges from tropical moist-deciduous forests in the foothills to alpine meadows above treeline (Champion and Seth 1968). About 6% of Indian human population lives in the IHR. The growing population and consequent anthropogenic pressures in the region have exerted considerable pressure on various ecosystems (Badola and Hussain 2003). Further, changing land use patterns, unsustainable use of natural resources, and unplanned infrastructure developments have led to habitat loss, ecological degradation, and forest fragmentation resulting to changes in species range, diversity, and numbers, and their local extinctions. In addition to this, climate change–induced impacts are predicted to affect the critical ecosystem goods and services provided by the IHR.
Target species
Rhizocarpon geographicum (L.) DC. (the map lichen; Family, Rhizocarpaceae) is a crustose lichen, which grows on exposed rock surface in higher mountain region (Fig. 2). This lichen species is broadly distributed in the North American range, sub-Antarctic islands, Antarctic Peninsula, Andes of Peru, Colombia, and South Asia (Caseldine and Baker 1998). In India it is distributed throughout the Himalayan region (Jammu and Kashmir, Himachal Pradesh, Uttarakhand, Sikkim, and Arunachal Pradesh) at 3000–4300-m altitude (Bajpai et al. 2016b). It grows at substantially lower rates than other foliose and crustose lichen, especially in arctic and alpine environment (Armstrong 2005). Having circular or roughly circular yellow green thalli, this lichen species is widely used in determining the relative age of deposits, e.g., moraine systems, thus revealing evidence of glacial advances, through lichenometry (Beschel 1973; Armstrong 2004). Lichenometry is the geomorphic method of geochronologic dating that uses lichen growth to determine the age of exposed rock, based on a presumed specific rate of increase in radial size of thalli over the time.
Species presence data
In total 159 species presence records of R. geographicum were incorporated in this investigation (Supplementary Material; Table S1). This includes 117 records from Lichen herbaria of CSIR-National Botanical Research Institute (NBRI), Lucknow, India, and 42 records from field survey in different natural habitats across IHR. The point’s record from each herbarium specimens was transformed into geographical coordinates using Google Earth and ArcGIS (version 10.3) software.
Environmental predictors
A total of 19 bioclimatic variables (Hijmans et al. 2005) from WorldClim database (http://www.worldclim.org) at a resolution of approximately 1 km2 were used for distribution modeling of R. geographicum. The variables derived from monthly mean temperature and precipitation data (Table 1) had a high conformity rate in species distribution modeling, even for those having few distribution records (Pearson and Dawson 2003; Khanum et al. 2013; Hamid et al. 2019; Kumar et al. 2019). In addition to 19 bioclimatic variables, topographic variable (altitude) of 30 arc second (approx. ~ 1 km) spatial resolution was utilized as additional predictor variables for modeling current potential distribution of R. geographicum. The future climate data from IPCC 5th assessment for the years 2050 and 2070 were used from http://www.ccafs-climate.org/ web-source to project the future potential distribution of the target species.
For reducing the uncertainty in model prediction, we used three different global climate models (GCMs), i.e., MRI-CGCM3, CSIRO-MK3.6, and HadGEM2-CC. Each GCM was tested under future greenhouse gas concentration trajectories, called representative concentration pathways (RCPs), i.e., RCP 4.5, RCP 6.0, and RCP 8.5, for the periods 2050 and 2070. The RCP 4.5 (“intermediate stabilization pathways”) was developed by Pacific Northwest National Laboratory, USA. It is relatively stable scenario in which greenhouse gases (GHG) will stabilize due to greenhouse technologies and total radiative forcing reaches 4.5 W/m2 and stabilized shortly after 2100, without overshooting the long-run radiative forcing target level (Smith and Wigley 2006; Wise et al. 2009). RCP 6.0 was developed by the National Institute for Environmental Studies, Japan, and it is a stabilization scenario where total radiative forcing is stabilized after 2100 without overshoot by employment of a range of technologies and strategies for reducing GHG (Fujino et al. 2006; Hijioka et al. 2008). However, RCP 8.5 was developed by International Institute for Applied Systems Analysis, Austria, and it represents relatively extreme scenarios where GHG will continuously increase throughout 2100, at which time radiative force will reach 8.5 W/m2 (Riahi et al. 2007; Meinshausen et al. 2011). For future potential distribution predictions, we ran averaging the results (ensemble approach) from the CGCM3, CSIRO-MK3, and HadGEM2-CC future climate models (Araújo and New 2007).
To reduce multi-collinearity among the 20 environmental variables, highly correlated variables were eliminated from further modeling using pair-wise Pearson’s correlation coefficient (r ≥ 0.85). This reduction of predictor variables resulted in the inclusion of seven variables for models (Supplementary Material; Table S2). These variables included annual mean temperature (Bio1), mean diurnal range temperature (Bio2), isothermality (Bio2/Bio7)*100 (Bio3), temperature seasonality (Bio4), mean temperature of wettest quarter (Bio8), precipitation of driest month (Bio14), and altitude (Alt) (Table 1).
Predictive MaxEnt modeling
For predictive modeling, we used maximum entropy–based techniques, i.e., MaxEnt ver. 3.3.3k (http://www.cs.princeton.edu/~schapire/MaxEnt/), because of their better performance with small sample sizes relative to other modeling algorithms (Elith et al. 2011). MaxEnt program is based on maximum entropy theory and uses only presence records data for predicting species distribution (Phillips et al. 2006). The probability output response of MaxEnt devised by Phillips et al. (2006) can either be raw, logistic, or cumulative, and for this study logistic response was selected, following Phillips and Dudik (2008).
In total, twenty replicate runs were set for modeling and averaged the result. In replicate run, cross-validation technique was selected, in which samples are divided into replicate folds and each fold is used to test data. To estimates relative influence of different predictor variables, jackknife test (Pearson et al. 2007; Shcheglovitova and Anderson 2013), response curve, and relative percentage contribution of variables (Rajpoot et al. 2020; Kumar et al. 2021) were used. We used the area under ROC (receiver operating characteristics) curve (AUC) to estimates model performance and accuracy. The value of AUC ranged from 0 to 1 (Fielding and Bell 1997). An AUC value closer to 0.5 indicates that the model did not perform better than random, whereas value closer to 1.0 indicates perfect and more accurate prediction (Swets 1988). The thresholds values adopted for interpreting the AUC were as follows: 0.5–0.6, fail; 0.6–0.7, poor; 0.7–0.8, fair; 0.8–0.9, good; and 0.9–1, excellent (Lobo et al. 2008). Further, model performance was assessed based on the partial AUC metrics (Lobo et al. 2008) calculated using online NicheToolBox (http://shiny.conabio.gob.mx:3838/nichetoolb2/). The distribution of AUC ratios was plotted by executing 5% omission with 500 bootstrap iterations, followed by comparison between mean value of AUCrandom and AUCpartial to test whether the model performed better than random predictions. For conformity of model robustness, we executed 10 percentile training presence threshold rules over a twenty replicates of model runs (Pearson et al. 2004). Other setting was set to default as the MaxEnt algorithms are already calibrated on a wide range of species datasets as suggested by Phillips and Dudik (2008).
Analysis of habitat change
We have analyzed the suitable habitat change area for future scenarios of 2050 and 2070 years under the RCP 4.5, 6.0, and 8.5. For the habitat suitability class maps, we categorized the final logistic output of MaxEnt model into the following classes: (i) unsuitable (0.00–0.25), (ii) low suitable (0.25–0.50), (iii) medium suitable (0.50–0.75), and (iv) high suitable (0.75–1.00) in DIVA GIS (version 7.5) (Hijmans et al. 2005).
Analysis of range contraction or expansion
By subtracting the binary map of current period with each of the future scenarios in ArcGIS (Version 10.3), climatic range shift maps of R. geographicum were generated for 2050 and 2070 years under RCP 4.5, 6.0, and 8.0. Each maps represent the following classes: (i) stable/no-change (predicted areas remain to suitable), (ii) unsuitable, (iii) lost (areas not predicted to remain suitable in future/range contraction), and (iv) gained habitats (areas that are predicted to be suitable in future but currently not suitable/range expansion).
Results
Model performance
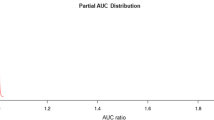
The model calibration test for R. geographicum yielded highly satisfactory results, evident by average training and test AUC values of 0.984 ± 0.002 and 0.968 ± 0.030, respectively. The average partial AUC value at 5% omission with 500 bootstrap iterations was 0.9635±0.012 (Fig. 3). All the tests for model performance showed that the MaxEnt model for R. geographicum exhibit excellent consistency and represents more defined and restricted ecological niche in the study area with high accuracy.
Partial area under curve (AUC) distribution for Rhizocarpon geographicum generated by executing 5% omission with 500 bootstrap iterations in the receiver operating characteristic (ROC) space. Red curve represents the distribution of AUC ratios for random models, while the blue curve along with shaded bars show the frequency distribution of the ratios between AUC from model prediction and AUCrandom
Influencing predictor variables
Amongst the seven selected environmental predictors, altitude (alt) and annual mean temperature (Bio1) together contributed 77.5% to the model and appeared as most influential variables for current potential distribution of R. geographicum (Table 1). Precipitation of driest month (Bio14) and mean diurnal range temperature (Bio2) made the lowest contribution to the predictive model. Considering the permutation importance, annual mean temperature (Bio1) had the maximum influence on model and contributed to 86.6%, followed by altitude (alt), i.e., 4.5% (Table 1). The climatic profiles (based on the occurrence records of species: minimum, maximum, and mean values) of 20 environmental variables for the species under investigation are presented in Table S3 (Supplementary Material).
The response curves showed the changes in the logistic prediction when each predictor variable changed by keeping all other variables at their average sample value. The response curve of R. geographicum showed that the elevation (alt) influenced the habitat suitability area at certain range only (Fig. 4a), and a bell-shaped distribution curve was observed for annual mean temperature (Bio1) (Fig. 4b). Mean temperature of wettest quarter (Bio8) and temperature seasonality (Bio4) negatively influenced the logistic prediction (Fig. 4c,d). The jackknife test showed that the annual mean temperature (Bio1) and altitude (alt) are the main factors influencing the distribution pattern of R. geographicum and showed highest training gain (Fig. 4e).
Potential current prediction
By undertaking visual inspection of the potential current prediction of R. geographicum based on occurrence records, it is clear that the Eastern Himalayan region possesses highly suitable habitat (red) distributed through Sikkim to Arunachal Pradesh state of IHR, whereas in Western Himalayan region, Uttarakhand and Himachal Pradesh possess highly suitable habitat classes (Fig. 5a). The current suitable habitats of R. geographicum cover 27.7% of the total geographical area of IHR and are confined mainly to areas between 3000 and 4300 m. Such habitats occur throughout the Eastern Himalayan region and Western Himalayan region, except the westernmost part (Jammu & Kashmir and Ladakh) (Fig. 6a).
Habitat suitability classes under current and future climatic conditions
During the future climate change scenarios (2050 and 2070), some of the areas which are currently less suitable for R. geographicum will become suitable, and at the same time, some areas which are modeled to be highly suitable in the current climatic condition will become less suitable. In the Eastern Himalayan region, the highly suitable habitat area (red) is predicted to increase and shift towards higher elevations in future climatic scenarios (Fig. 5). In the Western Himalayan region, the loss of suitable habitat area is predicted more for all habitat classes, i.e., low (light blue), moderate (dark blue), and high (red), for the future climate change scenarios (Fig. 5).
Our analysis showed that in 2050, there will be a decrease in highly suitable habitat area (showed in red color) by − 1.69% (RCP 6.0) to − 5.50% (RCP 8.5). Similarly, the moderately suitable habitat area (dark blue color) showed considerable loss in suitable area in all scenarios, i.e., RCP 4.5, 6.0, and 8.5 by − 2.13%, − 4.48%, and − 0.50%, respectively. The low suitable habitat area (light blue) showed marginal decrease by − 1.39% in RCP 6.0 and a marginal increase of 0.83% and 0.25% in RCP 4.5 and RCP 8.5, respectively. Similarly, in 2070, it is expected that there will be a considerable decrease in highly suitable habitat area (red), by − 4.10% and − 6.46% under RCP 4.5 and RCP 8.5 and marginal decrease by − 1.48% under the RCP 6.0. The moderately suitable habitat area (dark blue) will also be lost by − 9.45%, − 2.71% and − 3.52% for RCP 4.5, 6.0, and 8.5, respectively. The low suitable habitat area (light blue) showed very small gain in suitable area by 0.11% for RCP 6.0 and considerable loss in suitable area by − 4.46% and − 2.77% for RCP 4.5 and RCP 8.5, respectively. The unsuitable area (white) showed an increase for all future scenarios (RCP 4.5, 6.0 and 8.5) over the years 2050 and 2070 (Table 2 and Fig. 5).
Range contraction or expansion
Area with suitable climatic conditions for R. geographicum is predicted to decline by the 2050s and the 2070s under all RCP scenarios. The averaged future prediction from three GCMs under the RCP 4.5 by year 2050 showed the loss of 17.03% (51,322 km2), suitable area (indicated in dark green color/ no change/ stable), and an increase by year 2070 (56,822 km2; 18.66%), from the current climatic suitable value as 27.96% (84,252 km2) (Table 3; Fig. 6). In RCP 6.0, the average future prediction for the year 2050 showed loss of suitable habitat area (49,791 km2; 16.52%), and further loss in suitable habitat area (46,763 km2; 15.53%) is predicted for the year 2070 (Table 3; Fig. 6). Whereas in higher scenarios (RCP 8.5) the loss of suitable habitat area predicted was greater than both the RCPs (4.5 and 6.0), and the predicted loss in suitable habitat area is from 14.11% (42,518 km2) to 11.38% (34,305km2) for the year 2050 and 2070 respectively (Table 3; Fig. 6). The loss in suitable habitat area in average model of RCP 4.5 and 6.0 was predicted to occur in patches in eastern Himalaya region (Sikkim and Arunachal Pradesh) and in large continuous blocks in Western Himalaya (Jammu and Kashmir, Ladakh, Himachal Pradesh and Uttarakhand) ([Fig. 6).
It is estimated that in 2050 there will be a huge range contraction (area indicated in red color) in current suitable area of R. geographicum towards north-west side of Western Himalaya (Jammu and Kashmir). Conversely, there will be range expansion (area showed in purple color) in the habitat by 15,394 km2 (5.11%) in RCP 6.0 to 23,166 km2 (7.69%) in RCP 8.5, towards the northern side of Eastern Himalaya (Sikkim and Arunachal Pradesh) and north-eastern side of Western Himalaya (Himachal Pradesh and Uttarakhand). The unsuitable area (area indicated in light green color) varied from 1,94,144 km2 (64.42%) to 2,01,916 km2 (67.0%). Likewise, by 2070, range contraction (red) of current suitable area for R. geographicum varied from 27,430 km2 (9.10%) under RCP 4.5 to 49,747 km2 (16.51%) under RCP 8.5 in lower elevation zone of Eastern and Western Himalayan regions, and there will be a range expansion (purple) in suitable habitat from 24,310 km2 (8.07%) in RCP 6.0 to 39,856 km2 (8.50%) in RCP 8.5, towards the north-eastern parts of Western Himalayan states (Jammu & Kashmir, Ladhakh Himachal Pradesh and Uttarakhand). The unsuitable area (light green) by 2070 also varied from 1,77,454 km2 (58.88%) in RCP 6.0 to 1,93,000 km2 (64.04%) in RCP 8.5. In summary both scenarios (2050 and 2070) showed similar trends for range contraction and expansion, and collectively the range expansion/contraction reached peak under the RCP 8.5 scenario for the year 2070 (Table 3 and Fig. 6).
Discussion
ENM tools have become increasingly appreciable in ecological and bio-geographical research globally (Guisan and Zimmermann 2000), mostly because ecologists need ways of rapidly assessing potential distribution and impact of climate change on large numbers of species, with only few distribution records (Araújo and Rahbek 2006). Several critical opinions on ENM analysis have been presented over the years, and MaxEnt seems to be the most reliable application for modeling species distribution (Bradie and Leung 2016), and its usefulness was also tested in the case of rare species (Kumar and Stohlgren 2009; Rebelo and Jones 2010). In the present investigation, the MaxEnt modeling result suggests that the suitable habitat area for R. geographicum would shrink under the predicted levels of climate change. The MaxEnt model performed better than random, with high satisfactory mean test AUC value. MaxEnt predicted the loss of suitable habitat area more in Western Himalayan region than in Eastern Himalayan region. It was also predicted that the species may shift upward (i.e., more than 4000-m-high elevated zones), mostly in Western Himalayan region (Uttarakhand, Himachal Pradesh, and Jammu and Kashmir) (Fig. 6).
Field observations suggest the R. geographicum is highly dependent on local seasonal climate and generally colonized on the exposed rock surfaces in treeline ecotones to alpine meadows between 3000 and 3900 m in Western Himalaya and 3500 and 4400 in Eastern Himalaya. Among the temperature-related climatic variables, the mean annual temperature, mean temperature of wettest quarter, and temperature seasonality have shown significant contribution in model prediction. Currently, R. geographicum is more abundant (highly suitable habitat area) towards the Eastern Himalayan region, possibly because of low mean temperature of wettest quarter and higher mean temperature of warmest month. The low winter temperature at treeline elevations in Western Himalayan region seems to restrict its abundance (Sharma et al. 2009; Ranjitkar et al. 2014; Tewari et al. 2017).
The slow growth rate and longevity of Rhizocarpon species are valuable tools to estimate the exposure age of rock surface, i.e., in lichenometry (Locke et al. 1979; Innes 1985; Mathews 1994; Benedict 2009; Bajpai et al. 2016b). Thus, considering the importance of R. geographicum in lichenometry, the present investigation of MaxEnt model showed that the predicted suitable habitat area under the current climate condition would become unsuitable in the predicted future climate change scenarios, indicating direct evidence for declining or melting of glacier/ice cover in future climate change scenarios.
Considering climate sensitivity of the species, the seasonality (such as temperature, rainfall, and humidity) exposure to it leads to a regular, periodic changes in the growth of thalli and availability of resources (Armstrong 2006; Armstrong 2011). Innes (1985) found that close to snow patches, the thalli of R. geographicum were smaller than expected. Similarly, Pitman (1973) observed that the thallus diameter increased away from the center of snow patches due to a reduced growing season and ground instability. All of these factors are known to combine in trigging the morphological and physiological changes in species (Armstrong 2011). Then, the species may be capable of adapting to future climate condition through physiological changes or through adaptation to microclimatic conditions responsible for its survival in natural conditions.
In the current investigation, the MaxEnt model output of R. geographicum showed the range expansion of suitable habitat towards higher elevation areas of Himalaya in future climate scenarios, which may be possible through adaption towards the changes in local climatic condition. These range expansions were generally more pronounced for the extreme future scenarios (RCP 8.5) than for the moderate and intermediate climate scenarios (RCP 4.5 and RCP 6.0). Rubio-Salcedo et al. 2017) predicted similar results of adaption and range expansion for forty one lichen species in Iberian Peninsula in response to the climate change, using ensemble climatic modeling approach, whereas Ellis et al. (2014) projected the suitable climate space for 382 epiphytic lichen species in Brittan and showed that 38% of the species losing and 62% are gaining the suitable habitat area, by 2080. Furthermore, a study across twenty-six mountains in Switzerland demonstrated upward (towards the higher elevation) range extension of the alpine vegetation (Pauli et al. 1996). A similar study by Moiseev and Shiyatov (2003) showed the upward shift of treeline in Siberia. Several studies mainly showed the significant loss of suitable habitat under climate change scenarios for many lichen species in various geographical regions (Hauck 2009; Binder and Ellis 2008; Lang et al. 2012; Ellis 2013; Allen and Lendemer 2016; Rubio-Salcedo et al. 2017; Ellis 2019). Recently, Allen and Lendemer (2016) and Devkota et al. (2019) have shown that climate change poses a significant threat to montane lichen species in higher elevation area. Devkota et al. (2019) predicted the loss of suitable habitat for endemic lichen Lobaria pindarensis in the Hindu Kush Himalayan (HKH) region under the future climate change scenarios. Our model also predicted a contraction of suitable habitat range (10.86–16.51 %) for R. geographicum in the IHR (part of HKH region).
In the present investigation, the overall MaxEnt model predicted that the suitable area of R. geographicum may expand towards higher altitudes of Western Himalayan region, while it may contract towards lower altitude in both Western and Eastern Himalayan regions (Fig. 6). These results can be attributed to a rapid deglaciation in the Western Himalaya, which started much before the global average of degradation and may prevail in future climatic conditions (Shekhar et al. 2017). Both range expansion and contraction peaks were observed in RCP 8.5 of future climate scenarios for the year 2070. Such results are in concordance with studies of lichen in HKH (Devkota et al. 2019), and other parts of the world (Binder and Ellis 2008; Ellis et al. 2007a; Ellis et al. 2009; Ellis 2015; Ellis et al. 2014; Allen and Lendemer 2016; Nascimbene et al. 2016; Rubio-Salcedo et al. 2017; Fačkovcová et al. 2017), which also exhibited both expansion and contraction of potential habitat in response to climate change.
Conclusion
Conservation of pioneer and vulnerable ecosystems at species level has often failed owing to lack of proper knowledge about target species in terms of their habitat stabilities. The application of ENM to extract the basic inventory data of species, i.e., only presence records, to provide prediction of species distribution under changing climate at regional and global scale, may be further used to make recommendations for policymakers and conservationists dealing with impact of climate change and its ecosystem functioning (Sanchez et al. 2011). In lichens, species distribution modeling with MaxEnt extended in to a new tools and it would be especially useful in reconstructing their distribution and potential migratory routes. In the present investigation, we used the maximum entropy–based modeling algorithms, as implemented in ENM software MaxEnt (Phillips et al. 2006), to characterize the environmental niche of the R. geographicum and to predict the changes in distribution under climate change scenarios. So far, MaxEnt has not implemented and linked with in any lichenometry-based crustose lichen species to predict the changes in alpine ecosystem. MaxEnt accounts some consequences; for example, it does not estimate directly the probability of occurrence, and rather, it estimates the environmental suitability for the species and can be mapped in a geographical space (Royle et al. 2012). Despite the limitations, it is most and widely used algorithms with good predictive power capabilities. It may be pointed out that actual distribution of any climate sensitive species at regional scale depends upon its variability in physiological tolerance; thus, trait coupled niche modeling analysis is required for its effective conservation. In addition, a long-term species-specific investigation is required at representative sites to develop mitigation measures under the changing climate. Since Nepal constitutes a significant part of the species distributional range, collaborative efforts would go a long way in conserving these critical ecosystems. Trans-boundary cooperation among the Himalayan countries is of critical importance in addressing conservation issues of the Himalayan region.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AUC:
-
area under the curve
- CCAFS:
-
CGIAR Research Program on Climate Change, Agriculture and Food Security
- CMIP5:
-
Coupled Model Intercomparison Project Phase 5
- ENM:
-
ecological niche modeling
- GCM:
-
global climate model
- GHG:
-
greenhouse gases
- IHR:
-
Indian Himalayan Region
- IPCC:
-
Intergovernmental Panel on Climate Change
- MaxEnt:
-
maximum entropy
- MRI-CGCM3:
-
Meteorological Research Institute Coupled Global Climate ModelVersion-3
- RCP:
-
representative concentration pathway
- ROC:
-
receiver operating characteristic curve
References
Allen JL, Lendemer JC (2016) Climate change impacts on endemic, high-elevation lichens in a biodiversity hotspot. Biodivers Conserv 25:555–568. https://doi.org/10.1007/s10531-016-1071-4
Anderson RP, Martinez-Meyer E (2004) Modeling species’ geographic distributions for preliminary conservation assessments: an implementation with the spiny pocket mice (Heteromys) of Ecuador. Biol Conserv 116:167–179. https://doi.org/10.1016/S0006-3207(03)00187-3
Araújo MB, New M (2007) Ensemble forecasting of species distribution. Trends Ecol Evol 22:42–47. https://doi.org/10.1016/j.tree.2006.09.010
Araújo MB, Rahbek C (2006) How does climate change affect biodiversity? Science 313:1396–1397. https://doi.org/10.1126/science.1131758
Armstrong RA (2004) Lichens, lichenometry and global warming. Microbiologist 5(32):e35
Armstrong RA (2005) Radial growth of Rhizocarpon section Rhizocarpon lichen thalli over six years at Snoqualmie Pass in the Cascade Range, Washington State. Arct Antarct Alp Res 37(4):411–415. https://doi.org/10.1657/1523-0430(2005)037[0411:RGORSR]2.0.CO;2
Armstrong RA (2006) Seasonal growth of the crustose lichen Rhizocarpon geographicum (L.) DC. in South Gwynedd, Wales. Symbiosis 41:97–102
Armstrong RA (2011) The biology of the crustose lichen Rhizocarpon geographicum. Symbiosis 55:53–67. https://doi.org/10.1007/s13199-011-0147-x
Badola R, Hussain SA (2003) Conflict in paradise: women and protected areas in the Indian Himalayas. Mount Res Dev 23(3):234–237
Bajpai R, Mishra S, Dwivedi S, Upreti DK (2016a) Change in atmospheric deposition during last half century and its impact on lichen community structure in Eastern Himalaya. Sci Rep 6(1):1–14. https://doi.org/10.1038/srep30838
Bajpai R, Singh CP, Shukla P, Upreti DK (2016b) Preliminary lichenometric studies in Eastern and North-Western Himalaya. J Geol Soc India 87:535–538. https://doi.org/10.1007/s12594-016-0427-4
Benedict JB (2009) A review of lichenometric dating and its applications to archaeology. Am Antiq 74:143–172. https://doi.org/10.2307/25470542
Beschel RE (1973) Lichen as a measure of the age of recent Moraines. Arct Antarct Alp Res 5(4):303–309. https://doi.org/10.1080/00040851.1973.12003739
Binder MD, Ellis CJ (2008) Conservation of the rare British lichen Vulpicida pinastri: changing climate, habitat loss and strategies for mitigation. Lichenologist 40:63–79. https://doi.org/10.1017/S0024282908007275
Bradie J, Leung B (2016) A quantitative synthesis of the importance of variables used in MaxEnt species distribution models. J Biogeogr 44(6):1344–1361. https://doi.org/10.1111/jbi.12894
Branquinho C, Matos P, Pinho P (2015) Lichens as ecological indicators to track atmospheric changes: future challenges. In: Lindenmayer D, Barton P, Pierson J (eds) Indicators and surrogates of biodiversity and environmental change. CSIRO Publishing, CRC Press, Melbourne
Cameron RP, Neily T, Clayden SR (2011) Distribution prediction model for Erioderma mollissimum in Atlantic Canada. Bryologist 114:231–238. https://doi.org/10.1639/0007-2745-114.1.231
Carlsen T, Bendiksby M, Hofton TH, Reiso S, Bakkestuen V, Haugan R, Kauserud H, Timdal E (2012) Species delimitation, bioclimatic range, and conservation status of the threatened lichen Fuscopannaria confusa. Lichenologist 45(5):565–575. https://doi.org/10.1017/S0024282912000199
Caseldine C, Baker A (1998) Frequency Distributions of Rhizocarpon geographicum s.l., Modeling, and Climate Variation in Tröllaskagi, Northern Iceland. Arct Antarct Alp Res 30(2):175–183
Champion HG, Seth SK (1968) A revised survey of the forest types of India. Government of India, New Delhi
Devkota S, Dymytrova L, Chaudhary RP, Werth S, Scheidegger C (2019) Climate change-induced range shift of the endemic epiphytic lichen Lobaria pindarensis in the Hindu Kush Himalayan region. Lichenologist 51(2):157–173. https://doi.org/10.1017/S002428291900001X
Elith J, Leathwick JR (2009) Species distribution models: ecological explanation and prediction across space and time. Annu Rev Ecol Evol Syst 40:677–697. https://doi.org/10.1146/annurev.ecolsys.110308.120159
Elith J, Phillips SJ, Hastie T, Dudik M, Chee YE, Yates CJ (2011) A statistical explanation of MaxEnt for ecologists. Divers Distrib 17:43–57. https://doi.org/10.1111/j.1472-4642.2010.00725.x
Ellis CJ (2013) A risk-based model of climate change threat: hazard, exposure, and vulnerability in the ecology of lichen epiphytes. Botany 91(1):1–11. https://doi.org/10.1139/cjb-2012-0171
Ellis CJ (2015) Ancient woodland indicators signal the climate change risk for dispersal-limited species. Ecol Indic 53:106–114. https://doi.org/10.1016/j.ecolind.2015.01.028
Ellis CJ (2019) Climate change, bioclimatic models and the risk to lichen diversity. Diversity 11(4):54. https://doi.org/10.3390/d11040054
Ellis CJ, Coppins BJ, Dawson TP, Seaward MRD (2007a) Response of British lichens to climate change scenarios: trends and uncertainties in the projected impact for contrasting biogeographic groups. Biol Conserv 140:217–235. https://doi.org/10.1016/j.biocon.2007.08.016
Ellis CJ, Coppins BJ, Dawson TP (2007b) Predicted response of the lichen epiphyte Lecanora populicola to climate change scenarios in a clean-air region of Northern Britain. Biol Conserv 135(3):396–404. https://doi.org/10.1016/j.biocon.2006.10.036
Ellis CJ, Yahr R, Coppins BJ (2009) Local extent of old-growth woodland modifies epiphyte response to climate change. J Biogeogr 36(2):302–313. https://doi.org/10.1111/j.1365-2699.2008.01989.x
Ellis CJ, Eaton S, Theodoropoulos M, Coppins BJ, Seaward MRD, Simkin J (2014) Response of epiphytic lichens to 21st Century climate change and tree disease scenarios. Biol Conserv 180:153–164. https://doi.org/10.1016/j.biocon.2014.09.046
Fačkovcová Z, Senko D, Svitok M, Guttová A (2017) Ecological niche conservatism shapes the distributions of lichens: geographical segregation does not reflect ecological differentiation. Preslia 89:63–85. https://doi.org/10.23855/PRESLIA.2017.063
Fielding AH, Bell JFL (1997) A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ Conserv 24(1):38–49. https://doi.org/10.1017/S0376892997000088
Forrest JL, Wikramanayake E, Shrestha R, Areendran G, Gyeltshen K, Maheshwari A, Mazumdar S, Naidoo R, Thapa GJ, Thapa K (2012) Conservation and climate change: assessing the vulnerability of snow leopard habitat to treeline shift in the Himalaya. Biol Conserv 150:129–135. https://doi.org/10.1016/j.biocon.2012.03.001
Fujino J, Nair R, Kainuma M, Masui T, Matsuoka Y (2006) Multi-gas mitigation analysis on stabilization scenarios using AIM global model. Energy J 3:343–354. https://doi.org/10.5547/ISSN0195-6574-EJ-VolSI2006-NoSI3-17
Garcia K, Lasco R, Ines A, Lyon B, Pulhin F (2013) Predicting geographic distribution and habitat suitability due to climate change of selected threatened forest tree species in the Philippines. Appl Geogr 44:12–22. https://doi.org/10.1016/j.apgeog.2013.07.005
Green TGA, Sancho LG, Pintado A (2011) Ecophysiology of desiccation/rehydration cycles in mosses and lichens. In: Lüttge U, Beck E, Bartels D (eds) Plant Desiccation Tolerance. Ecological Studies (Analysis and Synthesis), vol 215. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-19106-0_6
Guisan A, Zimmermann NE (2000) Predictive habitat distribution models in ecology. Ecol Model 135:147–186. https://doi.org/10.1016/S0304-3800(00)00354-9
Hamid M, Khuroo A, Charles B, Ahmad R, Singh CP, Aravind NA (2019) Impact of climate change on the distribution range and niche dynamics of Himalayan birch, a typical treeline species in Himalayas. Biodivers Conserv 28:2345–2370. https://doi.org/10.1007/s10531-018-1641-8
Hauck M (2009) Global warming and alternative causes of decline in arctic-alpine and boreal-montane lichens in North-Western Central Europe. Glob Chang Biol 15(11):2653–2661. https://doi.org/10.1111/j.1365-2486.2009.01968.x
Hijioka Y, Matsuoka Y, Nishimoto H, Masui M, Kainuma M (2008) Global GHG emissions scenarios under GHG concentration stabilization targets. J Glob Environ Eng 13:97–108
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978. https://doi.org/10.1002/joc.1276
Innes JL (1985) A standard Rhizocarpon nomenclature for lichenometry. Boreas 14:83–85. https://doi.org/10.1111/j.1502-3885.1985.tb00890.x
ISFR (2019) India State of Forest Report 2019 by Forest Survey of India. Ministry of Environment and Forests & Climate Change, Government of India, Dehradun, India. Assessed 21 Mar. 2021. https://fsi.nic.in/isfr-volume-i?pgID=isfr-volume-i
Khanum R, Mumtaz AS, Kumar S (2013) Predicting impacts of climate change on medicinal asclepiads of Pakistan using Maxent modelling. Acta Oecol 49:23–31. https://doi.org/10.1016/j.actao.2013.02.007
Kong F, Tang L, He H, Yang F, Tao J, Wang W (2021) Assessing the impact of climate change on the distribution of Osmanthus fragrans using Maxent. Environ Sci Pollut Res 28:34655–34663. https://doi.org/10.1007/s11356-021-13121-3
Krishnan R, Shrestha AB, Ren G, Rajbhandari R, Saeed S, Sanjay J, Syed MA, Vellore R, Xu Y, You Q, Ren Y (2019) Unravelling climate change in the Hindu Kush Himalaya: rapid warming in the mountains and increasing extremes. In: Wester P, Mishra A, Mukherji A, Shrestha AB (eds) The Hindu Kush Himalaya Assessment-Mountains, Climate Change, Sustainability and People. Springer, Cham., pp 57–97. https://doi.org/10.1007/978-3-319-92288-1_3
Kukwa M, Kolanowska M (2016) Glacial refugia and the prediction of future habitat coverage of the South American lichen species Ochrolechia austroamericana. Sci Rep 6(1):1–9. https://doi.org/10.1038/srep38779
Kumar P (2012) Assessment of impact of climate change on Rhododendrons in Sikkim Himalayas using Maxent modelling: limitations and challenges. Biodivers Conserv 21:1251–1266. https://doi.org/10.1007/s10531-012-0279-1
Kumar S, Stohlgren TJ (2009) Maxent modeling for predicting suitable habitat for threatened and endangered tree Canacomyrica monticola in New Caledonia. J Ecol Nat Environ 1(4):094–098
Kumar D, Singh M, Sharma S (2019) Fate of important medicinal plants in the eastern Himalaya in changing climate scenarios: a case of Panax pseudoginseng Wall. Appl Ecol Environ Res 17(6):13493–13511. https://doi.org/10.15666/aeer/1706_1349313511
Kumar D, Rawat S, Joshi R (2021) Predicting the current and future suitable habitat distribution of the medicinal tree Oroxylum indicum (L.) Kurz in India. J Appl Res Med Aromat Plants 23:100309. https://doi.org/10.1016/j.jarmap.2021.100309
Lang SI, Cornelissen JHC, Shaver GR, Ahrens M, Callaghan TV, Molau U, Ter Braak CJF, Hölzer A, Aerts R (2012) Arctic warming on two continents has consistent negative effects on lichen diversity and mixed effects on bryophyte diversity. Glob Chang Biol 18(3):1096–1107. https://doi.org/10.1111/j.1365-2486.2011.02570.x
Lavorel S, Ganier E (2002) Predicting changes in community composition and ecosystem functioning from plant traits: revisiting the Holy Grail. Funct Ecol 16(5):545–556. https://doi.org/10.1046/j.1365-2435.2002.00664.x
Lobo JM, Jiménez-Valverde A, Real R (2008) AUC: a misleading measure of the performance of predictive distribution models. Glob Ecol Biogeogr 17:145–151. https://doi.org/10.1111/j.1466-8238.2007.00358.x
Locke WW, Andrews JT, Webber PJ (1979) A manual for lichenometry. British Geomorphological Research Group Technical Bulletin 261:47 pp
Manish K, Telwala Y, Nautiyal DC, Pandit MK (2016) Modelling the impacts of future climate change on plant communities in the Himalaya: a case study from Eastern Himalaya, India. Model Earth Syst Environ 2(2):1–12. https://doi.org/10.1007/s40808-016-0163-1
Mathews JA (1994) Lichenometric dating: a review with particular reference to ‘Little Ice Age’ moraines in southern Norway. In: Beck C (ed) Dating in surface context. New Mexico, Albuquerque, pp 185–212
Meinshausen M, Smith SJ, Calvin K, Daniel JS, Kainuma MLT, Lamarque J-F, Matsumoto K, Montzka SA, Raper SCB, Riahi K, Thomson A, Velders GJM, van Vuuren DPP (2011) The RCP greenhouse gas concentrations and their extensions from 1765 to 2300. Clim Chang 109:213–241. https://doi.org/10.1007/s10584-011-0156-z
Miądlikowska J, Kauff F, Högnabba F, Oliver JC, Molnár K, Fraker E, Gaya E, Hafellner J, Hofstetter V, Gueidan C, Kukwa M, Lücking R, Björk C, Sipman HJM, Burgaz AR, Thell A, Passo A, Myllys L, Goward T, Fernández-Brime S, Hestmark G, Lendemer J, Thorsten Lumbsch H, Schmull M, Schoch C, Sérusiaux E, Maddison DR, Elizabeth Arnold A, Lutzoni F, Stenroos S (2014) A multigene phylogenetic synthesis for the class Lecanoromycetes (Ascomycota): 1307 fungi representing 1139 infrageneric taxa, 317 genera and 66 families. Mol Phylogenet Evol 79:132–168 https://doi.org/10.1016/j.ympev.2014.04.003
Mittermeier RA, Turner WR, Larsen FW, Brooks TM, Gascon C (2011) Global biodiversity conservation: the critical role of hotspots. In: Zachos F, Habel J (eds) Biodiversity Hotspots. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-20992-5_1
Moiseev PA, Shiyatov SG (2003) Vegetation dynamics at the treeline ecotone in the Ural Highlands, Russia. In: Nagy L, Grabherr G, Körner C, DBA T (eds) Alpine Biodiversity in Europe. Ecological Studies (Analysis and Synthesis), vol 167. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-18967-8_26
Nascimbene J, Casazza G, Benesperi R, Catalano I, Cataldo D, Grillo M, Isocrono D, Matteucci E, Ongaro S, Potenza G, Puntillo D, Ravera S, Zedda L, Giordani P (2016) Climate change fosters the decline of epiphytic Lobaria species in Italy. Biol Conserv 201:377–384. https://doi.org/10.1016/j.biocon.2016.08.003
Nash TH III, Reiner A, Demmig-Adams B, Kilian E, Kaiser WM, Lance OL (1990) The effect of atmospheric desiccation and osmotic water stress on photosynthesis and dark respiration of lichens. New Phytol 116:269–276. https://doi.org/10.1111/j.1469-8137.1990.tb04714.x
Pandit MK, Babu CR (1998) Biology and conservation of Coptis teeta Wall: an endemic and endangered medicinal herb of Eastern Himalaya. Environ Conserv 25(3):262–272. https://doi.org/10.1017/S0376892998000320
Pandit MK, Sodhi NS, Koh LP, Bhaskar A, Brook BW (2007) Unreported yet massive deforestation driving loss of endemic biodiversity in Indian Himalaya. Biodivers Conserv 16:153–163. https://doi.org/10.1007/s10531-006-9038-5
Pauli H, Gottfried M, Grabherr G (1996) Effects of climate change on mountain ecosystems - upward shifting of alpine plants. World Resour Rev 8:382–390
Pearson RG, Dawson TP (2003) Predicting the impacts of climate change on the distribution of species: Are bioclimate envelope models useful? Glob Ecol Biogeogr 12:361–371. https://doi.org/10.1046/j.1466-822X.2003.00042.x
Pearson RG, Dawson TP, Liu C (2004) Modelling species distributions in Britain: a hierarchical integration of climate and land-cover data. Ecography 27:285–298. https://doi.org/10.1111/j.0906-7590.2004.03740.x
Pearson RG, Raxworthy CJ, Nakamura M, Peterson AT (2007) Predicting species distributions from small numbers of occurrence records: a test case using cryptic geckos in Madagascar. J Biogeogr 34:102–117. https://doi.org/10.1111/j.1365-2699.2006.01594.x
Pereira HM, Leadley PW, Proença V, Alkemade R, Scharlemann JPW, Fernandez-Manjarrés JF, Araújo MB, Balvanera P, Biggs R, Cheung WWL, Chini L, Cooper HD, Gilman EL, Guénette S, Hurtt GC, Huntington HP, Mace GM, Oberdorff T, Revenga C, Rodrigues P, Scholes RJ, Sumaila UR, Walpole M (2010) Scenarios for global biodiversity in the 21st century. Science 330:1496–1501. https://doi.org/10.1126/science.1196624
Peterson AT, Soberón J, Pearson RG, Anderson RP, Martínez-Meyer E, Nakamura M, Araújo MB (2011) Ecological niches and geographic distributions. Princeton University Press, Princeton
Phillips SJ, Dudik M (2008) Modelling of species distributions with MaxEnt: new extensions and a comprehensive evaluation. Ecography 31(2):161–175. https://doi.org/10.1111/j.0906-7590.2008.5203.x
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modelling of species geographic distributions. Ecol Model 190:231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Pinho P, Dias T, Cruz C, Sim Tang Y, Sutton MA, Martins-Loução MA, Máguas C, Branquinho C (2011) Using lichen functional diversity to assess the effects of atmospheric ammonia in Mediterranean woodlands. J Appl Ecol 48:1107–1116. https://doi.org/10.1111/j.1365-2664.2011.02033.x
Pitman GTK (1973) A lichenometrical study of snowpatch variation in the Frederikshåb district, southwest Greenland and its implication for the study of climatic and glacial fluctuations. Bull Grønl Unders 104:1–31
Rajpoot R, Adhikari D, Verma S, Saikia P, Kumar A, Grant KR, Datanandan A, Kumar A, Khare PK, Khan ML (2020) Climate models predict a divergent future for the medicinal tree Boswellia serrata Roxb. in India. Glob Ecol Conserv 23:e01040. https://doi.org/10.1016/j.gecco.2020.e01040
Ranjitkar S, Kindt R, Sujakhu NM, Hart R, Guo W, Yang X, Shrestha KK, Xu J, Luedeling E (2014) Separation of the bioclimatic spaces of Himalayan tree rhododendron species predicted by ensemble suitability models. Glob Ecol Conserv 1:2–12. https://doi.org/10.1016/j.gecco.2014.07.001
Raxworthy CJ, Martinez-Meyer E, Horning N, Nussbaum RA, Schneider GE, Ortega-Huerta MA, Peterson AT (2003) Predicting distributions of known and unknown reptile species in Madagascar. Nature 426:837–841. https://doi.org/10.1038/nature02205
Rebelo H, Jones G (2010) Ground validation of presence-only modelling with rare species: a case study on barbastelles Barbastella barbastellus (Chiroptera: Vespertilionidae). J Appl Ecol 47(2):410–420. https://doi.org/10.1111/j.1365-2664.2009.01765.x
Riahi K, Gruebler A, Nakicenovic N (2007) Scenarios of long-term socio-economic and environmental development under climate stabilization. Technol Forecast Soc Change 74(7):887–935. https://doi.org/10.1016/j.techfore.2006.05.026
Royle JA, Chandler RB, Yackulic C, Nichols JD (2012) Likelihood analysis of species occurrence probability from presence-only data for modelling species distributions: Likelihood analysis of presence-only data. Methods Ecol Evol 3(3):545–554. https://doi.org/10.1111/j.2041-210X.2011.00182.x
Rubio-Salcedo M, Psomas A, Prieto M, Zimmermann NE, Martínez I (2017) Case study of the implications of climate change for lichen diversity and distributions. Biodivers Conserv 26:1121–1141. https://doi.org/10.1007/s10531-016-1289-1
Sanchez AC, Osborne PE, Haq N (2011) Climate change and the African baobab (Adansonia digitata L.): the need for better conservation strategies. Afr J Ecol 49:234–245. https://doi.org/10.1111/j.1365-2028.2011.01257.x
Saran S, Joshi R, Sharma S, Padalia H, Dadhwal VK (2010) Geospatial modeling of Brown oak (Quercus semecarpifolia) habitats in the Kumaun Himalaya under climate change scenario. J Indian Soc Remote Sens 38:535–547. https://doi.org/10.1007/s12524-010-0038-2
Sharma E, Chettri N, Tse-ring K, Shrestha AB, Jing F, Mool P, Eriksson M (2009) Climate change impacts and vulnerability in the Eastern Himalayas. International Centre for Integrated Mountain Development (ICIMOD), Kathmandu
Shcheglovitova M, Anderson RP (2013) Estimating optimal complexity for ecological niche models: a jackknife approach for species with small sample sizes. Ecol Model 269:9–17. https://doi.org/10.1016/j.ecolmodel.2013.08.011
Shekhar M, Bhardwaj A, Singh S, Ranhotra PS, Bhattacharyya A, Pal AK, Roy I, Martín-Torres FJ, Zorzano MP (2017) Himalayan glaciers experienced significant mass loss during later phases of little ice age. Sci Rep 7(1):1–14. https://doi.org/10.1038/s41598-017-09212-2
Shrestha UB, Bawa KS (2014) Impact of climate change on potential distribution of Chinese caterpillar fungus (Ophiocordyceps sinensis) in Nepal Himalaya. PLoS One 9(9):e106405. https://doi.org/10.1371/journal.pone.0106405
Shrestha UB, Gautam S, Bawa KS (2012) Widespread Climate Change in the Himalayas and Associated Changes in Local Ecosystems. PLoS One 7(5):e36741. https://doi.org/10.1371/journal.pone.0036741
Smith SJ, Wigley TML (2006) Multi-Gas Forcing Stabilization with the MiniCAM. Energy J 27:373–391
Stocker TF, Qin D, Plattner GK, Tignor MM, Allen SK, Boschung J et al (2014) Climate change 2013: the physical science basis. Cambridge University Press, Contribution of Working Group I to the Fifth Assessment Report of IPCC the Intergovernmental Panel on Climate Change
Swets J (1988) Measuring the accuracy of diagnostic systems. Science 240:1285–1293. https://doi.org/10.1126/science.3287615
Szczepańska K, Pruchniewicz D, Kossowska M (2015) Modeling the potential distribution of three lichens of the Xanthoparmelia pulla group (Parmeliaceae, Ascomycota) in Central Europe. Acta Soc Bo Pol 84(4):431–438. https://doi.org/10.5586/asbp.2015.035
Telwala Y, Brook BW, Manish K, Pandit MK (2013) Climate-induced elevational range shifts and increase in plant species richness in a Himalayan biodiversity epicentre. PLoS One 8(2):e57103. https://doi.org/10.1371/journal.pone.0057103
Tewari VP, Verma RK, von Gadow K (2017) Climate change effects in the Western Himalayan ecosystems of India: evidence and strategies. For Ecosyst 4(1):1–9. https://doi.org/10.1186/s40663-017-0100-4
Thuiller W, Broennimann O, Hughes G, Alkemade JRM, Midgley GF, Corsi F (2006) Vulnerability of African mammals to anthropogenic climate change under conservative land transformation assumptions. Glob Change Bio 12(3):424–440. https://doi.org/10.1111/j.1365-2486.2006.01115.x
Walther GR, Post E, Convey P, Menzel A, Parmesan C, Beebee TJ et al (2002) Ecological responses to recent climate change. Nature 416:389–395. https://doi.org/10.1038/416389a
Wang T, Campbell EM, O’Neill GA, Aitken SN (2012) Projecting future distributions of ecosystem climate niches: uncertainties and management applications. For Ecol Manag 279:128–140. https://doi.org/10.1016/j.foreco.2012.05.034
Wilson RJ, Gutiérrez D, Gutiérrez J, Martínez D, Agudo R, Monserrat VJ (2005) Changes to the elevational limits and extent of species ranges associated with climate change. Ecol Lett 8(11):1138–1146. https://doi.org/10.1111/j.1461-0248.2005.00824.x
Wise MA, Calvin KV, Thomson AM, Clarke LE, Bond-Lamberty B, Sands RD, Smith SJ, Janetos AC, Edmonds JA (2009) Implications of Limiting CO2 Concentrations for Land Use and Energy. Science 324:1183–1186. https://doi.org/10.1126/science.1168475
Wolseley P, James PW, Theobald MR, Sutton MA (2006) Detecting changes in epiphytic lichen communities at sites affected by atmospheric ammonia from agricultural sources. Lichenologist 38:161–176. https://doi.org/10.1017/S0024282905005487
Zobel DB, Singh SP (1997) Himalayan forests and ecological generalizations. BioScience 47(11):735–745. https://doi.org/10.2307/1313096
Żołkoś K, Kukwa M, Afranowicz-Cieślak R (2013) Changes in the epiphytic lichen biota in Scots pine (Pinus sylvestris) stands affected by a colony of grey heron (Ardea cinerea): a case study from northern Poland. Lichenologist 45:815–823. https://doi.org/10.1017/S0024282913000558
Acknowledgements
This study is part of projects entitled “Timberline and Altitudinal Gradient Ecology of Himalayas, and Human Use Sustenance in a Warming Climate” under National Mission on Himalayan Studies (NMHS) Program (Grant# NMHS/2015-16/LG03/03), and an Institute In-House Project#4 entitled “Mainstreaming Himalayan Biodiversity for Sustainable Development” funded by Ministry of Environment Forest & Climate Change (MoEF&CC), New Delhi, India. The authors are grateful to the Directors, G.B. Pant National Institute of Himalayan Environment (NIHE), Almora, Uttarakhand, and CSIR-National Botanical Research Institute (NBRI), Lucknow, U.P., India, for their encouragement and supports.
Author information
Authors and Affiliations
Contributions
DK and AP collected field data. DK, RB, and DKU compiled data from Herbarium. DKU, SPS, DK, and RB designed the MS. DK, SPS, AP, SR, and MJ wrote the first draft of the manuscript, and all authors contributed to subsequent revisions. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 31 kb)
Rights and permissions
About this article
Cite this article
Kumar, ., Pandey, A., Rawat, S. et al. Predicting the distributional range shifts of Rhizocarpon geographicum (L.) DC. in Indian Himalayan Region under future climate scenarios. Environ Sci Pollut Res 29, 61579–61593 (2022). https://doi.org/10.1007/s11356-021-15624-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15624-5