Abstract
The hazel bolete Leccinellum pseudoscabrum (Kallenb.) Mikšík 2017 specimens and beneath soil layer (0–10 cm) have been examined on the occasion of 210Po and 210Pb activity concentrations, the nuclide bioaccumulation potential by species and distribution in fruit bodies. Mushrooms and forest soils came from six geographically distant locations in the northern and central parts of Poland. The threat to humans from 210Po and 210Pb contained in mushrooms has been also assessed. The absolute values of the 210Po radioactivity, respectively, in caps and stems of fruit bodies were in the range 0.74 ± 0.06–8.59 ± 0.36 Bq kg−1 dry biomass and from 0.81 ± 0.06–8.23 ± 0.37 Bq kg−1 dry biomass, while the values of the 210Pb radioactivity in caps and stems were in the range 0.61 ± 0.04–6.33 ± 0.22 Bq kg−1 dry biomass and 0.83 ± 0.04–4.59 ± 0.24 Bq kg−1 dry biomass, respectively. A potential related effective dose assessment showed that mushrooms L. pseudoscabrum can contribute at 0.89–10.3 μSv kg−1 db from 210Po decay and 0.42–4.37 μSv kg−1 db from 210Pb decay.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The studied isotopes of 210Po (highly radiotoxic alpha emitter) and 210Pb (beta emitter) are widely present in the environment, both daughters of uranium 238U and exist as a naturally occurring radioactive material (NORM). Their half-lives are 138.38 days for 210Po and 22.3 years for 210Pb, while effective dose coefficients for ingestion, which measure a hazard of nuclear material given by its radiotoxicity arising from its radioactive characteristic, are 1.2 μSv Bq−1 and 0.69 μSv Bq−1, respectively (Persson and Holm 2011; ICRP 2012). Their occurrence in the atmosphere is a result of 222Rn decay diffusing from the ground when the short-lived 222Rn daughters (218Po → 214Pb → 214Bi → 214Po, 210Tl) quickly attach to airborne particles, decay as 210Pb → 210Bi → 210Po, and end up in the biosphere through dry and wet deposition (Skwarzec et al. 2001). Both isotopes, 210Po and 210Pb, are also observed in abiotic media (soil, sediment, water) in varying concentrations (Skwarzec et al. 2006, 2012; Strumińska-Parulska et al. 2010; Persson and Holm 2011). Food, and to a fewer extent aspirated aerosols, are the main sources of 210Po and 210Pb in humans (Pietrzak-Flis et al. 1997). Both nuclides are trace elements while their physical and chemical properties and radiotoxicity lead to their significant contribution to the overall radiation dose from foodstuffs. Hence, 210Po is classified as one of the most important nuclides among the naturally occurring radioisotopes for which humans are exposed (Pietrzak-Flis et al. 1997; Persson and Holm 2011). 210Po and 210Pb content in foodstuffs of plant origin and mushrooms depends on the geological structure of lithosphere, climate, and agronomic conditions (Persson and Holm 2011; Strumińska-Parulska and Olszewski 2018).
Wild growing mushrooms take part in soil weathering and decomposition of the organic substrate, and cycling the elements also constitute a source of food for forest animals and human (Falandysz and Treu 2017). Mycelium of many mushrooms well take-up from the soil substrata and bioconcentrate in flesh of fruit body various chemical elements including toxic mercury, cadmium, lead, and radioactive compounds that might rebound on health of human and animals, including game animals (Škrkal et al. 2015; Steinhauser and Saey 2016; Falandysz 2017). Among the persistent radionuclides, the most often studied in mushrooms is 137Cs, the artificial gamma emitter coming from the nuclear weapons use and testing and from nuclear power plant accidents, and the second is natural 40K (Mietelski et al. 2010; Steinhauser et al. 2014; Chatterjee et al. 2017; Cocchi et al. 2017; Prand-Stritzko and Steinhauser 2017; Tuo et al. 2017). Other radionuclides and some extremely toxic, mainly alpha emitters compounds, i.e., 210Po, 210Pb, 234,238U, 228,230,232Th, 238Pu, and 239 + 240Pu, which are much more laborious to study are with much less data in mushrooms (Mietelski et al. 2002; Vaaramaa et al. 2009; Guillén and Baeza 2014; Strumińska-Parulska et al. 2016, 2017; Szymańska et al. 2018).
Hazel bolete (Leccinellum pseudoscabrum (Kallenb.) Mikšík 2017) is a common, edible, and tasty mushroom, widespread in northern and central Europe. Despite its common name, hazel bolete is most often found close to hornbeam trees, although sometimes occurs with hazel trees.
The L. pseudoscabrum has no previous data on the radionuclides accumulated in mushrooms. This study intended to investigate the 210Po and 210Pb content, their bioconcentration potential by species and distribution in fruit bodies, and possible hazard of exposure to human consumers.
Materials and methods
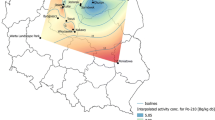
The hazel bolete (Leccinellum pseudoscabrum (Kallenb.) Mikšík 2017) fruit bodies and topsoil (0–10-cm layer) samples were collected from forests in the northern and central parts of Poland in 2000–2008. Mushrooms were collected in five places (Gołdap, Ciechocinek, Sławno, Złotów, and Borkowo) from the northern region, and one place (Bugaj) from the central region of the country, while topsoil samples from three locations (Sławno, Złotów, and Borkowo) (Fig. 1). Depending on the site, were collected from 3 to 18 composite samples of mushrooms (from 3 to 30 fruit bodies per a discrete sample). The places from the north of Poland, where the samples of L. pseudoscabrum were collected, have the acidic, leached brown soils—dystrophic autogenous soils formed from acidic rocks poor, containing 2–3% of organic matter, rich in potassium, calcium, and phosphorus. The Bugaj sampling location has podzolic soils with an organic matter content at 1.5–2% (Jadczyszyn et al. 2015).
All fruit body samples were separated for caps and stems, cut, and dried at 65 °C to constant weight. Further, the analyzed fungal materials were grinded up for a fine powder in a porcelain mortar. The topsoil samples were ridded of noticeable organisms and dried at 65 °C to stable mass as well. Next, the soil samples were sieved through a plastic sieve. Samples about 4–5 g of mushrooms and 2 g of soil substrate were taken and beforehand the radiochemical analysis, 9.5 mBq of 209Po was added as a yield tracer. All fungal materials prepared were digested using a solution of concentrated 65% HNO3 and further heated until acid has evaporated. According to soil samples, they were also digested using concentrated 65% HNO3 and HCl. Thanks to this we obtained, and were able to determine, the labile fraction of 210Po and 210Pb (not connected with the crystal structure of soil), available for living organisms. The dried residues of mushroom and soil samples were dissolved in 0.5 M HCl and a pinch of ascorbic acid was added. The polonium measurement discs were prepared by its autodeposition on pure 100% silver, the solution was transferred to PTFE vessels equipped with silver sheet bottom disc, and Po was autodeposited at 90 °C for 4 h. The activities of 209Po and 210Po were measured using an alpha spectrometer (Skwarzec 1997; Strumińska-Parulska et al. 2015, 2016). The 210Pb determination method was based on its indirect measurement via its daughter 210Po activity measurement (Strumińska-Parulska et al. 2015, 2016). All samples after the first 210Po deposition were evaporated and the dry residue was heated very strongly with 30% H2O2 added to remove potential polonium residues. Next, the samples were stored for 6 months to allow for 210Po increment from 210Pb. The deposition of 210Po on silver disc was repeated and the activities of ingrowing 210Po were measured in alpha spectrometer (Alpha Analyst S470, Canberra-Packard). The 210Po activity in analyzed samples was corrected for decay to the day of polonium deposition (time of separation 210Po from 210Pb). 210Pb activity at the time of sample collection was calculated using the simplified form of the Bateman equation (Skwarzec 1997). The chemical analysis efficiency of 210Po and 210Pb determination ranged 95–98%, while the results were given with standard deviation (SD) calculated for 95% confidence intervals.
An important question in chemical substances analysis of biota is their uptake and bioconcentration from soil, water, or air; bioaccumulation and biomagnification from the food net relationships; and distribution (fate) in a body. In the case of fungi (mushrooms), such information parameters could be described by the calculated values of bioconcentration factor (BCF) and, less frequently, the discrimination ratio (DR). The reduced assessment of soil-mushroom (through mycelium) radionuclide bioconcentration level could deliver the BCF (Eq. 1) (Gadd 2007; Strumińska-Parulska et al. 2016). Baeza and collaborators gave the discrimination ratio (DR) that could describe the different accumulations of the radionuclides in the cap + gills and stem (Eq. 2). As they wrote “a value of DR > 1 therefore indicates that the fruiting bodies accumulate this radionuclide in the cap+gills better than in the stem” (Baeza et al. 2006). The mushroom DR value might reflect the level of radionuclide uptake and its distribution into the caps and stems; however, the value of DR > 1 could also indicate the atmospheric fallout impact on radionuclides’ presence in the cap surface. In the end, a large variation of the DR values or its high values could be a result not only of the mushroom metabolism but also of the atmospheric deposition of 210Po and 210Pb onto the cap. It was not possible to differentiate these processes that impact on the DR value. The BCFs and DRs were calculated using presented formulas (Baeza et al. 2006; Strumińska-Parulska et al. 2016, 2017):
Results and discussion
210Po and 210Pb activity concentrations in hazel bolete and topsoil
The results of 210Po and 210Pb activity concentrations measured in L. pseudoscabrum and topsoil were given in Tables 1 and 2. Greater 210Po and 210Pb activity concentrations were noticed in the hazel bolete cap samples collected from the Borkowo place (Bo1): 8.59 ± 0.36 Bq 210Po kg−1 dry biomass (db) and 6.33 ± 0.22 Bq 210Pb kg−1 db than from other places. Obtained data on 210Po and 210Pb activity concentrations in caps and stems and knowledge on their biomass share within the whole fruit bodies enabled for calculations of the nuclide activity concentrations for entire mushrooms. Mushrooms collected from the location Bo1 (Borkowo) showed on the greatest activity concentration for both nuclides: 5.47 ± 1.04 Bq 210Po kg−1 db and 4.06 ± 0.63 Bq 210Pb kg−1 db than elsewhere. Also, forest topsoil from the Borkowo (Bo1) place was higher in 210Po and 210Pb, i.e., respectively, with 102 ± 2 Bq kg−1 dry mass (dm) and 100 ± 4 Bq kg−1 dm than elsewhere. The lowest 210Po and 210Pb activity concentrations were observed in hazel bolete caps from Sławno (Sł2): 0.74 ± 0.06 Bq 210Po kg−1 db and 0.63 ± 0.06 Bq 210Pb kg−1 db, while the lowest 210Po and 210Pb activity concentrations in topsoil were from the forests of the Złotów region, i.e., 18.8 ± 1.3 Bq 210Po kg−1 dm and 11.2 ± 0.4 Bq 210Pb kg−1 dm. The differences in 210Po and 210Pb content in soil, similarly to other elements, depended on its type, permeability, and fertility, and dark soil had greater sorption possibilities. It was noticed the organic soil contained three times more 210Po than mineral soil (Karanakura i in., 2000).
The basic statistical analysis showed on significant differences in 210Po or 210Pb activity concentrations among the all mushrooms sampling locations (the H test (Kruskal-Wallis) p value 0.003 for 210Po and 0.0004 for 210Pb), while not between caps and stems of fruit bodies at every place sampled (the Mann-Whitney U test p value 0.08–1 for 210Po and 0.15–1 for 210Pb). In the case of the soil samples, the 210Po and 210Pb activity concentrations were diversified according to the sampling sites (H test p value was 0.06 for 210Po and 0.06 for 210Pb). The cluster analysis has been carried out also (Ward’s method) (Fig. 2). The resulting dendrogram shows that the clusters are linked at increasing levels of dissimilarity and with a strong connection between 210Po and 210Pb in soil samples. Also, the 210Po and 210Pb content of the whole mushrooms depended on their amount in stems (Fig. 2). The principal component analyses (PCA) has been carried out to demonstrate any possible spatial variations in the radionuclide activity concentrations between the Sławno, Złotów, and Borkowo places (Fig. 3). The analysis was based on the correlation matrix, and a fiducial significance level of p < 0.05 was chosen. The PCA data uncovered that 90.45% of information regarding the radionuclide compositional variability for three places could be described by three varifactors. The first varifactor (V1) described 51.26% of the total variance and loaded heavily on the positively correlated variables, describing 210Po and 210Pb in stems and whole mushrooms (loadings were 0.965, 0.956, 0.859, and 0.782, respectively). The second varifactor (V2) was loaded primarily by positively correlated 210Po and 210Pb in soil (loadings were 0.726. and 0.906, respectively) and explained 20.33% of the total variance (Fig. 3). Both analyses (cluster and PCA) showed that 210Po concentration in the soil samples related basically to 210Pb activity concentration in soil. The activity concentrations of 210Po and 210Pb in analyzed hazel bolete mushrooms were mostly similar to the other studies on genus Leccinum (e.g., foxy bolete, orange oak bolete, slate bolete, red-capped scaber) (Vaaramaa et al. 2009; Strumińska-Parulska et al. 2016; Szymańska et al. 2018), although much lower than determined in birch bolete (Leccinum scabrum) and orange birch bolete (Leccinum versipelle) collected in Norway (Gwynn et al. 2013).
Although Guillén and Baeza (2014) stated the most important feature in polonium accumulation in mushrooms was its species, our results confirmed previous observations (confirmed by inter-species studies)—the most important aspects in 210Po and 210Pb distribution are local climate and geological conditions, e.g., atmospheric fallout, geological structure, natural radioactivity (Vaaramaa et al. 2009; Gwynn et al. 2013; Strumińska-Parulska et al. 2016; Strumińska-Parulska and Olszewski 2018; Szymańska et al. 2018). Besides, in the forest area, the trees might decrease the influence of wet and dry deposition on soils and mushrooms (Kirchner and Daillant 1998; Strumińska-Parulska et al. 2017).
210Po and 210Pb BCFs and DRs
On the ground of 210Po and 210Pb activity concentrations in mushrooms and topsoil (Table 1), the values of BCF and DR were calculated (Table 3). The formula of soil-to-mushroom BCF (Eq. 1) gave information about the elements bioconcentration level. The values of BCF ranged 0.04–0.11 for 210Po and 0.01–0.14 for 210Pb (Table 2) and were lower when compared with the previously studied Leccinum spp. Low BCF values might be connected to soil substrate, namely brown acidic soil. Previous studies showed the highest BCF values were calculated for podzols while the lowest for brown soil (Strumińska-Parulska et al. 2016; Szymańska et al. 2018). Next, in the study, the 210Pb BCF values were lower when compared with 210Po BCF values (the Mann-Whitney U test p = 0.10), which confirmed the previous reports of stable lead (Pb) low bioconcentration (BCF < 1) capabilities by mushrooms (Jarzyńska et al. 2011; Strumińska-Parulska et al. 2016; Szymańska et al. 2018). Gadd (2007) has already noticed fungi may mobilize some metals, can methylate metalloids (e.g., selenium, tellurium, arsenic), or efficiently accumulate and immobilize some forms of metals (e.g., nickel, zinc, cadmium, lead). The L. pseudoscabrum due to the BCF values below unity (BCF < 1) is considered as excluder of 210Po and 210Pb, similarly to other mushrooms studied so far such as Leccinum sp. and parasol mushroom (Macrolepiota procera) (Strumińska-Parulska et al. 2016, 2017).
The estimated values of discrimination ratio (DR) (Eq. 2) might show the radionuclide differentiation in cap and stem and the impact of airborne particle fallout. The calculated DR values ranged 0.17–4.79 for 210Po and 0.27–5.34 for 210Pb (Table 2) and were similar to the previously studied Leccinum sp. mushrooms (described as “translocation factor”) (Strumińska-Parulska et al. 2016; Szymańska et al. 2018). The cluster analysis showed that 210Pb DR value was connected to 210Pb activity concentration in the cap, while 210Po DR depended on its BCF (Fig. 2).
Annual effective radiation doses
One of the important parts of food analysis is its safety evaluation. In order to estimate the potential radiotoxicity from ingested 210Po and 210Pb, the effective radiation doses for adult members of the public due to analyzed mushrooms consumption were calculated (Fig. 4) (ICRP 2012). According to the analyzed L. pseudoscabrum, the consumption of 1 kg of dried caps, stems, and/or whole fruiting bodies (equivalent to 10 kg of fresh mushrooms; assuming moisture content 90%) would give the effective radiation dose at 0.89–9.18 μSv from 210Po decay and 0.42–4.37 μSv from 210Pb decay. The annual effective dose from the total natural radiation in Poland was estimated at 2.1–2.6 mSv, while 210Po and 210Pb taken with food and water give 54 μSv (Pietrzak-Flis et al. 1997). Therefore, the evaluated effective doses from the intake of uncooked L. pseudoscabrum in volume equivalent to 1 kg of dried fruit bodies seemed to be low when compared with other domestic food products available in Poland (Strumińska-Parulska 2015; Strumińska-Parulska et al. 2016; Strumińska-Parulska and Olszewski 2018; Olszewski et al. 2019). Lead leaks at a high rate out of the fruit bodies in the course of the household treatment (e.g., blanching, pickling) or industrial processing (canning) (Drewnowska et al. 2017; Pankavec et al. 2019). Therefore, the intake rates of 210Pb from a mushroom meal (blanched-parboiled and further cooked or blanched/pickled) will result in the lower effective radiation dose than can be assessed for unprocessed produce. Basically, there is no data available on 210Po and 210Pb in cooked mushrooms or an impact of culinary processing.
Conclusions
The studies indicated that 210Po and 210Pb are weakly bioconcentrated (BCF < 1) by hazel bolete (L. pseudoscabrum). The activity levels of 210Po and 210Pb in forest topsoil were low and together with low BCF values for both nuclides accounted for their low activity levels in fruit bodies of L. pseudoscabrum. Based on the data for raw mushrooms, consumption of L. pseudoscabrum could lead to effective doses at 0.89–9.18 μSv kg−1 db from 210Po decay and 0.42–4.37 μSv kg−1 db from 210Pb decay, which are safe from the radiological point of view.
References
Baeza A, Guillen J, Salas A, Manjon JL (2006) Distribution of radionuclides in different parts of a mushroom: influence of the degree of maturity. Sci Total Environ 359:255–266. https://doi.org/10.1016/j.scitotenv.2005.05.015
Chatterjee S, Sarma MK, Deb U, Steinhauser G, Walther C, Gupta DK (2017) Mushrooms: from nutrition to mycoremediation. Environ Sci Pollut Res 24:19480–19493. https://doi.org/10.1007/s11356-017-9826-3
Cocchi L, Kluza K, Zalewska T, Apanel A, Falandysz J (2017) Radioactive caesium (134Cs and 137Cs) in mushrooms of the genus Boletus from the Reggio Emilia in Italy and Pomerania in Poland. Isot Environ Health Stud 53:620–627. https://doi.org/10.1080/10256016.2017.1337761
Drewnowska M, Falandysz J, Chudzińska M, Hanć A, Saba M, Barałkiewicz D (2017) Leaching of arsenic and sixteen metallic elements from Amanita fulva mushrooms after food processing. LWT - Food Sci Technol 84:861–866. https://doi.org/10.1016/j.lwt.2017.04.066
Falandysz J (2017) Mercury accumulation of three Lactarius mushroom species. Food Chem 214:96–101. https://doi.org/10.1016/j.foodchem.2016.07.062
Falandysz J, Treu R (2017) Fungi and environmental pollution. J Environ Sci Health B 52(3):147. https://doi.org/10.1080/03601234.2017.1261535
Gadd GM (2007) Geomycology: biogeochemical transformations of rocks, minerals, metals and radionuclides by fungi, bioweathering and bioremediation. Mycol Res 111(1):3–49. https://doi.org/10.1016/j.mycres.2006.12.001
Guillén J, Baeza A (2014) Radioactivity in mushrooms: A health hazard? Food Chem 154:14–25. https://doi.org/10.1016/j.foodchem.2013.12.083
Gwynn JP, Nalbandyan A, Rudolfsen G (2013) 210Po, 210Pb, 40K and 137Cs in edible wild berries and mushrooms and ingestion doses from high consumption rates of these wild foods. J Environ Radioact 116:34–41. https://doi.org/10.1016/j.jenvrad.2012.08.016
ICRP The International Commission on Radiological Protection (2012) Compendium of dose coefficients based on ICRP Publ 60. ICRP Publ 119, Ann ICRP 41(Suppl). Ottawa
Jadczyszyn J, Pudełko R, Doroszewski A, Łopatka A (2015) Zintegrowany System Informacji o Rolniczej Przestrzeni Produkcyjnej (ZSIRPP), Konferencja naukowa „GIS w rolnictwie”, Starachowice, 25–27.11.2015
Jarzyńska G, Gucia M, Kojta AK, Rezulak K, Falandysz J (2011) Profile of trace elements in parasol mushroom (Macrolepiota procera) from Tucholskie Forest. J Environ Sci Health B 46:741–751
Kirchner G, Daillant O (1998) Accumulation of 210Pb, 226Ra and radioactive cesium by fungi. Sci Total Environ 222:63–70. https://doi.org/10.1016/S0048-9697(98)00288-5
Mietelski JW, Baeza AS, Guillen J, Buzinny M, Tsigankov N, Gaca P, Jasińska M, Tomankiewicz E (2002) Plutonium and other alpha emitters in fruit bodies from Poland, Spain and Ukraine. Appl Radiat Isot 56:717–729. https://doi.org/10.1016/S0969-8043(01)00281-0
Mietelski JW, Dubchak S, Błażej S, Anielska T, Turnau K (2010) 137Cs and 40K in fruiting bodies of different fungal species collected in a single forest in southern Poland. J Environ Radioact 101:706–711. https://doi.org/10.1016/j.jenvrad.2010.04.010
Olszewski G, Szymańska M, Westa M, Moniakowska A, Block K, Strumińska-Parulska D (2019) On the extraction efficiency of highly radiotoxic 210Po in Polish herbal teas and possible related dose assessment. Microchem J 144:431–435
Pankavec S, Hanć A, Barałkiewicz D, Dryżałowska A, Zhang J, Falandysz J (2019) Mineral constituents of conserved white button mushrooms: similarities and differences. Roczn Państw Zakł Hig (Ann Nat Inst Hyg) 71 in press
Persson BRR, Holm E (2011) Polonium-210 and lead-210 in the terrestrial environment: a historical review. J Environ Radioact 102:420–429. https://doi.org/10.1016/j.jenvrad.2011.01.005
Pietrzak-Flis Z, Chrzanowski E, Dembińska S (1997) Intake of 226Ra, 210Pb and 210Po with food in Poland. Sci Total Environ 203:157–165. https://doi.org/10.1016/S0048-9697(97)00144-7
Prand-Stritzko B, Steinhauser G (2017) Characteristics of radiocesium contaminations in mushrooms after the Fukushima nuclear accident: evaluation of the food monitoring data from March 2011 to March 2016. Environ Sci Pollut Res 25:2409–2416. https://doi.org/10.1007/s11356-017-0538-5
Škrkal J, Rulik P, Fantinova K, Mihalik J, Timkova J (2015) Radiocaesium levels in game in the Czech Republic. J Environ Radioact 139:18–23. https://doi.org/10.1016/j.jenvrad.2014.09.016
Skwarzec B (1997) Radiochemical methods for the determination of polonium, uranium and plutonium in environment. Chem Anal 42:107–115
Skwarzec B, Strumińska DI, Ulatowski J, Gołębiowski M (2001) Determination and distribution of Po-210 in tobacco plants from Poland. J Radioanal Nucl Chem 250(2):319–322. https://doi.org/10.1023/A:1017908031690
Skwarzec B, Strumińska DI, Boryło A (2006) Radionuclides of iron (55Fe), nickel (63Ni), polonium (210Po), uranium (234U, 235U, 238U) and plutonium (238Pu, 239+240Pu, 241Pu) in Poland and Baltic Sea environment. Nukleonika 51(2):45–51
Skwarzec B, Strumińska-Parulska DI, Boryło A, Kabat K (2012) Polonium, uranium and plutonium radionuclides in aquatic and land ecosystem of Poland. J Environ Sci Health A 47:1–18
Steinhauser G, Saey PRJ (2016) 137Cs in the meat of wild boars: a comparison of the impacts of Chernobyl and Fukushima. J Radioanal Nucl Chem 307(3):1801–1806. https://doi.org/10.1007/s10967-015-4417-6
Steinhauser G, Brandl A, Johnson TE (2014) Comparison of the Chernobyl and Fukushima nuclear accidents: a review of the environmental impacts. Sci Total Environ 470-471:800–817. https://doi.org/10.1016/j.scitotenv.2013.10.029
Strumińska-Parulska DI (2015) Determination of 210Po in calcium supplements and the possible related dose assessment to the consumers. J Environ Radioact 150:121–125. https://doi.org/10.1016/j.jenvrad.2015.08.006
Strumińska-Parulska DI, Olszewski G (2018) Is ecological food also radioecological? – 210Po and 210Pb studies. Chemosphere 191:190–195. https://doi.org/10.1016/j.chemosphere.2017.10.051
Strumińska-Parulska DI, Skwarzec B, Tuszkowska A, Jahnz-Bielawska A, Borylo A (2010) Polonium (Po-210), uranium (U-238) and plutonium (Pu-239+240) in the biggest Polish rivers. J Radioanal Nucl Chem 286(2):373–380. https://doi.org/10.1007/s10967-010-0716-0
Strumińska-Parulska DI, Szymańska K, Skwarzec B (2015) Radiolead 210Pb and 210Po/210Pb activity ratios in dogs hair. J. Environ. Sci. Health A, 50:1180–1186. https://doi.org/10.1080/10934529.2015.1047675
Strumińska-Parulska DI, Szymańska K, Krasińska G, Skwarzec B, Falandysz J (2016) Determination of 210Po and 210Pb in red-capped scaber (Leccinum aurantiacum): bioconcentration and possible related dose assessment. Environ Sci Pollut Res 23:22606–22613. https://doi.org/10.1007/s11356-016-7473-8
Strumińska-Parulska DI, Olszewski G, Falandysz J (2017) 210Po and 210Pb bioaccumulation and possible related dose assessment in parasol mushroom (Macrolepiota procera). Environ Sci Pollut Res 24(34):26858–26864. https://doi.org/10.1007/s11356-017-0458-4
Szymańska K, Falandysz J, Skwarzec B, Strumińska-Parulska D (2018) 210Po and 210Pb in forest mushrooms of genus Leccinum and topsoil from Northern Poland and its contribution to the radiation dose. Chemosphere 213:133–140. https://doi.org/10.1016/j.chemosphere.2018.09.022
Tuo F, Zhang J, Li W, Yao S, Zhou Q, Li Z (2017) Radionuclides in mushrooms and soil-to-mushroom transfer factors in certain areas of China. J Environ Radioact 180:59–64. https://doi.org/10.1016/j.jenvrad.2017.09.023
Vaaramaa K, Solatie D, Aro L (2009) Distribution of 210Pb and 210Po concentrations in wild berries and mushrooms in boreal forest ecosystems. Sci Total Environ 408:84–91. https://doi.org/10.1016/j.scitotenv.2009.09.005
Acknowledgments
Technical assistance by Joanna Gozdek, Anna Dryżałowska, Renata Arszyło, Michalina Mazur, Magdalena Lenarczyk, and Katarzyna Szałabska is acknowledged.
Funding
This study was partly supported by the Ministry of Sciences and Higher Education under grant no. DS/530-8635-D745-18 and 8636-D506-18.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Georg Steinhauser
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Szymańska, K., Strumińska-Parulska, D. & Falandysz, J. Isotopes of 210Po and 210Pb in Hazel bolete (Leccinellum pseudoscabrum) – bioconcentration, distribution and related dose assessment. Environ Sci Pollut Res 26, 18904–18912 (2019). https://doi.org/10.1007/s11356-019-05376-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-05376-8