Abstract
This study was performed to determine the concentrations of copper (Cu), zinc (Zn), and lead (Pb) in the gill, liver, muscle, and tail fin tissues of Euthynnus affinis, Katsuwonus pelamis, and Thunnus albacares from Oman Sea. All samples were analyzed using a flame atomic absorption spectrophotometer and the results were expressed as μg g−1 dry weight. Metal concentrations were significantly higher in the liver than other tissues in three species (with some exceptions) (p < 0.05). The concentrations of metal accumulation in tissues of tuna species followed the Zn > Cu > Pb. Correlation matrix and principal component analysis (PCA) revealed that Zn and Pb have anthropogenic sources. Estimated daily intake (EDI) in three tuna species for heavy metals were below the tolerable daily intake (TDI). Also, the mean target hazard quotient (THQ) based on studied metals in three tuna species was below 1, which suggests that consumption of these fish can be safe for human health in the Oman Sea.

.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fish contain high protein, Omega-3 and Omega-6 fatty acids, vitamins, and other essential minerals; therefore, fish consumption is necessary for human health (Kris-Etherton et al. 2002; Mziray and Kimirei 2016) and should be consumed in a weekly diet. Fish consumption, at least two meals per week, could prevent cardiovascular diseases (Miniadis-Meimaroglou et al. 2007; Mori 2017). It is well known that heavy metals are deliberately or accidentally discharged into aquatic environments. In the aquatic environment, the main sources of heavy metal contaminants are anthropogenic activities, such as industries and agriculture (Fallah et al. 2011). Human use of heavy metals in industries and other activities has caused marine environmental pollution and adverse effects on the health of marine organisms (Castro-Gonzalez and Mendez-Armenta 2008). Marine organisms, especially fish, are a good indicator of metal pollution monitoring in the aquatic environment (Rashed 2001; Kojadinovic et al. 2007). Since the fish are situated at the top of the food chain, contaminants could accumulate in fish tissues and transferred to humans (Yilmaz et al. 2010). Different organs in fish are used for pollution biomonitoring. The gill has a large surface area with water in fish, therefore it absorbed right amount of metal ions (Dhaneesh et al. 2012). The amount of concentrations of heavy metals in gills indicates the heavy metal concentrations in the waters (Romeo et al. 1999). The liver is an essential organ in heavy metal accumulation due to its role in fish metabolism (Squadrone et al. 2013). Muscle is significant in the human diet and used as an excellent tool for the health risk assessment in heavy metal pollution (Yi et al. 2017; Sadeghi et al. 2019). Tail fin can be used to heavy metal biomonitoring without killing fish (Mziray and Kimirei 2016) that needs more studies. Fish is one of the most critical marine products in the south Iran. Tuna fish is one of the leading fish used in Iranian canning plants. Tuna fish is widespread use in human food; therefore, determination amount of pollutant accumulation in tuna tissues is necessary and is also essential to assess the potential of exposures to humans (Kojadinovic et al. 2007; Hart et al. 2008; Percin et al. 2011). There are several major fishing centers for tuna fish in the Iranian coasts of the Oman Sea (Hamzeh et al. 2013). Previous studies on contamination in harbors have shown high levels of heavy metal concentration in the sediments of this area (Hamzeh et al. 2013; Pakzad et al. 2014; Sadeghi et al. 2015). Therefore, the heavy metal contamination in harbors can affect on the fish quality. According to reports by IOTC (2017), the tuna and tuna-like fishes catch is 234,000 Mt in the Indian Ocean areas. Since the Oman Sea leads to the Indian Ocean, the amount of tuna catches in this sea is very high.
Euthynnus affinis (kawakawa or mackerel tuna), Katsuwonus pelamis (skipjack tuna), and Thunnus albacares (yellowfin tuna) are three species of Scombridae family. These species are distributed in the Indo-West Pacific region, Indian and Atlantic Oceans, and also tropical and subtropical waters (Collette 2001; Hart et al. 2008; Langley et al. 2009). Since these species are located at the top of the marine food chain and most commonly used in canned tuna, the accumulation of heavy metals throughout the food web would tend to a severe risk for human health (Kojadinovic et al. 2007). The USEPA (2000) has proposed target hazard quotient (THQ) and total THQ (TTHQ) to evaluate the non-carcinogenic human health risk for heavy metal with fish consumption (Varol et al. 2017). To the best of our knowledge, very few studies have been done to determine the bioaccumulation of heavy metals in species of Scombridae family (tuna fish) in Oman Sea (Al-Busaidi et al. 2011; Ahmed and Bat 2015). Also there is no previous study of metal accumulation and risk assessment for human consumption in the tuna fish on the Iranian regions of Oman Sea. Keeping the above facts in mind, the aim of this study were (1) to determine the concentrations of copper (Cu), zinc (Zn), and lead (Pb) in the gill, liver, muscle, and tail fin tissues of Euthynnus affinis, Katsuwonus pelamis, and Thunnus albacares from Oman Sea, (2) to assess principal component analysis (PCA) for distribution of heavy metals in the different tissues, (iii) to evaluated EDI (estimated daily intake) of heavy metals and compare it with TDI (tolerable daily intake), and (4) to assess potential human health risks by consumption of tuna fish based on THQ (target hazard quotient) and TTHQ (total target hazard quotient).
Material and methods
Sample collection
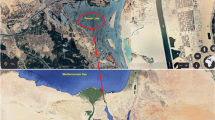
Three fish species, namely Euthynnus affinis, Katsuwonus pelamis, and Thunnus albacares, which belong to the family Scombridae, were selected for this study (n = 30 for each species). These species are selected because they are commercially valued and used in high quantities along Iran and other parts of the world. Fish samples were collected from the Oman Sea (Geographic coordinates: 60° 74′ 56″ E and 25° 26′ 79″ N) in May 2017. The samples were kept in a cool box with ice and immediately transported to the laboratory.
Sample preparation
In the laboratory, samples were washed with distilled water. Total fish length (Cm) and weight (g) were measured before sample dissection. Gills, liver, muscle (from the dorsal region), and tail fin tissues of each three species were collected, stored in clean plastic polyethylene bags and frozen at − 20 °C before chemical analysis (Staniskiene et al. 2006).
Chemical analysis and quality assurance/control
Before the analysis, all the laboratory equipment were cleaned with nitric acid for 48 h and then rinsed with distilled water and dried in the oven at 150 °C. For metal concentration, fish tissues were dried in an oven at 105 °C to constant weight. A total of 1 g of each dried tissues was digested with 10 mL of 4:1 (v/v) HNO3-HCLO4 at 150 °C on a hot plate. After cooling solutions at room temperature, samples were filtered through a filter paper (Whatman No. 42) and diluted to 25 mL with double distilled water (AOAC 2005). All samples were analyzed three times for heavy metal concentration. Concentrations of copper (Cu), zinc (Zn), and lead (Pb) were analyzed using an atomic absorption spectrophotometer (GBC-932 Australia). All results of heavy metal concentration were expressed as μg g−1 dry weight. Standard solutions of each metal were prepared from stock solutions (high quality and analytical grade chemicals: Merck, Darmstadt, Germany). Certified reference material, DORM-2 from the National Research Council Canada, was prepared for quality assurance and control. The recovery rates for Cu, Zn, and Pb were 101.3, 97.4, and 98.5%, respectively. Limits of detection (LOD) were 0.05 μg g−1 for Cu and Pb and 0.01 μg g−1 for Zn.
Health risk assessment for fish consumption
Estimated daily intake of heavy metals
The estimated daily intake (EDI) of heavy metals (Cu, Zn, and Pb) was determined according to the following equation reported by Bortey-Sam et al. (2015):
where MC is the mean concentration of heavy metal in muscle tissue of fish (μg/g); FDC is the average food daily consumption of fish muscle (g/person/day), which is 25.2 g/person/day in Iran (IFO 2015), and BW is the body weight (average 70 kg for adults). EDI was expressed as μg/kg bw/day.
Target hazard quotients
Target hazard quotients (THQ) were used to assess the risk of heavy metal contamination for human health. According to Yi et al. (2017), THQ is less than 1 which indicates that there are no adverse effects during a lifetime for human health. Also, THQ exceeded which suggests adverse health effects for consumers. THQ was calculated according to the following equation (USEPA 2000):
where EF is the exposure frequency (days per year); ED is the exposure duration (years); FIR is the food ingestion rate (g/person/day); C is the metal concentration in fish (μg/g); RFD is the oral reference dose (μg g−1/day). BW is the body weight (kg), and ATn is the average exposure time for non-carcinogens effects (days per year × ED). All parameters and values used in THQ estimation are shown in Table 1.
Total THQ (TTHQ) was calculated as the sum of the individual THQ for each metal in each species according to the method of Li et al. (2013):
Statistical analysis
Statistical analysis was performed using SPSS (Statistical Package for Social Sciences) software (version 22.0, SPSS Company, Chicago, USA). The data were first checked for normality using the Shapiro-Wilk test. Comparisons significant differences of heavy metal concentrations among three fish species and tissues were determined using one-way ANOVA followed by Duncan’s test. Comparisons of heavy metal concentrations on each tissue of fish species were tested using Kruskal–Wallis test. Spearman correlation rank tests were used to test relationships between concentrations of the same heavy metal in different tissues and between the concentrations of different heavy metals in the same tissues. Principal component analysis (PCA) (PAST 1.9 Windows) was used to investigate the distribution of heavy metals in the different tissues. A p < 0.05 was defined as statistically significant. Microsoft Office Excel (2013) was used to draw the diagrams.
Results and discussion
Heavy metal concentrations in tuna fish
The results of heavy metal determination accumulated (mean ± standard deviation) in gill, liver, muscle, and tail fin tissues of Euthynnus affinis, Katsuwonus pelamis, and Thunnus albacares, as well as the range of the weight and length of the fish sampled, are shown in Table 2. According to the results, Cu and Zn concentrations were significantly higher in the liver than in other tissues in three species (p < 0.05) (Table 2). Between the three measured heavy metal, Zn concentration detected in all tissues of Euthynnus affinis was significantly higher than in the other two species (p < 0.05). In addition, concentration levels from highest to the lowest for Zn in gill, muscle, and tail fin tissues were detected as follows: Euthynnus affinis > Katsuwonus pelamis > Thunnus albacares. This pattern was Euthynnus affinis > Thunnus albacares > Katsuwonus pelamis for Zn in liver tissue (Table 2). A comparison of heavy metals concentration in fish tissues showed that there was a significant difference between the amount of metal accumulation in liver tissue of all three fish species (with some exceptions)(p < 0.05). Also, Table 2 showed no significant difference found for Cu concentration in muscle and tail fin tissues of all three tuna species (p > 0.05).
In this study, the accumulation of heavy metals in the liver tissue of three fish species was recorded more than other tissues. Most authors reported similar results for high heavy metal concentrations in the liver (e.g., Kojadinovic et al. 2007; Ahmed and Yousuf 2013; El-Moselhy et al. 2014). Therefore, the liver is suitable organ for heavy metals monitoring in fish and aquatic environments. The highest concentrations of Cu were shown in the liver, while the lowest concentrations were recorded in the tail fin of three tuna species. Among tuna species, Euthynnus affinis contained the highest Cu concentration in gill, liver, and muscle tissues (Fig. 2). The level of Cu in the gill of Euthynnus affinis (37.93 ± 12.86 μg g−1 dry weight) was higher than those observed in this species from Karachi Pakistan by Ahmed and Yousuf (2013) (16.57 + 3.40 μg g−1 dry weight). Also, mean concentration of Cu for Euthynnus affinis in the literature reported 17.2 μg g−1 dry weight (Agusa et al. 2005), 6.63 + 1.65 μg g−1 dry weight (Ahmed and Yousuf 2013), 1.64 ± 0.04 μg g−1 dry weight (Sivaperumal et al. 2006) for muscle, and 59.07 + 24.59 μg g−1 dry weight (Ahmed and Yousuf 2013) for liver tissues. Cu level in the liver of Katsuwonus pelamis in the present research was found to be lower than levels reported by Kojadinovic et al. (2007) and Ahmed and Yousuf (2013). For the Katsuwonus pelamis, the Cu concentration in gill and muscle were slightly higher than those described in the samples of the previous researches (7.39 + 1.91; 1.02 ± 0.89 μg g−1 dry weight) (Ahmed and Yousuf 2013; Kojadinovic et al. 2007) and was slightly lower than those reported in Katsuwonus pelamis liver tissue from the Western Indian Ocean (93.6 ± 75.3 μg g−1 dry weight) (Kojadinovic et al. 2007). Cu level in liver tissue of Thunnus albacares was very lower than those reported by Kojadinovic et al. (2007) (121 ± 74 μg g−1 dry weight). Mean value of Cu detected in Thunnus albacares was higher than those observed in muscle of this species from Red Sea (0.35 ± 0.07 μg g−1 dry weight) (Al-Shwafi 2002), Mozambican channel, and Reunion Island (Indian Ocean) (0.97 ± 0.23 and 1.99 ± 1.47 μg g−1 dry weight) (Kojadinovic et al. 2007).
High levels of heavy metal in three tuna species tissues could be due to high concentrations of this metal in the water. Regarding the levels of heavy metals in different tissues of three tuna species, the highest values were recorded for Zn in Euthynnus affinis. Mean value of Zn detected in gill tissues of Euthynnus affinis was higher than those reported in this species from Karachi Pakistan by Ahmed and Yousuf (2013) (16.34 + 5.13 μg g−1 dry weight). The level of Zn in the present study was extremely lower from that reported in the muscle of Euthynnus affinis (953 μg g−1 dry weight) from coastal areas of Malaysia (Agusa et al. 2005). Mean concentration of Zn for Katsuwonus pelamis in the literature reported 208 ± 61 μg g−1 dry weight (Kojadinovic et al. 2007) and 68.1 + 21.13 μg g−1 dry weight (Ahmed and Yousuf 2013) for liver tissue. Comparing present results to those reported by Ahmed and Yousuf (2013), it can be concluded that Zn contents in the muscle of Euthynnus affinis from Karachi Pakistan (16.33 + 2.26 μg g−1 dry weight) are extremely lower than our data. Mean Zn concentration in the muscle of Katsuwonus pelamis was 30.57 ± 3.71 μg g−1 dry weight. Study conducted by Kojadinovic et al. (2007) reported higher concentration of Zn in Katsuwonus pelamis (125 ± 94 μg g−1 dry weight) as compared with that reported in this study. On the other hand, Zn concentration detected in Katsuwonus pelamis was higher than the maximum level of Zn (6.87 + 2.41 μg g−1 dry weight) observed in this species from Karachi Pakistan (Ahmed and Yousuf 2013). Mean value of Zn recorded in the liver and muscle tissues of Thunnus albacares were lower than those observed from Mozambican channel (Indian Ocean) (439 ± 254 and 64.1 ± 47.3 μg g−1 dry weight, respectively) (Kojadinovic et al. 2007). Bhoyroo et al. (2015) reported mean Zn concentrations in the Scombridae family, including Thunnus albacares in the EEZ (exclusive economic zone) of Mauritius between 1.34 and 10.03 mg/kg dry weight that was higher than the results in the present study. According to Hamzeh et al. (2013), Zn level is higher than the concentration of other contaminants in the surface sediments of several harbors of Oman Sea. Also, according to unpublished results of the previous work of the authors, the amount of Zn and Pb in the sediments of the Ramin, Beris, and Konarak harbors were higher than other heavy metals. Since tuna fish feed on small fish and other benthic organisms such as crustacean (Collette 2001), high levels of Zn accumulation in tuna tissues can be related to this case.
According to Ahmed and Yousuf (2013), Pb concentration in liver and gill tissues of Euthynnus affinis and Katsuwonus pelamis were lower than Pb in both species in this research. Agusa et al. (2005) reported mean Pb concentrations in the muscle of Euthynnus affinis from coastal areas of Malaysia 0.041 μg g−1 dry weight that was lower than those observed in the present study for this species (1.27 ± 0.22 μg g−1 dry weight). The highest lead concentration in the muscle of Euthynnus affinis recorded 0.4958 + 0.13641 μg g−1 dry weight in Karachi Fish Harbour, Pakistan (Ahmed and Bat 2015), which was less than the results of the present study. Mean value of Pb recorded in the muscle of Katsuwonus pelamis was higher than those observed in this species from Sultanate of Oman (0.0492 ± 0.197 μg g−1 dry weight) (Al-Busaidi et al. 2011) and the Mozambican channel (Indian Ocean) (0.07 ± 0.08 μg g−1 dry weight) (Kojadinovic et al. 2007). Ahmed and Yousuf (2013) reported maximum lead concentrations in the muscle of Katsuwonus pelamis from Karachi Pakistan 0.54 + 0.17 μg g−1 dry weight which was lower than results in present study. Among three tuna species, Thunnus albacares contained the lowest Pb concentration in muscle tissue with a mean concentration of 0.67 ± 0.16 μg g−1 dry weight. Mean concentration of Pb in Thunnus albacares from Western Indian Ocean, Mozambique Channel, in muscle tissue (0.09 ± 0.14 μg g−1 dry weight) (Kojadinovic et al. 2007) were lower than those found in this study (0.67 ± 0.16 μg g−1 dry weight).
Box plots (median, first and third percentiles, minimum and maximum values, and outliers) for the concentrations of Cu, Zn, and Pb in gill, liver, muscle, and tail fin tissues of tuna fish species from Oman Sea are presented in Fig. 1. The mean concentration of Cu, Zn, and Pb was lower in all tissues of Thunnus albacares (expect Zn in the liver).
The results indicated a significant number of positive correlations of heavy metals in tissues of three tuna species (Table 3). The highest positive correlation was found between gill and muscle tissues of Thunnus albacares for Cu (r = 0.754, p < 0.05). Also, a few significant negative correlations were observed for Thunnus albacares between gill and liver for Cu (r = − 0.745, p < 0.05) and Pb (r = − 0.855, p < 0.01) and between liver and muscle for Cu (r = − 0.900, p < 0.01). However, a significant negative correlation was shown between the liver and muscle of Katsuwonus pelamis (r = − 0.853, p < 0.01) for Pb. Comparing with published data, Burger and Gochfeld (2005) and Al-Busaidi et al. (2011) found no significant correlation between the heavy metal in fish tissues; while the significant correlation was found for the metal in tuna fish and dolphins from the Ecuadorian coast (Araujo and Cedeno-Macias 2016). There was no clear correlation between gill, liver, and muscle with tail fin as a non-destructive organ in three tuna species for pollution monitoring. However, a few positive correlations were recorded between gill and tail fin for Zn in Katsuwonus pelamis (r = 0.661, p < 0.05), muscle and tail for Cu and Zn in Euthynnus affinis (Cu: r = 0.697, Zn: r = 0.636 p < 0.05). A similar result for the relationship between heavy metal concentrations in tissues with tail fin in three marine fish species have been reported by Mziray and Kimirei (2016) which recommended that tail fin was not a good organ for all species and all metals, in pollution biomonitoring.
The correlation matrix was used for principal components extracted (Fig. 2). Two PC from three extracted PC had Eigenvalues greater than 0.6 that are used for assessment. Eigenvalue of PC1 and PC2 was 1.9 and 0.9, respectively. PC1 with 65.13% variance was highly influenced by Zn. The PC2 accounted for 32.55% variance and had high influenced with Pb. PC loading of Zn and Pb in tuna fish tissues suggested the anthropogenic source of heavy metals.
Health risk assessment
The EDI values compared with TDI values (respective tolerable daily intake) of Cu, Zn, and Pb through the tuna fish consumption were calculated and listed in Table 4. The results showed that the EDI in three tuna species for Cu, Zn, and Pb were below the tolerable daily intake, suggesting that consumption of Euthynnus affinis, Katsuwonus pelamis, and Thunnus albacares has no risks for people in the Oman Sea. Also, the lowest and highest daily intakes were recorded for Pb and Zn, respectively, at three tuna species. The mean THQ values of Cu, Zn, and Pb (based on the mean concentration) for average 70 kg adult humans at Euthynnus affinis, Katsuwonus pelamis, and Thunnus albacares were below 1 (Table 4). Higher THQ of Zn showed in Euthynnus affinis (0.11). Also, the higher THQ in Katsuwonus pelamis and Thunnus albacares recorded for Pb (0.11 and 0.06). Based on USEPA (2011) recommendation, THQ < 1 indicates no risk effect on human health. Therefore, the consumption of three tuna species from Oman Sea may be safe. Total THQ value of all heavy metals followed the descending order of Katsuwonus pelamis > Euthynnus affinis > Thunnus albacares. Similarly, consumption of tuna species from the eastern Pacific Ocean (Ordiano-Flores et al. 2011), the Indian Ocean around Sri Lanka (Jinadasa et al. 2018), and Galicia-Spain (Núñez et al. 2018) were found to be safe for human consumption without no threat to health.
Conclusions
The results of the present study showed new information on the concentration of heavy metals in the gill, liver, muscle, and tail fin tissues of the Scombridae family from the Oman Sea, Iran. Accumulation of heavy metals in the liver tissue of Euthynnus affinis, Katsuwonus pelamis, and Thunnus albacares were recorded more than other tissues. The mean heavy metal concentration in muscle tissue of three tuna species followed the descending order of Zn > Cu > Pb. Zn concentration was significantly higher than in all tissues of Euthynnus affinis other both species. According to the results, estimated daily intakes for Cu, Zn, and Pb were lower than tolerable daily intake in all species. The target hazard quotient and total target hazard quotient for all heavy metal in muscle tissues of three tuna species were less than 1. Thus, the consumption of these species from Oman Sea has no threat to human health. The results showed in this research will provide baseline information for future studies on bioaccumulation of heavy metal contamination and human risk assessment in commercial fish species of the Oman Sea.
References
Agusa T, Kunito T, Yasunaga G, Iwata H, Subramanian A, Ismail A, Tanabe S (2005) Concentrations of trace elements in marine fish and its risk assessment in Malaysia. Mar Pollut Bull 51:896–911
Ahmed Q, Bat L (2015) Heavy metal levels in Euthynnus affinis (cantor 1849) Kawakawa fish marketed at Karachi Fish Harbour, Pakistan and potential risk to human health. J Black Sea/Mediterranean Environ 21(1):35–44
Ahmed Q, Yousuf F (2013) Determination of heavy metals in Indian mackerel fishes (family-Scombridae) from Karachi Coast (doctoral dissertation) . https://doi.org/10.13140/RG.2.1.1188.6481
Al-Busaidi M, Yesudhason P, Al-Mughairi S, Al-Rahbi WAK, Al-Harthy KS, Al-Mazrooei NA, Al-Habsi SH (2011) Toxic metals in commercial marine fish in Oman with reference to national and international standards. Chemosphere 85(1):67–73
Al-Shwafi NAA (2002) Heavy metal concentration levels in some fish species in the Red Sea and Gulf of Aden-Yemen. Qatar Univ Sci J:171–176
AOAC (2005) Official methods of analysis, 18th edn. Association of Official Analytical Chemists, Gaithersburg
Araujo CVM, Cedeno-Macias LA (2016) Heavy metals in yellowfin tuna (Thunnus albacares) and common dolphin fish (Coryphaena hippurus) landed on the Ecuadorian coast. Sci Total Environ 541:149–154
Bhoyroo V, Soobratty N, Lalljee B (2015) Detection of heavy metals bio-accumulation in scombrids for the determination of possible health hazard. Afr J Food Sci Technol 6(4):98–107. https://doi.org/10.14303/ajfst.2015.041
Bortey-Sam N, Nakayama SM, Ikenaka Y, Akoto O, Baidoo E, Yohannes YB, Mizukawa H, Ishizuka M (2015) Human health risks from metals and metalloid via consumption of food animals near gold mines in Tarkwa, Ghana: estimation of the daily intakes and target hazard quotients (THQs). Ecotoxicol Environ Saf 111:160–167
Burger J, Gochfeld M (2005) Heavy metals in commercial fish in New Jersey. Environ Res 99:403–413
Castro-Gonzalez MI, Mendez-Armenta M (2008) Heavy metals: implications associated to fish consumption. Environ Toxicol Pharmacol 26:263–271
Collette BB (2001) Scombridae. Tunas (also, albacore, bonitos, mackerels, seerfishes, and wahoo). In: Carpenter KE, Niem V (eds) FAO Species Identification Guide for Fishery Purposes. The Living Marine Resources of the Western Central Pacific. Bony Fishes (Labridae to Latimeriidae), Estuarine Crocodiles. FAO, Rome, pp 3721–3756
Dhaneesh KV, Gopi M, Ganeshamurthy R, Kumar TTA, Balasubramanian T (2012) Bio-accumulation of metals on reef associated organisms of Lakshadweep Archipelago. Food Chem 131(3):985–991
El-Moselhy KM, Othman AI, Abd El-Azem H, El-Metwally MEA (2014) Bioaccumulation of heavy metals in some tissues of fish in the Red Sea, Egypt. Egypt J Basic Appl Sci:97–105
Fallah AA, Saei-Dehkordi SS, Nematollahi A, Jafari T (2011) Comparative study of heavy metal and trace element accumulation in edible tissues of farmed and wild rainbow trout (Oncorhynchus mykiss) using ICP-OES technique. Microchem J 98(2):275–279
Hamzeh MA, Shah-hosseini M, Naderi BA (2013) Effect of fishing vessels on trace metal contamination in sediments of three harbors along Iranian Oman Sea coast. Environ Monit Assess 185:1791–1807
Hart K, Kannan K, Tao L, Takahashi S, Tanabe S (2008) Skipjack tuna as a bioindicator of contamination by perfluorinated compounds in the oceans. Sci Total Environ 403:215–221
IOTC–SC20 (Indian Ocean Tuna Commission) (2017) Report of the 20th Session of the IOTC Scientific Committee. Seychelles, 30 November −4 December 2017. IOTC–2017–SC20–R[E]: 232 pp
Iran Fisheries Organization (IFO) (2015) Annual Iranian Fisheries Statistics 2013 and 2014. Fisheries design and program office, Tehran, Iran P: 33
Jinadasa BKKK, Chathurika GS, Jayaweera CD, Jayasinghe GDTM (2018) Mercury and cadmium in swordfish and yellowfin tuna and health risk assessment for Sri Lankan consumers. Food Addit Contam Part B:1–6. https://doi.org/10.1080/19393210.2018.1551247
Kojadinovic J, Potier M, Le Corre M, Cosson RP, Bustamante P (2007) Bioaccumulation of trace elements in pelagic fish from the Western Indian Ocean. Environ Pollut 146:548–566
Kris-Etherton P, Harris W, Appel L (2002) Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 106:2747–2757
Langley A, Wright A, Glenn Hurry G, Hampton J, Aqorua T, Rodwell L (2009) Slow steps towards management of the world’s largest tuna fishery. Mar Policy 33(2):271–279
Li J, Huang ZhY HY, Yang H (2013) Potential risk assessment of heavy metals by consuming shellfish collected from Xiamen, China. Environ Sci Pollut Res 20(5):2937–2947
Miniadis-Meimaroglou S, Dimizas C, Loukas V, Moukas A, Vlachos A, Thomaidis N, Paraskevopoulou V, Dasenakis M (2007) Proximate composition, fatty acids, cholesterol, minerals in frozen red porgy. Chem Phys Lipids 146:104–110
Mori TA (2017) Marine OMEGA-3 fatty acids in the prevention of cardiovascular disease. Fitoterapia. 123:51–58
Mziray P, Kimirei IA (2016) Bioaccumulation of heavy metals in marine fishes (Siganus sutor, Lethrinus harak, and Rastrelliger kanagurta) from Dar es Salaam Tanzania. Reg Stud Mar Sci 7:72–80
Núñez R, Garcia MA, Alonso J, Melgar MH (2018) Arsenic, cadmium and lead in fresh and processed tuna marketed in Galicia (NW Spain): risk assessment of dietary exposure. Sci Total Environ 627:322–331
Ordiano-Flores A, Galván-Magaña F, Rosiles-Martínez R (2011) Bioaccumulation of mercury in muscle tissue of yellowfin tuna, Thunnus albacares, of the Eastern Pacific Ocean. Biol Trace Elem Res 144:606–620
Pakzad HR, Pasandi M, Soleimani M, Kamali M (2014) Distribution and origin of heavy metals in the sand sediments in a sector of the Oman Sea (the Sistan and Baluchestan province, Iran). Quat Int 345:138–147
Percin F, Sogut O, Altinelataman C, Soylak M (2011) Some trace elements in front and rear dorsal ordinary muscles of wild and farmed Bluefin tuna (Thunnus thynnus L. 1758) in the Turkish part of the eastern Mediterranean Sea. Food Chem Toxicol 49(4):1006–1010
Rashed MN (2001) Monitoring of environmental heavy metals in fish from Nasser Lake. Environ Int 27(1):27–33
Romeo M, Siau Y, Sidoumou Z, Gnassia-Barelli M (1999) Heavy metal distribution in different fish species from the Mauritania coast. Sci Total Environ 232:169–175
Sadeghi P, Kazerouni F, Savari A, Movahedinia A, Safahieh A, Ajdari D (2015) Application of biomarkers in Epaulet grouper (Epinephelus stoliczkae) to assess chromium pollution in the Chabahar Bay and Gulf of Oman. Sci Total Environ 15(518–519):554–561
Sadeghi P, Loghmani M, Afsa E (2019) Trace element concentrations, ecological and health risk assessment in sediment and marine fish Otolithes ruber in Oman Sea, Iran. Mar Pollut Bull 140:248–254
Sivaperumal P, Sankar TV, Nair PGV (2006) Heavy metal concentrations in fish, shellfish and fish products from internal markets of India vis-avis international standards. Food Chem 102:612–620
Squadrone S, Prearo M, Brizio P, Gavinelli S, Pellegrino M, Scanzio T, Guarise S, Benedetto A, Abete MC (2013) Heavy metals distribution in muscle, liver, kidney and gill of European catfish (Silurus glanis) from Italian Rivers. Chemosphere 90:358–365
Staniskiene B, Matusevicius P, Budreckiene R, Skibniewska KA (2006) Distribution of heavy metals in tissues of freshwater fish in Lithuania. Pol J Environ Stud 15(4):585–591
USEPA (U.S. Environmental Protection Agency) (2000) Guidance for assessing chemical contaminant data for use in fish advisories, volume II. Risk assessment and fish consumption limits. (EPA 823-B-00-008). United States Environmental Protection Agency, Washington, DC
USEPA (United States Environmental Protection Agency) (2011) USEPA Regional screening level (RSL) summary Table: November 2011. (Available at: http://www.epa.gov/regshwmd/risk/human/Index.htm)
USEPA, Risk-Based Concentration Table (2010) http://www.epa.gov/reg3hwmd/risk/human/index.htm
Varol M, Kaya KG, Alp A (2017) Heavy metal and arsenic concentrations in rainbow trout (Oncorhynchus mykiss) farmed in a dam reservoir on the Firat (Euphrates) River: risk-based consumption advisories. Sci Total Environ 599–600:1288–1296
Yi Y, Tang C, Yi T, Yang Z, Zhang S (2017) Health risk assessment of heavy metals in fish and accumulation patterns in food web in the upper Yangtze River, China. Ecotoxicol Environ Saf 145:295–302
Yilmaz AB, Sangun MK, Yaglioglu D, Turan C (2010) Metals (major, essential to non-essential) composition of the different tissues of three demersal fish species from Iskenderun Bay, Turkey. Food Chem 123:410–415
Acknowledgments
The authors would like to thank the Chabahar Maritime University for the support of this research and anonymous reviewers for providing helpful comments that improved the quality of this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sadeghi, P., Loghmani, M. & Frokhzad, S. Human health risk assessment of heavy metals via consumption of commercial marine fish (Thunnus albacares, Euthynnus affinis, and Katsuwonus pelamis) in Oman Sea. Environ Sci Pollut Res 27, 14944–14952 (2020). https://doi.org/10.1007/s11356-020-07907-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-07907-0