Abstract
Forest harvesting activities on peatlands have long been associated with nutrient leaching and deterioration of downstream water quality. This study aims to assess the effect of grass seeding practice on harvested blanket peatlands to immobilize N and reduce its export to water courses. First, a plot-scale field experiment was conducted by seeding with two grass species (Holcus lanatus and Agrostis capillaris) to study the N uptake potential from a harvested area. Secondly, a simulated rainfall experiment was conducted to study the effect of these grasses on reducing N leaching from surface peat using laboratory flume approach. In the end, the role of seeded grasses in removing N from nutrient-rich throughflow water was assessed using simulated overland flow experiment. The results showed that the seeded grasses had the potential to uptake over 30 kg ha−1 of N in the first year after seeding on harvested peatlands, whereas it takes over 2.5 years to establish the same level of N uptake by natural re-vegetation (non-grassed). In the simulated rainfall experiment, the inorganic N (NH4+-N and NO3−-N) leaching in surface runoff from grassed flumes was 72% lower (453 mg m−2) than non-grassed flumes (1643 mg m−2). In the simulated overland flow experiment, the N retention by grassed flumes was significantly higher (98%) as compared to non-grassed flumes (70%) in the simulated overland flow experiment. Comparatively higher concentrations of NH4+-N and NO3−-N in soil porewaters of non-grassed flumes suggest that this N retention by non-grassed flumes is less sustainable and is likely to be leached in runoff in subsequent flow events. The results from all three experiments in this study suggest that seeded grasses are a major sink of N on harvested blanket peatland forests. Immobilization of N onsite using the grass seeding and mini-buffer practice could be an efficient and a feasible mean of reducing N export from harvested blanket peatland forests in order to protect the sensitive water courses. However, the sustainability of retention and immobilization of N by grasses needs to be studied further in long-term field-scale experiments on multiple peatland sites.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The deterioration of the water quality of peatland-fed headwaters has gained increasing interest in recent years. Often, these headwaters are oligotrophic by nature and feed waters containing salmonids and freshwater pearl mussels (one of the most endangered species in the world and protected under the EU Habitats Directive within Special Areas of Conservation), which are very sensitive to changes in water quality (O’Driscoll et al. 2012). Forestry-related activities such as drainage, afforestation, fertilization, and harvesting are seen as one of the major sources of pollutants in these upland areas as other human activities are generally limited in these areas. It is estimated that approximately 218,000 ha of the upland peat catchments in western Ireland were afforested since the 1950s (Renou-Wilson and Byrne 2015) and most of these forests have reached harvestable age and are currently or due to be harvested in the near future. The progression of harvesting of these upland peat forests will lead to the deterioration of downstream water quality due to nutrient leaching (Rodgers et al. 2010; O’Driscoll et al. 2016).
Increased input of phosphorus (P) and nitrogen (N) can lead to oxygen depletion (O’Driscoll et al. 2016) and eutrophication of water bodies. Several studies have reported an increase in leaching of P (Nisbet et al. 1997; Lundin 1998; Ahtiainen and Huttunen 1999; Nieminen 2003; Cummins and Farrell 2003; Rodgers et al. 2010; Asam et al. 2014a; O’Driscoll et al. 2014a; Kaila et al. 2014; Marttila et al. 2018) and N (Rosén and Lundmark-Thelin 1987; Lundin 1999; Nieminen 1998, 2003, 2004; Asam et al. 2014a; O’Driscoll et al. 2014a; Nieminen et al. 2020a) after harvesting of peatland forests. There could be several reasons for the increased leaching, but the most immediate is the disruption of nutrient cycling due to the reduction of tree uptake.
Disturbance of surface peat due to the movement of heavy machinery during harvesting operations (O’Driscoll et al. 2014a) and increase in the decomposition of organic matter due to alteration in soil moisture and temperature (LIoyd and Taylor 1994; Davidson et al. 1998; Roberts et al. 2005) after harvesting can also increase the release of nutrients from surface peat. In addition, the harvest residue remaining onsite, in the form of brash mats and windrows, after conventional stem-only harvesting (Rodgers et al. 2010), is a potential source of nutrients. Interestingly, studies have reported negligible release of N from decomposing harvest residue during the initial years (Palviainen et al. 2004; Kaila et al. 2012; Asam et al. 2014a; Asam et al. 2014b), but the decomposing harvest residue is also reported to facilitate soil microbial activity by providing additional nutrients and fresh organic matter to the soil microbes (Fontaine et al. 2004, 2007) and may result in increased decomposition and N release from surface peat.
Several studies have reported significantly higher concentrations of N (NH4+-N and NO3−-N) in soil or porewater under harvest residue piles/windrows when compared to the residue-free areas (Rosén and Lundmark-Thelin 1987; Nieminen 1998; Asam et al. 2014a). Using the laboratory flume approach, Asam et al. (2014a) reported significantly higher N release (in the form of NH4+-N and NO3−-N) from peat with harvest residue (brash) than the peat without harvest residue. Similarly, O’Driscoll et al. (2014a), in a mini-catchment–scale plot study on peatlands, also reported significantly higher N release from plots with harvest residue (brash) than those without.
One of the widely recommended methods to protect water courses from the negative impacts of forest harvesting activities is to divert the runoff through a natural or restored buffer area before it enters the water courses (Silvan et al. 2004a; Nieminen et al. 2005; Väänänen et al. 2008; Vikman et al. 2010). However, in Ireland and the UK, the establishment of riparian buffer areas was not considered during the afforestation of the majority of upland blanket peat catchments and trees were planted up to the edges of streams (Ryder et al. 2011). The restoration work required to create buffer areas on peatlands results in a rise in the water table, leading to anoxic conditions in surface peat, which increases nutrient leaching from peat during the initial few years (Kaila et al. 2016). It was recently reported by Nieminen et al. (2020b) that the restoration of forestry-drained peatlands, to create buffer areas for water quality protection, resulted in a significant increase in nutrient, carbon, and heavy metal exports to water courses. The restoration-induced export of nutrients may be so high during the initial couple of years that the whole concept of creation of buffer areas by restoring peatlands to protect downstream water quality becomes controversial (Nieminen et al. 2020b).
Well-established vegetation is required for efficient retention of N in peatland buffer areas (Silvan et al. 2004a), which can take several years to establish (O’Driscoll et al. 2011). So, unless the buffer areas are created a number of years prior to harvesting of upstream forests, they may not be effective for inorganic N retention (Nieminen et al. 2020b), which is generally released during the initial few years after harvesting of peatland forests. The uneven topography and established preferential flow channels contribute to the difficulties in the creation of effective buffer areas in upland blanket peat catchments in Ireland. Additionally, even if established, they may not be very effective in retaining nutrients as more than 70% of the nutrients are released during storm events from harvested blanket peat sites (Rodgers et al. 2010). During storm events, the hydraulic loading entering the buffer areas is generally very high and nutrient retention efficiency of buffer areas is very low (Silvan et al. 2005; Väänänen et al. 2008; Vikman et al. 2010; Asam et al. 2012). O’Driscoll et al. (2014b) reported only 18% retention of P by buffer area created in a blanket peatland forest in the west of Ireland.
Whole-tree harvesting (WTH), involving the removal of stem and the harvest residue, is another potential method to reduce nutrient leaching after harvesting peatland forests (Nisbet et al. 1997; Nieminen 2004; Rodgers et al. 2010; Asam et al. 2014a), but considering the local climatic conditions and characteristics of blanket peatlands in Ireland, the removal of all the harvest residue may not be practically possible as the part of harvest residue will still be required in the form of brash mats for extraction and transport of tree stems (Rodgers et al. 2010). In their study of forested upland blanket peatlands, O’Driscoll et al. (2011) reported a novel method to immobilize P, which attempted to determine the likely impacts of seeding of the entirety of the harvested peatland immediately post-harvesting with native grasses. Asam et al. (2012), in a study using the mini-buffer approach (Fig. 1), suggested that these native grasses retained nearly 90% of P from nutrient-rich throughflow water, simulating the corresponding release from the brash windrows in harvested blanket peat sites.
Photo of a clear-felled peatland forest site (a), schematic presentation of current (normal) situation of a clear-felled area and buffer area (b), and proposed mini-buffer approach by re-orienting the brash windrows (c) (Asam et al. 2012). Reprinted figure with permission from Elsevier. Copyright©2012. License Number: 4922400445350
The present study aims to investigate the mechanism and role of seeded native grasses’ ability to immobilize N on harvested blanket peatland forests in order to reduce N export to receiving water bodies. Firstly, a plot-scale experiment was established on a peatland site immediately after harvesting to investigate the N uptake potential of seeded grasses (Holcus lanatus and Agrostis capillaris), alongside field surveys conducted on 9 different blanket peat sites, harvested over the previous 1–5 years, to study the N uptake by natural re-vegetation. Secondly, a simulated rainfall experiment was conducted using laboratory flumes to examine the leaching of N in surface runoff from surface peat seeded with grasses (grassed flumes) and non-seeded control (non-grassed flumes) with natural re-vegetation. Finally, the retention of N by well-established grassed flumes and non-grassed flumes (control with natural re-vegetation) was assessed by subjecting them to N-rich overland flow events, simulating the N released from brash mats and windrows on harvested blanket peatlands. It was hypothesized that seeded grasses would significantly improve N uptake during the initial year after harvesting and immobilize higher amount of N onsite as compared to natural re-vegetation. It was also hypothesized that the seeded grasses (grassed flumes) would have higher N uptake from N-rich throughflow water as compared to natural re-vegetation (non-grassed control flumes).
Materials and methods
Nitrogen in biomass of grass-seeded plots and natural re-vegetation
Grass-seeding plot-scale study was conducted in the Glennamong river catchment (53° 58′ N, − 9° 37′ E, 69 m a.s.l.), where 1 ha forest area was harvested in August 2009 (Table 1). Three plots, approximately 100 m2 (plot 1), 360 m2 (plot 2), and 660 m2 (plot 3) were selected, and all harvest residue was removed from these plots. Each of the selected plots was seeded with a 50:50 ratio of two grass species namely, Holcus lanatus and Agrostis capillaris, at a rate of 36 kg ha−1 in October 2009 (O’Driscoll et al. 2011). A non-seeded area beside the seeded plots was used as an experimental control (non-grassed). Soil characteristics of the Glennamong site are presented in Table 2. For the natural re-vegetation survey, a total of nine blanket peatland sites were selected in close proximity (within 10 km) to the Glennamong site, which were harvested in the previous 1–5 years (Table 1). All the sites had similar soil type, climatic and hydrological conditions and received approximately 2000 mm of precipitation annually. For more detail on the study sites, see O’Driscoll et al. (2011) and Asam et al. (2012, 2014b).
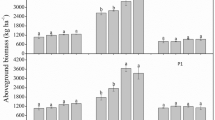
Estimation of aboveground vegetation biomass from the seeded grass plots (plots 1, 2, and 3) along with the nine selected peatland sites was conducted in August 2010, using the standard procedure explained in O’Driscoll et al. (2011). Following the estimation of biomass (Fig. 2), all dried vegetation and grass samples were separately stored in the laboratory prior to further analysis of N content. In order to observe the ability of seeded grasses to assimilate N available in the root zone, soil porewater samples were collected from 3 grass-seeded plots and 1 non-grassed control plot using suction samplers attached to a removable syringe (Rhizon MOM 10 cm moisture filters) (Asam et al. 2012; Asam et al. 2014a). The porewater was collected from the top 0–10-cm layer of peat at three randomly selected points in each of the four plots in August 2010 and subsequently transported to the laboratory and stored at 4 °C until further analysis.
Aboveground vegetation biomass (dry mass) of natural re-vegetation in surveyed blanket peatland sites (reproduced from O’Driscoll et al. 2011).
Simulated rainfall and laboratory flume experiment
Flume preparation
The flume samples were prepared from Srahrevagh river catchment (54° 00′ N, − 9° 32′ E, 220 m a.s.l.), before forest harvesting. Soil characteristics of the Srahrevagh site are presented in Table 2. To prepare the laboratory flumes, 0.1-m–deep slabs of surface peat were collected using a rectangular flume sampler (Asam et al. 2014a). The flume sampler was constructed from galvanized steel with dimensions of 1 m length, 0.225 m width, and 0.1 m depth. To collect the peat slabs, the flume sampler was inserted upside down and vertically through the peat surface and extracted intact with the aid of a wide flat spade. After collection, the flume samplers containing the peat were transported to the laboratory where the peat within the sampler was transferred into 2-m–long, 0.225-m–wide, and 0.13-m–deep flume so that two slabs of peat (surface layer up) were placed tightly against each other to form a continuous peat surface (Mulqueen et al. 2006, Asam et al. 2012, 2014a; Fig. 3).
Using this method, six flume samples were prepared in the lab. Three out of 6 flume samples were seeded with Holcus lanatus and Agrostis capillaris (50:50) grasses using the same seeding rate of 36 kg ha−1 as for the grass-seeded plots at Glennamong site and the remaining three flume samples were retained as non-grassed controls. After seeding of grasses and for the duration of the rainfall and overland flow study, the flumes were stored in an unheated glass house and subjected to local weather conditions, except rainfall (Fig. 3; Asam et al. 2012; Asam et al. 2014a).
Simulated rainfall experiment
Rainfall events were applied using a rainfall simulator constructed in the laboratory, which was calibrated to achieve an intensity of 30 mm h–1 at 85% uniformity. Further detail about the rainfall simulator is available in Asam et al. (2014a). The source water for rainfall was potable tap water with measured P, NH4+-N, and NO3−-N concentrations of 0.005 mg L−1, 0.026 mg L−1, and 0.12 mg L−1, respectively. The measured electrical conductivity of source water was 0.435 dS m−1, and pH was 7.7. The stored grassed and non-grassed flume samples were transferred to the laboratory for irrigation with simulated rainfall once in a week for the first 6 weeks and once in a fortnight for the subsequent time period. The slope of the flumes was set at 5° during the whole experiment, simulating the average hill slope gradient in blanket peatlands.
Every rainfall event lasted for 1 h after the start of runoff into the collection bucket (Fig. 4), and it took 10 to 15 min of additional rain to saturate the soil in flume samples before the runoff started during those rainfall events. Runoff water volume was measured from the runoff collection bucket at the end of each rainfall event, and a subsample was filtered using 0.45 μm syringe filters, which was stored at 4 °C until further analyses. The simulated rainfall experiment continued for 48 weeks (340 days, from June 2010 to May 2011). During this period, 24 rain events were applied with a total rainfall sum of 880 mm. In addition to runoff samples, soil porewater samples were also collected from these flumes to examine the relationship of N concentration (mg L−1) in porewater with the N concentration (mg L−1) in the runoff. The porewater filters were inserted at three different points along the length of the flumes at distances of 0.5 m, 1.5 m, and 1.75 m measured from the flume inlet (Fig. 3), and porewater samples were again collected using suction samplers as explained in the previous section. The porewater samples were collected 1–2 h before applying the rainfall and stored at 4 °C until further analysis.
Grass was well established on the grassed flumes by the end of October 2010 (21 weeks since seeding) and continued to improve after that. Some growth of vegetation (mainly mosses) was also observed in non-grassed flumes at the end of the simulated rainfall study (week 48). Growth of vegetation in grassed and non-grassed flumes at different stages of simulated rainfall study is presented in Fig. 3.
Simulated overland flow experiment
The grassed and non-grassed flumes established after the 48 week simulated rainfall experiment (Fig. 3) were re-deployed for use in this part of the study. This experiment was conducted in June–July 2011. Its purpose was to examine the effectiveness of seeded grasses (grassed flumes) to uptake N from nutrient-rich throughflow water. This might demonstrate how using grass seeding as a management technique could immobilize N onsite rather than letting it progress downslope to buffer areas where retention efficiency is reported to be low (O’Driscoll et al. 2014b). This experiment was simulating the grassed and non-grassed flumes as mini-buffers (Fig. 1) established between the brash mats/windrows, and different inlet NH4+-N concentrations in overland flow in this study simulate the potentially higher NH4+-N concentrations released from soils underneath the brash mats/windrows (Asam et al. 2014a).
The flumes were subjected to 8 different overland flows with different NH4+-N concentrations (Fig. 4). The hydraulic loading for all the flows was kept low and constant at 30 mm h−1, simulating the generally low-flow scenario observed in areas between brash mats/windrows as compared to normal buffer areas downstream of the catchments where hydraulic loading is generally higher (Asam et al. 2012; Fig. 1). The concentrations of NH4+-N were prepared in a feeding tank by dissolving NH4Cl in the same source water used in the simulated rainfall study. The concentrations of NH4+-N in the range of 0–2 mg L−1 in runoff from harvested peatland forests, using artificial brash windrows experiment, have been reported by Asam et al. (2014a). As the hydraulic loading selected for this experiment was low (30 mm h−1), the higher concentrations of NH4+-N were used in order to give reasonable N loading to the flumes. The prepared NH4+-N concentrations were 1.0 (C1), 2.0 (C2), 4.0 (C3), and 6.0 (C4) mg L−1 with an equivalent NH4+-N loadings of 30, 60, 120, and 180 mg m−2, respectively. Each NH4+-N concentration was applied twice, and the order of addition was as follows: C1, C2, C3, C4, C4, C3, C2, and C1. The NO3−-N was not added in the nutrient-rich water as NO3−-N concentration released from harvested blanket peatlands is generally low (Asam et al. 2014a). However, the concentration naturally present in the source water was 0.12 mg L−1 resulting in NO3−-N loading of 3.6 mg m−2 during each flow event.
During all overland flow events, the slope of the flumes remained set at 5°. The N-rich water, prepared in the feeding tank, was pumped into the small chamber at the upslope end of the flume, which subsequently filled and flowed over the surface of peat through the 2-m length of flume and ultimately into the effluent collection bucket at the downslope end of the flume (Fig. 4). Runoff volume collected in the bucket was measured at the end of each flow event, and a subsample was filtered using a 0.45-μm syringe filter, which was stored at 4 °C until further analyses. The 8 overland flow tests were conducted in 2 months, and the total hydraulic load applied during this whole period was 240 mm. There was a gap of 1 week between each flow event, and the flumes were stored in the glass house during that time. The selected hydraulic loading was calculated from an equivalent rainfall of 2000 mm year−1, which is typical in the blanket peatland sites in the west of Ireland and assuming that approximately 75% of this rainfall results into runoff based on the finding (70 to 80%) reported by Labadz et al. (2010) regarding the peatlands. Runoff water quantity of 240 mm applied in 2 months (30 mm in every flow event) is equivalent to 1440 mm year−1 which is quite reasonable to compare with the on-field situation on selected blanket peat sites.
To understand if the vegetation was effectively taking up N from peat in flumes, soil porewater samples were collected from both the grassed and non-grassed flumes using the same process described in “Simulated rainfall and laboratory flume experiment.” Porewater samples were collected within 1 h after each overland flow event and again after 1 week. At the end of simulated overland flow experiment, the above ground vegetation from both the grassed and non-grassed flumes was harvested, dried at 60 °C until constant mass, weighed for biomass, and stored until further analysis.
Laboratory analysis and calculations
The stored runoff and porewater samples from all the experiments were analyzed for NO3−-N and NH4+-N within 1 week of collection using a Konelab 20 auto analyzer (Asam et al. 2014a; O’Driscoll et al. 2014a). The harvested and dried vegetation and grass samples from “Grass-seeded plots and natural re-vegetation” and “Simulated overland flow experiment” were milled to a particle size of approximately 2 mm and analyzed for total N using a Leco CHN 1000 analyzer. In the simulated rainfall experiment, the total amounts (mg m−2) of NH4+-N and NO3−-N exiting the flumes were calculated by multiplying the respective concentrations with the volume of water collected in the bucket at the end of each rainfall event. The individual loads from all the 24 rainfall events were added together to calculate the total export of NH4+-N and NO3−-N (mg m−2) during the 48 weeks of simulated rainfall study.
Similarly, during the simulated overland flow experiment, the total loads of NH4+-N and NO3−-N exiting the flumes were calculated by multiplying the respective concentrations measured in the outlet with the water volume collected in the bucket at the end of each flow event. The difference in concentration of inlet and outlet during each flow event was used to calculate the N retention both in terms of % age and mg m−2. The results of all the 8 flow events were added together to calculate the overall difference of N retention between grassed and non-grassed flumes during the whole overland flow study period. Statistical analysis was done by using the Statistical Package for the Social Sciences (SPSS) 21.0 and Microsoft Excel 2016. Student’s t test was employed, and p < 0.05 was considered statistically significant for comparing different responses of NH4+-N and NO3−-N concentrations both in grassed and non-grassed flumes.
Results and discussion
Nitrogen uptake by seeded grasses and natural re-vegetation on harvested blanket peatlands
Total N content in aboveground biomass of grass-seeded study plots 1, 2, and 3 was 44.7, 12.1, and 33.2 kg ha−1, respectively (Fig. 5), with an average of 30 kg ha−1. In contrast, the total N measured in non-seeded control plot was only 0.4 kg ha−1 (Fig. 5). The NH4+-N concentration measured in porewater samples of control plot was 1.65 mg L−1, which was higher than respective concentrations in grass-seeded plot 1 (0.06 mg L−1), plot 2 (0.20 mg L−1), and plot 3 (0.10 mg L−1) (Fig. 5). Nitrate-N was not detected in any of the porewater samples from grass-seeded and control plots.
The total N content (kg ha−1) of natural re-vegetation in different surveyed blanket peatland sites is presented in Fig. 6. There was no measurable vegetation in the first year after harvesting of surveyed sites as the vegetation started to establish after about 1.5 years post-forest harvesting on blanket peatlands (O’Driscoll et al. 2011; Fig. 2). Therefore, there was no nitrogen uptake shown by natural vegetation during the first year after harvesting. The total N content in the aboveground vegetation started to appear in the second-year post-harvesting and increased linearly with vegetation biomass and reached approximately 62 kg ha−1 at 4 years post-harvesting (Fig. 6). The N content in the aboveground biomass of grass-seeded plots was significantly higher (p = 0.05) when compared with control plots and surveyed blanket peatlands harvested 1 year prior to the survey.
This study supports the hypothesis that seeded grasses (H. lanatus and A. capillaris) have the potential to accumulate a significant amount of N during the first year of seeding soon after the forest harvesting on blanket peatlands (Fig. 5). The accumulated N by seeded grasses in plot scale study is almost equal to a cumulative of about 2.5 years by natural re-vegetation on harvested blanket peatlands (Fig. 6). This higher N accumulated in grassed plots is mainly due to the significantly higher vegetation biomass production in grass-seeded plots as compared to the natural re-vegetation 1 year post-harvesting (O’Driscoll et al. 2011).
This is important to note that on upland peat catchments in Ireland, the forests planted with lodgepole pine trees are very dense (2800 stems ha−1) as compared to peatlands forests in countries such as Sweden and Finland (1000 stem ha−1) (Rodgers et al. 2010). These tree stands with little self-pruning, lack of thinning operations, thick ground-needle layer, and limited light penetration at the forest floor (Rodgers et al. 2010; Asam et al. 2014a), makes it very difficult for vegetation to grow on the forest floor. This is probably one of the major reasons that there is no or very little ground vegetation on the forest floor of blanket peatlands. It can take several years after harvesting before significant biomass of natural vegetation is developed (O’Driscoll et al. 2011) to support the nutrient cycling, including N, for its effective immobilization on site. The uptake of N by well-developed peatland forests varies between 26 and 49 kg ha−1 year−1 (Finér 1989; Nieminen 1998), and it was very encouraging that the N taken up by grasses in the plot-scale study on average was 30 kg ha−1 in the first year after seeding (Fig. 5).
Vegetation is one of the main factors for N uptake and immobilization in peatlands as it converts it into organic form, which has very low mobility (Silvan et al. 2004a, 2004b). Enhanced vegetation growth after harvesting of blanket peatland forests have been reported to immobilize P onsite and have the potential to reduce P in runoff (O’Driscoll et al. 2011, 2014a). The higher N content by grasses on seeded plots and corresponding lower concentrations of NH4+-N in the pore water (Fig. 5) as compared to control plots reflects the effectiveness of grasses in taking up inorganic N from peat soil that otherwise would potentially leach to the downstream water courses.
Nitrogen release dynamics from grassed and non-grassed flumes in simulated rainfall study
During the 48 weeks of simulated rainfall study, the non-grassed flumes released significantly (p = 0.01) higher concentrations of NH4+-N (Fig. 7) as compared to grassed flumes. Three distinct phases of NH4+-N release were observed during the whole period of simulated rainfall experiments. In the first phase (weeks 1–21, June to October 2010), there was a very low concentration range of NH4+-N released from grassed flumes (0.02–0.24 mg L−1) as compared to non-grassed flumes (0.02–2.94 mg L−1). During the second phase (weeks 22–38, November 2010 to February 2011), relatively higher concentration of NH4+-N was released from grassed flume as compared to the first phase. However, the concentrations of NH4+-N released during this phase from non-grassed flumes (2.40–3.17 mg L−1) were still significantly higher than grassed flumes (0.63–1.52 mg L−1) (Fig. 7). In the third phase (weeks 38–48, March to May 2011), the concentration of NH4+-N in runoff from grassed flumes decreased from 1.27 mg L−1 in March down to 0.01 mg L−1 in May. In the same period, the NH4+-N concentration from non-grassed flumes decreased from 2.07 down to 0.68 mg L−1. The total NH4+-N released from non-grassed flumes was significantly (p = 0.00) higher (1530 ± 31 mg m−2) than grassed flumes (453 ± 236 mg m−2) during 48 weeks of simulated rainfall study (Table 3).
Nitrate-N release from flumes followed a different pattern than NH4+-N (Fig. 7). Concentrations of NO3−-N released from non-grassed flumes were in the range of 0.0–0.87 mg L−1, whereas, no NO3−-N release (0.0 mg L−1) was observed from grassed flumes throughout the simulated rainfall study. A spike in the concentration of NO3−-N release from non-grassed flumes was observed in the start of spring 2011 (week 38) when it reached a maximum concentration of 0.87 mg L−1 and reduced to 0.33 mg L−1 again in April and 0.02 mg L−1 in May (Fig. 7). Overall, the export of NO3−-N from non-grassed flumes was 113 ± 91 mg m−2, which was significantly higher (p = 0.01) as compared to grassed flumes (0.0 mg m−2) (Table 3).
The porewater concentrations for the first 8 rainfall events are not shown as there was no measurement of porewater for these initial events. Porewater NH4+-N concentrations measured before applying the rainfall events were in the range of 1.7–4.7 mg L−1 in non-grassed flumes, which were significantly higher (p = 0.00) than in grassed flumes (0.04–3.0 mg L−1) (Fig. 8a). Similarly, porewater NO3−-N concentrations were in the range of 0.0–1.0 mg L−1 and 0.0–0.12 mg L−1, respectively, from non-grassed and grassed flumes (Fig. 8b) and were significantly (p = 0.00) different. Porewater NH4+-N concentrations measured from flumes were plotted against NH4+-N concentrations measured in runoff from flumes (Fig. 8c), which showed a strong linear correlation (R2 > 0.95). Similarly, porewater NO3−-N concentrations also showed a correlation with NO3−-N concentrations in runoff (Fig. 8d).
The simulated rainfall study using laboratory flumes also supported this hypothesis that seeded grasses will reduce leaching of N from surface peat after harvesting. The grassed flumes released almost 70% lower NH4+-N as compared to non-grassed flumes (Table 3). Combining NH4+-N and NO3−-N (Inorganic N), the release from grassed flumes is almost 72% lower than the non-grassed flumes (Table 3). The release of N observed in the simulated rainfall study is mainly in the form of NH4+-N, the likely reason is that blanket peatlands are acidic in nature and are saturated most of the time making conditions highly anaerobic and unfavourable for nitrification and hence NO3−-N production (Regina et al. 1996). However, higher concentrations of NO3−-N have been released by non-grassed flumes in early spring (Fig. 7), suggesting that nitrification may have occurred due to higher evaporation and creation of aerobic conditions in peat in flumes. The same release trend has not been seen in grassed flumes, possibly because grasses had taken up any NO3−-N produced in grassed flumes. Release of N from peat during the simulated rainfall study suggests that harvested peatlands are sources of N release in runoff irrespective of the harvest residue, similar findings have been reported by Asam et al. (2014a). However, the level of concentrations released in laboratory flume study could be higher than in the field due to more favourable conditions in the laboratory and glass-house for decomposition and other mechanisms facilitating the release of N. The significant reduction in N release by grassed flumes in this study clarifies that the grasses grown after harvesting of blanket peatland forests have a high potential of immobilizing N onsite.
Nitrogen uptake dynamics of grassed and non-grassed flumes during nutrient-rich simulated overland flow study
The NH4+-N retention by grassed and non-grassed flumes during the overland flow study is presented in Fig. 9 and Table 4. Grassed flumes removed 94–99% of NH4+-N during overland flow events, depending on NH4+-N inflow concentration (Table 4). The retention of NH4+-N by non-grassed flumes was 23–83% (Table 4) during the same events. Average retention (%) of NH4+-N by grassed flumes was 98%, which was significantly higher (p = 0.00) than non-grassed flumes (70%). Total NH4+-N retained during the whole overland flow experiment was 766 ± 11 mg m−2 and 551 ± 56 mg m−2, respectively, by grassed and non-grassed flumes (Table 4). Figure 9 shows that % NH4+-N retention by non-grassed flumes decreased with decrease in inlet NH4+-N concentration and the difference between grassed and non-grassed flumes to retain NH4+-N increased with decreasing inlet concentration of NH4+-N mainly because the grassed flumes consistently retained over 94% of added NH4+-N during all the overland flow events.
The concentration of NO3−-N was not added into the source water during different events, however, there was about 3.6 mg m−2 of NO3−-N available in each flow event due to natural background concentration (0.12 mg L−1) of NO3--N present in the source water. Leaching or retention of these NO3−-N concentrations by grassed and non-grassed flumes is presented in Table 5. In general, the grassed flumes retained most (77%) of the NO3−-N present in each of the flow events, whereas the non-grassed flumes did not retain NO3−-N, they even released additional amounts of NO3−-N into the outlet (Table 5). Total inflow of NO3−-N on to the flumes during 8 overland flow events was 28.8 mg m−2. Total outflow of NO3−-N from grassed flumes was 6.5 ± 0.4 mg m−2 (22.3 ± 0.4 mg m−2 or 77% retained) and non-grassed flumes was 42.9 ± 0.1 mg m−2 (14.1 mg m−2 additional leaching) (Table 5). This outflow of total NO3−-N from grassed flumes was significantly lower (p = 0.00) than non-grassed flumes.
The porewater concentrations of NH4+-N in samples taken within 1 h of each flow event were significantly higher (p = 0.00) in non-grassed flumes (1.22–2.04 mg L−1) as compared to grassed flumes (0.03–0.18 mg L−1) (Fig. 10). The porewater NH4+-N concentrations measured after 1 week of each flow event were still significantly higher (p = 0.00) in non-grassed flumes (1.10–1.85 mg L−1) as compared to grassed flumes (0.01–0.04 mg L−1), suggesting quick and effective uptake by vegetation in grassed flumes after each flow event (Fig. 10). There was no NO3−-N observed in the porewater of grassed flumes during the whole overland flow experiment; however, in non-grassed flumes, there was scattered presence of NO3−-N during different flow events which ranged from 0.0 to 0.35 mg L−1 and 0.0 to 0.22 mg L−1, respectively, within 1 h and 1 week of flow events (Fig. 10). This difference of porewater NO3−-N between grassed and non-grassed flumes was significant (p = 0.00). Mass of aboveground vegetation measured at the end of simulated overland flow study was 484 ± 139 g m−2 and 173 ± 28 g m−2, respectively, for grassed and non-grassed flumes. The total N content in aboveground biomass for grassed and non-grassed flumes was 5756 ± 1649 mg m−2 and 2199 ± 356 mg m−2, respectively.
The upland blanket peat catchments in Ireland were commonly prepared for afforestation by ploughing (Carling et al. 2001) which created drainage furrows at intervals of approximately 2.0 m that follow the slopes down to collector drains, collecting surface runoff waters from the catchment, prior to entering streams or rivers (Rodgers et al. 2010; O’Driscoll et al. 2014a). During forest harvesting on blanket peatlands, the harvest residue, consisting of tree crown and any side branches, is used as protective brash mats underneath the heavy harvesting machinery to maintain the integrity of the peat surface and to facilitate the extraction of stems from the harvested site to the road (Rodgers et al. 2010). To facilitate the replanting of the site, some site preparation is required and the brash mats are collected together and re-shaped to form lines of forest residue called windrows. Generally, these windrows follow the main slope and drainage furrows in parallel rows to about 20 m from the edge of streams (Rodgers et al. 2010; Asam et al. 2014a). It was recommended by Asam et al. (2012) and O’Driscoll et al. (2014a) in their studies on blanket peat that brash windrows may be formed across the slope on harvested sites rather than along the slope and direction of furrows in order to maximize the retention of P by surface layer of peat and vegetation/grasses between brash windrows (Fig. 1; Asam et al. 2012; O’Driscoll et al. 2014a). The brash free areas downslope and between brash mats/windrows will act like mini-buffers where hydraulic loading is low as compared to traditional buffer areas downslope the catchment, allowing sufficient retention time for effective nutrient uptake by soil and vegetation (Fig. 1).
The results of simulated overland flow study support the hypothesis that grasses grown in mini-buffers between brash mats/windrows would be an efficient means for N retention on-site from throughflow nutrient-rich water. During all the flow events, the grassed flumes retained over 94% of added NH4+-N irrespective of increase (C1–C4) or subsequent decrease (C4–C1) in the inlet concentration of NH4+-N in overland flows (Table 4) with overall average retention of over 98%. Whereas, the retention of NH4+-N by the non-grassed flumes was significantly lower (23%) as compared to grassed flumes (94%) when the inlet concentration was 1.0 mg L−1 (C1) and this % retention increased (from 23 to 83%) with an increase in inlet concentration (C1–C4). However, it started to decrease again (74 to 37%) when the inlet concentration was reduced from C4 to C1 in the second set of overland flow events (Table 4). This is highlighted that even though the overall retention by non-grassed flumes is high (70%), they will not be very effective at lower concentrations (Table 4; Fig. 9).
Although the N uptake by grassed flumes is significantly higher, the non-grassed flumes have also shown a reasonable N uptake during the nutrient-rich overland flow study. One likely reason for this considerable N uptake by non-grassed flumes is the significant growth of natural vegetation even in non-grassed flumes as all the flumes were kept in the glass-house during the simulated rainfall and overland flow study, where there were favorable conditions for the growth of vegetation. The biomass of vegetation harvested from non-grassed flumes at the end of overland flow study was 173 g m−2 with an overall N content of 2199 mg m−2 in comparison the grassed flumes produced 484 g m−2 grass biomass with a total N content of 5756 mg m−2 during the same period (“Simulated overland flow experiment”). Secondly, the mechanisms other than vegetation uptake could also be responsible for N uptake in non-grassed flumes such as the microbial biomass. Silvan et al. (2003), in their study on peatland buffer areas, reported that about 15% of added inorganic N was immobilized by microbial biomass.
The importance of grasses in N immobilization is further signified with the facts that the concentrations of NH4+-N in pore water of non-grassed flumes are consistently higher than grassed flumes even after 1 week of application of nutrient-rich overland flows (Fig. 10), indicating that the retention by non-grassed flumes is not long term and sustainable. It has a high risk of subsequent leaching in runoff as the concentration of NH4+-N in pore water has a strong correlation with a corresponding concentration in runoff (Fig. 8). Whereas in grassed flumes, the significantly lower concentrations in porewater suggest effective uptake of NH4+-N soon after the application of nutrient-rich overland flow (Fig. 10). It is further noted that higher export of NO3−-N is measured in outflow from non-grassed flumes as compared to grassed flume in each overland flow test (Table 5). This concentration of NO3−-N is higher than even the default input of NO3−-N in nutrient-rich overland flow suggesting that a part of NH4+-N retained by non-grassed flumes is converted to NO3−-N. Higher concentration of N is reported to enhance the N mineralization in peat, which leads to an increase in the nitrification process (Mäkiranta et al. 2012). On the other hand, the grassed flumes effectively removed NH4+-N from the nutrient-rich flow and porewater, allowing less time for N mineralization and NO3−-N formation.
Vegetation can also increase the retention of nutrients from throughflow water by making the soil more porous and diverting more water towards the root zone. Several studies have shown that vegetation can effectively uptake nitrogen (NH4+-N and NO3−-N) from soils and throughflow water (Huttunen et al. 1996; Bedard-Haughn et al. 2004; Silvan et al. 2004a; Silvan et al. 2004b; Hefting et al. 2005; Søvik and Syversen 2008; Bhattarai et al. 2009). In an artificial nutrient addition experiment by Silvan et al. (2004a) on a restored peatland buffer area, vegetation biomass was responsible for uptake of over 70% of the added 90 kg ha−1 of N. If the N taken up by the grasses and vegetation ends up as their organic structural component and accumulates, the sustainable N immobilization is possible.
Conclusions
Given (1) the difficulties in the construction of buffer areas in blanket peatlands (Ryder et al. 2011; O’Driscoll et al. 2014b), (2) the negative effects of restoration work during the creation of buffer areas in peatland forestry and subsequent leaching of nutrients and heavy metals (Kaila et al. 2016; Nieminen et al. 2020b), (3) the low retention efficiencies during typical high hydraulic loadings in the blanket peatlands (Asam et al. 2012; O’Driscoll et al. 2014b), and (4) difficulties in removing all the harvest residue from blanket peatlands due to poor carrying capacity of the peat, the results of all the three experiments in this study suggest that seeded grasses are a major sink of N and immobilization of N onsite using the grass seeding and mini-buffer practice (by changing the orientation of brash windrows) could be an efficient and feasible means of reducing N export from harvested peatlands in order to protect sensitive water courses. However, the sustainability of retention and immobilization of N needs to be studied further in long-term field-scale experiments.
Data availability
Not applicable
References
Ahtiainen M, Huttunen P (1999) Long-term effects of forestry managements on water quality and loading in brooks. Boreal Environ Res 4:101–114
Asam Z, Kaila A, Nieminen M, Sarkkola S, O’Driscoll C, O’Connor M, Sana A, Rodgers M, Xiao L (2012) Assessment of phosphorus retention efficiency of blanket peat buffer areas using a laboratory flume approach. Ecol Eng 49:160–169
Asam Z, Nieminen M, Kaila A, Sarkkola S, O’Driscoll C, O’Connor M, Sana A, Rodgers M, Zhan X, Xiao L (2014a) Export of phosphorus and nitrogen from lodgepole pine (Pinus contorta) brash windrows on harvested blanket peat forests. Ecol Eng 64:161–170
Asam Z, Nieminen M, Kaila A, Laiho R, Sarkkola S, O’Connor M, O’Driscoll C, Sana A, Rodgers M, Zhan X, Xiao L (2014b) Nutrient and heavy metals in decaying harvest residue needles on drained blanket peat forests. Eur J Forest Res 133:969–982
Bedard-Haughn A, Tate K, Kessel C (2004) Using nitrogen-15 to quantify vegetation buffer effectiveness for sequestering N in runoff. J Environ Qual 33:2252–2262
Bhattarai R, Kalita P, Patel M (2009) Nutrient transport through a vegetative filter strip with subsurface drainage. J Environ Manag 90:1868–1876
Carling A, Irvine J, Hill A, Wood M (2001) Reducing sediment inputs to Scottish streams: a review of the efficiency of soil conservation practices in upland forestry. Sci Total Environ 265:209–227
Cummins T, Farrell EP (2003) Biogeochemical impacts of clearfelling and reforestation on blanket peatland streams I. Phosphorus. For Ecol Manag 180:545–555
Davidson EA, Belk E, Boone RD (1998) Soil water content and temperature as independent or confounded factors controlling soil respiration in a temperate mixed hardwood forest. Glob Chang Biol 4:217–227
Finér L (1989) Biomass and nutrient cycle in fertilized and unfertilized pine, mixed birch and pine and spruce stands on a drained mire. Acta For Fennia 208:1–63
Fontaine S, Bardoux G, Abbadie L, Mariotti A (2004) Carbon input to soil may decrease soil carbon content. Ecol Lett 7:314–320
Fontaine S, Barot S, Barre P, Bdioui N, Mary B, Rumpel C (2007) Stability of organic carbon in deep soil layers controlled by fresh carbon supply. Nature 450:277–280
Hefting M, Clement J, Bienkowski P, Dowrick D, Guenat C, Butturini A, Topa S, Pinay G, Verhoeven J (2005) The role of vegetation and litter in the nitrogen dynamics of riparian buffer zones in Europe. Ecol Eng 24:465–482
Huttunen A, Heikkinen K, Ihme R (1996) Nutrient retention in the vegetation of an overland flow treatment system in northern Finland. Aquat Bot 55:61–73
Kaila A, Asam Z, Sarkkola S, Xiao L, Laurén A, Vasander H, Nieminen M (2012) Decomposition of harvest residue needles on peatlands drained for forestry – implications for nutrient and heavy metal dynamics. For Ecol Manag 277:141–149
Kaila A, Sarkkola S, Laurén A, Ukonmaanaho L, Koivusalo H, Xiao L, O’Driscoll C, Asam Z, Tervahauta A, Nieminen M (2014) Phosphorus export from drained Scots pine mires after clear-felling and bioenergy harvesting. For Ecol Manag 325:99–107
Kaila A, Asam Z, Koskinen M, Uusitalo R, Smolander A, Kiikkilä O, Sarkkola S, O’Driscoll C, Kitunen V, Fritze H, Nousiainen H, Tervahauta A, Xiao L, Nieminen M (2016) Impact of re-wetting of forestry-drained peatlands on water quality - a laboratory approach assessing the release of P, N, Fe, and dissolved organic carbon. Water Air Soil Pollut 227:1–15
Labadz, J., Allott, T. Evans, M., Butcher, D., Billett, M., Stainer, S., yallop, A., Jones, P., Innerdale, M., Harmon, N., Maher, K, Bradbury, R., Mount, D., O’Brien, H., Hart, R., 2010. Peatland hydrology. Report to IUCN UK Peatland Programme. Edinburgh (funded by the North Pennines AONB Partnership).
LIoyd J, Taylor JA (1994) On the temperature dependence of soil respiration. Funct Ecol 8:315–323
Lundin L (1998) Alternative peatland forestry; impacts on hydrology and surface water chemistry. In: Proceedings of the International Peat Symposium, Jyväskylä, Finland, 7–9 September, vol 1998, pp 76–78
Lundin L (1999) Effects on hydrology and surface water chemistry of regeneration cuttings in peatland forests. Int Peat J 9:118–126
Mäkiranta P, Laiho R, Penttilä T, Minkkinen K (2012) The impact of logging residue on soil GHG fluxes in a drained peatland forest. Soil Biol Biochem 48:1–9
Marttila H, Karjalainen S-M, Kuoppala M, Nieminen M, Ronkanen A-K, Kløve B, Hellsten S (2018) Elevated nutrient concentrations in headwaters affected by drained peatland. Sci of The Tot Environ 643:1304–1313
Mulqueen J, Rodgers M, Marren N, Healy MG (2006) Erodibility of hill peat. Irish J Agric Food Res 45:103–114
Nieminen M (1998) Changes in nitrogen cycling following the clearcutting of drained peatland forests in southern Finland. Boreal Environ Res 3:9–21
Nieminen M (2003) Effects of clear-cutting and site preparation on water quality from a drained Scots pine mire in southern Finland. Boreal Environ Res 8:53–59
Nieminen M (2004) Export of dissolved organic carbon, nitrogen and phosphorus following clear-cutting of three Norway spruce forests growing on drained peatlands in southern Finland. Silva Fennica 38:123–132
Nieminen M, Ahti E, Nousiainen H, Joensuu S, Vuollekoski M (2005) Does the use of riparian buffer zones in forest drainage sites to reduce the transport of solids simultaneously increase the export of solutes? Boreal Environ Res 10:191–201
Nieminen M, Sarkkola S, Haahti K, Sallantaus T, Koskinen M, Ojanen P (2020a) Forestry on drained peatlands as a source of surface water nitrogen and phosphorus in Finland. Suo 71(1):1–13
Nieminen M, Sarkkola S, Tolvanen A, Tervahauta A, Saarimaa M, Sallantaus T (2020b) Water quality management dilemma: Increased nutrient, carbon, and heavy metal exports from forestry-drained peatlands restored for use as wetland buffer areas. For Ecol Manag 465. https://doi.org/10.1016/j.foreco.2020.118089
Nisbet T, Dutch J, Moffat A (1997) Whole-Tree Harvesting. A Guide to Good Practice. Forestry Commission Practice Guide, Forestry Commission, Edinburgh, UK
O’Driscoll C, Rodgers M, O’Connor M, Asam Z, De Eyto E, Poole R, Xiao L (2011) A potential solution to mitigate phosphorus release following clearfelling in peatland forest catchments. Water Air Soil Pollut 221:1–11
O’Driscoll C, de Eyto E, Rodgers M, O’Connor M, Asam Z, Xiao L (2012) Diatom assemblages and their associated environmental factors in upland peat forest rivers. Ecol Indic 18:443–451
O’Driscoll C, O’Connor M, Asam Z, de Eyto E, Poole R, Rodgers M, Zhan X, Nieminen M, Xiao L (2014a) Whole-tree harvesting and grass seeding as potential mitigation methods for phosphorus export in peatland catchments. For Ecol Manag 319:176–185
O’Driscoll C, O’Connor M, Asam Z, de Eyto E, Rodgers M, Xiao L (2014b) Creation and functioning of a buffer zone in an upland peat forest. Ecol Eng 62:83–92
O’Driscoll C, O’Connor M, Asam Z, de Eyto E, Brown LE, Xiao L (2016) Forest clearfelling effects on dissolved oxygen and metabolism in peatland streams. J Environ Manag 166:250–259
Palviainen M, Finér L, Kurka A-M, Mannerkoski H, Piirainen S, Starr M (2004) Decomposition and nutrient release from logging residues after clear-cutting of mixed boreal forest. Plant Soil 263:53–67
Regina K, Nykänen H, Silvola J, Martikainen PJ (1996) Fluxes of nitrous oxide from boreal peatlands as affected by peatland type, water table level and nitrification capacity. Biogeochem. 35:401–418
Renou-Wilson F, Byrne KA (2015) Irish peatland forests: Lessons from the past and pathways to a sustainable future. Restoration of Boreal and Temperate Forests, 2nd Edition. In: 321-335
Roberts S, Harrington C, Terry T (2005) Harvest residue and competing vegetation affect soil moisture, soil temperature, N availability, and Douglas-fir seedling growth. For Ecol Manag 205:333–350
Rodgers M, O’Connor M, Healy MG, O’Driscoll C, Asam Z, Nieminen M, Poole R, Muller M, Xiao L (2010) Phosphorus release from forest harvesting on an upland blanket peat catchment. For Ecol Manag 260:2241–2248
Rosén K, Lundmark-Thelin A (1987) Increased nitrogen leaching under piles of slash – a consequence of modern forest harvesting techniques. Scand J For Res 2:21–29
Ryder L, de Eyto E, Gormally M, Sheehy-Skeffington M, Dillane M, Poole R (2011) Riparian zone creation in established coniferous forests in Irish upland peat catchments: physical, chemical and biological implications. Biol Environ: Proc. R Irish Acad 111B(1):41–60
Silvan N, Vasander H, Karsisto M, Laine J (2003) Microbial immobilisation of added nitrogen and phosphorus in constructed wetland buffer. Appl Soil Ecol 24:143–149
Silvan N, Vasander H, Laine J (2004a) Vegetation is the main factor in nutrient retention in a constructed wetland buffer. Plant Soil 25:179–187
Silvan N, Tuittila E-S, Vasander H, Laine J (2004b) Eriphorium vaginatum plays a major role in nutrient immobilization in boreal peatlands. Ann Bot Fenn 41:189–199
Silvan N, Sallantaus T, Vasander H, Laine J (2005) Hydraulic nutrient transport in a restored peatland buffer. Boreal Environ Res 10:203–210
Søvik A, Syversen N (2008) Retention of particles and nutrients in the root zone of a vegetative buffer zone - effect of vegetation and season. Boreal Environ Res 13:223–230
Väänänen R, Nieminen M, Vuollekoski M, Nousiainen H, Sallantaus T, Tuittila E-S, Ilvesniemi H (2008) Retention of phosphorus in peatland buffer zones at six forested catchments in southern Finland. Silva Fennica 42:211–231
Vikman A, Sarkkola S, Koivusalo H, Sallantaus T, Laine J, Silvan N, Nousiainen H, Nieminen M (2010) Nitrogen retention by peatland buffer areas at six forested catchments in southern and central Finland. Hydrobiol. 641:171–183
Acknowledgements
The authors gratefully acknowledge NUI Galway, Trinity College Dublin, University of Gujrat, Coillte Teoranta, and the Marine Institute for their help and allowing the use of their facilities.
Funding
Financial support from the Department of Agriculture, Food, and the Marine in Ireland, under SANIFAC (RSF07 552) and ForSite (11/C/208) projects is acknowledged.
Author information
Authors and Affiliations
Contributions
ZZA conducted the experimental work, compiled all the results and wrote the manuscript first draft. COD & MOC facilitated in experimentation and extensive field work. MA significantly contributed in data analysis and interpretation using statistical tools and also wrote the relevant sections in the manuscript first draft. MW helped in drawing figures and tables. MR contributed in writing the manuscript. ASN contributed in editing and improving the overall manuscript. LX supervised this study and contributed in all aspects including experimentation, analysis and writing the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests
Ethical approval
Not applicable
Consent to participate
Not applicable
Consent to publish
Not applicable
Additional information
Responsible Editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Asam, ZuZ., O’Driscoll, C., Abbas, M. et al. Mechanism and role of seeded native grasses to immobilize nitrogen on harvested blanket peat forests for protection of water courses. Environ Sci Pollut Res 28, 24756–24770 (2021). https://doi.org/10.1007/s11356-020-11433-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-11433-4