Abstract
The current research aimed at collecting detailed information about the consequences of cropping history on the accumulation of soil organic carbon (SOC) within different soil depths, i.e., 0–10, 10–20, 20–30 and 30–60 cm. The study site is located at El Gabal El Asfar area (Egypt) whose soils were irrigated with raw sewage effluent as a sole source of irrigation for different periods extended up to 80 years. SOC increased progressively with increasing cropping time, and on the other hand, decreased noticeably with increasing soil depth. The increases significantly correlated with both of the silt and clay contents in soils which increased with time. Soil bulk density and the hydraulic conductivity significantly and negatively correlated with SOC, respectively. Fractions of SOC, i.e., water soluble C, hot water C and soil biomass C in the surface soil layer (0–10 cm), increased progressively with increasing time of land use. Such pools significantly correlated with SOC on one hand and with each other on the other hand. Active (labile) organic carbon fraction increased with time. This fraction also significantly correlated with the different C pools. In conclusion, the hypothesis that SOC is physically protected against soil microbes within the soil requires more investigations to clarify such results obtained herein because this study highlighted the presence of a dynamic equilibrium among the different fractions or pools of the SOC.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soil organic carbon (SOC) is the potential sink of atmospheric carbon (Brar et al. 2013); hence, it deserves special attention (Vieira et al. 2007; De Bona et al. 2008). It is probably the major carbon reservoir in the terrestrial ecology (Zhou et al. 2019) that directly affects soil quality (Shaffer et al. 2001) and health (Francaviglia et al. 2019). Moreover, carbon sequestration in soil can minimize climate change (Chen et al. 2010; Schlesinger and Amundson 2019). However, the distribution and stability of organic matter in soils seemed to be largely indistinguishable (Zhao et al. 2019); therefore, further researches are needed to highlight such pathways in soils. It is hypothesized that soil aggregates can potentially protect SOM from being decomposed by soil microbes (Tan et al. 2007; Goebel et al. 2009; Wiesmeier et al. 2012; D’Acqui et al. 2017). According to this theory, the labile form of soil C finds its way among soil particles (Whalen et al. 2000; Sheng et al. 2015; Tobiašová et al. 2016) to exist in too small pores that cannot be entered by soil biota (Balesdent et al. 2000), thus comprising partially protected C fraction in soil (Goebel et al. 2009). These findings probably indicate that the carbon cycle seemed to be incomplete and that the residual or non-labile C fractions as well as the total contents of SOC are expected to be in continuous increase with time. In spite of that, SOC content seemed to be extremely low in many arable lands of arid and semi-arid zones that were cultivated with different crops for hundreds of years and are still in use (Nieder and Benbi 2008; Tarnocai et al. 2009; Bassouny and Abbas 2019; Farid et al. 2019). The alternative assumption is that the by-products of soil biota are probably the stable organic forms (Schmidt et al. 2011) that exist mainly in the organic-mineral aggregates (Strosser 2010), hence improving soil aggregation (Weigel et al. 2011). These two assumptions should be further investigated while reconsidering the dynamics of building up of SOC, especially in arid soils.
Some factors might contribute to building up of SOC, e.g., climate conditions and the fine mineral fraction in soil. In this concern, high temperature is thought to accelerate the decomposition of SOC in soils (Moinet et al. 2018); hence, the decomposition cycle of SOC seemed to be high in the arid (Mlih et al. 2016) and semi-arid region soils (Maia et al. 2019). Also, the fine mineral fraction in soil is another potential component that determines SOC stabilization in most soils (Wiesmeier et al. 2019). It is therefore thought that following up the changes in soil texture (especially through the changes that might occur due to the clay content in soil) might draw attention to the pathways of carbon buildup in soils. Furthermore, soil cropping accelerates the degradation of SOM; thus, only recalcitrant materials remain (Paul et al. 1997). Also, cropping practices are important factors affecting SOC stocks (Chopin and Sierra 2019).
To investigate the dynamics of SOC in the arid zones, the changes that occur within the different fractions of SOC with time should be monitored. Water-soluble organic carbon (WSC) is the most dynamic C pool in the soil (Peregrina et al. 2012). It is the source of C energy for soil biota (Charest et al. 2004). Hot water carbon (HWC) is also considered a labile pool of soil carbon (Ghani et al. 2003), which contains readily available nutrients for plants (SrdjanŠeremešić et al. 2013); however, it is less thermally stable than the cold water–extracted OC (Landgraf et al. 2006). Stock of SOC is affected by soil management techniques (Post and Kwon 2000) and the labile fraction of SOC is an important marker determining the success of management practices (de Oliveira et al. 2016). Therefore, determination of the labile fractions of SOC in terms of active carbon (Blair et al. 1995) might be helpful for calculating carbon management index (CMI). This indicator finds out whether organic carbon management could result in variations in the organic C content of the investigated soils or not (Leal et al. 2016).
One of the main challenges facing crop production in the arid and semi arid-zone countries is fresh water scarcity (Abbas and Bassouny 2018; Zolti et al. 2019). These countries were and still forced toward using industrial and municipal wastewaters to satisfy their water needs (Gatta et al. 2018), after however considering appropriate wastewater treatments to diminish the levels of water contaminants (Galvis et al. 2018; Zolti et al. 2019). The financial aspects of reclaiming wastewaters are the recovery of water and nutrients, while diminishing the pollution discharges into water bodies (Zhang and Shen 2019); yet, inappropriate wastewater treatments might result in negative ecological impacts, e.g., growth of resistant soil bacteria (Bougnom et al. 2019), pathogens (El-Motaium and El-Seoud 2007), organic pollutants (El-Motaim and Hashim 2009), and accumulation of potentially toxic elements in soil (Abdel-Shafy and Abdel-Sabour 2006; Abdelhafez et al. 2015) which were accumulated mainly in the topsoil after being retained with the organic residues present in wastewaters (Abbas and Bassouny 2018). The arable lands of El Gabal El Asfar (Egypt) were selected for this study because they represent sandy soil texture (El-Hassanin et al. 1993) of the arid and semi-arid regions (Williams 1999). These soils are low in their organic carbon content (less than 50 g kg−1) (Abdel-Shafy and Abdel-Sabour 2006) even with successive applications of organic amendments for many years (Abbas and Bassouny 2018) because the decomposition of organic C under the arid and semi-arid climates prevailing in Egypt is thought to be high (Kirschbaum 2000; Rodeghiero et al. 2009). Accordingly, the consequences of amending these soils with organic amendments might be monitored clearly. These soils have received sewage effluents rich in organic C (Su et al. 2010) as a sole source for irrigation (rich in the suspended organic materials) for more than 80 years (El-Motaium 2000; Abdel-Shafy and Abdel-Sabour 2006); besides, they received sludge as a source of nutrients (Casado-Vela et al. 2006) which might contribute to increasing its SOC. It is then thought that these soils are the ideal model for studying the dynamics of OC in the irrigated soils of different cropping histories.
The current research aims at investigating the consequences of successive inputs of organic matter through sewage effluent on the accumulation of SOC and its different fractions, i.e., water-soluble organic carbon, hot water organic carbon and microbial biomass carbon within the different depths (0–10, 10–20, 20–30 and 30–60 cm) of the arid soils of El Gabal El Asfar area, Egypt. This study hypothesized the following two assumptions: the different fractions of SOC are correlated significantly and positively with each other (H1) and this assumption indicates that all SOC fractions are in continuous and dynamic equilibrium in soil. The organic residues are bound to the fine fractions of soil minerals (especially clay and silt) and this might increase soil bulk density and probably change soil texture on the long run because these organics trap the suspended minerals that may exist in the sewage effluent (H2). The consequences of these organic amendments on soil chemical characteristics and carbon management index were also investigated. It is thought that the results of such an investigation will be helpful in providing more knowledge about the global carbon cycle.
Materials and methods
Location of study and soil sampling
The study area was El Gabal El Asfar (Egypt) located between latitudes 30° 13′ 9″ and 30° 16′ 20″ N and longitudes 31° 22′ 10″ and 31° 24′ 10″ E (Fig. 1).
Seventeen farms were selected from the El Gabal El Asfar area (cultivated with vegetables and field crops) to represent soils of different cropping histories irrigated with raw effluent sewage water (water characteristics are presented in Table 1). The studied soils comprised ≤ 5 year (one farm), 5–10 years (2 farms), > 10–20 years (4 farms), > 20–30 years (3 farms), > 30–40 years (4 farms), > 40–50 (one farm) and > 50–80 years (2 farms). Soil samples were collected in triplicate from the 17 farms at four different depths, i.e., 0–10 cm, 10–20 cm, 20–30 cm and 30–60 cm, in three successive seasons (summer 2015, winter 2015/2016, and summer 2016). The collected soil samples were air dried, crushed with wooden mallet and sieved to pass through a 2-mm stainless steel sieve. The soils under study are classified as Typic Torripsamments according to the soil taxonomy of Soil Survey Staff (2010).
Soil analysis
Physical and chemical characteristics of the investigated soils were determined according to Klute (1986) and Page et al. (1982) as follows: particle size distribution by the pipette method, hydraulic conductivity in undisturbed soil sample, cation exchange capacity (CEC) by ammonium acetate method, and total organic carbon (TOC) according to the Walkley and Black method.
The different pools of SOC were determined according to Safařík and Šantrůčková (1992) and Ghani et al. (2003) as follows:
Cold and hot water soluble C
Soil portions (equivalent to 3 g oven dry weight each) were added to 50-mL polypropylene centrifuge tubes together with 30 mL distilled water. The suspension was then shaken for 30 min at 30 rpm, centrifuged for 20 min at 3500 rpm, and then filtered through a 0.45-μm cellulose nitrate membrane filter into separate vials for carbon analysis. The supernatant of this extract represented the cold water–soluble carbon (WSC). Another 30-mL aliquot of distilled water was added to the sediments in each tube and then shaken on a vortex shaker for 10 s to suspend soil in water. Tubes were capped and left on a hot water bath (80 °C) for 16 h; afterwards, tubes were shaken for 10 s on a vortex shaker, centrifuged for 20 min at 3500 rpm to release hot water C (HWC) from the SOM, and then filtered. Total carbon in the first and second extracts was determined by a Shimadzu Total Organic Carbon (TOC) Analyzer, model 5000A.
Microbial biomass carbon (MBC)
Two groups of moist soil samples (10 g each on dry weight basis) were placed into 50-mL beakers. The beakers were then placed inside 1-L glass jars. Small beakers, containing 10 mL each of chloroform, were placed to a group of soils previously placed in beakers, whereas the second group was left without chloroform to serve as a control treatment. The jars were sealed and allowed to stand at room temperature for 48 h immediately after fumigation and a 100-mL aliquot of K2SO4 solution 0.5 M was used to extract microbial biomass carbon. The aliquot was left until dryness and the dichromate oxidation method was used to determine microbial biomass carbon.
Active carbon (AC)
Soil portions (1 g on dry weight basis each) were added to centrifuge tubes together with aliquots of 20 mL KMnO4 (0.333 M). Suspensions were then shaken for 15 min at 200 rpm, centrifuged for 5 min at 3000 rpm, and transferred volumetrically using distilled water up to 10 mL. Thereafter, the active carbon was determined colorimetrically in solution at 550-nm light wave using spectrophotometer model Uni Cam UV 300.
Data processing
The CMI was calculated according to Blair et al. (1995) as follows:
Where CPI is the Carbon Pool Index calculated as the ratio between total soil C in soils of study and the corresponding one in the reference soil. The farm of cropping history < 5 years cropping was taken as a reference farm in the calculations of CPI, LI and CMI for the studied farms varying in their cropping histories.
LI, the liability index, was calculated as follows:
Results
Soil organic carbon
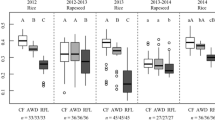
Data represented by Fig. 2 reveal that SOC increased progressively in soils with increasing time of land use. The values of soil organic carbon (SOC) within the surface soil layer ranged from 10.1 to 29.1 g kg−1. The corresponding concentrations in deeper soil layers were entirely lower.
The increases in SOC with increasing time of land use (within the different soil depths) were investigated by fitting to 3 mathematical models, i.e., linear model, quadratic model and a logarithmic one; and the fitting parameters are presented in Table 2. Based on the highest r2 values, accumulation of SOC seemed to follow the quadratic model with two slopes. The positive slope, of relatively high values, probably indicates accumulation of organic C in soils, while the negative one, of relatively lower values, refers to a decomposition phenomenon of the organic carbon in soil.
Organic carbon pools
The different pools of SOC were a matter of concern in the current study taking into consideration the variations that occurred only within the uppermost soil layers of 0–10 cm soil depth. Three carbon pools, namely, water soluble C, hot water C and soil biomass C besides the residual one, were considered. The values of the different pools ranged in the investigated soils from 1.41 to 3.26 mg kg−1 for the WSC, 1.69 to 3.81 mg kg−1 for the HWC and 2.60 to 5.16 mg kg−1 for the MBC (Fig. 3A). These pools of SOC increased gradually in soils with increasing inputs through sewage effluent; however, their concentrations seemed to be slight comparable to the residual SOC whose content increased in soil with increasing time of cropping (Fig. 3B).
Labile versus non-labile C fractions
Labile (active C) and non-labile (stable) carbon fractions have proven to be suitable tools for classifying SOC (Weigel et al. 2011). Values of the active carbon (AC) in the studied soils ranged from 4.37 to 6.34 mg kg−1 (Fig. 4A).
Active versus passive organic carbon (total-active) fractions were then considered in calculating the carbon management index to investigate the changes that took place among the different soil locations of study. The calculated CMI ranged from 101.83 to 139.79 (unit less) with an average value of 120.81 (Fig. 4B). These values probably indicate that carbon management index improved in soils with increasing organic inputs through sewage effluent.
Mutual relations among pools of SOC and their relation to soil physical and chemical characteristics
Table 3 reveals that SOC increased significantly and positively with increasing time of land use. The different C pools correlated significantly with SOC. Moreover, these different pools significantly correlated with each other. The active C fraction was significantly correlated with the different C pools, i.e., water-soluble C, hot water C and microbial biomass C besides the residual organic C. It is worthy to mention that the residual fraction of the organic C was highly correlated with the passive organic C (P = 1.000). On the other hand, soil bulk density and the hydraulic conductivity correlated significantly and negatively with the different C pools in soil, respectively. Also, the increases in SOC and its fractions or pools significantly correlated with each of the silt and clay contents in soils. In this concern, the soil of cropping history > 80 years recorded the highest increase in SOC (2.7-folds higher than the control soil of cropping history < 5 years). On the other hand, the increases that occurred in both the clay and silt contents in this soil were 19.5- and 30.2-folds, respectively, higher than the control.
There were 4 textural classes, i.e., sand, sandy loam, loamy sand and loam for soils within the area of study (Fig. 5). The original soil texture is thought to be sand. With time of land use, silt and clay contents increased in soil to comprise heavier textured soils. Thus, the texture of the surface top soils turned out to be a loam within a land use period of 50 years or more. Cation exchange capacity (CEC) of the investigated soils also correlated positively and significantly with the different C pools in soil besides the total content of soil organic carbon. As shown in Fig. 3, CEC values ranged from 3.22 to 24.75 cmolckg−1 soil. The increases in CEC are supposed to comprise 4 phases along the cropping history. The first and the fourth phases showed slight and, at the same time, steady increases. The second phase revealed progressive increases in soil CEC within the period extending from 30 to 40 years of cropping followed by a third phase of relatively stable values of soil CEC, probably indicating high soil buffering action.
Discussion
Organic matter increased progressively and significantly in the investigated arid soils with increasing cropping history. Such increases were pronounced mainly within the surface (0–10 cm) layer while decreasing considerably in deeper soil layers. Probably, the plant residues from the previous seasons accounted for such increases (Kögel-Knabner 2002), besides the raw sewage effluent (rich in suspended organic materials) which was used as a sole source for irrigating the soils (El-Motaium 2000; Abbas and Bassouny 2018). On the other hand, the conditions of drought were more considerable in the surface soil layers compared with the subsurface ones (Fierer et al. 2003); consequently, soil microbes and enzymes activities increased in the deeper soil layers (Madejón et al. 2009). Therefore, decomposition rates in deeper soil layers seemed to be higher. In spite of that, Paul et al. (1997) recorded that, by using 14C dating and 13C/12C measurements, the subsurface soil layers preserved more resistant organic carbon than that which exists in the surface ones. Probably, soil microorganisms, upon decomposition of organic matter mainly within the surface soil layer, built up more resistant organic C which was the byproducts of soil microbes (Rumpel and Kögel-Knabner 2011; Abdelhafez et al. 2018). Generally, the accumulation of SOC in the investigated soils followed a quadratic model with a positive slope, of relatively high values, indicating accumulation of SOC, and a negative one, of relatively low values, representing a decomposition phenomenon.
Organic carbon pools
Three carbon pools, i.e., water-soluble C, hot water C and soil biomass C besides the residual one, were considered in the current study to investigate the dynamics of soil organic carbon in Typic Torripsamment soils. Results obtained herein indicate that the values of the WSC ranged in soil from 1.41 to 3.26 mg kg−1. Although, it is only a small fraction of the SOC according to Ma et al. (2010), however, this fraction is quickly used by microbes and can reflect the turnover rate of soil organic matter (Gregorich et al. 2000). The other two C pools were a little bit higher than WSC while the residual organic carbon (ROC) seemed to be the largest organic C pool in soil. Generally, concentrations of WSC, HWC, MBC and ROC increased progressively in soil with increasing cropping history. Similar results revealed that application of sewage biosolids to soils of Poland improved significantly soil contents of water soluble carbon within a period of 1 year (Kalisz et al. 2012). It is thought that the water-soluble C pool is the metabolic potential of soil biota; consequently, MBC pool increased (Bastida et al. 2007). Soil organic carbon can also be classified into labile and non-labile carbon fractions (Weigel et al. 2011). The active carbon is the labile pool of the SOC (Blair et al. 1995). This fraction also increased progressively in the area of study with increasing cropping history; however, such concentrations seemed to be relatively low compared with the passive fraction.
Carbon management index
The labile C fraction is taken as an indicator in the monitoring programs of the arid regions (Oyonarte et al. 2007). It comprises water-soluble C (Zou et al. 2005; Sun et al. 2015), hot water C (Ghani et al. 2003; Akinsete and Nortcliff 2014) and, to some extent, microbial biomass C (Buchanan and King 1992; Sheng et al. 2015). The calculated CMI in this study ranged from 101.8 to 139.8. These values are higher than the corresponding ones calculated by Ghosh et al. (2016), which ranged from 47 to 59 at a soil depth of 0–5 cm. Nevertheless, there is no ideal value of CMI (Blair et al. 1995). Any changes in these labile fractions indicate either degradation or improvement in the management practices (Weil et al. 2003; Jinbo et al. 2006; Sodhi et al. 2009; Nahrawi et al. 2012). The CMI values obtained herein were in continuous increases indicating better soil quality (Leal et al. 2016). Accordingly, there were considerable positive changes in the management practices of SOC in the investigated farms owing to irrigation with raw sewage for a long time.
Organic carbon and soil aggregation
Since most of the variations in SOC occurred within the surface soil layer, accordingly, the different C pools, i.e., WSC, HWC, MBC and ROC, were estimated within this layer. These fractions of SOC were in continuous increases in soils with increasing cropping history (Kaur et al. 2008). Moreover, significant correlations were detected among the different C pools, i.e., WSC, HWC, MBC and ROC, and this probably indicates the existence of equilibrium among the different C pools in soil and that ROC was probably the reservoir of the other soil organic C forms. The significant correlations that exist also between the labile (used up by soil biota) and the non-labile C fractions in soil supported the existence of dynamics between the different C pools. Our findings support the assumption which indicates that the labile C is respired first and then recalcitrant or “stable” forms occurred. These results were similar to those found by Xu et al. (2011) who indicated the presence of significant positive correlations among all indicators of the organic C pools with each other. Likewise, de Souza et al. (2016) recorded positive correlations between the labile C fraction and each of the different C pools in soil. Accordingly, we accept the first hypothesis.
Implications of the organic carbon on soil characteristics
Physical and chemical characteristics of the investigated soils were also studied in relation to SOC within the surface top soil (0–10 cm). Although, RSC did not record the highest significant correlations with the investigated physical and chemical characteristics of the soil, its relative stability in soil may suggest that this fraction probably affects soil characteristics in the long run. Generally, physical and chemical characteristics of the investigated soils improved when soils were amended with sewage effluents. Such improvements were more significant with increasing time of land use. In this concern, soil bulk density of the studied soils decreased significantly with increasing SOC. This might have occurred because SOC was incorporated in the formation of soil aggregates (Balesdent et al. 2000; Six et al. 2002; Tan et al. 2007; Goebel et al. 2009; Wiesmeier et al. 2012; Farid et al. 2014; D’Acqui et al. 2017; Farid et al. 2018). On the other hand, significant correlations were detected between the different pools of organic carbon (especially the active and MBC) and the fine soil particles of silt and clay. This probably confirms that the by-products of soil biota are temporarily incorporated in the formation of the organic-mineral aggregates. Such a mechanism is thought to be responsible for soil organic carbon persistence in temperate soils (Barré et al. 2018). Moreover, the relation between SOM and soil particle size might grow with time to become a two-way relation that occurs simultaneously rather than a one-way relation between a dependent variable (SOC) and an independent one (particle size fraction). For example, soils of higher organic C content might also retain more clay and silt in soil, thus changing their textural classes. Accordingly, we accept the second assumption.
Likewise, CEC increased in the investigated soils along the cropping period following four phases of increases. The increment of CEC showed different patterns: slight increases up to 30 years, then progressive increases within the period extending from 30 to 40 years of cropping followed by a phase of relatively stable CEC values, probably indicating high soil buffering action, and finally slight increases occurred thereafter.
In conclusion, the hypothesis that SOC is physically protected against soil microbes within the soil aggregates or even chemically protected through sorption on clay minerals became not valid or at least requires more investigations to clarify such results obtained herein because this study highlighted the presence of a dynamic equilibrium among the different fractions or SOC pools. Thus, soil organic carbon acts as a potential sink of atmospheric carbon and the global carbon cycle seems to be complete.
References
Abbas MHH, Bassouny M (2018) Implications of long term irrigation with wastewater on the contents and retention kinetics of potentially toxic elements in Typic Torripsamment soils. Egypt J Soil Sci 58(3):337–357. https://doi.org/10.21608/ejss.2018.4232.1183
Abdelhafez AA, Abbas MHH, Attia TMS (2015) Environmental monitoring of heavy-metals status and human health risk assessment in the soil of Sahl El-Hessania area, Egypt. Pol J Environ Stud 24(2):459–467
Abdelhafez AA, Abbas MHH, Attia TMS, El Bably W, Mahrous SE (2018) Mineralization of organic carbon and nitrogen in semi-arid soils under organic and inorganic fertilization. Environ Technol Innov 9:243–253. https://doi.org/10.1016/j.eti.2017.12.011
Abdel-Shafy HI, Abdel-Sabour MF (2006) Wastewater reuse or irrigation on the desert sandy soil of Egypt: long-term effect. In: Hlavinek P, Kukharchyk T, Marsalek J, Mahrikova I (eds) Integrated urban water resources management. Springer Netherlands, Dordrecht, pp 301–312. https://doi.org/10.1007/1-4020-4685-5_31
Akinsete SJ, Nortcliff S (2014) Storage of total and labile soil carbon fractions under different land-use types: a laboratory incubation study. In: Hartemink AE, McSweeney K (eds) Soil carbon. Springer International Publishing, Cham, pp 197–207. https://doi.org/10.1007/978-3-319-04084-4_21
Balesdent J, Chenu C, Balabane M (2000) Relationship of soil organic matter dynamics to physical protection and tillage. Soil Tillage Res 53:215–230. https://doi.org/10.1016/S0167-1987(99)00107-5
Barré P, Quénéa K, Vidal A, Cécillon L, Christensen BT, Kätterer T, Macdonald A, Petit L, Plante AF, van Oort F, Chenu C (2018) Microbial and plant-derived compounds both contribute to persistent soil organic carbon in temperate soils. Biogeochemistry 140:81–92. https://doi.org/10.1007/s10533-018-0475-5
Bassouny MA, Abbas MHH (2019) Role of biochar in managing the irrigation water requirements of maize plants: the pyramid model signifying the soil hydro-physical and environmental markers. Egypt J Soil Sci 59(2):99–115. https://doi.org/10.21608/EJSS.2019.9990.1252
Bastida F, Moreno JL, Hernández T, García C (2007) The long-term effects of the management of a forest soil on its carbon content, microbial biomass and activity under a semi-arid climate. Appl Soil Ecol 37:53–62. https://doi.org/10.1016/j.apsoil.2007.03.010
Blair GJ, Lefory RD, Lisle L (1995) Soil carbon fractions based on their degree of oxidation, and the development of a carbon management index for agricultural systems. Aust J Agric Res 46:1459–1466. https://doi.org/10.1071/AR9951459
Bougnom BP, Thiele-Bruhn S, Ricci V, Zongo C, Piddock LJV (2019) Raw wastewater irrigation for urban agriculture in Africa increases the diversity of transferable antibiotic resistances genes in soil, including those encoding ESBLs. BioRxiv 615922. https://doi.org/10.1101/615922
Brar BS, Singh K, Dheri GS, Balwinder K (2013) Carbon sequestration and soil carbon pools in a rice–wheat cropping system: effect of long-term use of inorganic fertilizers and organic manure. Soil Tillage Res 128:30–36. https://doi.org/10.1016/j.still.2012.10.001
Buchanan M, King LD (1992) Seasonal fluctuations in soil microbial biomass carbon, phosphorus, and activity in no-till and reduced-chemical-input maize agroecosystems. Biol Fertil Soils 13:211–217. https://doi.org/10.1007/bf00340578
Casado-Vela J, Sellés S, Navarro J, Bustamante MA, Mataix J, Guerrero C, Gomez I (2006) Evaluation of composted sewage sludge as nutritional source for horticultural soils. Waste Manag 26:946–952. https://doi.org/10.1016/j.wasman.2005.07.016
Charest MH, Antoun H, Beauchamp CJ (2004) Dynamics of water-soluble carbon substances and microbial populations during the composting of de-inking paper sludge. Bioresour Technol 91:53–67. https://doi.org/10.1016/S0960-8524(03)00155-X
Chen L-WA, Verburg P, Shackelford A, Zhu D, Susfalk R, Chow JC, Watson JG (2010) Moisture effects on carbon and nitrogen emission from burning of wildland biomass. Atmos Chem Phys 10:6617–6625. https://doi.org/10.5194/acp-10-6617-2010
Chopin P, Sierra J (2019) Reduced tillage and organic amendments can offset the negative impact of climate change on soil carbon: a regional modelling study in the Caribbean. Soil Tillage Res 192:113–120. https://doi.org/10.1016/j.still.2019.05.009
D’Acqui LP, Bonetti A, Pini R, Certini G (2017) Physical protection of organic matter in minesoils assessed by low-temperature ashing (LTA). Geoderma 288:120–129. https://doi.org/10.1016/j.geoderma.2016.11.009
De Bona FD, Bayer C, Dieckow J, Bergamaschi H (2008) Soil quality assessed by carbon management index in a subtropical Acrisol subjected to tillage systems and irrigation. Aust J Soil Res 46:469–475. https://doi.org/10.1071/SR08018
de Oliveira FÉR, Oliveira JDM, Xavier FA (2016) Changes in soil organic carbon fractions in response to cover crops in an orange orchard. Rev Bras Cienc Solo 40:e0150105. https://doi.org/10.1590/18069657rbcs20150105
de Souza GP, de Figueiredo CC, de Sousa DMG (2016) Relationships between labile soil organic carbon fractions under different soil management systems. Sci Agric (Piracicaba Braz) 73:535–542. https://doi.org/10.1590/0103-9016-2015-0047
El-Hassanin AS, Labib TM, Dobal AT (1993) Potential Pb, Cd, Zn and B contamination of sandy soils after different irrigation periods with sewage effluent. Water Air Soil Pollut 66:239–249. https://doi.org/10.1007/bf00479848
El-Motaim R, Hashim ME (2009) Fate and behaviour of toxic organic pollutants in plant, soil and irradiated sewage sludge. In: Bahadir AM, Duca G (eds) The role of ecological chemistry in pollution research and sustainable development. Springer, Chistinau, pp 209–219. https://doi.org/10.1007/978-90-481-2903-4
El-Motaium RA (2000) Alleviation of environmental pollution using nuclear techniques recycling of sewage water and sludge in agriculture: a case study. Proceeding of ICEHM2000 (Cairo University, Egypt), pp. 323–332
El-Motaium RA, El-Seoud MAA (2007) Irradiated sewage sludge for production of fennel plants in sandy soil. Nutr Cycl Agroecosyst 78:133–142. https://doi.org/10.1007/s10705-006-9079-x
Farid IM, Abbas MHH, Beheiry GS, Elcossy SAE (2014) Implications of organic amendments and tillage of a sandy soil on its physical properties and C-sequestration as well as its productivity of wheat and maize grown thereon. Egypt J Soil Sci 54(2):177–194. https://doi.org/10.21608/ejss.2014.132
Farid IM, Abbas MHH, El-Ghozoli A (2018) Implications of humic, fulvic and K—humate extracted from each of compost and biogas manure as well as their teas on faba bean plants grown on Typic Torripsamments and emissions of soil CO2. Egypt J Soil Sci 58(3):275–298. https://doi.org/10.21608/ejss.2018.4232.1183
Farid I, Abbas MHH, Bassouny M, Gameel A, Abbas HH (2019) Indirect impacts of irrigation with low quality water on the environmental safety. Egypt J Soil Sci. https://doi.org/10.21608/ejss.2019.15434.1294
Fierer N, Allen AS, Schimel JP, Holden PA (2003) Controls on microbial CO2 production: a comparison of surface and subsurface soil horizons. Glob Chang Biol 9:1322–1332. https://doi.org/10.1046/j.1365-2486.2003.00663.x
Francaviglia R, Di Bene C, Farina R, Salvati L, Vicente-Vicente JL (2019) Assessing “4 per 1000” soil organic carbon storage rates under Mediterranean climate: a comprehensive data analysis. Mitig Adapt Strateg Glob Chang 24:795–818. https://doi.org/10.1007/s11027-018-9832-x
Galvis A, Jaramillo MF, van der Steen P, Gijzen HJ (2018) Financial aspects of reclaimed wastewater irrigation in three sugarcane production areas in the Upper Cauca river Basin, Colombia. Agric Water Manag 209:102–110. https://doi.org/10.1016/j.agwat.2018.07.019
Gatta G, Gagliardi A, Disciglio G, Lonigro A, Francavilla M, Tarantino E, Giuliani MM (2018) Irrigation with treated municipal wastewater on artichoke crop: assessment of soil and yield heavy metal content and human risk. Water 10(3):255. https://doi.org/10.3390/w10030255
Ghani A, Dexter M, Perrott KW (2003) Hot-water extractable carbon in soils: a sensitive measurement for determining impacts of fertilisation, grazing and cultivation. Soil Biol Biochem 35:1231–1243. https://doi.org/10.1016/S0038-0717(03)00186-X
Ghosh BN, Meena VS, Alam NM, Dogra P, Bhattacharyya R, Sharma NK, Mishra PK (2016) Impact of conservation practices on soil aggregation and the carbon management index after seven years of maize–wheat cropping system in the Indian Himalayas. Agric Ecosyst Environ 216:247–257. https://doi.org/10.1016/j.agee.2015.09.038
Goebel MO, Woche SK, Bachmann J (2009) Do soil aggregates really protect encapsulated organic matter against microbial decomposition? Biologia 64:443–448. https://doi.org/10.2478/s11756-009-0065-z
Gregorich EG, Liang BC, Drury CF, Mackenzie AF, McGill WB (2000) Elucidation of the source and turnover of water soluble and microbial biomass carbon in agricultural soils. Soil Biol Biochem 32:581–587. https://doi.org/10.1016/S0038-0717(99)00146-7
Jinbo Z, Changchun S, Wenyan Y (2006) Land use effects on the distribution of labile organic carbon fractions through soil profiles. Soil Sci Soc Am J 70:660–667. https://doi.org/10.2136/sssaj2005.0007
Kalisz B, Lachacz A, Glazewski R, Klasa A (2012) Effect of municipal sewage sludge under Salix plantations on dissolved soil organic carbon pools. Arch Environ Prot 38:87–97. https://doi.org/10.2478/v10265-012-0030-8
Kaur T, Brar BS, Dhillon NS (2008) Soil organic matter dynamics as affected by long-term use of organic and inorganic fertilizers under maize–wheat cropping system. Nutr Cycl Agroecosyst 81:59–69. https://doi.org/10.1007/s10705-007-9152-0
Kirschbaum MUF (2000) Will changes in soil organic carbon act as a positive or negative feedback on global warming? Biogeochemistry 48:21–51. https://doi.org/10.1023/a:1006238902976
Klute A (1986) Part 1. Physical and mineralogical methods. In: Part 1. Physical and mineralogical methods, 2nd edn. ASA-SSSA-Agronomy, Madison
Kögel-Knabner I (2002) The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter. Soil Biol Biochem 34:139–162. https://doi.org/10.1016/S0038-0717(01)00158-4
Landgraf D, Leinweber P, Makeschin F (2006) Cold and hot water–extractable organic matter as indicators of litter decomposition in forest soils. J Plant Nutr Soil Sci 169:76–82. https://doi.org/10.1002/jpln.200521711
Leal OA, Castilhos RMV, Pinto LFS, Pauletto EA, Lemes ES, Kunde RJ (2016) Initial recovery of organic matter of a grass-covered constructed soil after coal mining. Rev Bras Cienc Solo 40:1–16. https://doi.org/10.1590/18069657rbcs20150384
Ma XZ, Chen LJ, Chen ZH, Wu ZJ, Zhang LL, Zhang YL (2010) Soil glycosidase activities and water soluble organic carbon under different land use types. R.C. Suelo Nutr Veg 10:93–101. https://doi.org/10.4067/S0718-27912010000200001
Madejón E, Murillo JM, Moreno F, López MV, Arrue JL, Alvaro-Fuentes J, Cantero C (2009) Effect of long-term conservation tillage on soil biochemical properties in Mediterranean Spanish areas. Soil Tillage Res 105:55–62. https://doi.org/10.1016/j.still.2009.05.007
Maia SMF, Gonzaga GBM, Silva LKDS, Lyra GB, Gomes TCDA (2019) Soil organic carbon temperature sensitivity of different soil types and land use systems in the Brazilian semi-arid region. Soil Use Manag 35:433–442. https://doi.org/10.1111/sum.12508
Mlih R, Bol R, Amelung W, Brahim N (2016) Soil organic matter amendments in date palm groves of the middle eastern and north African region: a mini-review. J Arid Land 8:77–92. https://doi.org/10.1007/s40333-015-0054-8
Moinet GYK, Hunt JE, Kirschbaum MUF, Morcom CP, Midwood AJ, Millard P (2018) The temperature sensitivity of soil organic matter decomposition is constrained by microbial access to substrates. Soil Biol Biochem 116:333–339. https://doi.org/10.1016/j.soilbio.2017.10.031
Nahrawi H, Husni MHA, Radziah O (2012) Labile carbon and carbon management index in peat planted with various crops. Commun Soil Sci Plant 43:1647–1657. https://doi.org/10.1080/00103624.2012.681736
Nieder R, Benbi DK (2008) Carbon and nitrogen in the terrestrial environment. Springer Netherlands, Dordrecht. https://doi.org/10.1007/978-1-4020-8433-1
Oyonarte C, Mingorance MD, Durante P, Piñero G, Barahona E (2007) Indicators of change in the organic matter in arid soils. Sci Total Environ 378:133–137. https://doi.org/10.1016/j.scitotenv.2007.01.039
Page AL, Miller RH, Keeney DR (1982) Methods of soil analysis part 2-chemical and microbiological properties. Part II. ASA-SSSA. Agronomy, Madison
Paul EA, Follett RF, Leavitt SW, Halvorson A, Peterson GA, Lyon DJ (1997) Radiocarbon dating for determination of soil organic matter pool sizes and dynamics. Soil Sci Soc Am J 61:1058–1067. https://doi.org/10.2136/sssaj1997.03615995006100040011x
Peregrina F, Pérez-Álvarez EP, Colina M, García-Escudero E (2012) Cover crops and tillage influence soil organic matter and nitrogen availability in a semi-arid vineyard. Arch Agron Soil Sci 58:SS95–SS102. https://doi.org/10.1080/03650340.2011.648182
Post WM, Kwon KC (2000) Soil carbon sequestration and land use change: processes and potential. Glob Chang Biol 6:317–327. https://doi.org/10.1016/10.1046/j.1365-2486.2000.00308.x
Rodeghiero M, Heinemeyer A, Schrumpf M, Bellamy P (2009) Determination of soil carbon stocks and changes. In: Kutsch WL, Bahn M, Heinemeyer A (eds) Soil carbon dynamics: an integrated methedology. Cambridge University Press, Cambridge, pp 251–256. https://doi.org/10.1017/CBO9780511711794.005
Rumpel C, Kögel-Knabner I (2011) Deep soil organic matter—a key but poorly understood component of terrestrial C cycle. Plant Soil 338:143–158. https://doi.org/10.1007/s11104-010-0391-5
Safařík IVO, Šantrůčková H (1992) Direct determination of total soil carbohydrate content. Plant Soil 143:109–114. https://doi.org/10.1007/BF00009135
Schlesinger WH, Amundson R (2019) Managing for soil carbon sequestration: let’s get realistic. Glob Chang Biol 25:386–389. https://doi.org/10.1111/gcb.14478
Schmidt MWI, Torn MS, Abiven S, Dittmar T, Guggenberger G, Janssens IA, Kleber M, Kogel-Knabner I, Lehmann J, Manning DAC, Nannipieri P, Rasse DP, Weiner S, Trumbore SE (2011) Persistence of soil organic matter as an ecosystem property. Nature 478:49–56. https://doi.org/10.1038/nature10386
Shaffer MJ, Ma L, Hansen S (2001) Introduction to simulation of carbon and nitrogen dynamics in soils. In: Hansen S, Shaffer MJ, Ma L (eds) Modeling carbon and nitrogen dynamics for soil management. CRC Press, Florida, pp 1–10. https://doi.org/10.1201/9781420032635.ch1
Sheng H, Zhou P, Zhang Y, Kuzyakov Y, Zhou Q, Ge T, Wang C (2015) Loss of labile organic carbon from subsoil due to land-use changes in subtropical China. Soil Biol Biochem 88:148–157. https://doi.org/10.1016/j.soilbio.2015.05.015
Six J, Feller C, Denef K, Ogle SM, Sa JCDM, Albrecht A (2002) Soil organic matter, biota and aggregation in temperate and tropical soils - effects of no-tillage. Agronomie 22:755–775. https://doi.org/10.1051/agro:2002043
Sodhi GPS, V B, Benbi DK (2009) Using carbon management index to assess the impact of compost application on changes in soil carbon after ten years of rice–wheat cropping. Commun Soil Sci Plant Anal 40:3491–3502. https://doi.org/10.1080/00103620903326024
Soil Survey Staff (2010) Key to soil taxonomy, 11th edn. USDA- Natural Resources Conservation Service, Washington, D.C.
SrdjanŠeremešić DM, Sekulić P, Nešić L, Ćirić V (2013) Total and hot-water extractable carbon relationship in chernozem soil under different cropping systems and land use. Šeremešić 14:1496–1504. https://doi.org/10.5513/JCEA01/14.4.1382
Strosser E (2010) Methods for determination of labile soil organic matter: an overview. J Agrobiol 27:49–60. https://doi.org/10.2478/s10146-009-0008-x
Su Y, Wang J, Peng SC, Yue ZB, Chen TH, Jin J (2010) Rice straw and sewage sludge as carbon sources for sulfate-reducing bacteria treating acid mine drainage. Huan Jing KeXue 31:1858–1863
Sun SQ, Cai HY, Chang SX, Bhatti JS (2015) Sample storage-induced changes in the quantity and quality of soil labile organic carbon. Sci Rep 5:17496. https://doi.org/10.1038/srep17496
Tan Z, Lal R, Owens L, Izaurralde RC (2007) Distribution of light and heavy fractions of soil organic carbon as related to land use and tillage practice. Soil Tillage Res 92:53–59. https://doi.org/10.1016/j.still.2006.01.003
Tarnocai C, Canadell JC, Schuur EAG, Kuhry P, Mazhitova G, Imov S (2009) Soil organic carbon pools in the northern circumpolar permafrost region. Glob Biochem Cycles 23:GB2023. https://doi.org/10.1029/2008GB003327
Tobiašová E, Barančíková G, Gömöryová E, Makovníková J, Skalský R, Halas J, Koco Š, Tarasovičová Z, Takáč J, Špaňo M (2016) Labile forms of carbon and soil aggregates. Soil Water Res 11:259–266. https://doi.org/10.17221/182/2015-SWR
Vieira FCB, Bayer C, Zanatta JA, Dieckow J, Mielniczuk J, He ZL (2007) Carbon management index based on physical fractionation of soil organic matter in an Acrisol under long-term no-till cropping systems. Soil Tillage Res 96:195–204. https://doi.org/10.1016/j.still.2007.06.007
Weigel A, Eustice T, van Antwerpen R, van Naidoo G, Schulz E (2011) Soil organic carbon (SOC) changes indicated by hot water extractable carbon (HWEC). 84th Annual Congress of the South African Sugar Technologists’ Association, 17–19 August 2011. 84, Durban, South Africa, pp 210–222
Weil RR, Islam KR, Stine MA, Gruver JB, Samson-Liebig SE (2003) Estimating active carbon for soil quality assessment: a simplified method for laboratory and field use. Am J Altern Agric 18:3–17. https://doi.org/10.1079/AJAA200228
Whalen JK, Bottomley PJ, Myrold DD (2000) Carbon and nitrogen mineralization from light- and heavy-fraction additions to soil. Soil Biol Biochem 32:1345–1352. https://doi.org/10.1016/S0038-0717(00)00040-7
Wiesmeier M, Steffens M, Mueller CW, Kölbl A, Reszkowska A, Peth S, Horn R, Kögel-Knabner I (2012) Aggregate stability and physical protection of soil organic carbon in semi-arid steppe soils. Eur J Soil Sci 63:22–31. https://doi.org/10.1111/j.1365-2389.2011.01418.x
Wiesmeier M, Urbanski L, Hobley E, Lang B, von Lützow M, Marin-Spiotta E, van Wesemael B, Rabot E, Ließ M, Garcia-Franco M, Wollschläger U, Vogel HJ, Kögel-Knabner I (2019) Soil organic carbon storage as a key function of soils - a review of drivers and indicators at various scales. Geoderma 333:149–162. https://doi.org/10.1016/j.geoderma.2018.07.026
Williams WD (1999) Salinisation: a major threat to water resources in the arid and semi-arid regions of the world. Lakes Reserv Res Manag 4:85–91. https://doi.org/10.1046/j.1440-1770.1999.00089.x
Xu M, Lou Y, Sun X, Wang W, Baniyamuddin M, Zhao K (2011) Soil organic carbon active fractions as early indicators for total carbon change under straw incorporation. Biol Fertil Soils 47:745–752. https://doi.org/10.1007/s00374-011-0579-8
Zhang Y, Shen Y (2019) Wastewater irrigation: past, present, and future. WIREs Water 6:e1234. https://doi.org/10.1002/wat2.1234
Zhao Z, Wei X, Wang X, Ma T, Huang L, Gao H, Fan J, Li X, Jia X (2019) Concentration and mineralization of organic carbon in forest soils along a climatic gradient. For Ecol Manag 432:246–255. https://doi.org/10.1016/j.foreco.2018.09.026
Zhou Y, Hartemink AE, Shi Z, Liang Z, Lu Y (2019) Land use and climate change effects on soil organic carbon in North and Northeast China. Sci Total Environ 647:1230–1238. https://doi.org/10.1016/j.scitotenv.2018.08.016
Zolti A, Green SJ, Mordechay EB, Hadar Y, Minz D (2019) Root microbiome response to treated wastewater irrigation. Sci Total Environ 655:899–907. https://doi.org/10.1016/j.scitotenv.2018.11.251
Zou XM, Ruan HH, Fu Y, Yang XD, Sha LQ (2005) Estimating soil labile organic carbon and potential turnover rates using a sequential fumigation–incubation procedure. Soil Biol Biochem 37:1923–1928. https://doi.org/10.1016/j.soilbio.2005.02.028
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Kitae Baek
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elcossy, S.A.E., Abbas, M.H.H., Farid, I.M. et al. Dynamics of soil organic carbon in Typic Torripsamment soils irrigated with raw effluent sewage water. Environ Sci Pollut Res 27, 8188–8198 (2020). https://doi.org/10.1007/s11356-019-07526-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-07526-4