Abstract
The aim of the present study was to investigate whether curcumin (CUR) can ameliorate cadmium-induced reproductive toxicity and its mechanism. A total of 48 male mice were equally divided into 4 groups: control, CdCl2 (2 mg/kg, intraperitoneally inject) curcumin (50 mg/kg, intraperitoneally inject), co-treatment with curcumin (50 mg/kg), and CdCl2 (2 mg/kg) for 10 days. The results demonstrated that CdCl2 reduces sperm motility, decreases the sperm density and serum testosterone content, and significantly improves the rate of sperm deformity. CdCl2 increased the level of testicular total superoxide dismutase (T-SOD), glutathione peroxidase (GSH-Px) activity, and glutathione (GSH), and CdCl2 declined the level of malondialdehyde (MDA). However, the semen quality of the mice in the curcumin intervention group was improved. Moreover, the testosterone content and antioxidant capacity were increased. In the Cd group mice, the expression of testicular Nrf2, as well as the mRNA and protein expressions of the downstream target molecules, glutathione peroxidase (GSH-Px), and γ-glutamylcysteine synthetase (γ-GCS) of Nrf2 declined, while the above genetic expressions elevated significantly in the curcumin intervention group. Our results suggested that curcumin could protect against Cd-induced testicular injury via activating the Nrf2/ARE signaling pathway.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cadmium (Cd) accumulates in the ecosystem and enters the food chain via polluted soil and water environment. Due to the prolonged biological half-life, long-term exposure to Cd can damage the liver ( Zhang et al. 2018), kidney (Zang et al. 2018; Ge J et al. 2019), bone (Taha et al. 2018), lung (El-Ebiary et al. 2016), cardiovascular system (Ghosh 2018), immune system (Alkharashi et al. 2017), and reproductive system (Zhao et al. 2017; Zhao et al. 2015; De Souza Predes et al. 2010). The testicular injury is one of the main toxic reactions of Cd poisoning. Cd can destroy the blood-testis barrier of testes (Siu et al. 2009; El-Demerdash et al. 2004) and induce the germ cell apoptosis, testicular edema, hemorrhage, necrosis, and infertility (Tremellen 2008; Turner and Lysiak 2008; Niknafs et al. 2015).
Previous studies have shown that natural antioxidants such as quercetin (Jia et al. 2011), sulforaphane (de Figueiredo et al. 2015), grape seed extract (Long et al. 2016; Sönmez and Tascioglu 2016), resveratrol (Eyb et al. 2006), saffron (Asadi et al. 2013), Crocus sativus L. (Yari et al. 2016), and onion and garlic extracts (Ola-Mudathir et al. 2008) can reduce the reproductive toxicity of Cd and reproductive damage. The antioxidants and the antioxidant defense systems play a critical role in exerting a protective effect on the male reproductive organs from oxidative damage. Curcumin is a biologically active substance extracted from the roots of Zingiberaceae, such as turmeric. It has a variety of pharmacological properties, including anti-cancer, anti-inflammatory, anti-bacterial, anti-viral, anti-fungal, and antioxidant effects (Araujo and Leon 2001; Aggarwal et al. 2007; Ciftci et al. 2010). Previous studies have also confirmed that curcumin can prevent the peroxidation of the sperm and testicular tissue, enhance the sperm motility, decline the sperm deformity, reduce the histological damage of the testis (Abd El-Fattah et al. 2016; Farombi et al. 2007), and inhibit the Cd-induced apoptosis in the testicular tissue in rats (Aktas et al. 2012). However, the specific mechanism is yet unknown.
The transcription factor NF-E2-related factor 2 (Nrf2) primarily regulates the cytotoxic and redox states (Niture et al. 2010). Curcumin reduces the disease-induced damage by inducing Nrf2 expression. It also reduces the brain edema and neurological dysfunction after ischemic-reperfusion injury (I/R) by upregulating the Nrf2 expression (Li et al. 2013). Furthermore, curcumin can activate the Nrf2 signals and, as a result, improve the antioxidant levels in the kidneys of rats with type 2 diabetes (Kim et al. 2016), increase the activation of Nrf2, enhance the activity of phase II antioxidant enzymes, and inhibit the lymphoma of the liver in mice (Das and Vinayak 2015). In addition, the level of Nrf2 mRNA expression is significantly lower in human males with low sperm motility (Chen et al. 2012), and the genetic deletion of Nrf2 increase disrupts spermatogenesis of testis (Nakamura et al. 2010).
Therefore, in this study, we investigated whether curcumin can alleviate the Cd-induced reproductive toxicity by activating the Nrf2/ARE pathway. Artificially established Cd-poisoned mouse models were intervened with curcumin to investigate whether the protective effects of curcumin on Cd-induced reproduction damage were associated with the activation of the Nrf2 signaling pathway.
Methods
Animals
Kunming mice (aged 9 weeks and weighed 45 ± 2 g) were purchased from China Medical University (Shenyang, China). The mice were placed in an isolated environment with a temperature of 22–24 °C and humidity of 40–60% in a 12-h light/dark cycle. The mice were provided with clean drinking water and chow. The mice were adaptively fed for 1 week before the final experiment that was conducted according to the European Community Council Directive on November 24, 1986 (86/609/EEC) in accordance with the requirements of the Ethics Committee of Shenyang Agricultural University (China) for the use of experimental animals (Permit No. 264 SYXK<Liao>2011-0001, 20 October, 2011).
Materials
For the current experiment, curcumin was purchased from Shanghai Haoyang Biotechnology Co., Ltd. (Shanghai, China). CdCl2 was purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China) with a purity ≥ 99%.
Experimental methods
A total of 48 male mice were equally divided into 4 groups. Control group: mice were intraperitoneally injected with double distilled water at 2 mg/kg. Cd group: mice were intraperitoneally injected with CdCl2 at 2 mg/kg (10% of LD50). CUR group: mice were intraperitoneally injected with curcumin at 50 mg/kg. This dosage scheme was chosen according to the related previous studies by Eyb et al. (2006). Cd + CUR group: mice were intraperitoneally injected with CdCl2 at 2 mg/kg together with curcumin at 50 mg/kg for 10 days consecutively. After the mice were injected with CUR solution for 4 h, they were injected with CdCl2 solution. The dose of 0.05 mL/10 g was based on the body weight. The mice were weighed once every 5 days, and the dose of the drugs was adjusted accordingly. Subsequently, the mice were sacrificed after anesthesia with ether. Blood samples were collected, and serum was separated and stored at − 20 °C. The epididymis was separated, and semen quality was tested immediately. The testicular tissues were stored at − 80 °C for later use. The method of sample collection was as that described previously (Oguzturk et al. 2012).
Semen quality parameters test
The sperm motility was measured. The epididymis was placed in 2 mL physiological saline at 37 °C to prepare a sperm suspension. The number of sperms (200 sperms) moving in a straight line was counted by a blood cell counting plate preheated at 37 °C. The concentration of sperm was determined as follows: 100 μL of the suspension was placed in a 60 °C water bath to kill the sperm, followed by counting using a blood cell counting plate. The sperm deformity rate was determined as follows: 500 μL of the above sperm suspension was stained for 1 min with 50 μL of 2% eosin solution, followed by smearing and fixing with methanol. The deformity of the sperm head or tail was observed under a microscope. A total of 1000 sperms were observed in each mouse. This experiment was based on the method described previously with some modifications (Ciftci et al. 2012).
Serum testosterone test
Serum testosterone was assessed by radioimmunoassay (RIA). The testosterone 125I radioimmunoassay kit (Atomic High-Tech Co., Ltd, Beijing, China) was used according to the protocol of the manufacturer. The radioactivity was measured using an automated γ-counter.
Oxidation and antioxidant test
In this study, the antioxidant capacity of the testicular tissue was evaluated by analyzing the T-SOD and GSH-Px activities as well as the GSH content. The degree of lipid peroxidation in the testis of mice was determined by detecting the MDA content. T-SOD, GSH-Px, MDA, and GSH kits (Nanjing Jiancheng Institute of Biotechnology, Nanjing, China) were used. All the operations were conducted based on the requirements of the kit manufacturers.
Pathological test
The testicular samples of the mice were fixed with 10% neutral formalin solution, followed by routine treatment and paraffin embedding. Subsequently, the testicular samples were stained with hematoxylin and eosin (HE) and observed using an optical microscope.
RT-PCR analysis
Total RNA was extracted from the testicular tissue of mice using TRIzol reagent. The cDNA was extracted from 1 μg of total RNA according to the RNA PCR kit (Sangon Biotech Institute Co., Ltd, Shanghai, China). Primers (Nrf2: NM_010902.3 GSH-Px: X03920.1 γ-GCS: U85414.1 β-actin: BC138614.1) were synthesized by Sangon Biotech as shown in Table 1. The specific operation of real-time PCR was based on our previous report (Long et al. 2017). The gene expression (mRNA transcript levels) was expressed relative to the geometric mean of the housekeeping gene (β-actin) using the Ct method. The 2−△△CT method was used for real-time PCR data analysis.
Western blotting
One hundred milligrams of testicular tissues was homogenized using RIPA lysate. The supernatant was obtained by centrifugation of the lysate at 12000×g for 10 min at 4 °C. The protein concentration was estimated by the BCA protein assay kit (Nanjing Jiancheng Institute of Biotechnology, Nanjing, China). The testis protein was separated by SDS-PAGE and transferred to the PVDF membrane that was blocked with 5% skimmed milk for 1 h at room temperature. Then, the membrane was probed overnight at 4 °C using the primary antibodies against Nrf2, GSH-Px, γ-GCS, and β-actin (Santa Fe, NM, USA). Subsequently, the membrane was incubated with the corresponding secondary antibody for 2 h at room temperature (Beijing Solarbio Science & Technology Co., Ltd, Beijing, China).
Statistical analysis
The experimental data were statistically analyzed using one-way ANOVA of the SPSS 17.0 statistical software (IBM, Almon, NY, USA) and Excel software. All the data were expressed as mean ± standard error (mean ± SD). A P value < 0.05 indicated a difference, and a P value < 0.01 indicated a significant difference.
Results
Analysis of semen quality
Cd significantly reduced the semen quality in mice, while curcumin alone did not exhibit any effect, suggesting that curcumin increased the semen quality in Cd-contaminated mice. Table 2 demonstrated that the sperm motility and density of mice in the Cd group were significantly lower (P < 0.01), while the sperm deformity rate was significantly higher (P < 0.01) as compared with the control group (Fig. 1 and Table 1). However, no significant difference was detected in the sperm motility, sperm density, and sperm deformity between the CUR group and the control group (P > 0.05). Compared with the Cd group, the motility and concentration of sperm in the mice in the Cd (2 mg/kg) + CUR (50 mg/kg) group increased significantly (P < 0.01), while the sperm deformity rate declined significantly (P < 0.01).
Analysis of serum testosterone
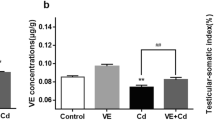
Figure 2 shows that Cd decreased the concentration of serum testosterone (P < 0.05) as compared with the control group, suggesting that curcumin can significantly increase the serum testosterone concentration (P < 0.01). Compared with the Cd group, concentration of serum testosterone in mice in the CdCl2 (2 mg/kg) + CUR (50 mg/kg) group increased significantly (P < 0.01).
Testicular antioxidant and oxidative damage results
According to Table 3, GSH-PX and T-SOD activities, as well as the GSH content, were significantly lower (P < 0.01, P < 0.05), while the MDA content was significantly higher than that of the control group (P < 0.01). In the CUR group, testis GSH-Px and T-SOD activities, as well as GSH content, were significantly higher (P < 0.05), while the MDA content was significantly lower than that of the control group (P < 0.05). In the CdCl2 (2 mg/kg) + CUR (50 mg/kg) group, the testicular antioxidant capacity was significantly higher (P < 0.05), while the MDA content was lower than that of the Cd group (2 mg/kg) (P < 0.05).
Histological changes in the testis
HE-stained testicular tissues of mice in different groups were observed. After HE staining, the nucleus of spermatogenic cells was stained blue, and the cytoplasm was stained red (Fig. 3). The interstitial cells and mature spermatozoa of the testes could be clearly distinguished from the slices. In addition, CdCl2 was found to significantly reduce the number of spermatogenic cells and mature spermatozoa in the seminiferous tubules as compared with the control group. Moreover, the number of Leydig cells was declined, the structure of the seminiferous tubules was destroyed, and the gap between the seminiferous tubules enlarged significantly (Fig. 3a–d). No significant difference was detected between the curcumin and control groups (Fig. 3e, f). Compared with the Cd group, the spermatogenic cells and mature spermatozoa in the testicular seminiferous tubules in the CUR + Cd group elevated significantly, the structure of the seminiferous tubules was intact, the Leydig cells were high in number, and the tubules were closely related (Fig. 3g, h).
Histopathological changes in the testis of mice. a, b Control group (100×, 400×). c, d Testicular tissue in the Cd group (100×, 400×). e, f Testicular tissue of the curcumin group (100×, 400×). g. h Testicular tissue of the CUR + Cd group (100×, 400×). SC, various levels of spermatogenic cells; MS, mature sperm; ST, seminiferous tubule; LC, interstitial cells; TM, testicular stroma
Expression levels of Nrf2, GSH-Px, and γ-GCS mRNA
The effects of curcumin on the expression of the related genes in the Nrf2 signaling pathway are shown in Fig. 4. Compared with the control group, the mRNA expression of Nrf2, GSH-Px, and γ-GCS in the Cd group decreased significantly (P < 0.05, P < 0.01). Conversely, the administration of curcumin alone significantly increased the mRNA expression of Nrf2, GSH-Px, and γ-GCS. Compared with the Cd group (2 mg/kg), the mRNA expression of Nrf2, GSH-Px, and γ-GCS in the CdCl2 (2 mg/kg) + CUR (50 mg/kg) group declined significantly (P < 0.05, P < 0.01).
Expression of Nrf2, GSH-Px, and γ-GCS proteins
Compared with the control group, the protein expressions of Nrf2 together with the downstream target genes γ-GCS and GSH-Px decreased significantly after addition of Cd (P < 0.05, P < 0.01) (Fig. 4). Treatment with curcumin alone can significantly increase the expression levels of Nrf2, γ-GCS, and GSH-Px proteins in the mouse testes. Compared with the Cd group, the expression of these proteins in the curcumin intervention group elevated significantly (P < 0.05, P < 0.01).
Discussion
In the present study, mice were treated with CdCl2 (2.0 mg/kg equivalent to ID50 10%) and curcumin (50 mg/kg). Compared with the control group, the concentration of serum testosterone and sperm density together with sperm motility in the epididymis of the Cd group declined significantly (P < 0.01), while the sperm deformity rate in the epididymis elevated significantly (P < 0.01). After curcumin treatment, serum testosterone concentration, sperm density, and sperm motility were significantly higher (P < 0.01), while the epididymal sperm deformity rate was significantly lower as compared with the Cd group. The HE staining of testicular tissues further confirmed our experimental results. In the Cd group, the gap between the spermatogenic tubules in the testis of mice enlarged significantly, the number of mesenchymal cells reduced significantly, and the number of spermatogenic cells and sperms in the seminiferous tubules declined significantly (Fig. 3a, b vs. Fig. 3c, d). However, after intervention with curcumin, the above changes improved significantly. The spermatogenic tubules were closely associated, and the seminiferous tubules were structurally intact. The number of spermatogenic cells, sperms, and interstitial cells were significantly higher than that in the Cd group (Fig. 3g, h), which was in agreement with the result from a previous study (Oguzturk et al. 2012), suggesting that the experimental models of reproductive intervention in mice can be used in subsequent research.
Furthermore, the detection of the antioxidant functions of the testicular tissues in the Cd group showed the decreased activity of SOD and GSH-Px, and the content of GSH decreased as well. As a major antioxidant enzyme, the decrease suggested the occurrence of oxidative damage in the testicular tissue. Thus, we speculated that the significant increase in the MDA content and the occurrence of lipid peroxidation in the testicular tissue were confirmed by detecting the changes of the MDA content in the testicular tissue. After treatment with curcumin, activities of SOD and GSH-Px increased, the content of GSH increased, and the content of MDA decreased, suggesting that curcumin can resist the Cd-induced oxidative damage. GSH is a tripeptide compound composed of glutamic acid, cysteine, and glycine. GSH-Px is a water-soluble tetrameric protein, and the active center of GSH-Px is selenocysteine (Juniper and Bertin 2013). The thiol group on cysteine is the active group of GSH and GSH-Px, and Cd has a strong affinity to sulfhydryl groups, which reduces the toxicity of Cd by combining with the thiol group on cysteine (Furia et al. 2013). However, the testicular GSH content and GSH-Px activity of mice treated by curcumin are increased significantly, indicating that curcumin can reduce the toxicity of Cd by increasing the content of GSH and activities of GSH-Px in the testicular tissue.
The Nrf2/ARE signaling pathway is a critical endogenous antioxidant system that plays a major role (Chen et al. 2012). For example, the knockout of the transcription factor Nrf2 mice interferes with sperm production (Nakamura et al. 2010). Nrf2 is resistant to heat stress-induced oxidative damage in mice testis (Li et al. 2013). The expression of Nrf2 in testicular tissue of 4-month-old mice with type 1 and type 2 diabetes declines significantly (Wang et al. 2014; Jiang et al. 2014). The current study found that Nrf2 mRNA and protein of the testicular tissues in the Cd group decreased. Moreover, the mRNA and protein expressions of GSH-Px and γ-GCS regulated by Nrf2 also decreased significantly (P < 0.05, P < 0.01) (Figs. 4 and 5). However, the expression of these genes in the mouse testicular tissue in the curcumin intervention group increased significantly (P < 0.05, P < 0.01), suggesting that curcumin can improve the endogenous antioxidant capacity of testicular tissues. The putative mechanism might be ascribed to the Cd-induced lipid peroxidation in testicular tissue (increased MDA content), causing pathological damage to the testicular tissue and inhibiting its endogenous antioxidant capacity. The mRNA and protein expression of Nrf2, GSH-Px, and γ-GCS in the testicular tissues decreased, thereby leading to reduced generation and utilization of testicular tissue GSH and accelerating the damage of the testicular tissues. However, curcumin translocated into the nucleus by activating Nrf2, which in turn, positively regulated the mRNA and protein expression of GSH-Px and γ-GCS and improved the production and utilization of GSH, thereby alleviating the Cd-induced reproductive toxicity in mice. GSH-Px and γ-GCS are important antioxidant enzymes which are regulated by Nrf2/ARE signaling pathway. These two antioxidant enzymes are expressed when Nrf2 binds to Maf protein to form heterodimer and then binding with ARE in the nucleus. Our results showed that the total Nrf2 expression of the testis tissue in the curcumin group was higher than that in the cadmium group; these results indicated that the translocation of Nrf2 into the nucleus is increased, but the specific mechanism needs to be further studied.
Effects of CUR on the Cd-induced expression of Nrf2, GSH-Px, and γ-GCS proteins in mice testis. a Nrf2. b GSH-Px. c γ-GCS. Compared with the control group, the difference was significant (aP < 0.05) or statistically significant (bP < 0.01). Compared with the Cd group, the difference was significant (cP < 0.05) or statistically significant (dP < 0.01). The mean ± SD was calculated for the 4 mice in each group
In summary, curcumin can counteract the cadmium-induced reproductive toxicity of mice by activating the Nrf2/ARE signaling pathway, thereby alleviating oxidative damage. The current study provided a novel idea for exploring the use of natural antioxidants against heavy metal toxicity.
Data availability statement
The data used to support the findings of this study are included within the article and readers can access the data supporting the conclusions of the study in this study.
References
Abd El-Fattah AA, Fahim AT, Sadik NA, Ali BM (2016) Resveratrol and curcumin ameliorate di-(2-ethylhexyl) phthalate induced testicular injury in rats. Gen Comp Endocrinol 225:45–54
Aggarwal BB, Sundaram C, Malani N, Ichikawa H (2007) Curcumin: the Indian solid gold. Adv Exp Med Biol 59(5):1–75
Aktas C, Kanter M, Erboğa M, Ozturk S (2012) Anti-apoptotic effects of curcumin on cadmium-induced apoptosis in rat testes. Toxicol Ind Health 28:122–130
Alkharashi NAO, Periasamy VS, Athinarayanan J, Alshatwi AA (2017) Sulforaphane mitigates cadmium-induced toxicity pattern in human peripheral blood lymphocytes and monocytes. Environ Toxicol Pharmacol 55:223–239
Araujo CC, Leon LL (2001) Biological activities of Curcuma longa L. Mem Inst Oswaldo Cruz 96:723–728
Asadi MH, Zafari F, Sarveazad A, Abbasi M, Safa M, Koruji M (2013) Saffron improves epididymal sperm parameters in rats exposed to cadmium. Nephrourol Mon 6(1):e12125
Chen K, Mai Z, Zhou Y, Gao X, Yu B (2012) Low NRF2 mRNA expression in spermatozoa from men with low sperm motility. Tohoku J Exp Med 228:259–266
Ciftci O, Tanyildizi S, Godekmerdan A (2010) Protective effect of curcumin on immune system and body weight gain on rats intoxicated with 2378-tetrachlorodibenzop-dioxin (TCDD). Immunopharmacol Immunotoxicol 32:99–104
Ciftci O, Aydin M, Ozdemir I, Vardi N (2012) Quercetin prevents 2378-tetrachlorodibenzo-p-dioxin-induced-testicular damage in rats. Andrologia. 44(3):164–173
Das L, Vinayak M (2015) Long term effect of curcumin in restoration of tumour suppressor p53 and phase-II antioxidant enzymes via activation of Nrf2 signalling and modulation of inflammation in prevention of cancer. PLoS One 10:e0124000
de Figueiredo SM, Binda NS, Nogueira-Machado JA, Vieira-Filho SA, Caligiorne RB (2015) The antioxidant properties of organosulfur compounds (sulforaphane). Recent Pat Endocr Metab Immune Drug Discov 9:24–39
De Souza Predes F, Diamante MA, Dolder H (2010) Testis response to low doses of cadmium in Wistar rats. Int J Exp Pathol 91:125–131
El-Demerdash FM, Yousef MI, Kedwany FS, Baghdadi HH (2004) Cadmium-induced changes in lipid peroxidationblood hematology biochemical parameters and semen quality of male rats protective role of vitamin E and beta-carotene. Food Chem Toxicol 42:1563–1571
El-Ebiary AA, El-Ghaiesh S, Hantash E, Alomar S (2016) Mitigation of cadmium-induced lung injury by Nigella sativa oil. Environ Sci Pollut Res 23(24):25356–25363
Eyb V, Kotyzova D, Koutensky J (2006) Comparative study of natural antioxidants-curcumin resveratrol and melatonin-in cadmium-induced oxidative damage in mice. Toxicology. 225:150–156
Farombi EO, Abarikwu SO, Adedara IA, Oyeyemi MO (2007) Curcumin and kolaviron ameliorate di-n-butylphthalate-induced testicular damage in rats. Basic Clin Pharmacol Toxicol 100:43–48
Furia E, Aiello D, di Donna L, Mazzotti F, Tagarelli A, Thangavel H (2013) Mass spectrometry and potentiometry studies of Pb(II)- Cd(II)-and Zn(II)-cystine complexes. Dalton Trans 43:1055–1062
Ge J, Zhang C, Sun YC, Zhang Q, Lv MW, Guo K, Li JL (2019) Cadmium exposure triggers mitochondrial dysfunction and oxidative stress in chicken (Gallus gallus) kidney via mitochondrial UPR inhibition and Nrf2-mediated antioxidant defense activation. Sci Total Environ. 689:1160–1171
Ghosh KNI (2018) Cadmium treatment induces echinocytosis DNA damage inflammation and apoptosis in cardiac tissue of albino Wistar rats. Environ Toxicol Pharmacol 59:43–52
Jia Y, Lin J, Mi Y, Zhang C (2011) Quercetin attenuates cadmium-induced oxidative damage and apoptosis in granulosa cells from chicken ovarian follicles. Reprod Toxicol 31:477–485
Jiang X, Bai Y, Zhang Z, Xin Y, Cai L (2014) Protection by sulforaphane from type 1 diabetes-induced testicular apoptosis is associated with the up-regulation of Nrf2 expression and function. Toxicol Appl Pharmacol 279:198–210
Juniper DT, Bertin G (2013) Effects of dietary selenium supplementation on tissue selenium distribution and glutathione peroxidase activity in Chinese Ring necked Pheasants. Animal. 7(4):562–570
Kim BH, Lee ES, Choi R, Nawaboot J, Lee MY, Lee EY (2016) Protective effects of curcumin on renal oxidative stress and lipid metabolism in a rat model of type 2 diabetic nephropathy. Yonsei Med J 57:664–673
Li Y, Huang Y, Piao Y, Nagaoka K, Watanabe G, Taya K, Li C (2013) Protective effects of nuclear factor erythroid 2-related factor 2 on whole body heat stress-induced oxidative damage in the mouse testis. Reprod Biol Endocrinol 11:23
Long M, Yang SH, Han JX, Li P, Zhang Y, Dong S (2016) The protective effect of grape-seed proanthocyanidin extract on oxidative damage induced by zearalenone in Kunming mice liver. Int J Mol Sci 17:E808
Long M, Yang S, Zhang Y, Li P, Han J, Dong S (2017) Proanthocyanidin protects against acute zearalenone-induced testicular oxidative damage in male mice. Environ Sci Pollut Res 24:938–946
Nakamura BN, Lawson G, Chan JY, Banuelos J, Cortes MM, Hoang YD, Luderer U (2010) Knockout of the transcription factor NRF2 disrupts spermatogenesis in an age-dependent manner. Free Radic Biol Med 49:1368–1379
Niknafs B, Salehnia M, Kamkar M (2015) Induction and determination of apoptotic and necrotic cell death by cadmium chloride in testis tissue of mouse. J Reprod Infertil 6:24–29
Niture SK, Kaspar JW, Shen J, Jaiswal AK (2010) Nrf2 signaling and cell survival. Toxicol Appl Pharmacol 244:37–42
Oguzturk H, Ciftci O, Aydin M, Timurkaan N, Beytur A, Yilmaz F (2012) Ameliorative effects of curcumin against acute cadmium toxicity on male reproductive system in rats. Andrologia. 44:243–249
Ola-Mudathir KF, Suru SM, Fafunso MA, Obioha UE, Faremi TY (2008) Protective roles of onion and garlic extracts on cadmiuminduced changes in sperm characteristics and testicular oxidative damage in rats. Food Chem Toxicol 46:3604–3611
Siu ER, Mruk DD, Porto CS, Yan CC (2009) Cadmium-induced testicular injury. Toxicol Appl Pharmacol 3:240–249
Sönmez MF, Tascioglu S (2016) Protective effects of grape seed extract on cadmium-induced testicular damage apoptosis and endothelial nitric oxide synthases expression in rats. Toxicol Ind Health 32:1486–1494
Taha MM, Mahdy-Abdallah H, Shahy EM, Ibrahim KS, Elserougy S (2018) Impact of occupational cadmium exposure on bone in sewage workers. Int J Occup Environ Health 24:101–108
Tremellen K (2008) Oxidative stress and male infertility-a clinical perspective. Hum Reprod Update 14:243–258
Turner TT, Lysiak JJ (2008) Oxidative stress: a common factor in testicular dysfunction. J Androl 29:488–498
Wang Y, Zhang Z, Guo W, Sun W, Miao X, Wu H (2014) Sulforaphane reduction of testicular apoptotic cell death in diabetic mice is associated with the upregulation of Nrf2 expression and function. Am J Physiol Endocrinol Metab 307:E14–E23
Yari A, Sarveazad A, Asadi E, Raouf Sarshoori J, Babahajian A, Amini N (2016) Efficacy of Crocus sativus L. on reduction of cadmium-induced toxicity on spermatogenesis in adult rats. Andrologia. 48:1244–1252
Zang Y, Devleesschauwer B, Bolger PM, Goodman E, Gibb HJ (2018) Global burden of late-stage chronic kidney disease resulting from dietary exposure to cadmium 2015. Environ Res 169:72–78
Zhang J, Wang Y, Fu L, Wang B, Ji YL, Wang H, Xu DX (2018) Chronic cadmium exposure induced hepatic cellular stress and inflammation in aged female mice. J Appl Toxicol. https://doi.org/10.1002/jat.3742
Zhao H, Liu W, Wang Y, Dai N, Gu J, Yuan Y, Liu X, Bian J, Liu ZP (2015) Cadmium induces apoptosis in primary rat osteoblasts through caspase and mitogen-activated protein kinase pathways. J Vet Sci 16:297–306
Zhao LL, Ru YF, Liu M, Tang JN, Zheng JF, Wu B, Gu YH, Shi HJ (2017) Reproductive effects of cadmium on sperm function and early embryonic development in vitro. PLoS One 2:1–12
Funding
This work was financially supported by the National Natural Science Foundation of China (grant no. 31772809; grant no. 31872538; grant no. 31640084; grant no. 31302152; grant no. 31201961), the National demonstration project on quality and safety of livestock and poultry products (grant no. 40010229), and the Key Research and Development Program of Shenyang (no. 17-165-3-00, no. 18-004-3-45).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The mice were adaptively fed for 1 week before the final experiment that was conducted according to the European Community Council Directive on November 24, 1986 (86/609/EEC) in accordance with the requirements of the Ethics Committee of Shenyang Agricultural University (China) for the use of experimental animals (Permit No. 264 SYXK<Liao>2011-0001, 20 October, 2011).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, SH., He, JB., Yu, LH. et al. Protective role of curcumin in cadmium-induced testicular injury in mice by attenuating oxidative stress via Nrf2/ARE pathway. Environ Sci Pollut Res 26, 34575–34583 (2019). https://doi.org/10.1007/s11356-019-06587-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-06587-9