Abstract
Mercury (Hg) bioaccumulation in fish poses severe threats to the food safety and human health. This study was conducted to assess Hg bioaccumulation in fish (n = 24) and scalp hair (n = 77) of the fishing communities at up- and downstream of the river Swat, Pakistan. The mean Hg concentration in upstream fish Salmo trutta fario (Brown trout) and Schizothorax plagiostomus (Swati fish) species was 34.7±18 μg kg−1 and 29.4±15 μg kg−1, respectively. The mean Hg concentration in downstream Swati fish, Crossocheilus diplochilus (Spena deqa), and Garra gotyla (Tora deqa) was 65±21 μg kg−1, 123±33 μg kg−1, and 326±53 μg Kg−1, respectively. The mean Hg concentration in scalp hair of the up- and downstream fishing communities was 658±125 μg kg−1 and 3969±791 μg kg−1, respectively. Independent T-test showed significant difference (p < 0.001) in the mean Hg concentration in scalp hair of the up- and downstream communities. The most prevalent health problems found in the fishing community were muscle pain, headache, visual impairment, arterial blood pressure, anemia, and kidney dysfunction. Multiple linear regression indicated that daily and weekly consumption of the fish significantly increase Hg accumulation in human scalp hair. Regular consumption of fruits and cruciferous and leafy vegetables were found to reduce Hg toxicity in the population. Further studies are recommended to identify the sources of Hg and welfare impact of fish contamination on the fishing community of river Swat.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mercury (Hg) is a widespread heavy metal of great environmental concern (Hsu-Kim et al. 2018; Selin 2018; Tang et al. 2020). Hg exists in elemental, inorganic, and or organic form in nature and poses severe threats to the food safety and human health (Hsiao et al. 2011; Sun et al. 2013) due to its toxicity and bioaccumulation in food webs (Dus et al. 2005). The chemical form of Hg largely affects its mobility and toxicity (ATSDR 2013). For instance, the organic form of Hg such as methyl mercury (MeHg) is more toxic than the elemental or inorganic form (Hasegawa et al. 2005). The MeHg can be produced by the methylation of elemental and inorganic Hg by bacteria in anaerobic conditions, even in the intestine of fish (Ali and Khan 2018). The MeHg retention time in fish depends on the physiological response of the fish such as homeostasis and detoxification (Ali et al. 2019). The toxic effects of MeHg were first realized when serious Hg poisoning occurred in Japan near the Minamata Bay due to consumption of contaminated fish and sea food (Björkman et al. 2007; Karabedian et al. 2009), as the MeHg was released from an industrial plant into the coastal area of Minamata.
Hg contamination in aquatic ecosystems arise from both natural and anthropogenic origin such as mineral deposits, forest fires (Camargo 2002), agricultural runoffs, mining activities, combustion of Hg containing fuels, and municipal and industrial wastewater discharges (Moiseenko and Gashkina 2016; Pavlish et al. 2003; Wang et al. 2004; WHO 2005). During the recent decades, urbanization, industrialization, and population growth has intensified the contamination of freshwater ecosystems. The fish living in polluted waters tend to accumulate Hg in their tissues and transfer across the food chain (Moiseenko and Gashkina 2016). The Hg can enter the fish body through its skin or gills and this mechanism is called bio-concentration, whereas the entry of Hg in fish body through ingestion of contaminated food/prey is called dietary accumulation (Ali and Khan 2018).
Human exposure to Hg occurs mainly through ingestion of the contaminated food and fish (ATSDR 2013; Fakour et al. 2010; Shah et al. 2016) and may cause acute or chronic effects ranging from severe disruption of tissue, shock, cardiovascular collapse, autism, gastrointestinal damage, renal and neurological disturbances, dermatitis, fatigue, and respiratory problems to life-threatening toxicity (Bastos et al. 2006; Eisler 2006; Lee et al. 2003; Riaz et al. 2016; WHO 2005). The Hg, when combined with other heavy metals, can have more toxicity due to the synergistic effect of the heavy metals mixture (Ali and Khan 2019). The Codex Alimentarius guideline levels of MeHg for predatory and non-predatory fish are 1000 μg kg−1 and 500 μgkg−1 wet weight (w/w), respectively (WHO 2007). The concentration of Hg in scalp hair is a widely used biomarker to evaluate Hg toxicity and contamination particularly for MeHg exposure from dietary consumption of fish and other foods (Agusa et al. 2007; Anwar et al. 2007; Díez et al. 2008; Feingold et al. 2020; Nuttall 2006) because hair sequesters mercury during its formation. Once incorporated into hair, Hg remains stable for a longer period as it binds to cysteine and can provide a history of MeHg exposure (Nuttall 2006).
The river Swat serves as a major fresh water body for a large portion of the Khyber Pakhtunkhwa province of Pakistan and has a significant role in fisheries and agricultural sector. The fish species found in river Swat provide significant contribution to the diet of the rural communities of Swat. However, river Swat receives most of the untreated wastewater (agricultural, industrial, and municipal) and a large proportion of solid wastes (hospital, industrial, and municipal) generated in the region. Emerald mining activities are also carried out in Swat and the local people take the mining debris to the bank of the river Swat for washing and search of emerald particles potentially contaminating the water body with different heavy metals. Furthermore, car batteries repairing shops and goldsmith shops present in the region are also likely to release heavy metals particularly Hg into the river Swat. As such due to the hazardous nature of Hg and its accumulation in food chain, it is imperative to monitor and quantify its levels in fish and fishing communities. This study is therefore conducted to (1) assess the Hg contamination in fish and scalp hair of the fishing community of river Swat, (2) assess the correlation between mercury accumulation in fish and human fish dietary patterns, and (3) assess the health effects of Hg exposure on the fishing community. To the best of our knowledge, no such study is conducted in the region previously.
Materials and methods
Study area
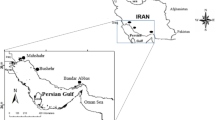
District Swat is mainly a mountainous terrain located between 34° 34′ to 35° 55′ N latitude and 72° 08′ to 72° 50′ E longitude in the Hindukush Himalaya range of Khyber Pakhtunkhwa Province Pakistan and covers a land area of 5337 km2 (Bacha et al. 2018; Iqbal et al. 2018). According to the Government of Pakistan census (2017), the total population of district Swat was 2.31 million. The main source of income for majority of the population is agriculture, forestry, hunting, and fishing (Qasim et al. 2013). The river Swat originates from Hindukush mountains and extend from Mahodand and Gabral at about 3,000 m elevation and drains the entire watershed of Swat valley (DAWN 2007). Sampling was carried out in June–July 2019 at the up- and downstream of the river Swat (Fig. 1).
According to the Fisheries department in Swat, Government of Khyber Pakhtunkhwa, Pakistan, twenty-five different fish species are found in river Swat. The classification of fish based on their habitat/locality and feeding habit is given in supplementary information (Table S1). The fish species found in river Swat are largely site specific, for instance, Brown trout fish species found in upstream water of river Swat is scarcely found in downstream area. Similarly, the downstream fish species are rarely available in upstream area (Table S1).
Fish sample collection and preparation
The fish were randomly caught from up- and downstream of the river Swat with the help of local fishermen, and four juvenile fish specimens (n = 24) obtained in comparatively high number were included as study samples. The upstream fish included Brown Trout and Swati fish. Downstream fish included Swati fish, Spena deqa, and Tora deqa. Brown trout is carnivorous while Swati fish, Tora Deqa, and Spena Deqa are omnivorous. The fish species were measured for its length and weight, packed in clean polythene zip lock bags with identification code, and transferred to the laboratory in iceboxes. The fish specimens were analyzed for mercury accumulation as individual samples on wet weight (w/w) basis.
In laboratory, the fish were washed with distilled water. Fish fillet [5 g (w/w)] excised from the pectoral region was thawed, washed with deionized water, and acid-wet digested (Ahmad et al. 2015). Briefly, the 5 g fish fillet were taken in Pyrex glass tube and added 2 mL mixture of analytical grade nitric acid manufactured by BDH AnalaR® and per chloric acid manufactured by Fischer® (HNO3-HClO4) with a ratio of 1:1, followed by 2 mL concentrated hydrochloric acid (HCl) manufactured by Sigma-Aldrich®, and the mixture was digested overnight in a digestion block at ambient temperature. Finally, 5 mL of concentrated sulfuric acid (H2SO4) manufactured by BDH AnalaR® was added and heated to 200 C° for 20 min until the digests were clear (Voegborlo and Adimado 2010). The extracts were cooled at room temperature, filtered into centrifuge tubes, diluted to 20 mL with double deionized water, and analyzed for T-Hg on the atomic absorption spectrometer (GBC 932 plus) via Hydride Generation technique.
Hair sample collection and preparation
The local residents living along river Swat and having fishing license issued by District Swat Fisheries Department, Government of Khyber Pakhtunkhwa, were considered as the fishing communities. Record of the fishing licenses issued by the concerned fisheries department was obtained through which we identify the fishing communities. Scalp hair samples (n=77) (42 upstream and 35 downstream) were collected from the fishing communities including children (age ≤17 years) and adults (age ≥ 18 years) of both sexes with prior written consent. The number of upstream participants involved in each experimental class included female children (n= 11), male children (n= 6), female adults (n = 9) and male adults (n= 16). Likewise, the participants from downstream included female children (n= 6), male children (n= 6), female adults (n = 11) and male adults (n= 12). The research protocol for this study was approved by the Ethics Committee of the University of Peshawar (Approval No. 919/UOP/ERB/2018) and performed according to the 1964 Declaration of Helsinki (Association WM 2000). The hair was cut from the occipital part of the head closest to the scalp using stainless steel scissors (Ahmad et al. 2018; Ohno et al. 2007). Female enumerators were involved to collect hair samples from female participants following social and cultural values of the study area. The hair samples were packed in separate polyethylene zip-lock bags and coded before transferring to laboratory for further analysis. In laboratory, the hair samples were thoroughly washed with distilled water and acetone manufactured by Sigma-Aldrich® using ultrasonic bath to remove the dirt and external contaminants (Ahmad et al. 2018; Al-Amodi et al. 2017). The clean hair samples were cut into small pieces and oven dried overnight at 50 Co. Dried hair samples (0.25 g) were weighed into clean Pyrex tubes, 2.0 mL of 1:1 HNO3: HClO4 was added, followed by the addition of 2 mL conc. HCl and the mixture was digested overnight in a digestion block at ambient temperature. Finally, 5 ml of concentrated H2SO4 was added and the tubes were heated at 200 °C for 20 minutes until the digests were clear (Koseoglu et al. 2017; Voegborlo and Adimado 2010). The digested solution was cooled at room temperature, filtered into centrifuge tubes, and diluted to 20 mL with double deionized water (Ahmad et al. 2018). The T-Hg concentration in prepared samples was assayed in triplicates using Atomic Absorption Spectrometer (GBC 932 plus) via hydride generation technique.
Household questionnaire survey
The household questionnaire survey comprised of contact and household information, economic status, dietary information, fish consumption habits, and health conditions including the prevalence of 21 acute and 10 chronic health effects of Hg toxicity reported in the literature. The inclusion criteria for the household questionnaire survey were interesting as only household involved in fishing were included. For the purpose, fishing villages were identified by computing the number of fishing licenses issued by the fisheries department Swat, Government of KPK, Pakistan. In upstream area, the villages selected for household survey and sampling were Koz Kalay, Tanqar, Shagai, and Jupin. Whereas in downstream area, the selected villages for sampling were Garho, Aligrama, and Maam Derai (Fig. 1). A total of 21 randomly selected households were surveyed among which 11 were from the upstream area and 10 from the downstream area. All the households were within 6 km distance from the river Swat.
Quality control
Rigorous quality control protocols were followed during sample preparation and analysis. The apparatus and glassware used were kept overnight in 5% HNO3 (v/v) solution, thoroughly washed with deionized water and oven dried at 60 °C prior to use (Ahmad et al. 2019a). Reagent blanks were prepared and analyzed with each batch of samples to check the possible contamination of chemicals and glassware used (contribution of reagents and vessels) (Ahmad et al. 2019b). All the reagents used were of analytical grade. Aqueous control samples were also run along with prepared samples and all samples were analyzed in triplicates. The percent recovery for Hg in aqueous control sample was 96.2%.
Statistical data analysis
Statistical data analysis was carried out using multiple linear regression and group mean difference t-test. Descriptive and multivariate statistical analysis were performed using STATA and OriginPro 2018.
The regression model equation can be expressed as Eq. 1 (Gujarati 2009).
where Y represents the dependent variable, X is independent variable with k = 1, 2 …….n. βo is constant or intercept and β1 is the slope variable or slope coefficient\( \Big(\ \frac{\Delta y}{\Delta x}= Slope \)). Error in the regression is represented by μi.
Group mean difference t-test can be expressed as Eq. 2.
Results and discussion
Hg concentration in fish
The mean Hg concentration observed in up- and downstream fish species is presented in Table 1. Generally, the concentration of Hg in downstream fish species was higher than upstream. The fish feeding habits, length, weight, and taxonomic considerations might have affected Hg accumulation in the studied fish in addition to the increasing input of the contaminant downstream in the river. Previously, many researchers found positive correlation between heavy metals accumulation with fish type, age, and weight (Ali and Khan 2018; Khan et al. 2012; Yousafzai et al. 2010). Likewise, high Hg concentration was observed in downstream fish species in Madre de Dios River (Peru) (Diringer et al. 2015). While the characteristics of Madre de Dios River are different from river Swat since Madre de Dior River flows in an area where artisanal and small scale gold mining is common, however, the results reflect site-specific contamination as high Hg concentration was observed in fish obtained from higher contaminated areas/sites. Conversely, some other researchers observed no significant variation of Hg concentration in up- and downstream fish (Michalak et al. 2014; Anwar et al. 2007). In present study, the high Hg concentration observed in downstream fish can be associated with anthropogenic inputs as our survey revealed that most of the agricultural, municipal, and industrial wastewater is directly discharged into the river Swat. Besides, a large proportion of the solid wastes (hospital, industrial, municipal) generated were also dumped into the river Swat.
The recommended guideline limits of Hg in predatory and non-predatory fish are 1000 μg kg−1 and 500 μg kg−1 (w/w), respectively (WHO 2007). In present study, the accumulation of Hg measured in the different fish species is below the limits allowed by law. However, higher Hg concentrations were observed for downstream fish species particularly Garra gotyla, and its excessive consumption must be limited by the local fishing communities to avoid excessive Hg body burdens.
Hg concentration in hair
The average Hg concentration in scalp hair of the up- and downstream fishing community was 658±125 μg kg−1 and 3969±791 μg kg−1, respectively. In upstream area, Hg concentration in scalp hair was above the WHO permissible limit of 1000 μg kg−1 for 19% subjects. Likewise, in downstream area, Hg concentration in scalp hair was above the WHO permissible limit for 51% subjects. Age and gender wise mean Hg concentration in scalp hair of the children (female and male) and adults (female and male) for up- and downstream fishing communities is presented in Fig. 2.
The horizontal line indicates the WHO guideline limit of 1000 μg kg−1 Hg for scalp hair. The mean Hg concentrations in scalp hair of the upstream fishing community were below the WHO guideline limit of 1000 μg kg−1 for children and adults (female and male). In contrast, the mean Hg concentrations in scalp hair of the downstream fishing community exceeded the WHO guideline limit for children and adults (Fig. 2). Independent T-test showed significant difference (p < 0.001) in the mean Hg concentration in scalp hair of the up- and downstream communities. However, no significant difference was observed for the Hg concentration in scalp hair of the male and female. Furthermore, research studies from Karachi, Hyderabad, and Lahore, Pakistan, reported high Hg concentration in scalp hair of the female compared to male (Anwar et al. 2007; Shah et al. 2016). Conversely, a research study from Negro River Basin, Brazil, reported significantly low Hg concentration in scalp hair of the females than male (Barbosa et al. 2001). In present study, no significant relationship was observed between Hg concentration in hair and age of the respondents. Similar findings were reported in some other studies (Karabedian et al. 2009; Michalak et al. 2014; Szynkowska and Pawlaczyk 2007). However, a positive correlation between Hg accumulation in hair and age of the respondents was reported by Wyatt et al. (2017) and Shah et al. (2016). The regression model indicated that literacy reduces the Hg accumulation in human hair; however, the result was not statistically significant, while Anwar et al. (2007) reported that individual having 11 or more years of education have relatively higher concentration of Hg in scalp hair.
Effect of dietary habits on Hg concentration
Swati fish was the most consumed fish species by the communities for being abundantly available in both up- and downstream areas of river Swat. Multiple linear regression results showed that in upstream area, daily and 2–3 times consumption of carnivorous fish (Brown trout) significantly (p < 0.01) increases the Hg concentration in scalp hair (Table S2). Similarly, consuming omnivorous fish (Swati fish) 2–3 times a week significantly (p < 0.1) increase the Hg concentration in scalp hair. In the downstream area, consuming omnivorous fish (Swati fish, Tora deqa, and Spina deqa) 2–3 times a week significantly (p < 0.5) increase the Hg concentration in scalp hair (Table S2). Previously, Feingold et al. (2020) and Díez et al. (2008) found a positive correlation between fish consumption and Hg level in hair, while Fakour et al. (2010) observed that 76.4% of the fish consumers have Hg levels exceeding the USEPA recommended level of 1000 μg kg−1. The variation of Hg accumulation in human scalp hair in relation to non-fish dietary habits is given in Table S3. The moderating effects of some of the non-fish dietary habits were also found (Table S3). The regression analysis showed that consuming dry fruits at least 1–2 times a month and/or 1–2 times a week lowers the Hg accumulation in scalp hair (p < 0.1). Previously, Wyatt et al. (2017) observed a decrease in hair Hg levels with consumption of grains and fruits. In contrast, Feingold et al. (2020) did not find any significant trend of hair Hg levels in relation to rice and fruits consumption.
Disease prevalence
The prevalence of acute and chronic diseases caused by Hg toxicity were assessed in the fishing community through the questionnaire survey (Fig. 3).
The most prevalent health problems recorded for the fishing community of up- and downstream were muscle pain (14.3%), depression (6.9%), headache (8.6%), nausea (4.8%), irritability (4.8%), low vision (4.8%), loss of hearing (5.7%), blood pressure (20.1%), anemia (9.5%), and nephropathy (7.1%). Previously, Shah et al. (2016) found back pain, headache, irritability, and loss of hearing as the common symptoms resulting from MeHg exposure due to fish consumption. We conducted literature review to identify the most common ailments caused by Hg toxicity and on the basis of dietary pattern associated the observed ailments to Hg toxicity since the respondents were fish consumers on regular basis. However, the observed ailments might have been caused by a multitude of other factors.
Conclusion
The concentration of Hg in up- and downstream fish species was within the WHO guideline limits of 1000 μg kg−1 and 500 μg kg−1 (w/w) for predatory and non-predatory fish, respectively. However, the mean Hg concentration in downstream fish species was 5 times higher than upstream fish. Likewise, the mean Hg concentration in scalp hair of the downstream fishing community was 6 times higher than upstream community and exceeded the WHO guideline limit of 1000 μg kg−1 for both children and adults. A positive correlation was found between fish consumption and Hg accumulation in scalp hair. The feeding habits, length, and weight as well taxonomic considerations might have affected Hg accumulation in the studied fish in addition to the increasing anthropogenic input of the contaminant downstream in the river. Various ailments associated with Hg toxicity were observed among the fishing communities. The higher Hg concentrations observed in river Swat fish species particularly in Brown trout indicated that its consumption must be limited to avoid excessive Hg ailments among the community. Further studies are recommended to identify the sources of Hg and welfare impact of fish contamination on the fishing community of river Swat.
Availability of data and materials
Summary of the data generated or analyzed during this study is included in the article. While detailed data of this study are available upon request.
References
Agusa T, Kunito T, Iwata H, Monirith I, Chamnan C, Tana TS, Subramanian A, Tanabe S (2007) Mercury in hair and blood from residents of Phnom Penh (Cambodia) and possible effect on serum hormone levels. Chemosphere 68:590–596
Ahmad H, Yousafzai AM, Siraj M, Ahmad R, Ahmad I, Nadeem MS, Ahmad W, Akbar N (2015) Muhammad K (2015): Pollution problem in River Kabul: accumulation estimates of heavy metals in native fish species. Biomed Res Int 2015:1–7
Ahmad I, Khan B, Khan S, Khan MT, Schwab AP (2018) Assessment of lead exposure among automobile technicians in Khyber Pakhtunkhwa, Pakistan. Sci Total Environ 633:293–299
Ahmad I, Khan B, Asad N, Mian IA, Jamil M (2019a) Traffic-related lead pollution in roadside soils and plants in Khyber Pakhtunkhwa, Pakistan: implications for human health. Int J Environ Sci Technol 16:8015–8022
Ahmad I, Khan B, Khan S, ur Rahman Z, Khan MA, Gul N (2019b): Airborne PM10 and lead concentrations at selected traffic junctions in Khyber Pakhtunkhwa, Pakistan: Implications for human health. Atmospheric Pollut Res 10, 1320-1325
Al-Amodi H, Adly H, ALrefai A, Zaghloul A (2017) Assessment of occupational exposure to mercury concentrations in hair and nail of dental staff at some dental clinics in Makkah Region. J Ergonomics 7:2
Ali H, Khan E (2018) Bioaccumulation of non-essential hazardous heavy metals and metalloids in freshwater fish. Risk to human health. Environ Chem Lett 16:903–917
Ali H, Khan E (2019) Trophic transfer, bioaccumulation, and biomagnification of non-essential hazardous heavy metals and metalloids in food chains/webs—Concepts and implications for wildlife and human health. Human Ecol Risk Assessment An Int J 25:1353–1376
Ali H, Khan E, Ilahi I (2019) Environmental chemistry and ecotoxicology of hazardous heavy metals: environmental persistence, toxicity, and bioaccumulation. J Chem 2019:6730305
Anwar M, Ando T, Maaz A, Ghani S, Munir M, Qureshi I, Naeem S, Tsuji M, Wakamiya J, Nakano A (2007) Scalp hair mercury concentrations in Pakistan. Environ Sci 14:167–175
Association WM (2000): Declaration of Helsinki, ethical principles for medical research involving human subjects. 52 nd WMA General Assembly, Edinburgh, Scotland
ATSDR 2013: Addendum to the toxicological profile for mercury (alkyl and dialkyl compounds), U.S. Department of Health and Human Services, Public Health Service, Atlanta, GA
Bacha MS, Nafees M, Hayat U, Nawab A, Rashid W, Khan MT (2018) Evaluating the local perceptions of climate change vulnerability in Hindukush Himalayan region of Pakistan. World J Environ Biosci 7:10–19
Barbosa A, Jardim W, Dorea J, Fosberg B, Souza J (2001) Hair mercury speciation as a function of gender, age, and body mass index in inhabitants of the Negro River basin, Amazon, Brazil. Arch Environ Contam Toxicol 40:439–444
Bastos WR, Gomes JPO, Oliveira RC, Almeida R, Nascimento EL, Bernardi JVE, de Lacerda LD, da Silveira EG, Pfeiffer WC (2006) Mercury in the environment and riverside population in the Madeira River Basin, Amazon, Brazil. Sci Total Environ 368:344–351
Björkman L, Lundekvam BF, Lægreid T, Bertelsen BI, Morild I, Lilleng P, Lind B, Palm B, Vahter M (2007) Mercury in human brain, blood, muscle and toenails in relation to exposure: an autopsy study. Environ Health 6:1–14
Camargo JA (2002) Contribution of Spanish–American silver mines (1570–1820) to the present high mercury concentrations in the global environment: a review. Chemosphere 48:51–57
DAWN (2007): Saving river Swat from growing pollution
Díez S, Montuori P, Pagano A, Sarnacchiaro P, Bayona JM, Triassi M (2008) Hair mercury levels in an urban population from southern Italy: fish consumption as a determinant of exposure. Environ Int 34:162–167
Diringer SE, Feingold BJ, Ortiz EJ, Gallis JA, Araújo-Flores JM, Berky A, Pan WK, Hsu-Kim H (2015) River transport of mercury from artisanal and small-scale gold mining and risks for dietary mercury exposure in Madre de Dios, Peru. Environ Sci Process Impacts 17:478–487
Dus L, Svobodová Z, Janous D, Vykusová B, Jarkovský J, Šmíd R, Pavlis P (2005) Bioaccumulation of mercury in muscle tissue of fish in the Elbe River (Czech Republic): multispecies monitoring study 1991–1996. Ecotoxicol Environ Saf 61:256–267
Eisler R (2006): Mercury Hazards to Living Organisms (1st ed.). . CRC Press., 27-28 pp
Fakour H, Esmaili-Sari A, Zayeri F (2010) Mercury exposure assessment in Iranian women’s hair of a port town with respect to fish consumption and amalgam fillings. Sci Total Environ 408:1538–1543
Feingold BJ, Berky A, Hsu-Kim H, Jurado ER, Pan WK (2020) Population-based dietary exposure to mercury through fish consumption in the Southern Peruvian Amazon. Environ Res 183:108720
Gujarati D (2009): Basic econometrics Tata McGraw-Hill Education
Hasegawa T, Asano M, Takatani K, Matsuura H, Umemura T, Haraguchi H (2005) Speciation of mercury in salmon egg cell cytoplasm in relation with metallomics research. Talanta 68:465–469
Hsiao H-W, Ullrich SM, Tanton TW (2011) Burdens of mercury in residents of Temirtau, Kazakhstan: I: Hair mercury concentrations and factors of elevated hair mercury levels. Sci Total Environ 409:2272–2280
Hsu-Kim H, Eckley CS, Achá D, Feng X, Gilmour CC, Jonsson S, Mitchell CP (2018) Challenges and opportunities for managing aquatic mercury pollution in altered landscapes. Ambio 47:141–169
Iqbal M, Akbar F, Ullah S, Anwar I, Khan MT, Nawab A, Bacha MS, Rashid W (2018) The effects of marble industries effluents on water quality in Swat, Northern Pakistan. J Biol Environ Sci 13:34–42
Karabedian S, Al-Imarah FJ, Ibraheem HK (2009) Determination of Mercury in hair samples of Iraqis population living in Safwan and Al-Zubair Southern Basrah. J Duhok Univ 12(Special issue):23–27
Khan B, Khan H, Muhammad S, Khan T (2012) Heavy metals concentration trends in three fish species from Shah Alam River (Khyber Pakhtunkhwa Province, Pakistan). J Nat Environ Sci 3(1):1–8
Koseoglu E, Koseoglu R, Kendirci M, Saraymen R, Saraymen B (2017) Trace metal concentrations in hair and nails from Alzheimer’s disease patients: Relations with clinical severity. J Trace Elem Med Biol 39:124–128
Lee DA, Lopez-Alberola R, Bhattacharjee M (2003) Childhood autism: a circuit syndrome? Neurologist 9:99–109
Michalak I, Chojnacka K, Saeid A, Mikulewicz M (2014) Research on mercury levels in scalp hair. Pol J Environ Stud 23
Moiseenko T, Gashkina N (2016) Bioaccumulation of mercury in fish as indicator of water pollution. Geochem Int 54:485–493
Nuttall KL (2006) Interpreting hair mercury levels in individual patients. Ann Clin Lab Sci 36:248–261
Ohno T, Sakamoto M, Kurosawa T, Dakeishi M, Iwata T, Murata K (2007) Total mercury levels in hair, toenail, and urine among women free from occupational exposure and their relations to renal tubular function. Environ Res 103:191–197
Pavlish JH, Sondreal EA, Mann MD, Olson ES, Galbreath KC, Laudal DL, Benson SA (2003) Status review of mercury control options for coal-fired power plants. Fuel Process Technol 82:89–165
Qasim M, Hubacek K, Termansen M, Fleskens L (2013) Modelling land use change across elevation gradients in district Swat, Pakistan. Reg Environ Chang 13:567–581
Riaz A, Khan S, Shah MT, Li G, Gul N, Shamshad I (2016) Mercury contamination in the blood, urine, hair and nails of the gold washers and its human health risk during extraction of placer gold along Gilgit, Hunza and Indus rivers in Gilgit-Baltistan, Pakistan. Environ Technol Innov 5:22–29
Selin NE (2018) A proposed global metric to aid mercury pollution policy. Science 360:607–609
Shah AQ, Kazi TG, Afridi HI, Arain MB (2016) A population assessment of mercury exposure from two cities of Pakistan with respect to freshwater and marine fish consumption. Toxicol Ind Health 32:1033–1041
Sun G, Li Z, Bi X, Chen Y, Lu S, Yuan X (2013) Distribution, sources and health risk assessment of mercury in kindergarten dust. Atmos Environ 73:169–176
Szynkowska M, Pawlaczyk A (2007) The influence of mercury content on the structural changes of bioindicator surfaces. Pol J Chem Technol 9:115–120
Tang W-L, Liu Y-R, Guan W-Y, Zhong H, Qu X-M, Zhang T (2020) Understanding mercury methylation in the changing environment: recent advances in assessing microbial methylators and mercury bioavailability. Sci Total Environ 714:136827
Voegborlo R, Adimado A (2010) A simple classical wet digestion technique for the determination of total mercury in fish tissue by cold-vapour atomic absorption spectrometry in a low technology environment. Food Chem 123:936–940
Wang Q, Kim D, Dionysiou DD, Sorial GA, Timberlake D (2004) Sources and remediation for mercury contamination in aquatic systems—a literature review. Environ Pollut 131:323–336
WHO 2005: Mercury in Drinking-water Background document for development of WHO Guidelines for Drinking-water Quality
WHO (2007): Evaluation of Certain Food Additives and Contaminants: Sixty-seventh Report of the Joint FAO/WHO Expert Committee on Food Additives, 67. World Health Organization
Wyatt L, Ortiz EJ, Feingold B, Berky A, Diringer S, Morales AM, Jurado ER, Hsu-Kim H, Pan W (2017) Spatial, temporal, and dietary variables associated with elevated mercury exposure in Peruvian riverine communities upstream and downstream of artisanal and small-scale gold mining. Int J Environ Res Public Health 14:1582
Yousafzai AM, Chivers DP, Khan AR, Ahmad I, Siraj M (2010) Comparison of heavy metals burden in two freshwater fishes Wallago attu and Labeo dyocheilus with regard to their feeding habits in natural ecosystem. Pakistan J Zool 42(5):537–544
Acknowledgements
The authors acknowledge South Asian Network for Development of Environmental Economics (SANDEE) and International Centre for Integrated Mountain Development (ICIMOD) for providing financial support to conduct this research work. The analytical support was provided by Pakistan Institute of Nuclear Science & Technology (PINSTECH) Islamabad, Pakistan.
Funding
This study was funded by the South Asian Network for Development of Environmental Economics (SANDEE) and International Centre for Integrated Mountain Development (ICIMOD).
Author information
Authors and Affiliations
Contributions
Conceptualization: BK, IAM, IA, MAM. Statistical analysis: IA, MR, MAM. Investigation: BK, MAM, SS, KN. Chemical analysis: SS, KN. Project administration: BK, IAM. Field expedition supervision: BK, IAM, MAM. Data interpretation: IA, BK, MR, MAM. Writing—review and editing: IA, BK, IAM, MAM, MR. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval and consent to participate
This study was conducted with prior approval from Ethical Research Committee, University of Peshawar. Written consent was obtained from all the sampling population involved in this study.
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 23 kb)
Rights and permissions
About this article
Cite this article
Munir, M.A., Khan, B., Mian, I.A. et al. Assessment of Hg accumulation in fish and scalp hair in fishing communities along river Swat, Pakistan. Environ Sci Pollut Res 28, 67159–67166 (2021). https://doi.org/10.1007/s11356-021-15348-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15348-6